Abstract
Keywords
Introduction
Salivary gland tumors are rare, heterogenous neoplasms that vary in terms of their location and histology. 1 They are often benign, with pleomorphic adenoma being most common, comprising approximately half of all salivary tumors. 2 The most common malignant salivary tumors are mucoepidermoid carcinoma (MEC) and adenoid cystic carcinoma (AdCC).1-3 The aggressiveness of salivary gland cancers (SGCs) depends on multiple factors including the histology of the tumor as they are grouped into low-grade, intermediate, or high-grade cases. 2
For tumors with benign or low-grade malignancies, surgery is the primary treatment modality. High-grade carcinomas and those with high-risk features, such as positive margins, are treated with surgery and adjuvant therapy.4,5 However, for locoregional recurrence or metastatic disease, treatment options and robust clinical evidence are limited.6,7 Systemic therapy is often explored in these cohorts because it can be administered, regardless of the local treatment history, such as prior radiotherapy. Targeted therapy following molecular characterization of tumors is also an alternative treatment option.7-9 Typically, the goal of systemic therapy is palliative, given minimal evidence that these therapies prolong (overall?) survival in metastatic tumors. 10
Immunotherapy has emerged as a new avenue since limited therapeutic options remain for recurrent or metastatic (R/M) SGCs. 11 High mutational burden in tumors like melanoma and non-small cell lung cancers are often referred to as “hot-tumors” that respond favorably to immune checkpoint inhibitors (ICIs). 12 A multicenter, open-label phase II KEYNOTE-158 study recruited patients with high mutational burden solid tumors and showed promising results with pembrolizumab. 13 Hence, ICIs are a potential option for the subset of salivary gland tumors that display a high mutational burden and high microsatellite instability or that are deficient in mismatch repair. 14 Several phase II trials have additionally emerged to investigate the therapeutic benefit of ICIs for SGC. However, due to heterogeneity of the disease, a concise recommendation has not been established, and the role of ICIs remains unknown. Therefore, the aim of this systematic review was to identify trials investigating ICIs for R/M SGCs and summarize the oncologic outcomes and adverse reactions associated with the treatment.
Methods
Search Strategy
Electronic databases PubMed (MEDLINE), Embase, and Scopus were accessed in January 2023 to identify eligible articles. Articles written in English were reviewed. There were no restrictions in publication year or article type. Studies with more than 3 patients with SGC of any histological type and treated with any systemic therapy combination that included immunotherapy (anti-PD1, anti-PDL1, anti-CTLA4) were included. A detailed search algorithm is provided in Supplemental Table 1.
Articles were excluded if treatment was for non-salivary gland tumors, for studies with less than 3 individuals, and if studies reported both SGC and non-SGC cases but did not provide subgroup SGC outcomes. When the full article was not available through the electronic databases, an author of the present study emailed the corresponding author. If there was no response, a follow-up email was sent to the corresponding author 1 month after the initial contact. After the third attempt, if no response was provided from the author, the article was deemed unavailable and excluded from the study, which occurred for 1 article. 15
After the initial search of the database, duplicated records were removed. Articles went through preliminary screening, then eligible articles underwent full-text review. Additionally, the reference list of articles that met the inclusion criteria were manually reviewed to identify additional eligible studies. The overall quality of evidence was made using the principles of the GRADE approach. 16
Studies were independently reviewed by 2 researchers (S.G.K., J.T.). Demographic information of average age, gender, and geographical location was extracted. Clinical outcome, tumor histology, and adverse event (AE) data were extracted. Disagreements regarding eligibility were resolved by either consensus or consultation with a third reviewer (C.B.S.).
Results
Seven hundred seventy studies were identified after pooling together results from all 3 databases (PubMed/MEDLINE, Embase, and Scopus; Figure 1). After excluding duplicates, 479 studies were screened by title and abstract and 30 studies were characterized as eligible, according to the prespecified eligibility criteria. All 30 full texts and their references were screened. Ultimately, 13 articles met the criteria and were included in the present analysis.
