Abstract
Objective:
To identify risk factors and develop a predictive model for the onset of external auditory canal stenosis (EACS) after endoscopic surgery.
Patients and Methods:
A retrospective analysis was conducted in 362 patients who underwent endoscopic surgery from January 2021 to September 2023. The patients were categorized into a training set (n = 217) and a test set (n = 145). A single-factor regression analysis was used to identify significant differences between the EACS and non-EACS groups within the training set. Least absolute shrinkage and selection operator (LASSO) regression analysis and multivariate logistic regression were employed to screen and develop predictive models, visualized in a nomogram. The predictive accuracy of the nomogram was assessed using receiver operating characteristic (ROC) curves, calibration plots, decision curve analysis (DCA), and clinical impact curves (CIC).
Results:
Univariate regression analysis of the training set revealed that the history of EACS, the history of ear surgery, the operative time, the levels of triglycerides (TG), the systemic immune-inflammation ratio (SIRI), and the albumin-to-creatinine score (AISI) were significant factors between the 2 groups (P < .05). Subsequently, these variables were included in the LASSO regression analysis, which identified 4 high-risk factors: history of ear surgery, operative time, TG levels, and SIRI. The model exhibited strong predictive capacity, with an area under the ROC curve of 0.89 (95% CI 0.82-0.95) in the training set and 0.88 (95% CI 0.72-1.00) in the validation set. Calibration curves, DCA, and CIC analyses further demonstrated the model’s excellent predictive value and clinical utility.
Conclusions:
The developed nomogram is a significant tool for predicting postoperative EACS in patients undergoing endoscopic surgery. It offers a valuable reference for the early identification of high-risk patients, facilitating timely clinical intervention and promoting personalized and precise treatment strategies.
Introduction
Acquired external auditory canal stenosis (EACS) is a subtype of EACS that arises from conditions such as surgery, inflammation, and burns. Medical interventions, including tympanoplasty, mastoidectomy, and meatoplasty, are significant contributors to the development of acquired EACS.
Li et al conducted a retrospective analysis of data from 131 cases (133 ears) of surgeries involving the outer and middle ear. The study revealed that 9 cases (10 ears) developed EACS, resulting in an incidence rate of 7.52%. 1 Acquired EACS can result in secondary surgeries, prolonged hospital stays, and increased healthcare costs, imposing additional physical and financial burdens on patients. Endoscopy, favored for its wide field of view, clear visualization, and minimally invasive nature in middle and outer ear surgeries, can increase the risk of skin defects in the external auditory canal and subsequent stenosis due to its approach through the external auditory canal. Despite these efforts, the etiology and pathogenesis of EACS remain unclear. Identifying high-risk patients preoperatively and implementing early postoperative measures could significantly reduce the incidence of stenosis. The primary objective of this study is to investigate and identify risk factors that contribute to postoperative EACS in otological procedures. By developing a robust risk prediction model, this research aims to provide a scientific basis for early clinical intervention and the formulation of tailored treatment plans, ultimately improving patient outcomes.
Methods
Study Design and Patient Selection
Data from electronic medical records were obtained for a cohort of 362 patients who underwent otoscopic surgery at the Department of Otorhinolaryngology of Xuzhou Medical University, over a period extending from January 2021 to September 2023. The patient cohort was randomly assigned to 2 groups: a training group comprising 217 participants and a test group with 145 participants, following a 6:4 ratio. The patients were stratified into the EACS group or the non-EACS group, depending on the presence or absence of EACS within a 6-month postoperative period. The inclusion criteria for the study were the following: (1) patients who underwent otoscopic middle ear or outer ear surgery and (2) those with symptom duration of at least 3 months who did not respond to conservative treatment. Exclusion criteria included: (1) patients who underwent microscopic middle ear surgery, (2) those in the acute phase of infection, (3) individuals with coagulation disorders, and (4) patients with severe systemic comorbidities. The ethics review board of the hospital granted the study with approval number XYFY2024-KL215-01. Patient consent was not required as this analysis was retrospective. All surgical procedures were performed by a single otologic surgeon with over 15 years of specialized experience in the field.
Data Collection
Based on the existing literature and clinical experience, the researchers conducted a thorough review of the participants’ medical records to extract essential data. This included fundamental demographic information such as name, sex, age, height, weight, duration of the disease, and medical history, specifically noting the presence of hypertension and diabetes mellitus. Consistent with NIH clinical trial guidelines, subjects were categorized into children (<18 years), adults (18-64 years), and the elderly (≥65 years) cohorts. In addition, the history of surgeries and EACS was documented. The study focused on 3 types of disease: noncholesteatomatous chronic otitis media, chronic cholesteatomatous otitis media, and external auditory canal atresia. In terms of surgical procedures, the 3 most frequently performed otoscopic surgeries in our institution were categorized and analyzed: simple tympanoplasty, mastoidectomy tympanoplasty, and meatoplasty.
After hospital admission, a comprehensive panel of laboratory tests was conducted to ascertain a range of hematologic and biochemical parameters. This included cholesterol and triglyceride (TG) levels, as well as a complete blood count to enumerate neutrophils, lymphocytes, monocytes, and platelets. Calculations for SIRI (Systemic Inflammatory Response Index), SII (Systemic Inflammation Index), AISI (Systemic Inflammation Synthesis Index), NLR (neutrophil/lymphocyte ratio), PLR (platelet/lymphocyte ratio), and MLR (monocyte/lymphocyte ratio) were performed, along with the calculation of the systemic inflammatory index, in the following manner: The SIRI is made up of neutrophils, monocytes, and lymphocytes; the SII is made up of neutrophils, platelets, and lymphocytes; and the AISI is made up of neutrophils, monocytes, platelets, and lymphocytes. The NLR is made up of neutrophil, granulocyte, and lymphocyte counts; the PLR is made up of platelets; and the MLR is made up of lymphocytes. Postoperative follow-up was systematically conducted for all patients. The first outpatient visit was scheduled 2 weeks after surgery, and subsequent follow-ups were performed regularly within 3 months postoperatively, depending on clinical symptoms. Each follow-up involved otoscopic cleaning of the operative cavity, morphological examination of the tympanic membrane and the external auditory canal, and documentation of otoscopic imaging data. The final assessment, conducted 6 months postoperatively, utilized a standardized measurement method: The external auditory canal isthmus was designated as the measurement point, and the diameter of the external auditory canal was quantitatively measured using Image J software (National Institutes of Health, Bethesda, MD), with a 2-mm diameter crochet/circumferential cutter as a reference (Figure 1).
