Abstract
Introduction
Necrotizing otitis externa (NOE) is a serious and life-threatening infection that affects the external auditory canal (EAC) and progressively extends to the temporal bone. 1 This condition predominantly affects immunosuppressed patients, particularly the elderly with diabetes. The pathogen most commonly implicated is Pseudomonas aeruginosa, accounting for 50% to 90%. 2
However, fungi causing NOE are rare, tend to be more invasive and challenging to diagnose and manage. The pathogens most frequently identified are Candida spp. and Aspegillus spp. 3 Fungal NOE is associated with substantial morbidity and mortality, mainly due to late diagnosis at the complications stage. 2 Early recognition and targeted treatment are critical to improve outcomes and prevent severe morbidity.
Our objectives were to describe the clinical characteristics and management approaches of fungal NOE and to identify the predictive factors of complications.
Materials and Methods
We conducted a retrospective and monocentric study that included all patients diagnosed with NOE and treated in our ENT department between 2006 and 2024.
The diagnosis of NOE was established using Cohen and Friedman criteria, 4 which include the following:
- Major signs: • Pain • Exudate • Edema • Granulations • Positive findings in temporal bone scan (bone erosion) or failure of local treatment after more than 1 week
- Minor signs: • Diabetes • Cranial nerve involvement • Debilitating condition • Old age
The fungal origin of the infection was determined based on the detection of fungal organisms in culture swabs (an EAC swab was performed for each patient).
A review of the patient’s chart was performed to evaluate epidemiologic, clinical, biological, microbiological, imaging, treatment, and outcomes data.
Our protocol of treatment for fungal NOE included hospitalization, intravenous antifungal (with regular monitoring of renal and liver function), daily local treatment during hospitalization (application of topical antiseptic agent: povidone iodine), regular monitoring of blood glucose levels, and resection of granulation tissue (local anesthesia).
An improvement was defined as the disappearance of pain, regression of the EAC inflammation, and normalization of the erythrocyte sedimentation rate (ESR).
An analytical study was conducted to identify the predictive factors of complications. The following variables were evaluated: age, sex, immunodeficiency, duration of diabetes diagnosis, and delay before consultation. Statistical analysis was performed using SPSS software, version 25 (IBM Corp., Armonk, NY, USA). Comparisons of qualitative variables were made using chi-square tests or Fisher’s exact test, where appropriate. For quantitative variables, the nonparametric Mann–Whitney test or Student’s test was applied depending on the data distribution. Statistical significance was assumed for P < .05.
Results
A total of 15 patients were diagnosed with fungal NOE. The mean age was 67.6 years. The sex ratio (Male/Female) was 2.7. All patients had diabetes with a mean diagnosis duration of 15.6 years. The median delay before consultation was 3.5 weeks (1-8 weeks). The affected side was the right ear in 7 cases (46.7%), and the left ear in 8 cases (53.3%). Symptoms included otalgia in all cases and otorrhea in 12 cases (80%). Physical examination revealed stenosis of the EAC in 12 cases (80%) and homolateral facial palsy in 5 cases (33.3%). The characteristics of the patients are summarized in Table 1.
Patients’ Characteristics.