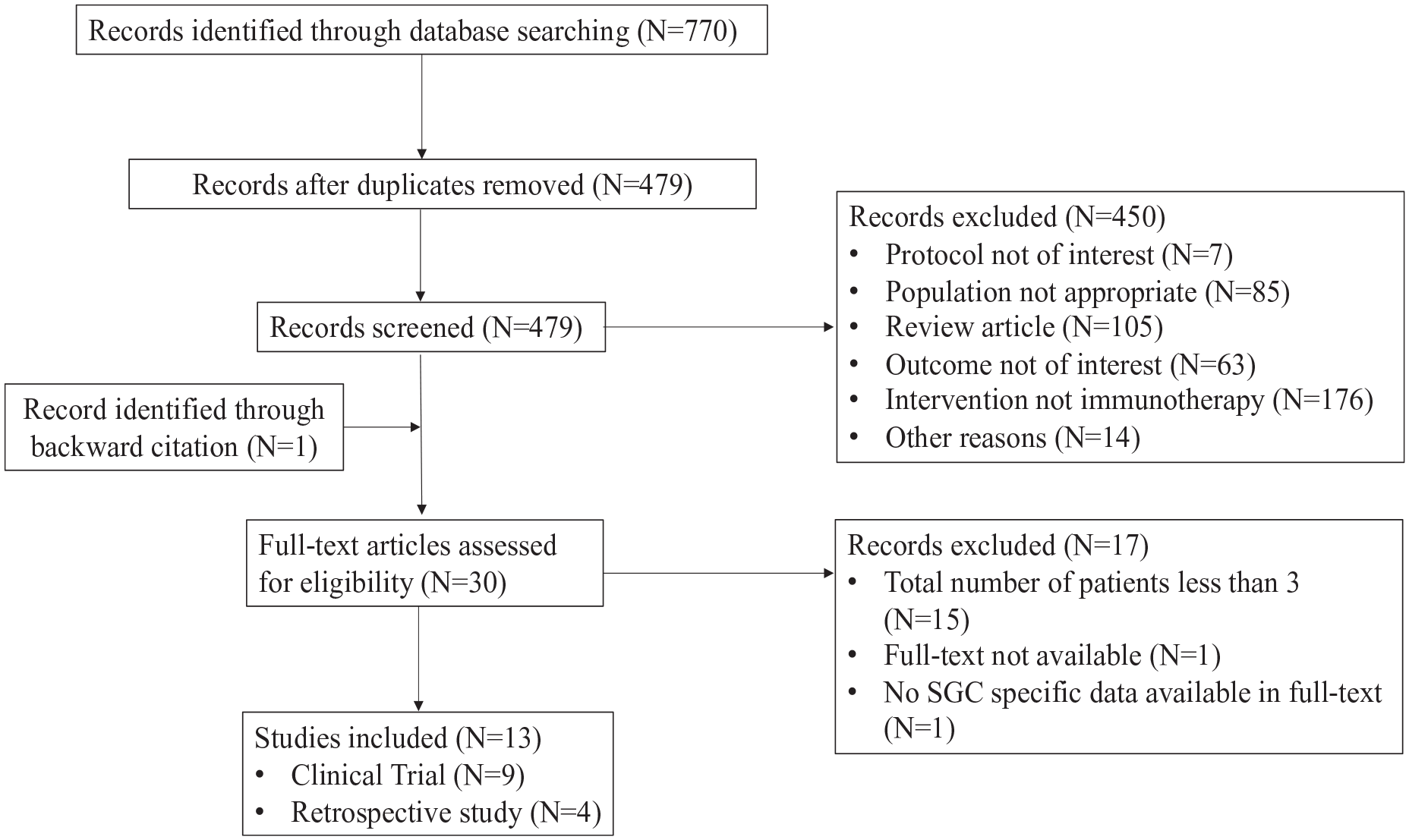
Flow diagram of search strategy.
Prospective Studies
Approximately two-thirds (9/13) of the studies reporting on 412 patients were prospective, with 2 being randomized clinical trials (Table 1). Most of the studies were conducted in the United States (7/9) with the remaining 2 studies coming from France and multiple countries, respectively. The average age was 61 ± 4 years old, and the male-to-female ratio was approximately 1.2:1.
