Abstract
Objective:
The use of mastoid pressure dressings (MPDs) after otologic surgery has been a standard practice for decades, believed to minimize complications such as hematoma and wound infection. However, the efficacy of MPDs in improving patient outcomes has been questioned. This study aimed to evaluate the necessity and impact of MPDs on postoperative pain, complications, and overall patient comfort.
Method:
In this prospective, randomized, controlled trial, 200 patients undergoing major ear surgeries were randomly assigned to receive either a MPD group or a non-MPD (NMPD) group. Postoperative pain was assessed using the Visual Analog Scale at the 3rd, 6th, and 24th hour. Complications, including hematoma, wound infection, and skin erythema, were systematically recorded.
Results:
Patients in the MPD group reported significantly-higher pain levels at all measured intervals than in the NMPD group (P < .05). Complications such as skin erythema and auricular bruising were more prevalent in the MPD group. Importantly, there was no significant difference between the two groups in the incidence of hematomas or wound infections.
Conclusion:
The findings suggest that MPDs may contribute to increased postoperative pain and discomfort without offering significant benefits in preventing complications. Given the lack of significant benefit and the increased patient discomfort observed with MPDs, their routine use in otologic surgery may warrant reconsideration. Future research should further explore optimal postoperative care strategies to enhance patient comfort and outcomes.
Keywords
Level of evidence of this article is Level 2.
Introduction
The mastoid pressure dressing (MPD), first described by Heermann in 1958 for ear surgeries, has maintained its traditional place despite the lack of sufficient literature support and the developments in surgical techniques and bleeding control throughout history. 1 In the conventional approach to middle ear and mastoidectomy surgeries, surgical incisions in the postauricular region or outer ear are typically sealed with pressure dressings. These dressings are believed to be necessary, particularly when surgical drains are not used, to minimize the risk of postoperative hematoma formation. 2
However, despite their long-standing use, the necessity and efficacy of MPDs have come under scrutiny. Pressure dressings can be uncomfortable, especially in hot weather, leading to significant discomfort, headaches, and skin reactions such as compressive, irritative, or allergic responses. Furthermore, the application of MPDs is based on the theoretical assumption that pressure will control bleeding and reduce the risk of hematoma development. Yet, with advances in surgical techniques and bleeding control methods such as bipolar cautery, the routine use of MPDs has become questionable. 3
Previous studies have yielded conflicting results regarding the benefits of MPDs. For instance, some research suggests that these dressings may not reduce the risk of hematoma or other complications.2,6 In contrast, others have suggested that dressings might be useful for reducing the risk of infection and providing psychological reassurance to patients, despite limited empirical evidence supporting these claims.7,8 Despite this, they continue to be used, possibly due to tradition rather than evidence-based practice. Studies such as those by O’Brien et al and Castelli et al have questioned the necessity of MPDs in the prevention of postoperative complications, suggesting that their use may not provide significant benefits.9,10
In light of these conflicting views, there is a pressing need to evaluate whether the continued use of MPDs is justified or whether their drawbacks outweigh the potential benefits. This study aimed to assess the impact of MPDs on postoperative pain, complications, and overall patient comfort in a randomized controlled trial. By comparing outcomes between patients who received MPDs and those who did not, this research seeks to determine whether the practice remains advantageous in modern otologic surgery and whether routine use should be reconsidered to optimize patient outcomes.
Materials and Methods
Study Design
This study was a prospective, randomized, controlled trial designed to assess the impact of MPDs on postoperative pain, complications, and patient comfort following otologic surgery. The study was conducted with ethics approval from the institutional ethics committee (protocol no. 2023-506).
Participants and Randomization
A total of 200 patients undergoing major ear surgery, including combined-approach tympanomastoidectomy, radical mastoidectomy, modified radical mastoidectomy, and revision mastoidectomy, were enrolled in the study. Patients requiring mastoidectomy were included to ensure a consistent surgical approach and to minimize the possibility of bias related to procedural variability. Patients were eligible for inclusion if they were adults aged 18 years or older, scheduled for elective otologic surgery, and provided informed consent. Exclusion criteria included a history of bleeding disorders, allergy to any materials used in the dressings, or inability to comply with study protocols and cases in which mastoidectomy was not required.