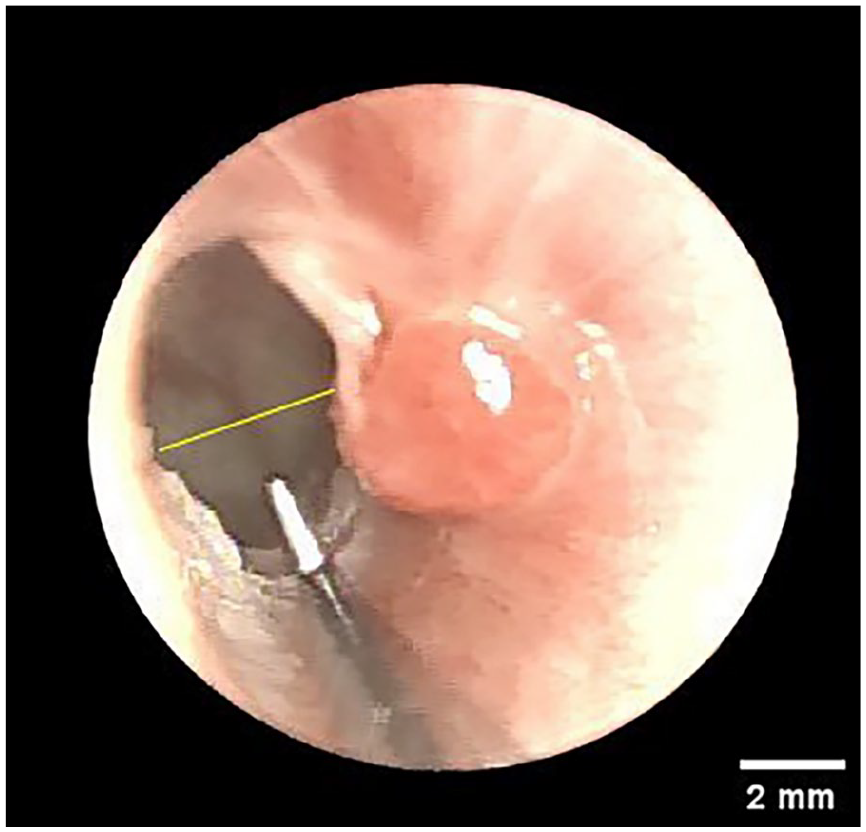
Images of the external auditory canal after otoscopic surgery.
Definition of EACS
In the context of our survey, EACS was operationally defined as a condition characterized by an opening diameter of fewer than 4 mm. Drawing from the seminal work of Jahrsdoerfer, it has been established that an external auditory canal diameter of fewer than 4 mm significantly increases the likelihood of exacerbation by cholesteatoma within the same canal. This association is particularly pronounced when the diameter of the external auditory canal is reduced to less than 2 mm. 2 In this research, the identification of EACS adhered to the specified criterion. In this study, postoperative External Auditory Canal (EAC) diameter at the isthmus was measured according to Jahrsdoerfer’s criteria for EACS (<4 mm) using ImageJ software with a 2-mm reference crochet for standardization (Figure 1). Since preoperative isthmus measurements were not routinely available, we implemented 2 safeguards: (1) intraoperative removal of stenotic tissue with bony canal enlargement to normal dimensions when preoperative stenosis was present and (2) definition of EACS as de novo stenosis (<4 mm) at 6-month follow-up. Patients with prior ear surgery were carefully screened to exclude pre-existing stenosis, ensuring observed EACS cases were attributable to the current endoscopic procedure.
Statistical Analysis
In the statistical analysis of our study, continuous variables were expressed as mean accompanied by standard deviation, with group comparisons performed using the t-test. Categorical variables were depicted through frequencies and percentages, with their significance evaluated via the Chi-square test or Fisher’s exact test, as appropriate. To facilitate statistical analysis and enhance clinical applicability, several continuous variables, including but not limited to surgery duration, disease duration, and various inflammatory indices such as SIRI, AISI, SII, NLR, PLR, MLR, as well as neutrophil counts, lymphocytes, monocytes, and platelets, were categorized. The optimal cutoff points for the transformation of continuous into categorical variables were determined using the ROC curve analysis, with the maximum Jordon index (sensitivity + specificity − 1) being utilized to identify these thresholds. Univariate analysis was conducted to identify variables with a P-value of less than .1 and of clinical relevance, which were subsequently subjected to lasso regression. Multivariate analysis was performed using the “lm” function in the R software (R Foundation for Statistical Computing, Vienna, Austria), incorporating factors selected from the lasso regression analysis, and nomograms were constructed using the “rms” package. The “pROC” package was employed to plot the ROC curves for both the training and testing datasets, and the concordance index (C-index) was calculated to assess the model’s predictive performance. Specifically, an AUC of 0.5 to 0.7 indicates a low predictive value, 0.7 to 0.8 indicates a moderate predictive value, 0.8 to 0.9 indicates a good predictive value, and greater than 0.9 indicates an excellent predictive value. Furthermore, the “rmda” package was utilized to carry out decision curve analysis (DCA) and clinical impact curves (CIC), which provided an evaluation of the predictive accuracy of the developed models. Statistical analyses were performed with SPSS (SPSS Inc., Chicago, IL) version 22.0 and R version 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria). A P-value of less than .05 was considered to indicate statistical significance.
Results
Population and Clinical Characteristics
Following the application of inclusion and exclusion criteria, the study sample comprised 362 participants, consisting of 166 males and 196 females, with ages ranging from 6 to 81 years and a median age of 46 years. The cohort exhibited an average duration of illness with an IQR of 12 months. The collected cases were stratified into a training group comprising 217 individuals and a testing group of 145 individuals, adhering to a 6:4 ratio. The study documented 29 cases of EACS, which constituted 7.70% of the total surgical procedures performed on otoscopic otitis media. Table 1 provides a comparative analysis of the clinical differences between the training and testing groups. Within the training group, EACS was observed in 21 patients, representing 9.68% of the group, while in the testing group, EACS occurred in 8 patients, representing 5.52% of that group.
Comparison of Characteristics Between the Training and Testing Sets.