Characteristics of Clinical Trials Including Patients with SGC Treated With ICIs.
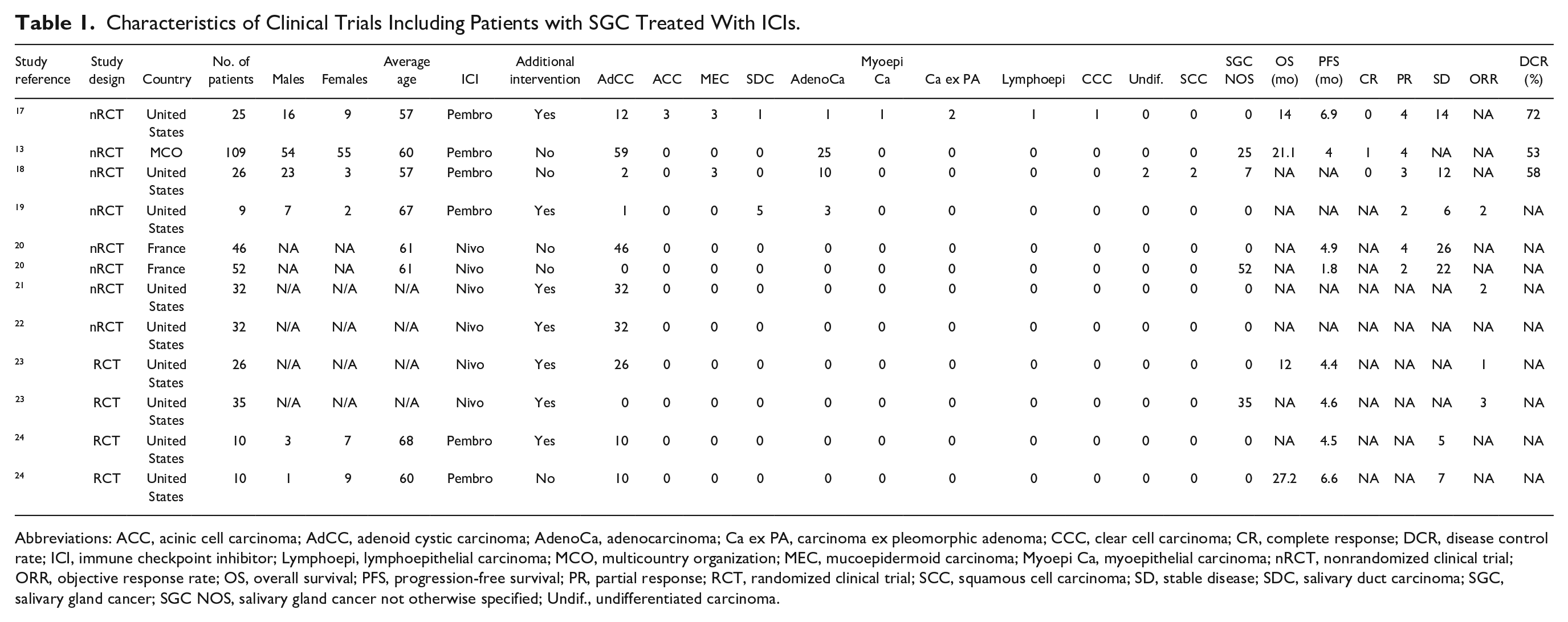
Abbreviations: ACC, acinic cell carcinoma; AdCC, adenoid cystic carcinoma; AdenoCa, adenocarcinoma; Ca ex PA, carcinoma ex pleomorphic adenoma; CCC, clear cell carcinoma; CR, complete response; DCR, disease control rate; ICI, immune checkpoint inhibitor; Lymphoepi, lymphoepithelial carcinoma; MCO, multicountry organization; MEC, mucoepidermoid carcinoma; Myoepi Ca, myoepithelial carcinoma; nRCT, nonrandomized clinical trial; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; PR, partial response; RCT, randomized clinical trial; SCC, squamous cell carcinoma; SD, stable disease; SDC, salivary duct carcinoma; SGC, salivary gland cancer; SGC NOS, salivary gland cancer not otherwise specified; Undif., undifferentiated carcinoma.