Participants were randomly assigned in a 1:1 ratio to either the MPD group or the non-MPD (NMPD) group. Randomization was performed using a computer-generated sequence, and allocation was concealed in sealed, opaque envelopes.
Informed Consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. Written informed consent was obtained from all participants prior to their inclusion in the study.
Surgical Procedure
All operations were performed by a single surgeon, ensuring consistency in surgical technique across all patients. Each patient underwent surgery through a retroauricular incision under hypotensive general anesthesia. Local infiltration with 1:200,000 adrenaline was applied post-aurally and inside of the ear canal. The wound closure was achieved using interrupted subcutaneous sutures with chromic catgut or polyglactin and either interrupted or continuous skin sutures with silk. Following surgery, the wound was cleaned with povidone-iodine (Baticon®, ADEKA, Turkiye). In the MPD group, a firm pressure dressing was applied over the wound, covering the entire mastoid area. The dressing was renewed the next day and removed at the end of the second day. In the NMPD group, only surgical strips were applied to the suture site, and patients did not receive a pressure dressing. Drains were not used in any patient group. All patients received systemic antibiotics (amoxicillin 875 + 125 mg clavulanic acid twice a day or 500 mg erythromycin if allergic) and local treatment (antibiotic ear drops with steroids for 2 weeks) 1 week after surgery. Postauricular stitches were removed from all patients in the first week.
Pressure Measurement of Mastoid Dressings
To ensure consistency in the application of MPDs, we measured the pressure exerted by the dressings using a balloon catheter coupled with a pressure transducer, as described in similar studies on head bandages in otologic surgery. 11 The balloon catheter was placed directly over the surgical site, separated from the wound by a single layer of sterile absorbent dressing. The pressure transducer was connected to a digital gauge calibrated in millimeters of mercury (mmHg) to provide real-time measurements of the exerted pressure.
Each dressing was tightened until it reached a target pressure of 38 to 46 mmHg, a range identified in the literature as sufficient to prevent hematoma formation while minimizing discomfort, such as headaches. This pressure was chosen based on studies that indicated it as the minimum necessary to control arterial bleeding at the surgical site, yet not so high as to induce significant patient discomfort. 11
Pressure measurements were recorded immediately after the dressing application and monitored to ensure consistency across all patients. The standardization of this pressure application allowed us to ensure that each patient received a consistent and optimal level of compression, aligning with best practices identified in the literature.
Outcome Measures
The primary outcome measure was the severity of postoperative pain, assessed using the Visual Analog Scale (VAS) at the 3rd, 6th, and 24th hour postsurgery. The VAS is a validated tool for measuring pain intensity, where patients rate their pain on a scale from 0 (no pain) to 10 (worst pain imaginable). 12 Secondary outcome measures included the incidence of postoperative complications such as hematoma, wound infection, skin erythema, auricular bruising, and wound dehiscence. The occurrence of these complications was monitored and recorded on postoperative day 1, at the time of dressing removal on postoperative day 2, and during follow-up visits at the 1st week postsurgery.
Patient Comfort and Satisfaction Assessment
Patient comfort and satisfaction were assessed using a structured questionnaire before discharge. Patients rated their comfort level on a five-point Likert scale, where 1 = very uncomfortable, 2 = uncomfortable, 3 = neutral, 4 = comfortable, and 5 = very comfortable. The responses were recorded and analyzed to compare the comfort levels between the MPD and NMPD groups. Additionally, patients’ complaints and reported benefits were assessed using an open-ended question in the same questionnaire. Patients were asked to freely describe any discomforts or benefits they experienced postoperatively. Their responses were recorded and categorized based on recurring themes such as tightness, pressure-related headaches, and ease of movement.
Statistical Analysis
Data were analyzed using IBM SPSS Statistics for Windows, version 26.0 (IBM Corp, Armonk, NY, USA). Descriptive statistics were presented as means and standard deviations for continuous variables and frequencies and percentages for categorical variables. The independent sample t-test was used to compare VAS scores between the MPD and NMPD groups. The Pearson chi-squared test was employed to compare the incidence of categorical complications between groups. A P < .05 was considered statistically significant.
