Abstract
Histoplasma capsulatum is a dimorphic soil-based fungus endemic to the Ohio and Mississippi River valleys and southeastern United States. An extremely rare presentation is the involvement of the mediastinum leading to vocal fold paralysis, with only 2 reported cases in the literature. This report presents the youngest patient to whom this pathology has been described and the third overall patient. In this case report, we present a 15-year-old boy with vocal fold paralysis secondary to left recurrent laryngeal nerve paralysis in the setting of confirmed histoplasmosis infection. On further workup, he was noted to have mediastinum lymphadenopathy on computed tomography (CT) chest. Pathology of the biopsy indicated granulomatous inflammation without the evidence of malignancy, prompting fungal serology testing resulting in a positive histoplasma antibody result. The patient received appropriate treatment, yet the dysphonia persisted. On flexible laryngoscopy, he was noted to have left vocal fold paralysis. After 2 years of ongoing paralysis, the decision was made to proceed with recurrent laryngeal nerve innervation for long-term benefits. After nerve innervation and fat laryngoplasty, the patient has had significant improvement in dysphonia and voice parameters.
Introduction
Histoplasmosis is caused by the dimorphic soil-based fungus Histoplasma capsulatum, which is endemic in the Ohio and Mississippi river valleys and southeastern United States. 1 H. capsulatum grows particularly well in areas containing bird and bat droppings and disruption of the soil leads to aerosolization of fungal spores, making infection more likely. 2 After infective particles enter the lungs, the local tissue responds by granuloma formation visible on chest radiographs, even in asymptomatic patients. 1
Histoplasmosis leading to vocal fold paralysis is an extremely rare presentation, with only 2 cases reported in the literature.3,4 The first documented case occurred in a 49-year-old man with a 48-pack-year smoking history who presented with 2 months of hoarseness secondary to left recurrent laryngeal paralysis. Computer tomography revealed a noncalcified left upper lobe mass, and wedge resection of the left lobe and lymph node biopsy confirmed budding yeast consistent with histoplasmosis. Additionally, skin testing was immunologically reactive to Histoplasma. 3 A second case occurred in 2022 in a 73-year-old man with a 50-pack-year smoking history who developed a 4-month history of dysphonia, dyspnea during phonation, and aspiration. 4 Videostroboscopy revealed right vocal fold paralysis, and chest CT displayed mediastinal and right paratracheal lymph node enlargement. Biopsy of these nodes confirmed a histoplasmosis infection.
In this report, we present the case of a 15-year-old boy with left recurrent laryngeal nerve paralysis after histoplasmosis infection. This is the first pediatric patient in whom this pathology has been described. The presentation of vocal fold paralysis due to histoplasmosis infection is extremely rare and no cases have been previously reported in a pediatric patient.
Case Report
A 15-year-old boy presented to an otolaryngology clinic with a 10 month history of dysphonia and vocal fold paralysis. Ten months prior, his whole family became ill with symptoms of cough and malaise that lasted ~1 month. He developed persistent dysphonia along with mediastinal, supraclavicular, and cervical lymphadenopathy, which was visible on CT (Figures 1 and 2). Fine needle aspiration of a supraclavicular lymph node was inconclusive, and the patient underwent right video-assisted thoracic surgery with mediastinal lymph node biopsy. Pathology indicated necrotizing granulomatous inflammation without evidence of malignancy (Figure 3). Biopsy results prompted fungal serology and Histoplasma antigen testing, which were positive for complement fixation of Histoplasma antibodies to both mycelia and yeast. The patient began 12 weeks of itraconazole and prednisone treatment. He had improvement in size of his mediastinal lymphadenopathy, but his dysphonia failed to improve. Dysphonia was believed to be secondary to compression from lymphadenopathy.