AdCC was the most common histology (56%), followed by salivary gland carcinoma not otherwise specified (29%), adenocarcinoma (9%), MEC (1%), salivary duct carcinoma (1%), acinic cell carcinoma (ACC; 1%), carcinoma ex-pleomorphic adenoma (<1%), undifferentiated carcinoma (<1%), squamous cell carcinoma (SCC; <1%), lymphoepithelioma (<1%), and clear cell carcinoma (<1%).
All patients had R/M disease, received anti-PD1 ICI (5 studies administering pembrolizumab, 4 studies administering nivolumab), and 6/9 studies also reported an additional intervention for all (n = 6) or some (n = 1) of the patients. Additional interventions included the following: anti-CTLA4 (ipilimumab; n = 3), histone deacetylase inhibitor (HDACi; ;vorinostat; n = 2), GnRH agonist (goserelin; n = 1), and radiotherapy (n = 1). The most common ICI treatment regimens were pembrolizumab 200 mg IV q3w or nivolumab 3 mg/kg IV q2w with a median follow-up time of 19.8 months. There was 1 complete response (CR), 19 partial responses (PRs), and 145 patients with stable disease (SD). The average objective response rate (ORR) was 2%. AEs were reported on average on 80% of all patients, with 15% of patients having an AE greater than or equal to grade 3 and no patients with a grade 5 AE (Table 2).
AEs Among Prospective Studies of Patients With Salivary Gland Cancer, Treated With ICIs.
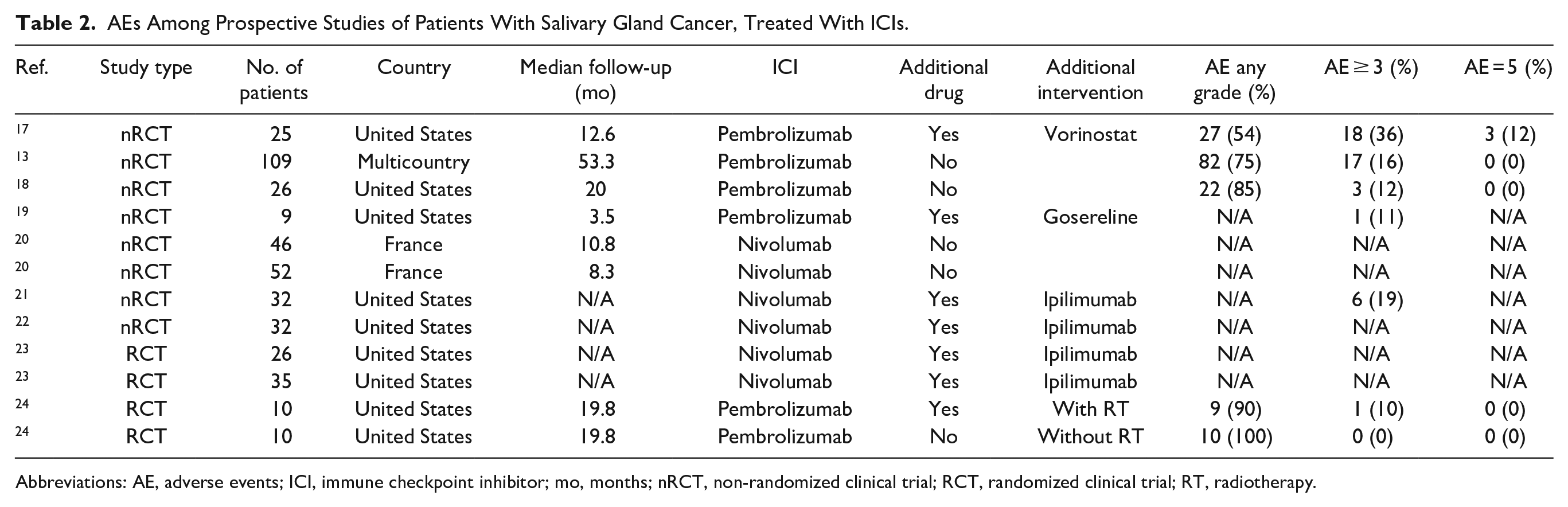
Abbreviations: AE, adverse events; ICI, immune checkpoint inhibitor; mo, months; nRCT, non-randomized clinical trial; RCT, randomized clinical trial; RT, radiotherapy.