Results
Patient Demographics and Surgical Details
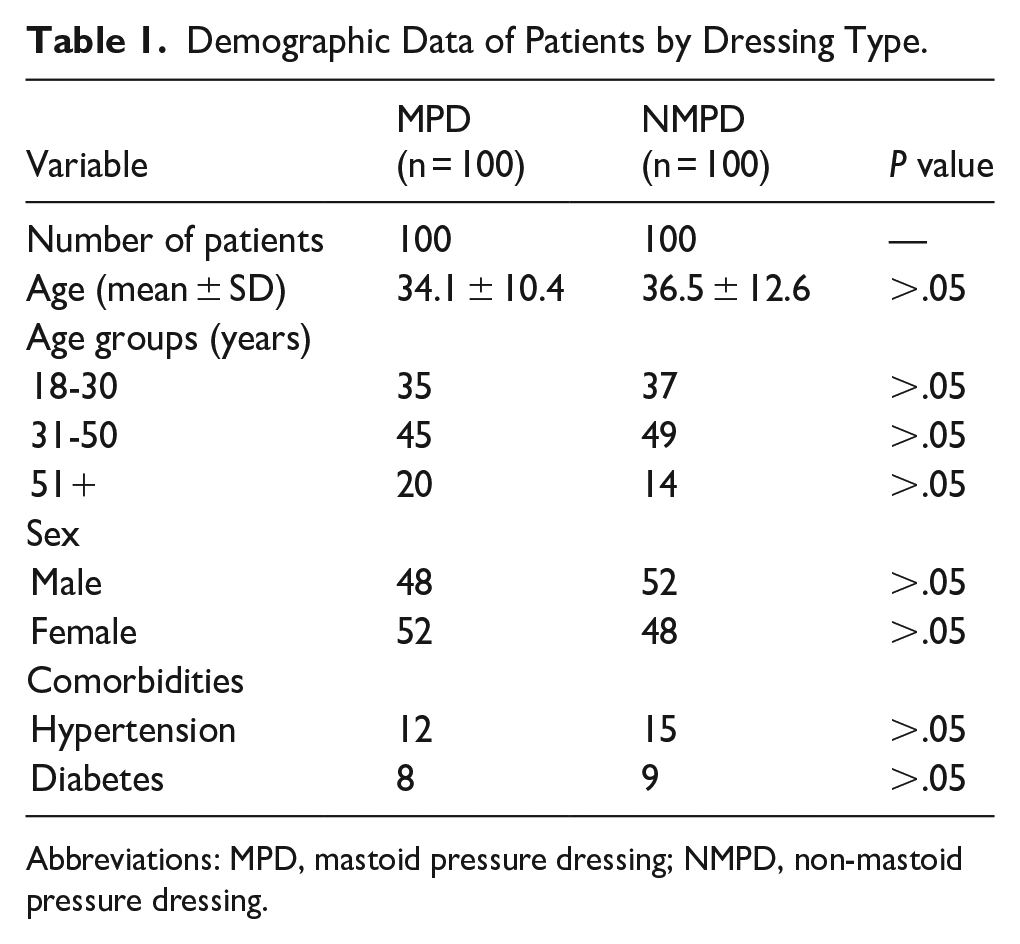
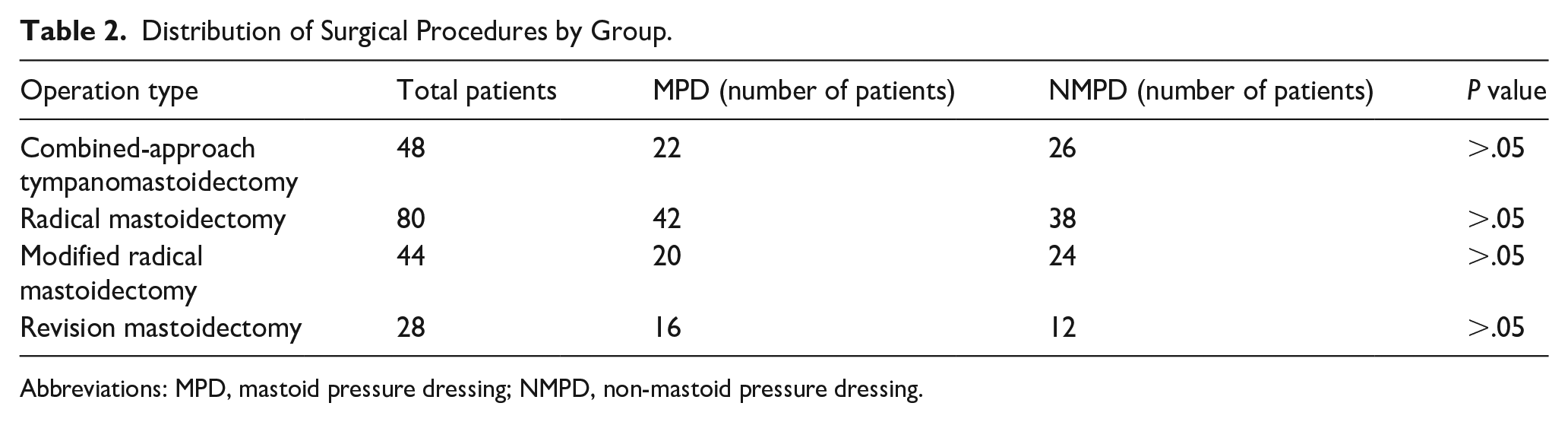
A total of 200 patients were enrolled in the study, with 100 patients assigned to the MPD group and 100 to the NMPD group. The baseline demographic characteristics, including age, gender, and comorbidities, were comparable between the 2 groups and are summarized in Table 1. The distribution of surgical procedures performed across the 2 groups was also similar and is detailed in Table 2.