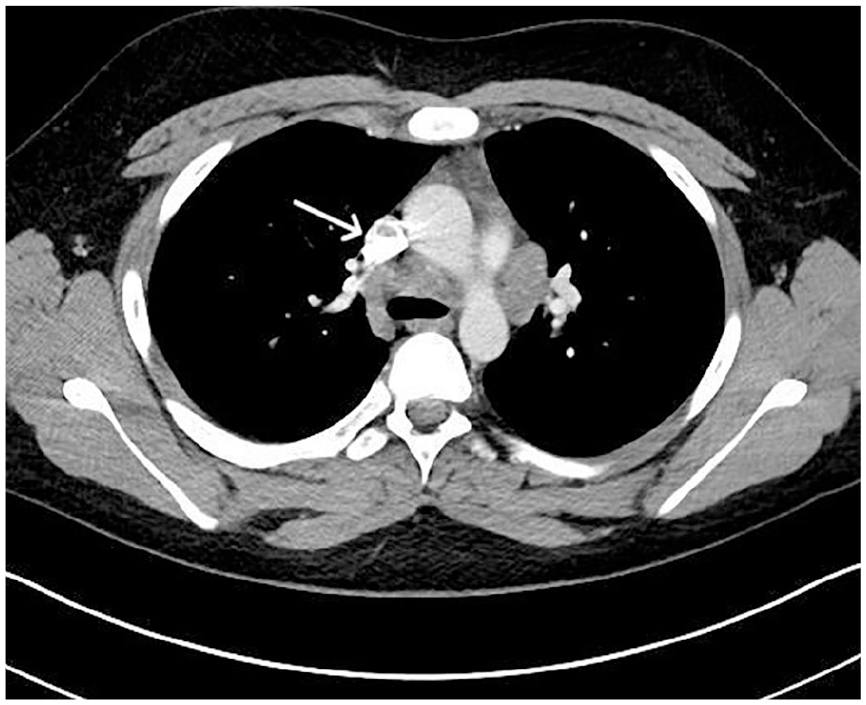
Postcontrast CT on soft tissue window with an arrow indicating mediastinal lymph node with calcification consistent with histoplasmosis.
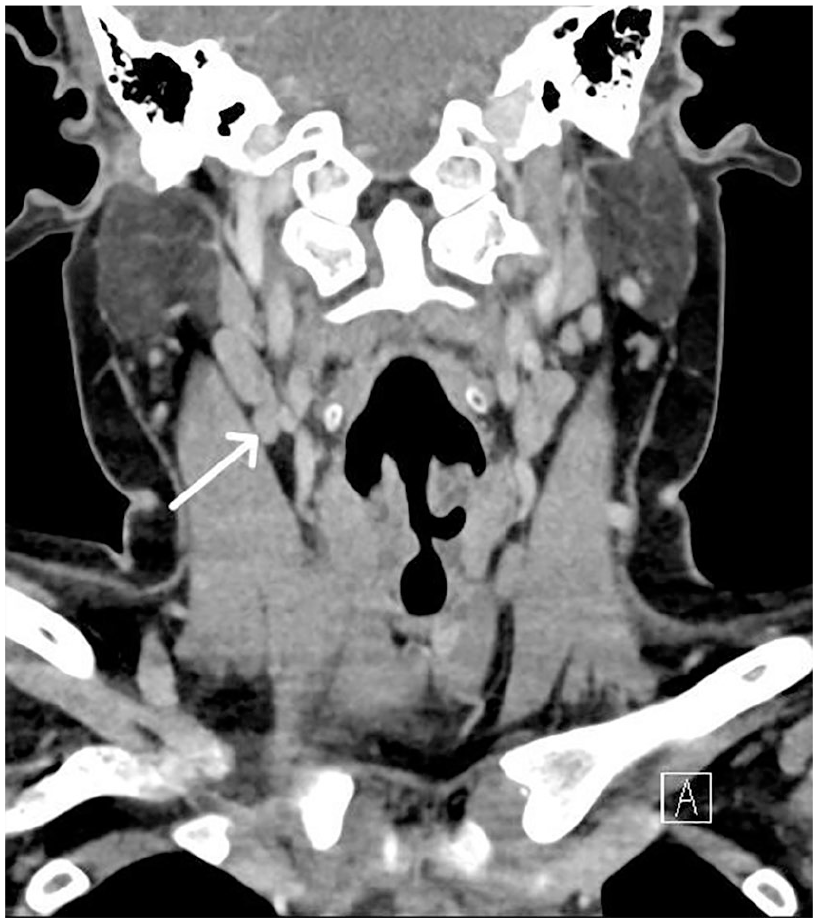
Postcontrast CT on soft tissue window with an arrow indicating right level 2A lymphadenopathy.