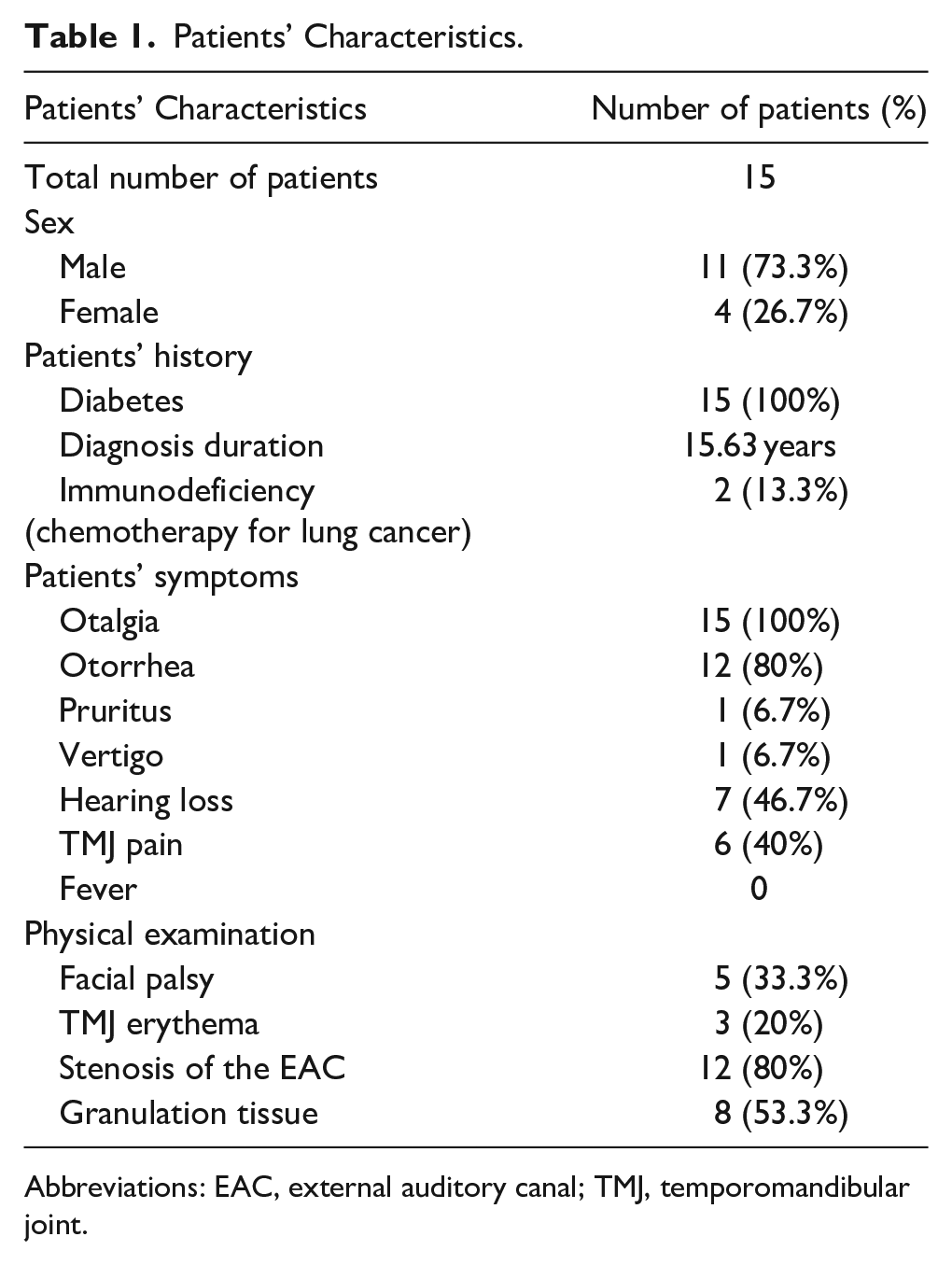
Abbreviations: EAC, external auditory canal; TMJ, temporomandibular joint.
Twelve patients received oral antibiotics prior to hospitalization. The biological tests showed an increase in ESR, with a mean of 105.2 ± 44.9 mm/hour, and a mean level of C-reactive protein of 53.5 ± 45 mg/L.
The pathogens identified included Candida albicans (3 patients), Candida tropicalis (1 patient), Aspergillus flavus (9 patients), Aspergillus niger (1 patient), and Aspergillus nidulans (1 patient). Two patients tested positive for both fungi (A. flavus) and Pseudomanas aruginosa.
All patients underwent a computed tomography (CT) scan, which revealed bone erosion in all cases. Imaging results are presented in Table 2. The complications revealed on imaging were extension to the temporomandibular joint (TMJ) (46.7%), involvement of the middle ear (60%), vascular thrombosis (26.6%), intracranial extension (13.3%), and soft tissue complications (retropharyngeal, parapharyngeal, and prevertebral abscesses) (33.3%).