Demographic Data of Patients by Dressing Type.
Abbreviations: MPD, mastoid pressure dressing; NMPD, non-mastoid pressure dressing.
Distribution of Surgical Procedures by Group.
Abbreviations: MPD, mastoid pressure dressing; NMPD, non-mastoid pressure dressing.
Postoperative Pain
One of the main focuses of this research was to compare pain and discomfort between the 2 groups. The severity of postoperative pain was measured using a VAS at the 3rd, 6th, and 24th hour postsurgery (Figure 1). The evaluation conducted at the end of the 3 hour period showed that pain and discomfort were significantly lower in the NMPD group (2.286 ± 0.98 vs 5.486 ± 1.28; P = .003). In the 6 hour evaluation, patients in the NMPD group reported being significantly-more comfortable than those in the MPD group (2.488 ± 1.01 vs 5.726 ± 1.31; P = .001). The 24 hour evaluation, performed before discharge and upon the removal of the pressure dressing, showed that VAS scores remained significantly higher in the MPD group (1.986 ± 0.84 vs 5.124 ± 1.26; P = .002).
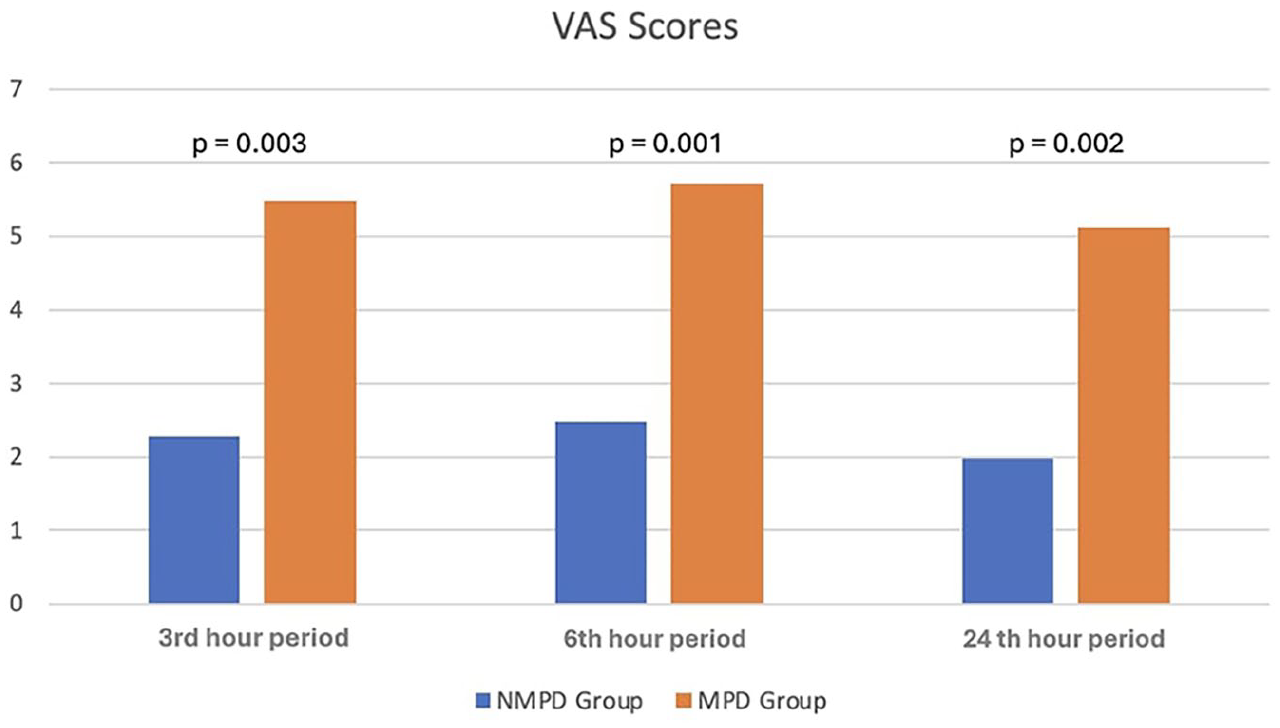
VAS scores for postoperative pain at the 3rd, 6th, and 24th hours in both groups. VAS, Visual Analog Scale.