Retrospective Studies
Four studies reporting on 61 patients were retrospective (Table 3). Three studies were completed in Japan, and 1 study was from Greece. The average age was 60 ± 11 years old, and the male-to-female ratio was 4:1. Salivary duct carcinoma was the most common histology (67%), followed by MEC (10%), AdCC (8%), SCC (5%), ACC (3%), and undifferentiated SGC (3%). Only 2 studies reported that all their patients had R/M disease. Median follow-up was 12.9 months. One patient reported a CR, 3 patients reported PR, and 11 patients reported SD. Median overall survival (OS) was 10.2 months, and median progression-free survival was 3.7 months. ORR was reported to be 10% in 1 study. Only one study reported on the total percentage of all AEs (92%), while the average percentage of AEs greater than or equal to grade 3 was 19%. No grade 5 AEs were reported (Table 4).
Characteristics of Retrospective Studies Including Patients with SGC Treated With ICIs.
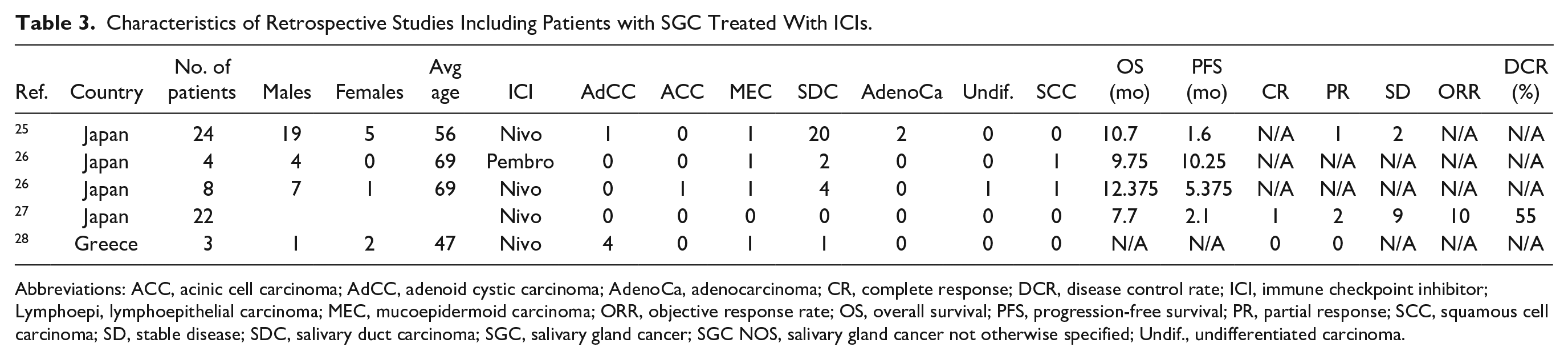
Abbreviations: ACC, acinic cell carcinoma; AdCC, adenoid cystic carcinoma; AdenoCa, adenocarcinoma; CR, complete response; DCR, disease control rate; ICI, immune checkpoint inhibitor; Lymphoepi, lymphoepithelial carcinoma; MEC, mucoepidermoid carcinoma; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; PR, partial response; SCC, squamous cell carcinoma; SD, stable disease; SDC, salivary duct carcinoma; SGC, salivary gland cancer; SGC NOS, salivary gland cancer not otherwise specified; Undif., undifferentiated carcinoma.
AEs Among Prospective and Retrospective Studies of Patients With Salivary Gland Cancer, Treated With ICIs.