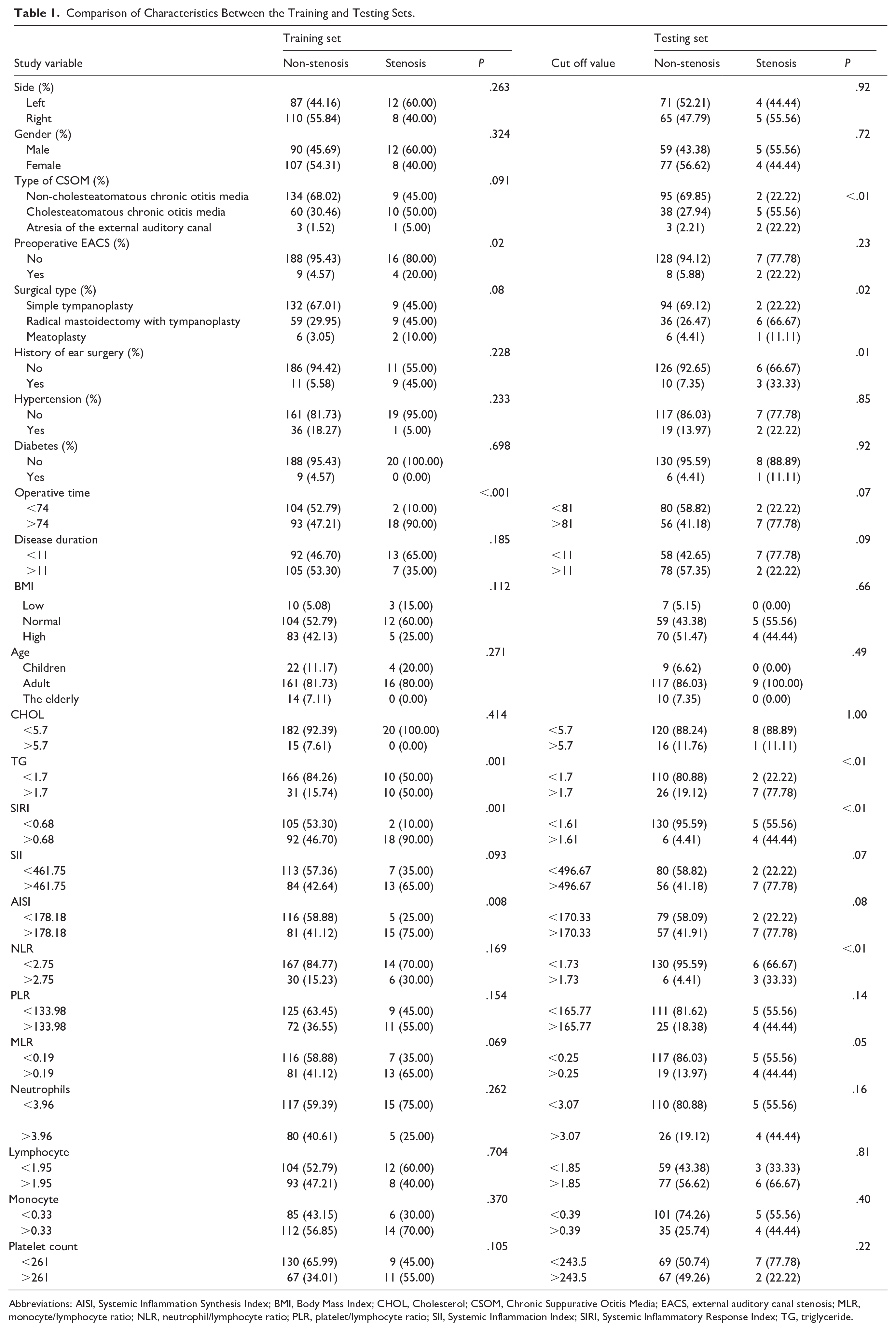
Abbreviations: AISI, Systemic Inflammation Synthesis Index; BMI, Body Mass Index; CHOL, Cholesterol; CSOM, Chronic Suppurative Otitis Media; EACS, external auditory canal stenosis; MLR, monocyte/lymphocyte ratio; NLR, neutrophil/lymphocyte ratio; PLR, platelet/lymphocyte ratio; SII, Systemic Inflammation Index; SIRI, Systemic Inflammatory Response Index; TG, triglyceride.
Screening Predictors and Building the Nomogram
Initial univariate analysis (P < .05) identified 7 potential predictors, which were subsequently analyzed using LASSO regression with EACS as the binary outcome (Figure 2). Through 10-fold cross-validation, we determined the optimal regularization parameter (λ = .012) that minimized the mean squared error. This process yielded 4 significant variables: prolonged operative time, elevated TG levels, increased SIRI, and a history of ear surgery (Figure 3). While these findings demonstrate predictive value, additional multivariate analyses are required to account for potential confounders and covariances, thereby enhancing the model’s clinical utility.
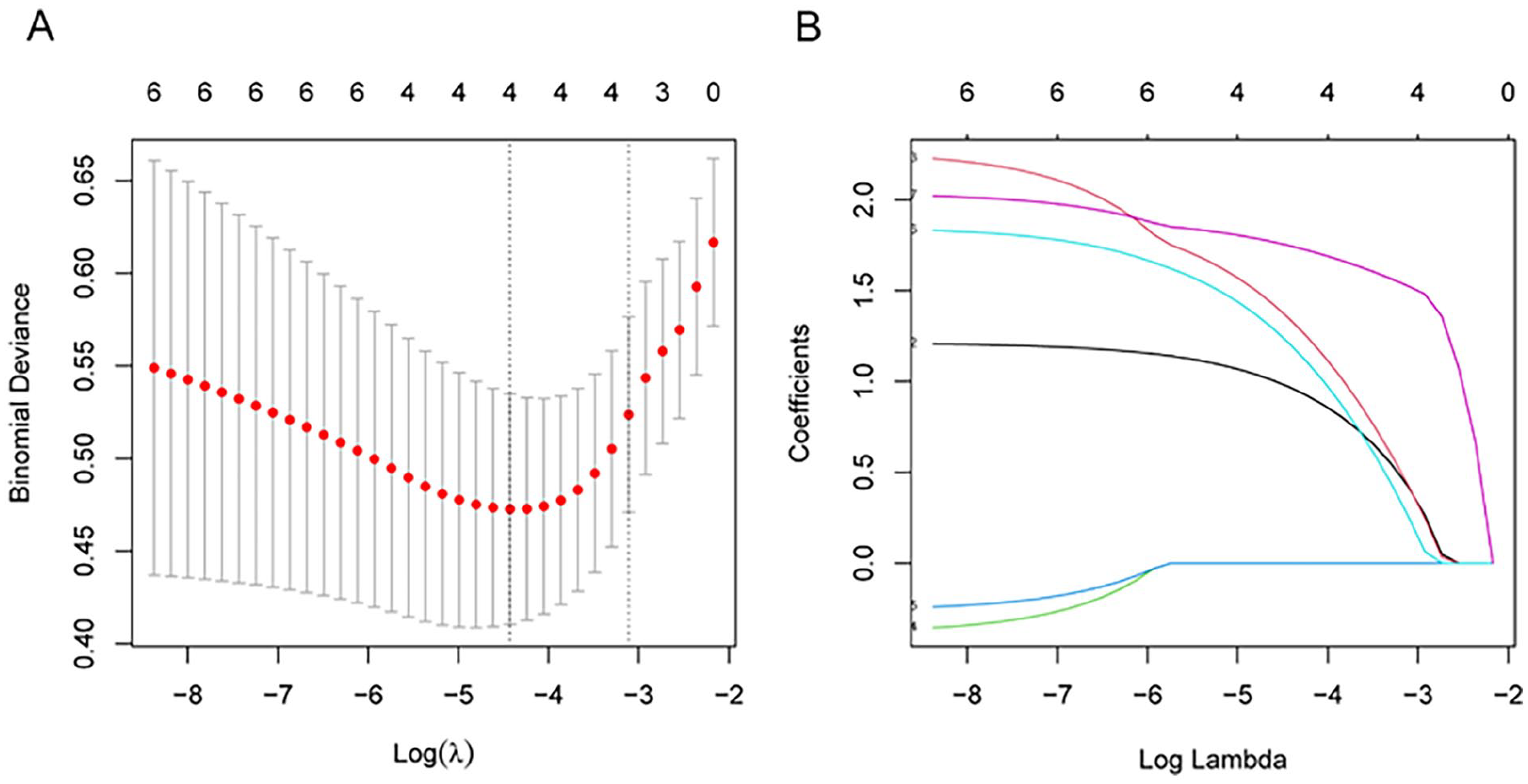
LASSO illustration of the regression analysis outcomes. (A) Applying LASSO regression to conduct a preliminary evaluation of clinical risk factors. (B) The coefficients of the risk factors were plotted using LASSO regression. LASSO, least absolute shrinkage and selection operator.