Postoperative Complications
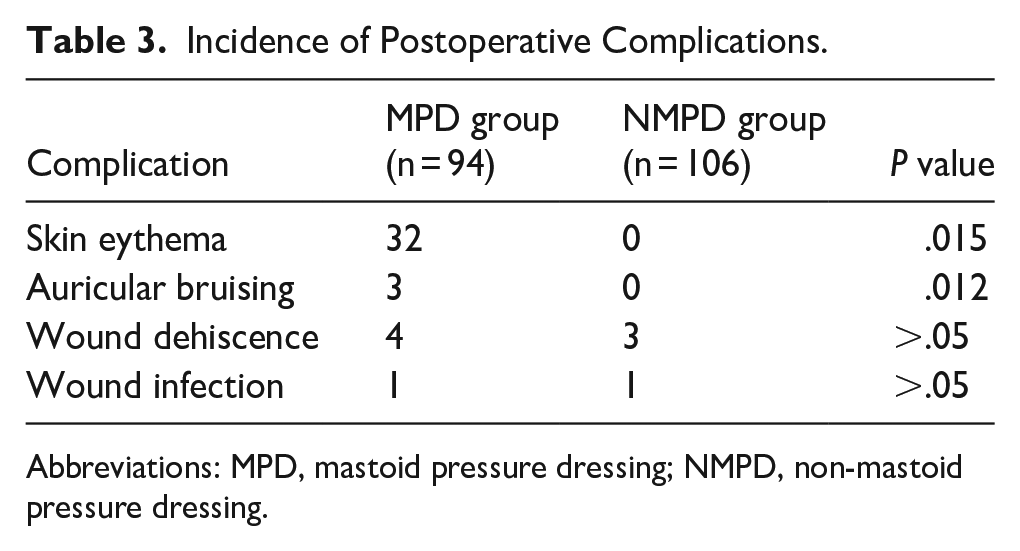
The occurrence of postoperative complications was monitored and recorded on postoperative day 1, at the time of dressing removal on postoperative day 2, and during follow-up visits at the 1st week postsurgery (Table 3). Bruising was observed in the auricle of 3 patients in the MPD group but did not require treatment and disappeared within a few days. Skin erythema was observed in 32 patients, especially in the forehead area in the MPD group; however, this also resolved within 1 to 2 days after the pressure dressing was removed (Figure 2). In 4 patients in the MPD group, slight wound opening was observed during the first week when the stitches were removed, but with local care, wound closure was achieved within a few days without the need for additional surgical intervention.
Incidence of Postoperative Complications.
Abbreviations: MPD, mastoid pressure dressing; NMPD, non-mastoid pressure dressing.