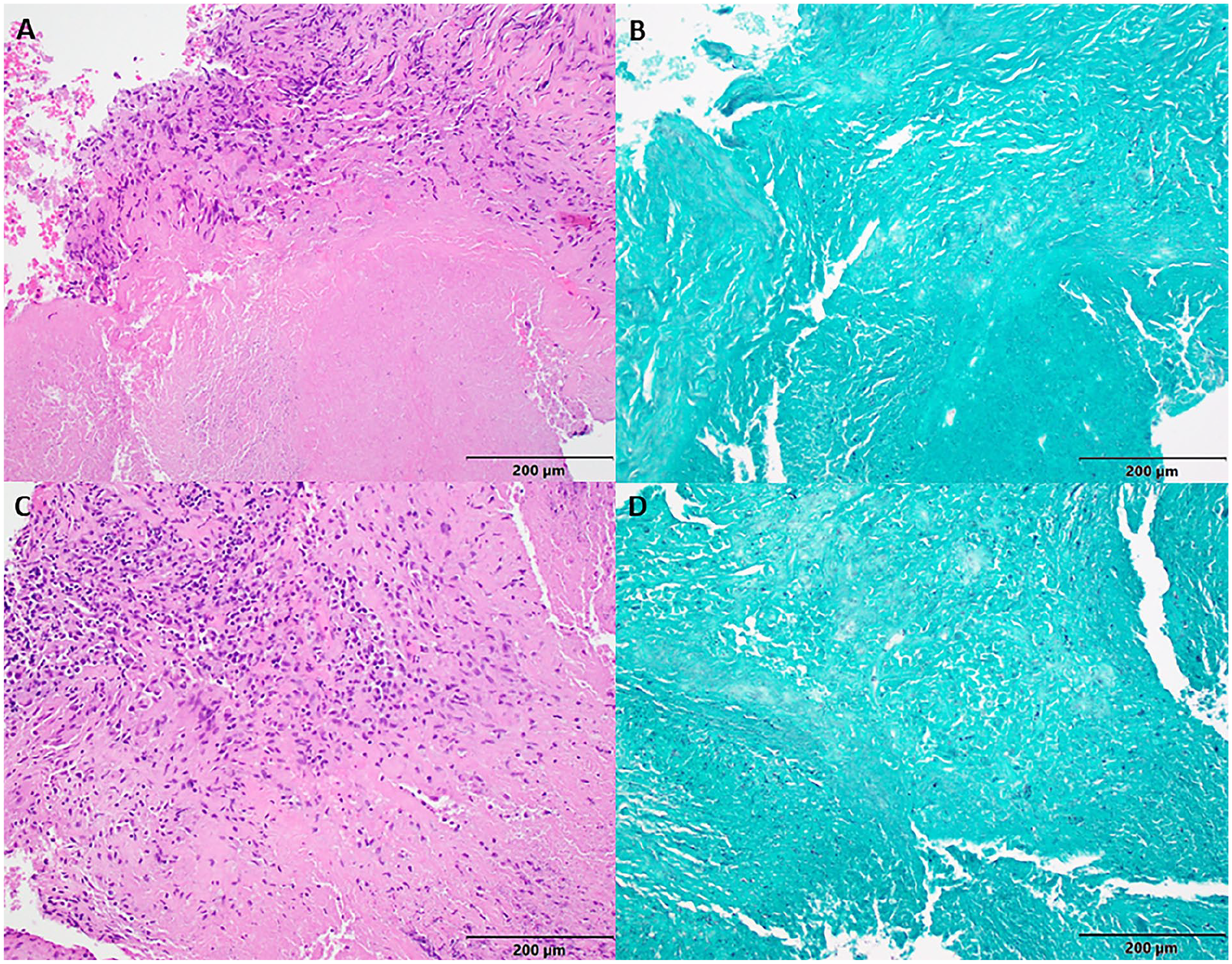
(A) H&E stain, 200× magnification, showing mediastinal lymph node showing a necrotizing granuloma with a mix of epithelioid and multinucleated giant cells, as well as lymphocytes surrounding a necrotic core. (B). GMS stain, 200× magnification of the same specimen as (A) showing nonspecific granular debris staining with GMS. No fungal organisms were seen on this stain; however, the sensitivity for identifying fungal organisms on a GMS-stained 5-micron tissue section is poor. (C) H&E stain, 200× magnification, separate lymph node showing necrotizing granulomatous inflammation and a robust lymphoplasmacytic reaction surrounding the necrosis. (D) GMS stain, 200× magnification, same specimen as (C) showing nonspecific granular debris staining with GMS.