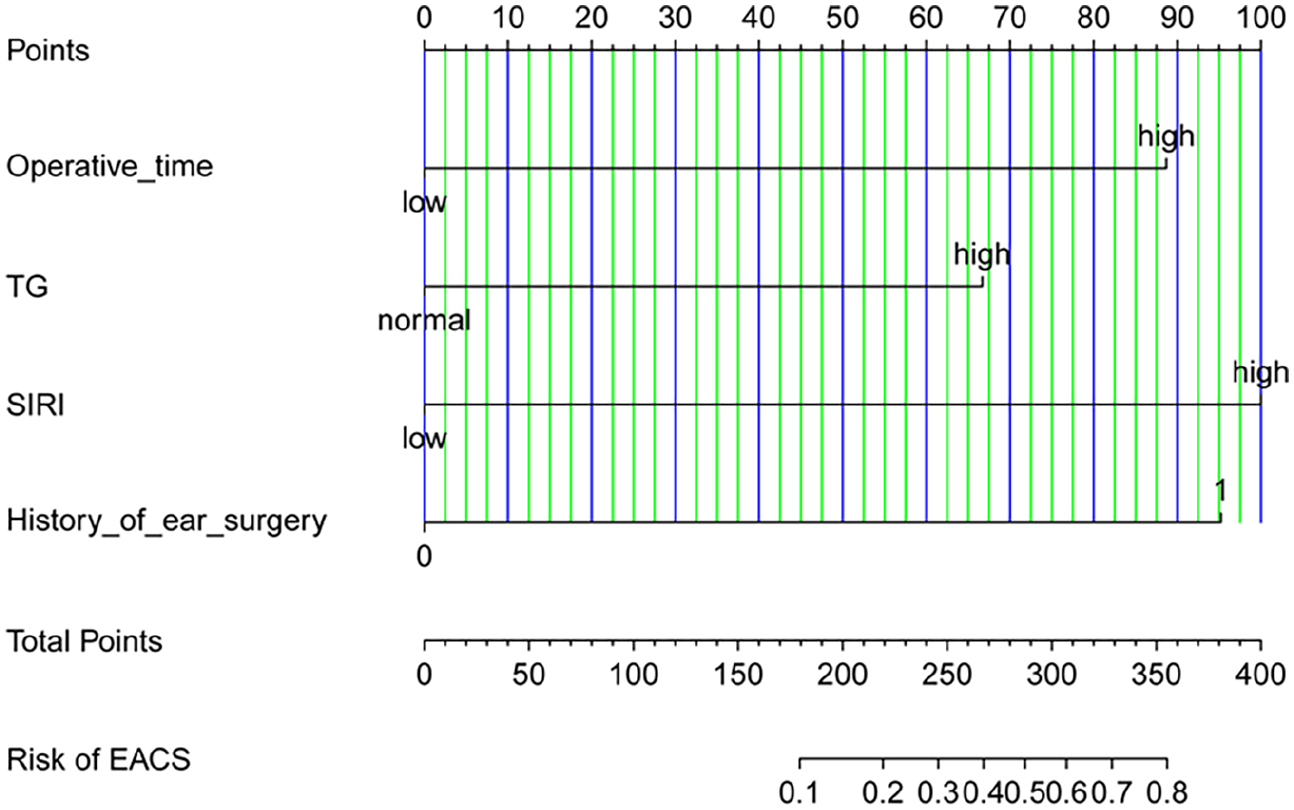
A nomogram is used to forecast the risk of EACS. Each predictor level represents a specific score. The total score is generated by aggregating the scores for each predictor variable. The total score corresponds to the EACS probability. EACS, external auditory canal stenosis.
In addition, a nomogram was developed based on these risk factors to estimate the probability of EACS. The nomogram allows the calculation of the total score by summing the values of all variables and then determining the estimated probability of EACS by drawing a vertical line from the total score axis to the outcome axis (see Table 2).
Risk Factors for EACS.
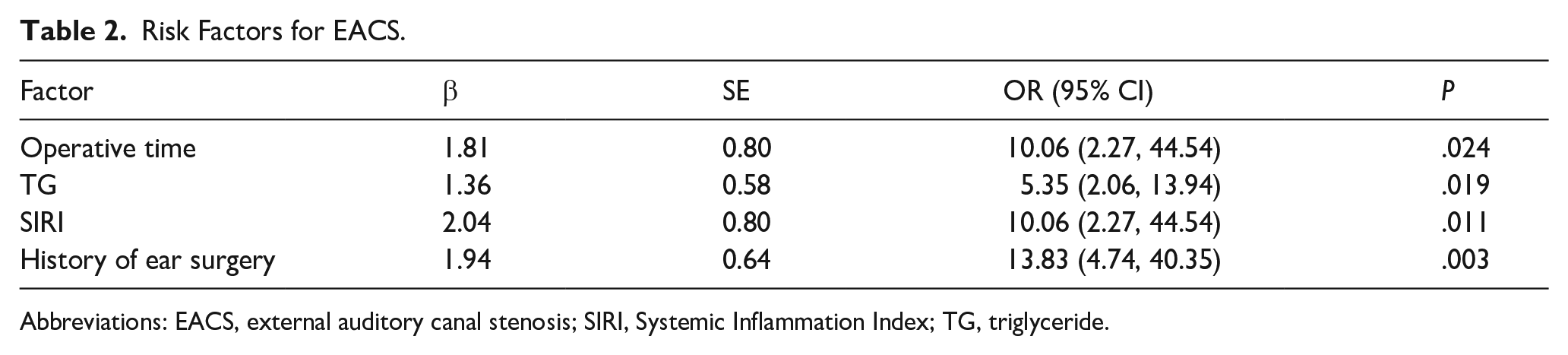
Abbreviations: EACS, external auditory canal stenosis; SIRI, Systemic Inflammation Index; TG, triglyceride.