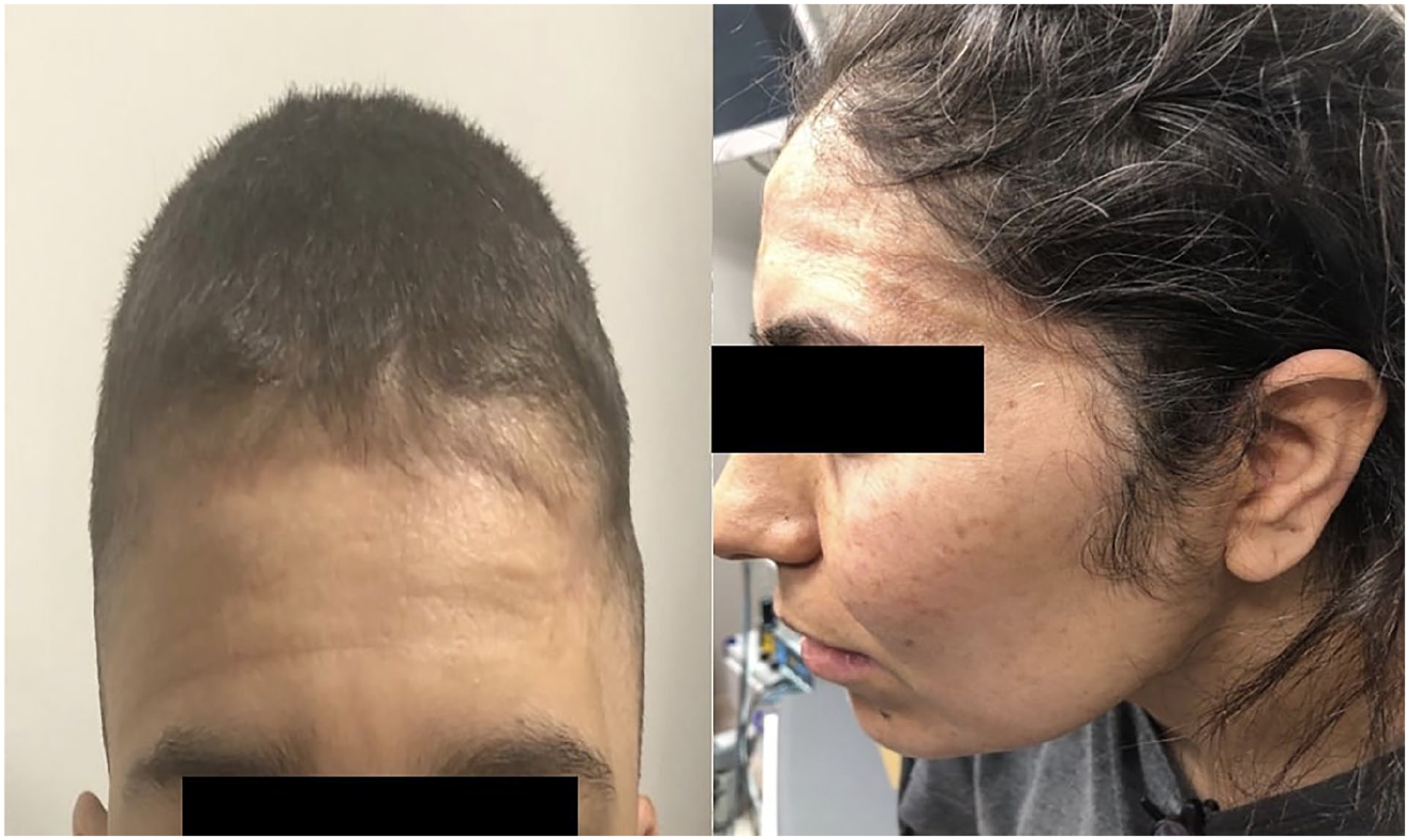
Skin erythema observed in patients with mastoid pressure dressings.
In contrast, no skin erythema or auricular bruising was observed in any patient in the NMPD group. However, in 3 patients in this group, slight wound opening was also observed during the first week when the stitches were removed. These wounds similarly closed within a few days with local care, without requiring further surgical intervention. The incidence of bruising and skin erythema in the auricle was statistically-significantly lower in the NMPD group (P = .012 for bruising and P = .015 for skin erythema).
One patient in each group developed a wound infection, which responded well to systemic antibiotic therapy. No hematoma or other wound infections developed in either group.
Stitch Removal and Postoperative Care
All patients received systemic antibiotics and local treatment as described in the Materials Methods section. Postauricular stitches were successfully removed from all patients during the first week of follow-up. No significant differences were observed in wound healing between the 2 groups at the 1st week follow-up visit.