Patients’ Imaging Findings.
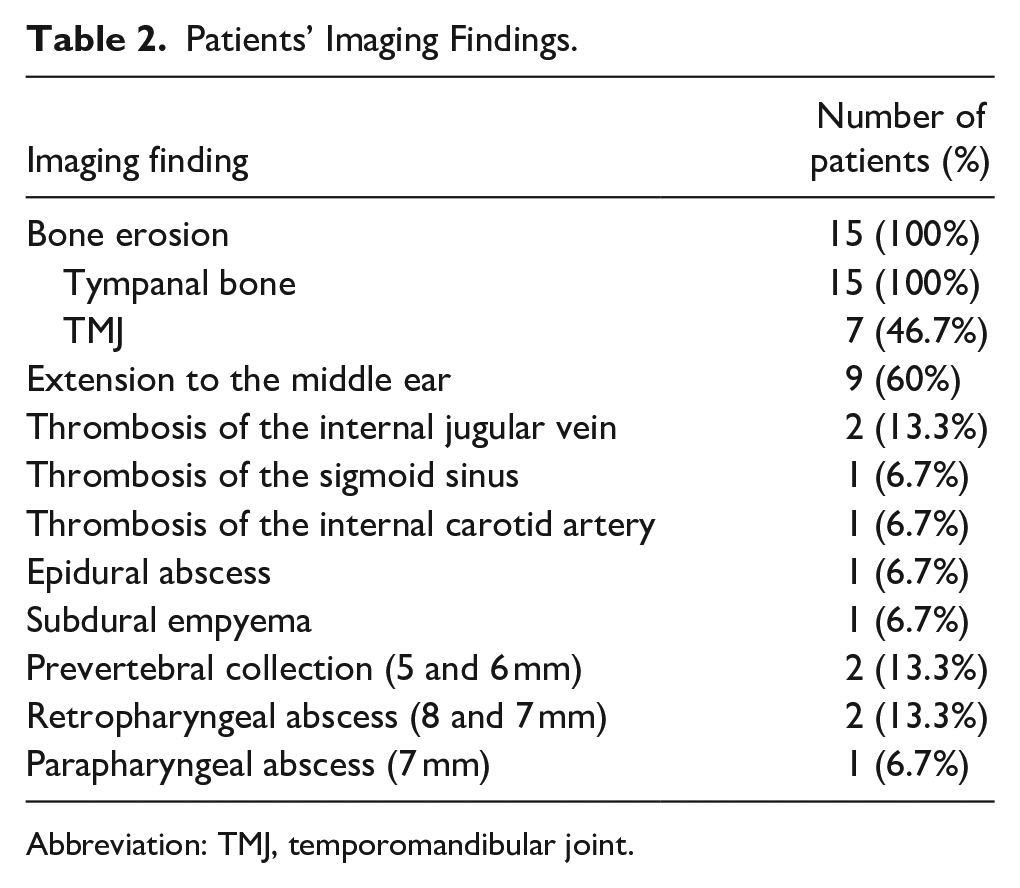
Abbreviation: TMJ, temporomandibular joint.
All patients received antifungal treatment, combined with antibiotics in 2 cases or as monotherapy in 13 cases. Among them, 12 patients were treated with Voriconazole, 2 patients received Fluconazole, and 1 patient was treated with Itraconazole. The mean duration of intravenous antifungal treatment was 40.5 ± 14 days. Oral switch therapy was administered in 11 cases (73.3%) with Voriconazole prescribed for 9 patients and antibiotics for 2 patients. The mean total duration (intravenous + oral) of antifungal treatment was 2 months (1.5-3 months). A daily local treatment was performed in all cases. Table 3 provides a summary of treatment combinations.
Treatment Combinations.