Evaluation and Testing of the Nomogram
We used calibration and discrimination curves to evaluate the performance of the nomogram in predicting the occurrence of EACS after otoscopic media otitis surgery (Figure 4). Calibration curve analysis demonstrated a good fit between predicted probabilities and the actual incidence of EACS in both the training and validation sets (Figure 5). The ROC curve for the model was 0.89 (95% CI 0.82-0.95) in the training set and 0.88 (95% CI 0.72-1.00) in the validation set, indicating good predictive discrimination. The ROC curve and the AUC are commonly used to assess the accuracy of diagnostic methods. Specifically, an AUC of 0.5 to 0.7 indicates low predictive value, 0.7 to 0.8 indicates moderate predictive value, 0.8 to 0.9 indicates good predictive value, and greater than 0.9 indicates excellent predictive value. The predicted probabilities of the EACS risk on the nomogram correlated well with the actual probabilities of the risk. The clinical utility of the model was further assessed using DCA and CIC curves. DCA revealed that, in the training cohort, the nomogram provided a superior net benefit when the threshold probability was between 0 and 0.7. DCA demonstrated the superior clinical utility of the nomogram across a wide probability threshold range. In the training cohort, the nomogram provided higher net benefit than both “treat-all” and “treat-none” strategies at probability thresholds of 0 to 0.8. This clinical advantage was maintained in the validation cohort with an extended threshold range of 0 to 0.85. These findings indicate that our predictive model offers robust clinical value for decision-making across most clinically relevant scenarios. CIC demonstrated excellent agreement between predicted and observed probabilities when the risk threshold exceeded 0.5 in the training cohort and 0.6 in the validation cohort. This threshold-dependent calibration accuracy was maintained across the clinically relevant range (0.5-0.9), with >90% of predictions falling within the 95% confidence intervals of observed event rates (Figures 6 and 7).
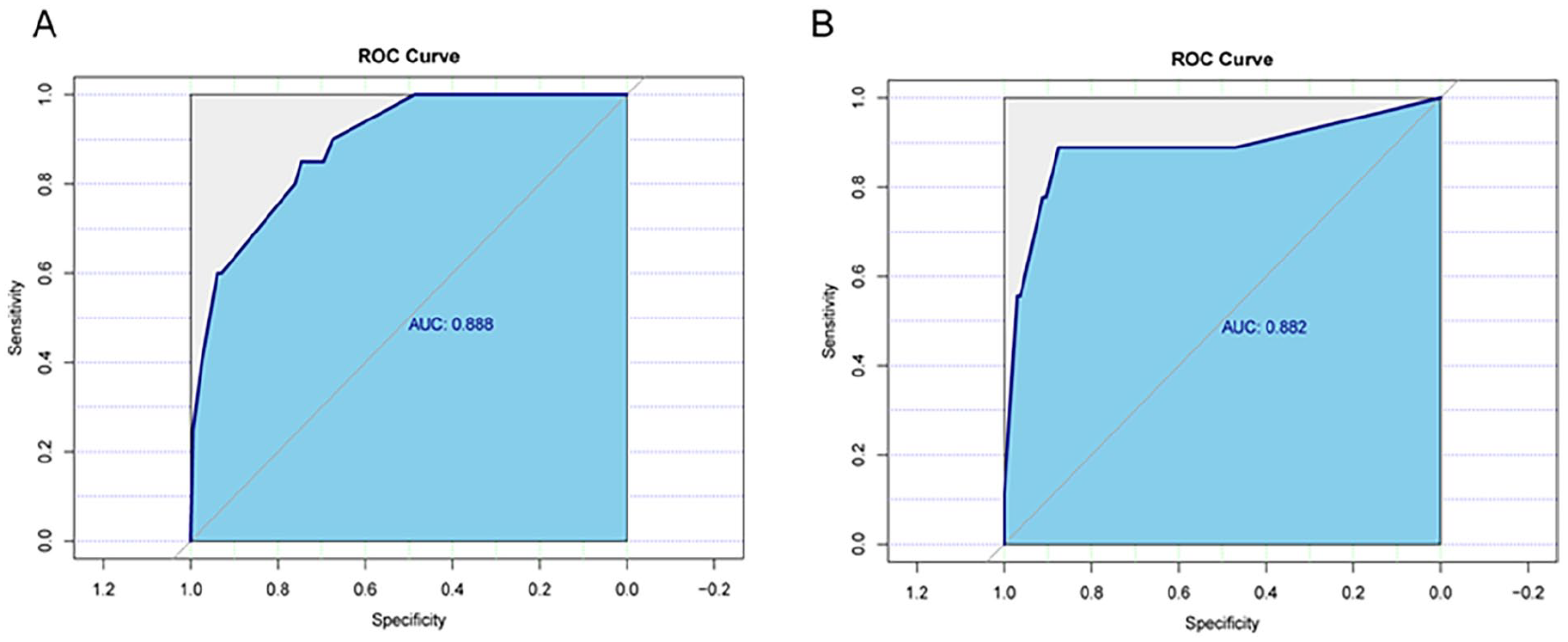
(A) Training set ROC curve and (B) testing set ROC curve. ROC, receiver operating characteristic.
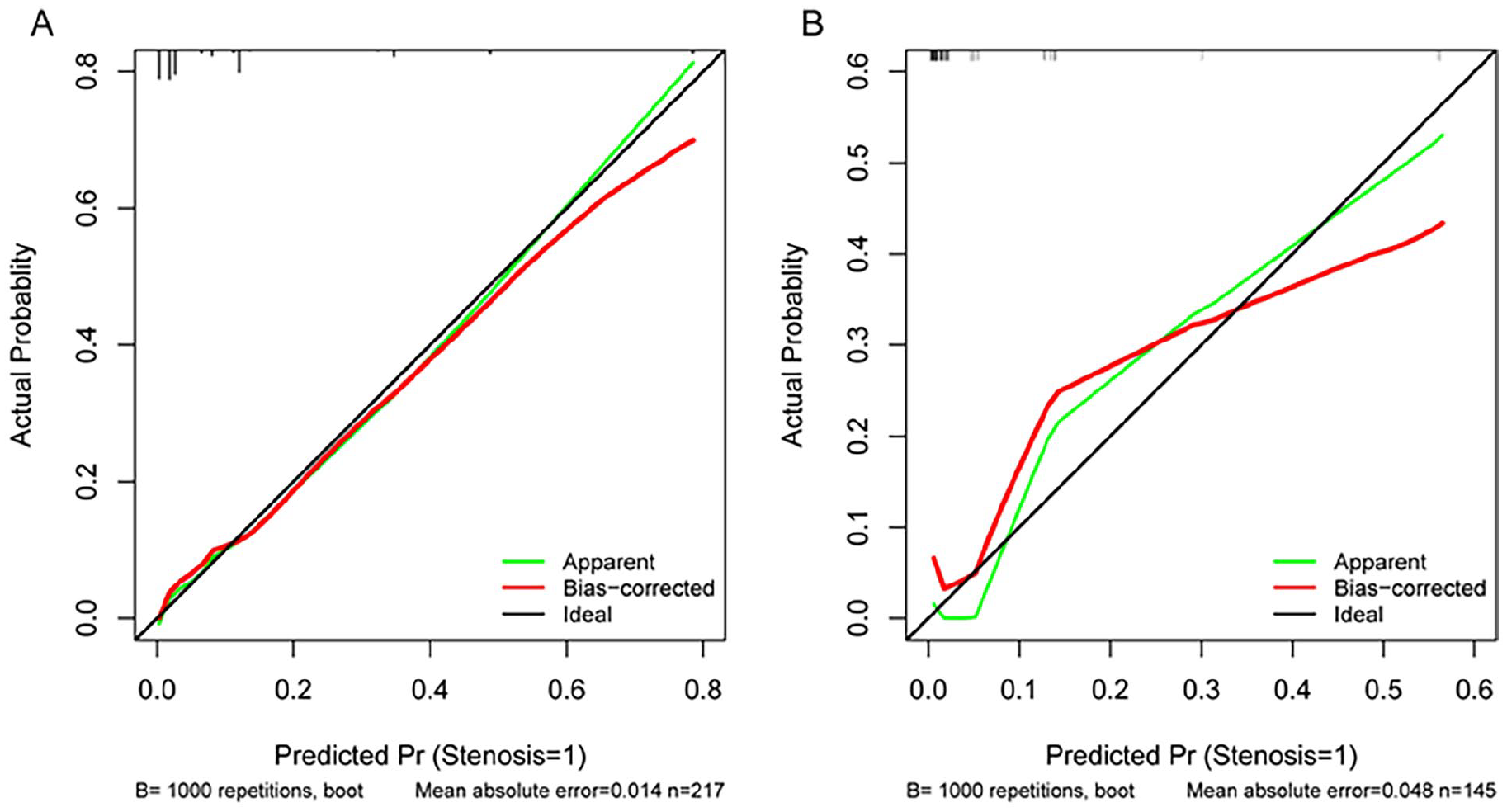
(A) Training set calibration curve and (B) testing set calibration curve; The x-axis is the predicted probability of the EACS. The y-axis is the observed EACS. The black line represents the perfect prediction of the ideal model. The green line represents the performance of the nomogram. The closer the green line is to the black line, the better the prediction performance. EACS, external auditory canal stenosis.