Patient Comfort and Satisfaction
In the NMPD group, 82% of patients (82/100) reported comfort scores of 4 or 5, whereas only 39% (39/100) in the MPD group reported similar scores (P < .001). Additionally, 29% (29/100) of MPD group patients reported discomfort levels of 1 or 2, compared with 7% (7/100) in the NMPD group (P = .007) (Figure 3). The most common complaints among MPD group patients were tightness (47%), pressure-related headaches (32%), whereas in the NMPD group, the most frequently-cited benefit was ease of movement (61%). These findings suggest that the absence of pressure dressings significantly improves patient comfort and reduces psychological distress related to bandage tightness.
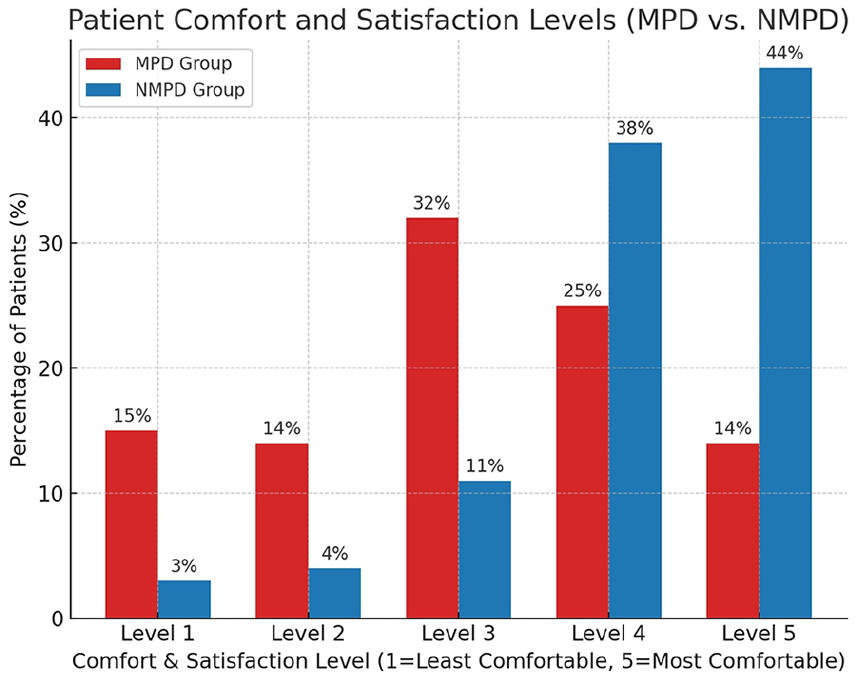
Patient comfort and satisfaction levels.
Discussion
The use of MPD after ear surgeries has long been a common practice, traditionally believed to prevent complications such as hematomas, control bleeding, and enhance wound healing. However, our study suggests that this conventional approach may not provide the anticipated benefits and may even result in more harm than good, particularly in terms of patient comfort and the risk of certain complications.
Our findings indicate that patients in the MPD group experienced significantly-higher levels of postoperative pain at all time points measured—3, 6, and 24 hours—than those in the NMPD group. This aligns with previous studies, such as those by Shinnawi et al, which found that the application of pressure dressings after otologic surgeries did not significantly reduce pain levels but rather increased patient discomfort due to the tightness and pressure exerted by the dressing. 13
Similarly, studies by O’Brien et al and Gurgel and Oghalai have reported that MPDs might lead to patient discomfort, including headaches and skin irritation, without significantly preventing complications like wound dehiscence or infection.2,9 Interestingly, while MPDs are designed to prevent postoperative complications, our study revealed that 32 patients in the MPD group developed skin erythema, particularly in the forehead area—a complication that was absent in the NMPD group. Additionally, 3 patients in the MPD group experienced auricular bruising, which also did not occur in the NMPD group. These findings suggest that the application of MPDs may contribute to certain adverse effects rather than mitigating them.
Importantly, this study is the first prospective study to standardize the pressure exerted by MPDs in otologic surgery, ensuring consistent application across all patients. By utilizing a balloon catheter and pressure transducer to measure the pressure exerted by the dressings, we maintained a target pressure range of 38 to 46 mmHg, as recommended in the literature. 11 This approach allowed us to objectively compare the outcomes between patients receiving MPDs and those who did not, providing a robust analysis of the effects of standardized pressure on postoperative complications.