For the management of complications, the 2 cases with intracranial extension were treated conservatively. After consulting the neurosurgical team, surgical intervention was not indicated. The TMJ involvement did not require special treatment. Anticoagulation was not indicated in the case of vascular thrombosis. Surgical treatment was not indicated in the cases of soft tissue abscesses due to their small size.
After a median follow-up of 6 months, all patients showed clinical improvement and resolution of symptoms, judged by their clinical progress and a decrease in sedimentation rates. Repeated imaging scans showed the resolution of complications in all cases. No cases of recurrence were observed.
We analyzed the predictors of the occurrence of complications. Older age and delay before consultation were significantly associated with soft tissue extension (P = .01, P < 0.01 respectively). Immunodeficiency and delay before consultation were significantly correlated with vascular thrombosis (P = 0.01 and P < .01 respectively). These factors are summarized in Table 4.
Predictive Factors of Complications.
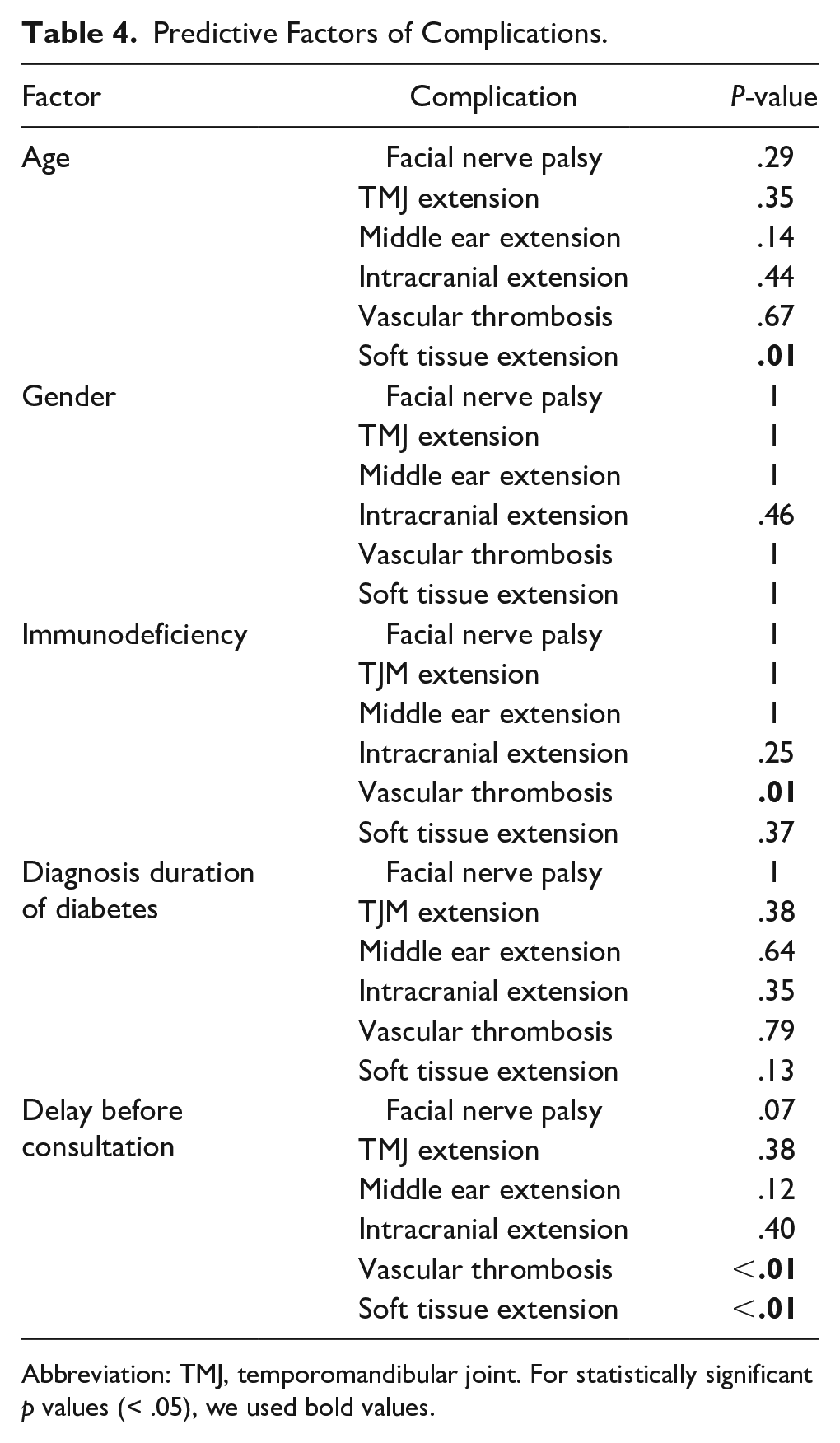
Abbreviation: TMJ, temporomandibular joint. For statistically significant p values (< .05), we used bold values.