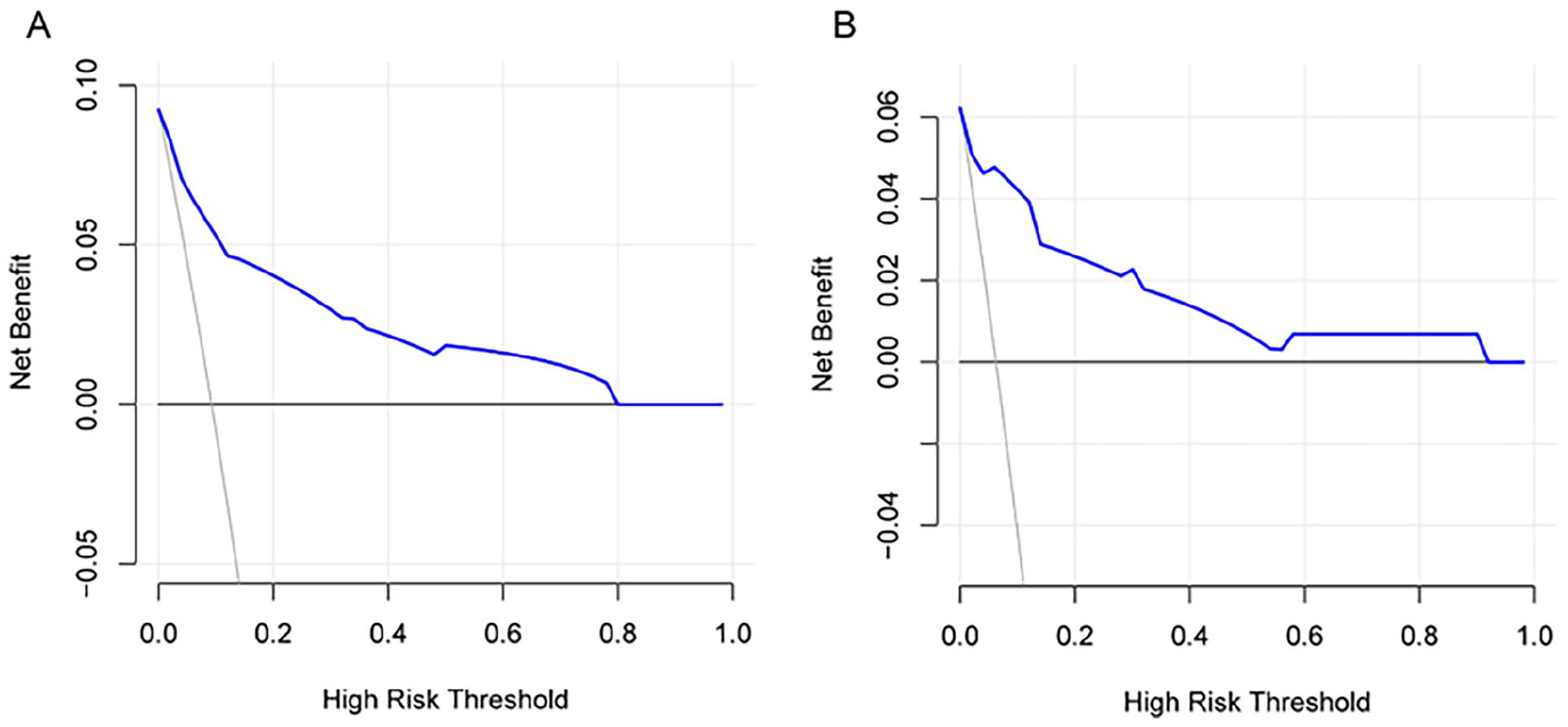
DCA for EACS nomogram. (A) Decision curve for the nomogram in significant cohorts and (B) verify the decision curve of the nomogram in the queue. On the x-axis, the probability threshold is depicted; the y-axis shows the overall advantage; the light gray curve of the negative slope signifies the occurrence of EACS in all patients with postoperative otitis media; and the black horizontal line indicates the absence of EACS in all patients with postoperative otitis media patients; the blue lines illustrate the predictive model’s decision-making advantage across various threshold probability levels. DCA, decision curve analysis; EACS, external auditory canal stenosis.