The patient was referred to Otolaryngology and Speech-Language Pathology due to unresolved dysphonia. The patient described the voice loss as stable without fluctuations of use. Glottic Function Index (GFI) was administered to estimate the degree of vocal fold dysfunction on a scale of 0 (least severe) to 20 (most severe), with the patient scoring 12. 5 Voice Handicap Index (VHI-10) was also used to assess the impact on quality of life, and the patient scored an overall score of 50 out of 120, with a score of 21, 10, and 18 in physical, emotional, and functional domains, respectively. Each domain contains a maximum score of 40, with higher scores indicating a greater negative psychosocial impact on quality of life due to voice complaints. 6 One year postinfection, he was found on flexible laryngoscopy to have a completely flaccid left vocal fold with significant atrophy and a large glottic gap that was not amendable to speech therapy alone, prompting surgical intervention. The patient underwent direct laryngoscopy with injection of 0.6 cc ProLaryn gel into the left vocal fold. At 1 month postoperative follow-up, the patient reported 30%-40% improvement in subjective vocal quality. At this time, GFI had improved from 12 to 11, and VHI-10 had improved from 50 to 34 out of 120.
After a few months of postoperative speech therapy, the patient’s voice had only slightly improved. He underwent a left recurrent laryngeal nerve reinnervation using the ansa cervicalis and a fat injection laryngoplasty. Briefly, the platysma, sternocleidomastoid, and omohyoid were dissected and skeletonized. Once retracted, the ansa cerivcalis was visualized. The recurrent laryngeal nerve was identified and divided distally. Both the recurrent laryngeal nerve and ansa cervialis were skeletonized and the 2 nerves were anastomosed. He tolerated the procedure well.
Two weeks postoperatively, the patient noticed a significant improvement in phonation, though diplophonia was present ~50% of the time during conversation. He continued with speech therapy, and 10 months postoperatively, he reported subjective return to baseline with a VHI-10 score of 11/120. On videostroboscopy, the patient had appropriate tone and bulk of the left vocal fold without a significant glottic gap (Figures 4 and 5). With continued speech therapy he has achieved a VHI-10 score of 0. He has agreed to follow-up in clinic as needed and has not reported relapse of his dypshonia, now 40 months postoperatively.
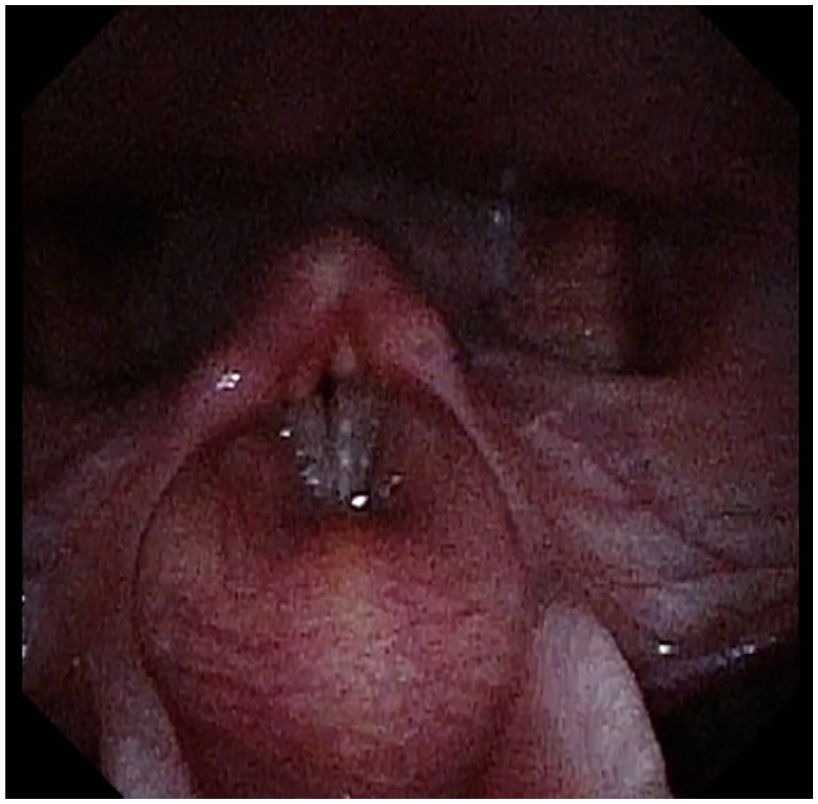
Videostroboscopy of patient adducting vocal folds 10 months post-left recurrent laryngeal nerve reinnervation.