Discussion
Fifteen patients diagnosed with fungal NOE were included in our study. All patients had diabetes. Otalgia was the most common symptom. Complications were noted in 80% of the cases with facial nerve palsy occurring in 5 cases (33.3%). CT imaging revealed TMJ extension (46.7%), middle ear involvement (60%), vascular thrombosis (26.6%), intracranial extension (13.3%), and soft tissue complications (33.3%). The identified fungi were Candida spp. (26.7%) and Aspergillus spp. (73.3%), with A. flavus being the most common, reported in 60% of cases. The mean total duration (intravenous + oral) of antifungal treatment was 2 months. Clinical improvement was observed in all cases. Older age and delay before consultation were significantly associated with soft tissue extension. Immunodeficiency and delay before consultation were significantly correlated with vascular thrombosis.
Fungal NOE is rare, representing 5% to 20% of NOE cases. A. fumigatus is the most commonly implicated pathogen, followed by C. albicans.2,3,5,6 In our series, A. flavus was the most reported pathogen. This is consistent with a recent systematic review revealing that A. flavus is more prevalent than A. fumigatus, a finding that contrasts with previous reports. 7
Although the exact pathophysiology of fungal NOE remains unclear, several critical risk factors have been identified. 2 These include long-term diabetes, immunosuppressive conditions (chronic renal failure, hematologic malignancies, and human immunodeficiency virus infection), prolonged systemic antibiotic therapy in patients initially treated for bacterial NOE, and the use of topical antibiotics.2,3,5,8,9
The diagnosis of fungal NOE is challenging and depends on a high degree of clinical suspicion. 2 It is based on a combination of clinical, microbiological, and imaging evaluations. Currently, there are no universally agreed upon criteria for establishing a definitive diagnosis. 9 The clinical presentation includes severe otalgia with nocturnal exacerbation, hearing loss, purulent otorrhea, and TJM pain. The infection extends from the EAC to the temporal bone through the fissures of Santorini. 3
Fungal NOE is typically more invasive than bacterial NOE, causing a higher incidence of complications and worse outcomes.2,3,10 This was supported by a case series of 60 patients with NOE, which confirmed that the fungal group is more invasive compared to the nonfungal group, with higher rates of facial nerve palsy, a younger mean age at presentation, and a lower 5-year survival rate. 11 Additionally, a retrospective study that included 48 NOE patients found that the majority of patients with a poor response to treatment were predominantly affected by fungal pathogens. 7 Complications can involve cranial neuropathy, with the facial nerve being the most commonly affected. In addition, the glossopharyngeal, vagus, spinal accessory and hypoglossal nerves can also be compromised, as well as the trigeminal and abducens nerves in rare cases. 3 A systematic review of 197 patients reported cranial nerve impairment in 27.4% of cases and TMJ involvement in 7.1% of cases. On the contrary, our study found a higher prevalence, with facial nerve palsy in 33.3% of cases and TMJ involvement in 46.7% of cases. To the best of our knowledge, this could be the first study to analyze predictive factors for fungal NOE complications. We found that older age and delay before consultation were significantly associated with soft tissue extension. Besides, immunodeficiency and delay before consultation were significantly correlated with vascular thrombosis.