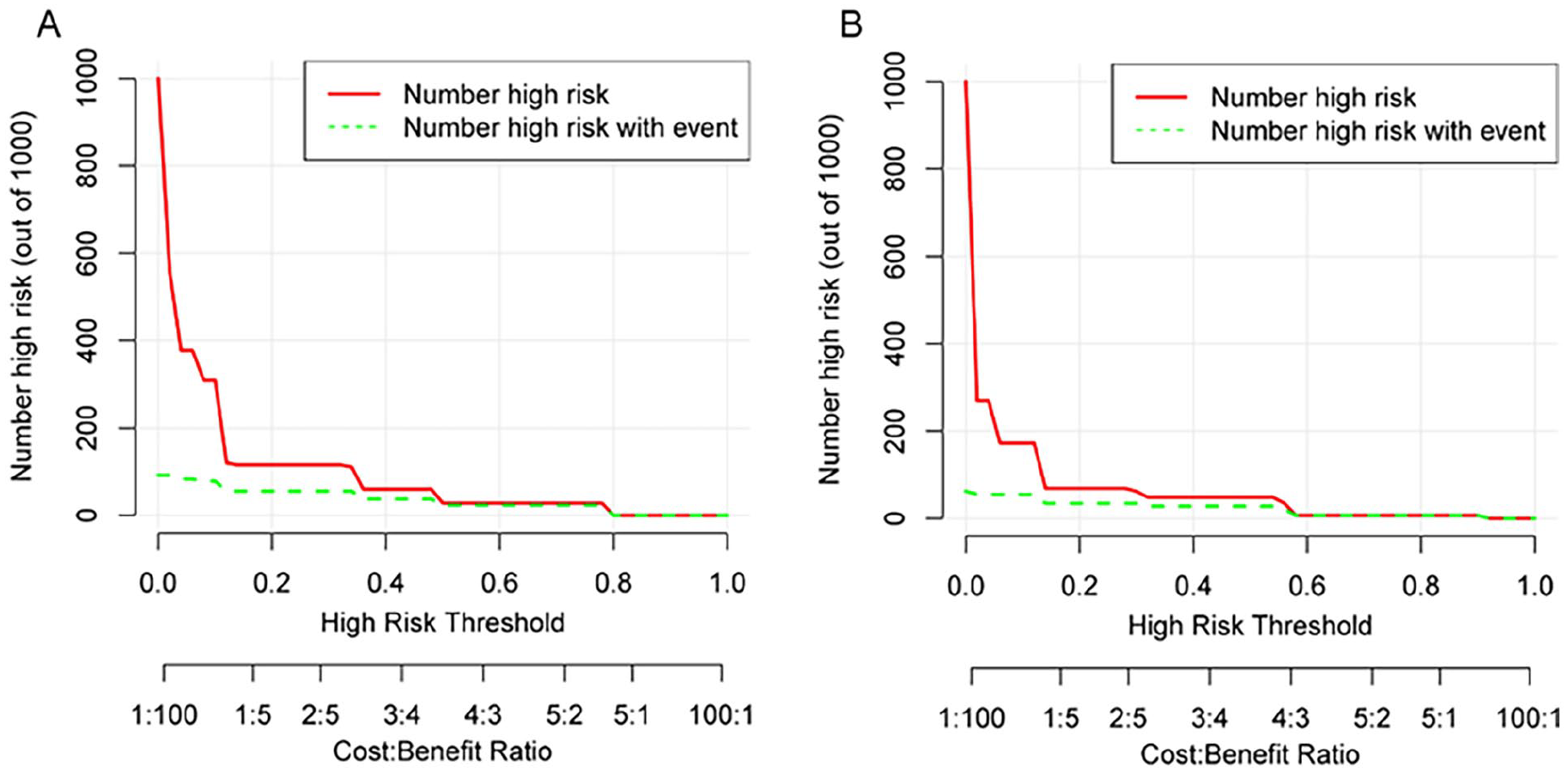
(A) Clinical impact curves for training sets and (B) clinical impact curves for testing sets. The red line (representing high-risk individuals) reflects the count of people identified as positive (high-risk) by the model at various threshold probabilities; the green curve (number of high-risk individuals with results) is the number of true positives at each threshold probability. Clinical impact graphs reveal significant net advantages for both types of nomograms, indicating their substantial predictive value in clinical settings.
Discussion
After otologic surgery, acquired EACS presents a significant clinical challenge. This condition may precipitate reduced ventilation and impaired self-cleansing of the external auditory canal, progression of atresia, complications due to cholesteatoma, and severe cases, involvement of the auditory ossicular chain leading to profound hearing loss.
The reported incidence rates differ slightly due to distinct study populations. The incidence rate of 7.52% in Li et al’s early work (n = 131) was used as a benchmark. In our larger cohort (n = 362), the overall incidence was 7.70% (29 cases). However, stratification revealed a higher incidence in the training set (9.68%, 21/217) than in the testing set (5.52%, 8/145). This discrepancy may stem from random variability in subgroup allocation or a marginally higher-risk profile in the training cohort. Importantly, the overall incidence remains consistent with prior findings, supporting the generalizability of our results while highlighting the utility of risk stratification in clinical prediction models.
The etiology and contributing factors of EACS remain to be elucidated. Nomograms are valuable tools in the diagnosis and prognosis of a variety of diseases, including cancer outcomes and surgical prognoses. They offer enhanced precision in estimating an individual’s risk of disease by incorporating multiple patient-specific variables, thereby facilitating the clinical decision-making process. This investigation introduces the novel application of nomograms for the prediction of EACS following otoscopic otitis media surgery. Utilizing multifactorial logistic regression analysis, the study identified operative duration, TG levels, history of ear surgery, and SIRI as independent risk factors for EACS and subsequently developed a predictive model based on the nomogram. The AUC of the predictive model was 0.89 in the training set and 0.88 in the testing set, indicating its robust predictive capacity. The goodness-of-fit test demonstrated a strong correlation between the predicted probabilities and actual outcomes. DCA further confirmed the clinical utility of the model across a broad spectrum of applications.
The study has identified hyperlipidemia as a substantial risk factor for the development of EACS. Nevertheless, the underlying mechanisms connecting hyperlipidemia to EACS are not yet fully understood.
A multitude of scholarly reviews have reported robust associations between hyperlipidemia and wound healing dynamics, as well as the occurrence of perioperative complications in a spectrum of surgical procedures. These studies have consistently indicated that elevated lipid levels may exert significant influences on the postoperative recovery process and the risk of adverse outcomes following surgery.3-5 Regarding direct heart surgery, Hosseinrezaei and colleagues noted an increased incidence of infections in sternal wounds among patients with hyperlipidemia. 6 Yamada et al observed that particular lipid species are present at concentrations that exceed 32 times in surgical specimens obtained from individuals afflicted with lumbar stenosis, compared to those devoid of the condition. 7 In an experiment of 41 cases of post-inflammatory medial meatal fibrosis (PIMMF), Mantsopoulos identified dense, scar-like fibrosis in approximately 3 quarters of EACS samples. This fibrosis closely paralleled the histopathological characteristics of keloid scar tissue. Moreover, in a substantial subset of cases, there was evidence of hyperproliferative or hyperkeratotic epithelium, a feature often associated with the scarring observed in keloid disease. These findings reinforce the hypothesis that there may be a shared pathophysiological basis between PIMMF-related EACS and keloid disease, suggesting a commonality in the mechanisms underlying the fibrotic processes in these conditions. 8
This study demonstrates that hyperlipidemia serves as a significant risk factor for the development of EACS. The pathogenic mechanism likely involves a synergistic interplay between inflammatory responses and fibrotic progression. Elevated TG levels were found to promote macrophage infiltration and stimulate the secretion of pro-inflammatory cytokines (including IL-6 and TNF-α) in vascular endothelial and soft tissues.9-11 Importantly, our clinical data revealed that EACS patients with hypertriglyceridemia consistently exhibited elevated systemic inflammatory markers, suggesting a potential cooperative effect between dyslipidemia and inflammatory pathways in EACS pathogenesis.