Moreover, our study observed that the discomfort and headaches often reported by patients in the MPD group are consistent with other research indicating that tight dressings can lead to compressive and irritative symptoms.9,14 The pressure exerted by these dressings not only causes physical discomfort but also delays the early detection of potential wound issues, which could be detrimental to the overall recovery process. 15
The higher VAS scores recorded in the MPD group at the 3rd, 6th, and 24th hour further emphasize the impact of these dressings on patient comfort. Despite the traditional rationale for using MPDs—primarily to reduce bleeding and support wound healing—the evidence from our study and previous literature suggests that these benefits do not outweigh the associated risks and discomfort. A recent study has also questioned the effectiveness of such dressings in postoperative care, particularly in terms of improving patient outcomes, which aligns with our findings. 16
In light of modern surgical techniques and advanced bleeding control methods, our findings suggest that the routine use of MPDs should be reexamined to ensure the best possible patient outcomes. The lack of significant differences in serious complications such as wound infections and hematomas between the MPD and NMPD groups supports the argument that MPDs may be unnecessary in many cases. This is further supported by recent literature, which indicates that with improved surgical techniques, the need for such dressings has diminished significantly. 17
In addition to traditional MPDs, the literature describes several alternative methods of postoperative compression and incision protection. For example, elastic headbands can provide gentle yet consistent support without exerting the same degree of tightness or discomfort typically associated with heavier gauze bandages. 18 Less intense compression options, such as padded or adjustable bandages, have also been used in select cases to minimize pressure-related pain and skin irritation. 19 These alternatives may be especially useful in patients who experience significant discomfort with conventional MPDs or who have a low to moderate risk of postoperative bleeding. Further comparative studies would help clarify which dressing type provides the optimal balance of patient comfort and clinical efficacy.
In certain clinical scenarios, the use of a pressure dressing may still be useful. Patients with inherent bleeding disorders—such as low platelet counts, coagulation factor deficiencies, or conditions like hemophilia—are at an increased risk of postoperative hemorrhage, 20 and the additional compression provided by a pressure dressing can help mitigate this risk. Similarly, procedures associated with significant vascularity or anticipated bleeding—such as resections for glomus tympanicum tumors or cases complicated by intraoperative vascular injuries (eg, to the sigmoid sinus or jugular vein)—may benefit from the adjunctive use of a pressure dressing. Moreover, other factors, including the use of antithrombotic therapy, uncontrolled hypertension, liver dysfunction, and the challenges of extensive dissection in revision surgeries, can further elevate the risk of bleeding.21,22 In these contexts, when used judiciously alongside modern hemostatic techniques, pressure dressings may still play a valuable role in enhancing postoperative safety and optimizing patient outcomes.
Pressure dressings may also be considered in cochlear implant procedures, particularly during the initial postoperative period when minimizing device movement can be critical to ensure proper healing and secure positioning of the implant. However, with advances in internal fixation techniques and improved implant design, many surgeons find that the risk of device displacement is substantially reduced, thus limiting the necessity of external compression.5,14 In certain cases involving challenging anatomy, complex revisions, or patient factors that elevate the risk of implant migration, a pressure dressing may still be useful for additional support.
However, this study does have certain limitations. The relatively-small sample size and single-center design may limit the generalizability of the findings. Additionally, the surgeries were performed by a single surgeon, which, while ensuring consistency, may introduce bias. The short follow-up period of 1 week also limits the ability to assess longer term outcomes. Future studies with larger, multicenter samples and longer follow-up periods are necessary to confirm these findings and to explore the long-term effects of MPDs on patient outcomes.
Conclusion
The findings of this study highlight the potential drawbacks of using MPDs after ear surgeries. Although MPDs have traditionally been believed to prevent postoperative complications such as hematoma and wound infection, our research suggests otherwise. Patients who did not receive MPDs reported significantly less pain and discomfort, as measured by the VAS at various postoperative intervals. Additionally, the incidence of complications such as bruising and skin erythema was notably lower in the NMPD group.
These results suggest that the routine use of MPDs may not only be unnecessary but may also contribute to increased postoperative discomfort and patient-perceived pain. Given these findings, the medical community should reconsider the routine application of MPDs and instead focus on optimizing hemostasis and wound healing through alternative approaches.