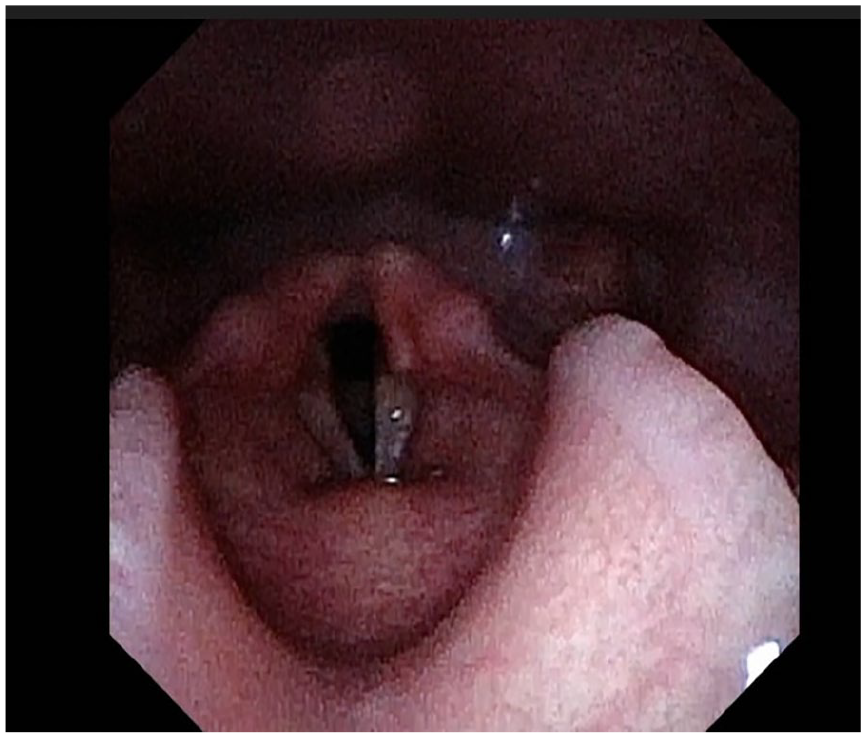
Videostroboscopy of patient abducting vocal folds 10 months post-left recurrent laryngeal nerve reinnervation.
Discussion
Histoplasmosis is a typically benign dimorphic fungus that is most concentrated in the Ohio and Mississippi River valleys. Histoplasma is found in soil and is the most common endemic fungal infection in the United States. 7
Areas of exposure frequently contain birds or bats in ~80% of histoplasmosis outbreak settings. 1 Eighty to 90% of individuals living within endemic areas have been exposed to Histoplasma, with an estimated incidence of 6.1 cases/100,000. 8 Previous estimates found that from 1938 to 2013, 2850 symptomatic cases were reported across the United States, comprising 105 reported histoplasmosis outbreaks.1,2 This is likely an underestimation of cases, as by 2016 only 10 states identified histoplasmosis as a reportable disease. 9 Another study reviewed data from reporting states between 2011 and 2014 and found 3409 histoplasmosis cases were reported as confirmed or likely infections. 9
The most common route of infection is inhalation of fungal spores after exposure to bird or cat droppings, with few patients developing symptoms during an infection. 2 In immunocompetent hosts, infections are typically asymptomatic or result in minimal signs and symptoms without the need to receive medical care. 7 When symptoms do arise from histoplasmosis, the host is usually immunocompromised and develops flu-like symptoms such as fever, malaise, chest discomfort, and dry cough. 8
Histoplasmosis severity varies based on various patient risk factors such as immunocompromised status, large inoculum, and extremes of age. 10 An estimated <1% of individuals infected with Histoplasma spp. develop symptoms, with immunocompetent hosts often only developing mild-flu-like symptoms such as fever, cough, chest pain, and malaise that resolve within 10 days of onset.2,10 When immunocompetent hosts develop pulmonary histoplasmosis, symptoms can include mediastinal adenopathy, severe dyspnea, and diffuse pulmonary infiltrates mimicking hypersensitivity pneumonitis. 10 Histoplasmosis infections are more likely to be severe in an immunocompromised state, with studies estimating 4% 90 day mortality in immunocompetent and 25% 90 day mortality in immunocompromised hosts who develop symptoms, respectively. 11 Immunocompromised patients are also more likely to develop progressive disseminated histoplasmosis symptoms such as fever, hepatosplenomegaly, lymphadenopathy, sepsis with hypotension, and disseminated intravascular coagulation. 12 Severe cases can manifest as acute pulmonary syndrome or disseminated disease of multiple organ system involvement. 13 Disseminated histoplasmosis can vary in presentation and involve every organ system, with the possibility of focal or systematic infection. 12 Possible manifestations include involvement of adrenal glands, testicular, bladder, and gastrointestinal tract.12,14,15