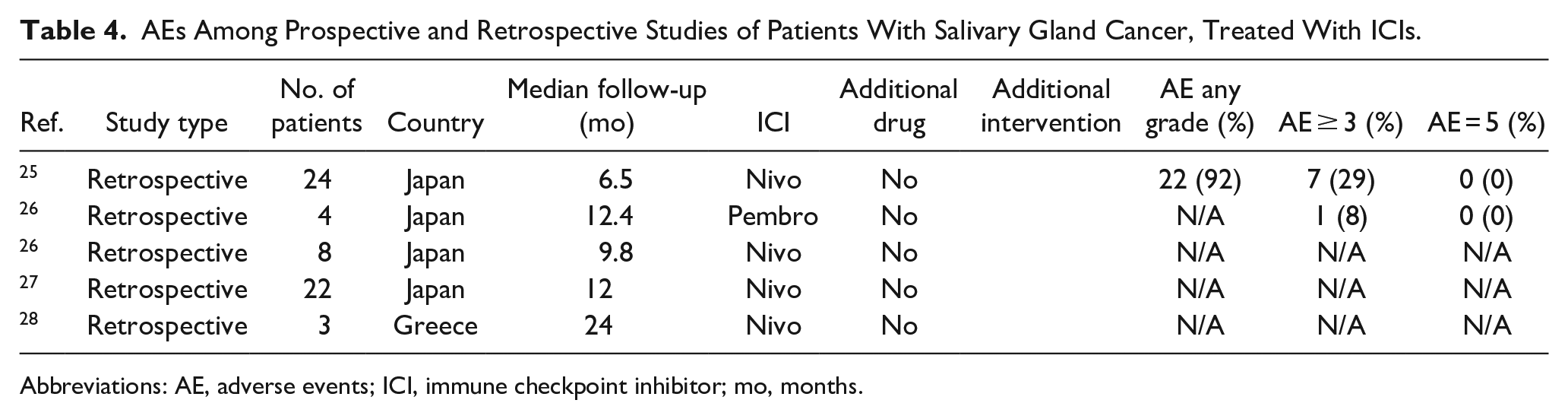
Abbreviations: AE, adverse events; ICI, immune checkpoint inhibitor; mo, months.
Discussion
Over recent years, ICIs have increasingly been utilized in solid tumor treatment paradigms. 29 To date, ICI’s role in advanced SGCs remains undefined. In recurrent, unresectable, or metastatic SGC, a multimodality treatment approach is often utilized as there are no established effective standardized therapies. 30 Evidence-based treatment regimens are limited due to the variability among SGC histologic subtypes and the heterogeneity in prior studies. Given these issues, our understanding of immune checkpoint therapy and malignant SGCs is lacking. Therefore, the current study systematically analyzes the role of immune checkpoint therapy, specifically anti PD-1 therapy, for R/M SGC.
In an analysis of 219 SGC tissue specimens, increased PD-1 expression was an independent predictor of poor disease-free survival. 31 In the present study, anti-PD1 therapy demonstrated an objective benefit in select patients. In both prospective and retrospective studies, 2 patients reported a CR, 22 reported a PR, and 156 had SDs. Disease control rate (DCR) and clinical benefit rate (CBR) are defined as the percentage of patients with advanced or metastatic cancer who have achieved CR, PR, and SD to a therapeutic intervention in clinical trials of anticancer agents. The DCR was calculated for studies that reported all 3 responses: references (13) 53%, (23) 72%, (24) 58%, (31) 55%. It is difficult to discern whether these responses were solely from immunotherapy; nevertheless, the outcomes warrant continual investigation with ICI in patients with R/M disease. Median OS also demonstrated moderate outcomes, with prospective studies reporting 17.55 months and retrospective studies showing 10.2 months.
Although response rates to ICI were relatively low, it is unclear whether this was related to poor susceptibility of SGCs to ICI in general, or the limited availability of reliable predictive biomarkers to identify responders. Only a subset of studies analyzed in this systematic review stratified ICI recipients by PD-L1 expression.13,18,24 In one of them, patients with a combined positive score (CPS) >1 had significantly-better ORR (10.7 vs 2.6 months). However, the majority of studies did not stratify by PD-L1 expression.