Fungal NOE differs from bacterial NOE in several significant aspects. A notable difference is the more pronounced elevation of the ESR in fungal NOE. 12 Additionally, fungal NOE is more likely to involve the TMJ and middle ear than bacterial NOE. 9
Swabs and cultures are crucial in isolating the causative pathogen, although initial samples may yield negative results. 3 A second sample is usually indicated in cases where there is a lack of clinical improvement despite intravenous antipseudomonal therapy. Fungal origin should be suspected in refractory culture negative NOE, and empiric antifungal treatment should be indicated. 13
CT plays an important role in the diagnosis of NOE by confirming temporal bone osteitis and detecting potential complications. It is the reference imaging modality, providing detailed information on cortical bone expansion, erosion, venous thrombosis, and intracranial extension. 3 Magnetic resonance imaging may also be indicated, as it provides a better analysis of the infratemporal region, skull base, and soft tissue involvement. 9 Second-line investigations are reserved for challenging diagnosis cases. Technecium 99 bone scanning offers a higher sensitivity for NOE detection, though it is not specific. 3 It shows areas of osteoblastic activity. 7 Gallium-67 scanning provides an advantage in monitoring disease activity, as it detects positive uptake of Ga-67 citrate during active infection, which normalizes as the infection resolves, making it useful for assessing treatment response. 14 Furthermore, biological markers such as ESR can be elevated and can serve as indicators of disease progression.
The baseline treatment for fungal NOE is prolonged antifungal therapy. Itraconazole, Fluconazole, Amphotericin B, and Voriconazole are the main treatment options. Voriconazole is recommended as first-line treatment for Aspergillus NOE. 7 It is also recommended for resistant Candida spp.1,3,7 In our study, Voriconazole was the most frequently prescribed antifungal agent. However, the ideal duration of therapy is still uncertain, with the literature suggesting a range of 6 weeks to several months.2,3,6 In our study, the mean duration of antifungal treatment was 2 months. Given the hepatotoxicity of many antifungal drugs, it is important to regularly evaluate liver and kidney functions.
The role of topical therapy (topical antifungal and/or antiseptic agents) remains controversial and is not well studied in the literature. 15 There is no consensus regarding the duration of use: 2 weeks, once the skin of the ear canal has returned to normal appearance or during hospitalization.13,15-17 In our series, we used povidone iodine for local treatment and applied daily during hospitalization.
In addition to antifungal treatment, it is crucial to manage diabetes and enhance immune function. 6 Regular aural lavage is also recommended. 3 Hyperbaric oxygen therapy is indicated in persistent cases. It is an efficient inhibitor of fungi growth, but it is not a standard treatment due to limited supporting evidence.3,7,11
Surgical intervention is recommended for the excision of granulation tissue and histopathological analysis. This approach helps confirm the fungal etiology and rules out differential diagnoses.3,6 However, when surgical treatment is used as a first-line option without prior stabilization of the disease using conservative methods, it can lead to adverse outcomes. 7
Mortality rates in fungal NOE vary between studies, with reported ranges between 2% and 11%. 7 In particular, no cases of mortality were observed in our study.
Conclusions
Fungal NOE is a rare, yet serious, condition associated with potential complications. Its diagnosis can be challenging and should be systematically considered in cases with poor response to antibacterial treatment. Unfortunately, the current literature provides limited guidance on optimal treatment and management protocols. The baseline treatment relies on antifungal therapy, but there is no consensus on the recommended duration of treatment, highlighting the need for further research to establish standardized guidelines.
Footnotes
Acknowledgements
None.
Author Contributions
Conceptualization: W.T. Data collection: Y.S. and A.B.M. Writing: W.T. and Y.S. All authors reviewed the manuscript.
Data Availability Statement
All data generated or analyzed during this research are included in this published article. Data supporting the findings of this study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