Laboratory studies suggestive of histoplasmosis include elevated alkaline phosphate levels, high lactate dehydrogenase, and increased ferritin expression. 12 Though these tests are suggestive, they are not sufficient for diagnosis. A diagnosis can be obtained through various laboratory techniques. The first technique, described in 1986, uses an immunoassay to detect circulating H. capsulatum polysaccharide antigen in urine and serum. 12 In pulmonary histoplasmosis, bronchoalveolar lavage samples detected antigens in 93% of cases, and adding bronchoalveolar lavage cytopathology increased the sensitivity to 97%. 8 In disseminated infection, mucocutaneous lesions, skin, and bone marrow usually reveal organisms such as yeasts within macrophages. 12
Vocal fold paralysis secondary to histoplasmosis is a rare presentation, with only 2 other cases reported in the literature.3,4 These patients were immunocompetent adult men with significant smoking histories. Patients reported 2 to 4 months of symptoms before presenting to clinic. The present case demonstrates the first case of pediatric vocal cord paralysis secondary to histoplasmosis. The first reported case was treated with ketoconazole (400 mg/daily) for 12 months, with no clinically significant improvement in hoarseness. 3 The second case was successfully treated with oral pozaconazole 300 mg twice daily for the first day, then 300 mg daily for 12 weeks. 4
Conclusion
Symptomatic histoplasmosis infections are rare in immunocompetent patients, and histoplasmosis presenting with vocal fold paralysis is exceedingly rare. Patients who develop symptoms most commonly report an acute course of mild flu-like symptoms. Rarely do patients develop further symptoms if they do not have additional morbidities such as being in an immunocompromised state.
Our patient was the first reported pediatric case of vocal fold paralysis secondary to histoplasmosis infection. Additionally, our patient was immunocompetent. His presentation demonstrates that histoplasmosis should be included in a broad differential diagnosis even in immunocompetent patients. Previous literature describes 2 cases, both including older patients who were immunocompetent, although they did each have an extensive smoking history. Our patient is unique even within this rare histoplasmosis presentation in that the patient was an adolescent without a smoking history.
Our patient’s vocal fold paralysis was successfully managed with a left recurrent laryngeal nerve reinnervation with autologous fat laryngoplasty. Although recurrent laryngeal nerve paralysis resulting from histoplasmosis appears to be exceedingly rare, providers should consider this etiology whether caring for adult or pediatric patients with dysphonias. Surgical and medical management in the small number of cases indicates a favorable prognosis in this rare presentation.
Footnotes
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Participate
Written informed consent was obtained from the patient’s legally authorized representative for the participation of this case report.
Consent for Publication
Written consent for this case report has been obtained from the patient’s legally authorized representative for the publication of this case report.
Author Contributions
Jacob Dylan Johnson: writing – original draft, writing – review and editing. Bronson C. Wessinger: Writing – review and editing. Derek B. Allison: writing, review and editing, figure creation. Caitlin E. Fiorillo: writing – review and editing, supervision.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