Six out of 9 prospective trials comprised some form of combination therapy, with either the addition of an anti-CTLA inhibitor, radiotherapy, or androgen deprivation. When radiotherapy is delivered in the treatment of cancer, not only is the local tumor treated but a systemic immune response is elicited known as the abscopal effect. 32 A phase II study examined the benefits of pembrolizumab ± radiotherapy. 24 An objective clinical response was not demonstrated outside of the radiation treatment field. For pembrolizumab plus radiotherapy, 50% reported SD compared with 70% pembrolizumab only group. Overall, there was no added benefit of XRT + PD1 with no increase in response rates outside of the radiation field.
Similarly, the role of anti PD-1 therapy and androgen deprivation therapy was assessed in a phase II trial with a group of 9 patients. 19 Androgen deprivation is often employed in select patients with advanced SGC when there is evidence of androgen receptor overexpression. The majority of the patients in this study had salivary ductal carcinoma, which is histologically similar to prostate cancer where androgen blockade is utilized for treatment.19,30 Two patients showed a partial beneficial response, and 6 patients had SD with a total CBR of 88%.
Any cancer-directed therapy, including ICI, is not without potential for unwarranted side effects and adverse reactions. In the present study, for prospective trials that reported AEs, the incidence of AE related to anti-PD1 therapy was noted to be greater than half of the patients, but AE of grade 3 or higher occurred in a minority (14.8%) of cases. This may suggest that while there are higher rates of AEs, severe or life-threatening consequences appear to be rare.
This study is not without many limitations. Most importantly, there was variability in histologic subtypes, which is a constant plague when trying to study large groups of patients with SGC. SGC vary significantly with regard to the underlying carcinogenesis mechanisms. The main SGCs have well-described, distinct pathobiology33,34 and tumor microenvironment. 35 Interestingly, we and others have recently described site-speciic differences within the same histological type, adding another layer of heterogeneity in SGC.36,37 The included studies had different outcome measures, which increases the difficulty of measuring appropriate treatment responses. In addition to an anti-PD1 therapy, a few studies included additional treatment modalities, which has the potential to confound objective outcomes. Nevertheless, the study provides a valuable insight into the current state of select immunotherapy regimens in advanced or recurrent malignant salivary gland tumors. Future improved study design, with more detailed clinical annotation and stratification by histological type is warranted to help elucidate if the treatment paradigm includes a role for immunotherapy in select patients with SGC.
Conclusion
Limited evidence exists regarding immunotherapy outcomes for metastatic or recurrent salivary gland tumor in the literature. Our systematic review revealed that while some oncological responses from anti-PD1 inhibitors such as nivolumab and pembrolizumab, caution should be exercised before drawing an overarching conclusion about the potential benefits of ICIs due to the heterogenous SGC histologies included and disparate treatment paradigms in each study. The DCR was promising, particularly in the absence of effective systemic therapies in the R/M setting for these cancers. However, future prospective studies that stratify by tumor histology, grade, and immunotherapy markers (eg, CPS, Tumor mutational burden (TMB)) are needed to provide definitive recommendations regarding immunotherapy for malignant salivary gland tumors.
Supplemental Material
sj-docx-1-ear-10.1177_01455613251324353 – Supplemental material for The Role of Immunotherapy in Salivary Gland Cancer: A Systematic Review
Supplemental material, sj-docx-1-ear-10.1177_01455613251324353 for The Role of Immunotherapy in Salivary Gland Cancer: A Systematic Review by Sul Gi Kim, Jason Tasoulas, Siddharth Sheth, Wendell G. Yarbrough, Trevor Hackman, Antonio L. Amelio and Christopher Blake Sullivan in Ear, Nose & Throat Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Sheth reported personal fees from Naveris and Medscape, and research funding from AstraZeneca, Merck, Inovio, Exelexis, and Regeneron. Dr Sheth belongs to the speaker’s bureau of Exelixis. A.L. Amelio reports personal fees from LG Life Sciences outside of the submitted work. No other disclosures were reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported in part by NIH/NIDCR R01DE030123 (to A.L. Amelio) and the NCI Center Core Support Grant 5P30CA016080-42 to the UNC Lineberger Comprehensive Cancer Center.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