Elevated triglyceride levels may stimulate fibroblast proliferation through lipid peroxidation products, particularly oxidized low-density lipoprotein.12,13 This finding is supported by Mantsopoulos’ histological analysis of 41 PIMMF cases, which revealed dense scar-like fibrosis in approximately 75% of EACS specimens. The fibrotic patterns observed in EACS exhibited remarkable histopathological similarities to keloid tissue, including characteristic epithelial hyperplasia and hyperkeratosis. 8 These parallel findings strongly suggest a shared pathophysiological basis between EACS and keloid disorders, potentially involving common fibrotic mechanisms in their disease processes.
Clinical studies consistently report that hyperlipidemia adversely affects wound healing and increases perioperative complication risks.3-5 Notable findings include Hosseinrezaei et al demonstrated a marked increase in sternal wound infection rates among hyperlipidemic patients. 6 Furthermore, Yamada’s comparative analysis revealed a striking 32-fold elevation in specific lipid species within surgical specimens from lumbar spinal stenosis patients compared to controls.
This study observed a significantly elevated prevalence of hyperlipidemia in the preoperative period among individuals who developed stenosis, compared to those who did not, a finding that corroborates the results of a US-based exploration of keloids. This association has led to the inference that hyperlipidemia may be an independent risk factor for the development of EACS subsequent to otitis media surgery. 14 A positive relationship was noted in this study between the length of surgical procedures and the rate of EACS. Endoscopic surgery involves the placement of the endoscope and surgical instruments within the constricted external auditory canal, which can lead to the endoscope’s repeated friction against the canal’s skin and the potential for surgical instruments to damage the skin flap. For cholesteatoma-type otitis media, an endoscopic hydro-mastoidectomy 15 is conducted, and the extended water impact, immersion, and abrasion of the external auditory canal bone with an electric drill may result in skin flap damage. Damage and loss of the skin of the external auditory canal are considered significant causes of acquired EACS. 16
Previous studies have identified ear surgery as a significant factor contributing to the development of EACS. For instance, Münir Demir Bajin reported that 13 out of 24 patients with acquired atresia of the external auditory canal had a history of prior ear surgery. 17 The underlying mechanism is likely related to the proliferation of surgical incisional scarring, 18 which aligns with our findings. Based on these observations, we hypothesize that patients who have undergone multiple ear surgeries are at a higher risk of developing EACS.
The BC investigation has revealed a significant association between acquired EACS and chronic inflammatory processes. Recurrent inflammation has been observed to lead to the deposition of fibrotic scar tissue, with the formation of fibrotic plugs that may extend laterally to the cartilaginous junction of the external auditory canal. 16 This finding implicates persistent inflammation as a key factor in the pathogenesis of acquired EACS, potentially influencing the structural integrity and function of the canal.
SIRI serves as a recognized indicator to evaluate systemic inflammatory conditions. These metrics are calculated using data obtained from a complete blood count (CBC), a diagnostic test known for its cost-effectiveness, ease of administration, and prompt delivery of results. The derivation of SIRI from the CBC results provides a convenient and effective method for medical professionals to assess systemic inflammatory activity in a spectrum of clinical scenarios. These indices are extensively applied in the clinical realm for the diagnosis, management, and prognostication of malignant and inflammatory disorders.19-21 Elbistanli et al discovered that NLR and PLR levels were elevated in individuals with exudative otitis media compared to the control group. Furthermore, these levels were found to be even more elevated in those with serous effusions than in those with mucoid discharges. 22 Zhang et al documented elevated levels of SII and NLR in individuals with sudden sensorineural hearing loss. These biomarkers have been found to possess predictive utility in inflammatory diseases. In addition, the study indicates that patients exhibiting heightened SII and NLR values are more likely to encounter a less favorable prognosis, suggesting a correlation between these inflammatory markers and the severity or progression of the auditory impairment. 23 This research represents the inaugural evaluations into the correlation between preoperative systemic inflammatory indices, namely SIRI, with the appearance of EACS. The results of the study have led to the formulation of a hypothesis suggesting that increased preoperative SIRI values could precipitate an exacerbated inflammatory response within the external auditory canal after surgery. This increased inflammatory condition is theorized to increase the propensity for the development of stenosis, thus implicating these inflammatory biomarkers as potential predictors of an individual’s risk of postoperative complications, specifically the formation of stenosis.
Our analysis of pediatric patients (n = 31, ages 6-18 years) revealed no significant difference in EACS incidence compared to adults (P > .05). Although younger children have inherently smaller anatomical dimensions, the surgical protocol incorporated proportional instrumentation and canal enlargement when indicated. All pediatric cases maintained postoperative diameters >4 mm, demonstrating that technical modifications can effectively address age-related anatomical constraints. These findings align with Bajin et al’s report that meticulous surgical technique can equalize stenosis risk across age groups. 17
Although elderly individuals typically exhibit elevated levels of CHOL, TG, and SIRI,24,25 our study observed no cases of EACS in this population. This apparent paradox may be attributed to several factors: First, age-related physiological adaptations, including diminished fibroblast proliferative capacity and attenuated inflammatory responses, may counteract the pro-fibrotic effects of metabolic dysregulation—a phenomenon consistent with the “inflamm-aging” hypothesis wherein chronic low-grade inflammation paradoxically limits acute postoperative fibrotic reactions. 26 Second, methodological limitations in our dichotomized classification of SIRI, cholesterol, and TG levels may have obscured subtle age-dependent risk gradients. Third, selection bias likely played a role as we excluded elderly patients with severe metabolic disorders (e.g., uncontrolled diabetes), thereby eliminating potential high-risk candidates from our cohort.
Research significance: To sum up, our nomogram provides otologists with a predictive instrument to estimate the probability of EACS following endoscopic otitis media surgery. For patients predisposed to stenosis, as identified by preoperative assessment with a nomogram, the adoption of preemptive intervention strategies is considered a viable approach. The utilization of diverse stenting methods post-surgically to mitigate the risks associated with EACS is a feasible and prudent intervention for patients identified as at elevated risk.
Conclusions
This research involved examining and assessing the risk elements associated with otitis media post-surgery following otoscopic otitis media and creating a related nomogram. This nomogram significantly predicts postoperative EACS in patients with otoscopic otitis media. It can provide a reference for the early detection of high-risk patients, provide clinical intervention, and promote individualized and precise treatment.
Footnotes
Acknowledgements
First, I would like to thank Prof. Li Wei for the conceptualization of this study and his valuable comments during the writing of the paper, and second, I would like to thank my fellow student, Yudi Shao, for his help during the analysis of the clinical data.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Graduate Research & Practice Innovation Program of Jiangsu Province (SJCX24_1545).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets analyzed during this study are protected under patient confidentiality regulations per ethical approval (No. XYFY2024-KL215-01). De-identified data may be requested from the corresponding author pending institutional review board approval.
