Abstract
A case of a 60-year-old male patient with thyroid cancer invading the trachea was presented in the current study. After the initial surgery via a sternocleidomastoid muscle (SCM) clavicular periosteal flap for tracheal reconstruction, the patient experienced flap necrosis. Subsequent debridement and tracheocutaneostomy were performed, followed by a successful repair of the tracheal defect using a platysma myocutaneous flap. The case underscores the critical role of preserving the SCM’s integrity to ensure adequate blood supply and reduce the risk of flap necrosis. A staged approach, involving initial debridement and tracheocutaneostomy, followed by secondary reconstruction with a platysma myocutaneous flap, proved effective in managing flap necrosis post-SCM myoperiosteal flap reconstruction. This strategy offers a safe and viable alternative for tracheal defect re-reconstruction.
Keywords
Introduction
Tracheal window resection followed by reconstruction using a sternocleidomastoid muscle (SCM) clavicular periosteal flap is an effective approach for the management of tracheal defects, with advantages such as abundant blood supply and minor anastomotic tension. However, its main complications include local airway stenosis and flap necrosis. 1 This article reports a case of tracheal window resection and reconstruction with SCM clavicular periosteal flap for thyroid cancer with tracheal invasion. The patient experienced flap necrosis after the first surgery. After initial debridement and tracheocutaneostomy, a secondary platysma myocutaneous flap was successfully used to repair the tracheal defect. We summarized a case where the reconstruction of the tracheal defect using an SCM myoperiosteal flap was unsuccessful. From this, we suggest that employing a platysma myocutaneous flap could serve as an effective and safe alternative for the re-reconstruction of the tracheal defect.
Case Report
A 60-year-old male patient presented with a 4-year history of right lateral cervical masses and a medical history of type 2 diabetes mellitus. Ultrasound revealed a 3.3 × 2.2 × 2.0 cm hypoechoic mass with indistinct borders in the right thyroid lobe, along with multiple abnormal lymph nodes in the right lateral cervical (Ⅱ, Ⅲ, Ⅳ, and Ⅵ regions) and supraclavicular areas, the largest measuring 4.0 × 3.5 × 2.0 cm. Contrast-enhanced computed tomography (CT) demonstrated uneven enhancement of the right thyroid mass, multiple enlarged lymph nodes in the right cervical region, and slight tracheal narrowing with leftward deviation (Figure 1A–D). The fibrolaryngoscope showed fixation of the right vocal cord and compression of the right tracheal wall (Figure 1E). Fine needle biopsy confirmed the histology of classical papillary thyroid carcinoma (PTC).
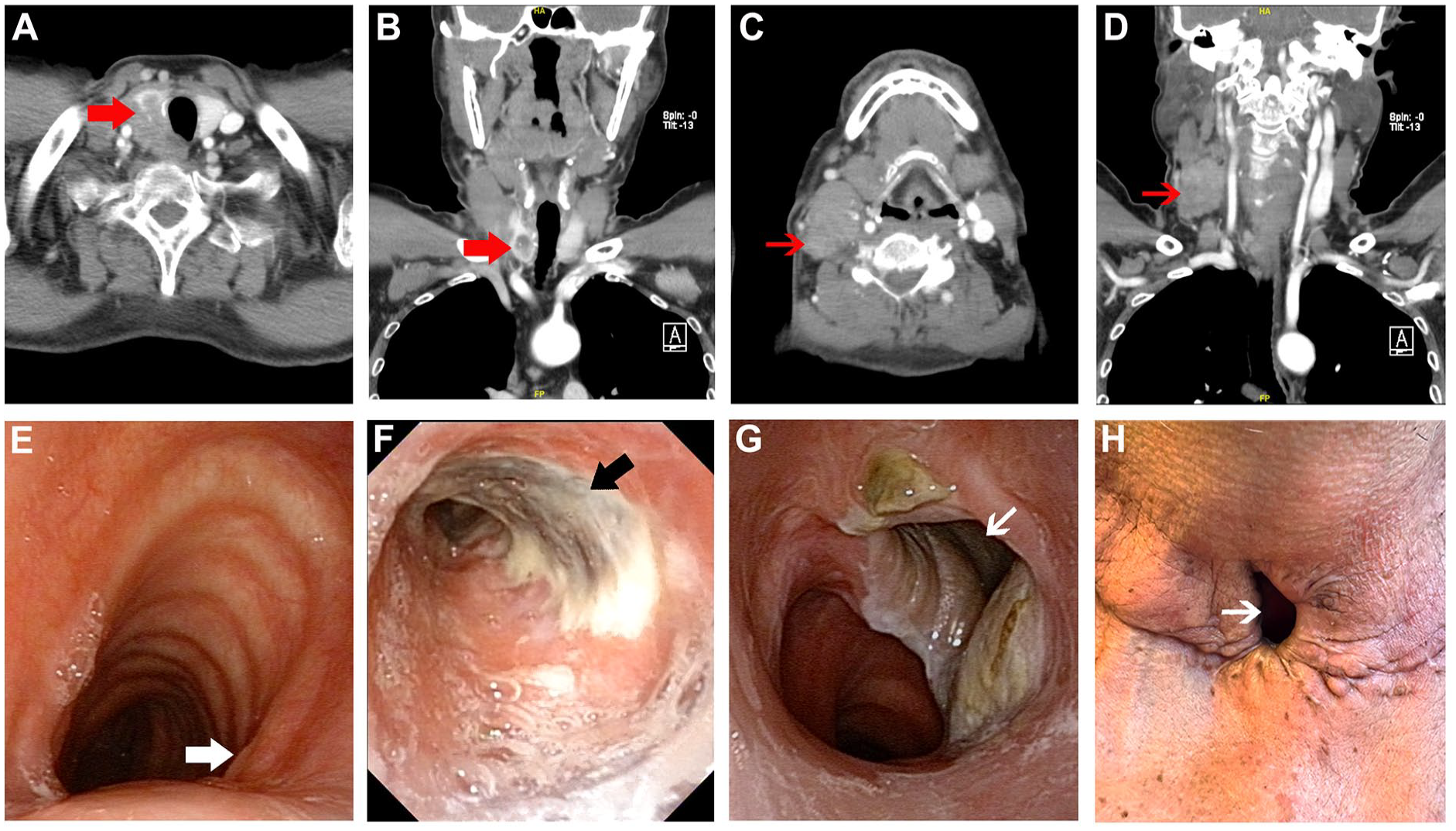
(A) and (B) Tumor invasion of the trachea before the first surgery (red thick arrows). (C) and (D) Enlarged lymph nodes adhered to the sternocleidomastoid muscle (red thin arrows). (E) Preoperative fibrolaryngoscope showed tumor invasion of the trachea (white thick arrow). (F) The fibrolaryngoscope after the first surgery indicated necrosis of the sternocleidomastoid muscle myoperiosteal flap (black thick arrow). (G) The fibrolaryngoscope and (H) outside view presented good healing of the tracheocutaneostomy site (white thin arrows) 8 months after the first stage of tracheocutaneostomy surgery.
It was found that the tumor had invaded the muscular layer of the esophagus and the 1st to 3rd tracheal rings, with significant adhesion of the enlarged right cervical lymph nodes to the SCM. A bilateral neck lymph node dissection (Ⅱ-Ⅵ regions) and total thyroidectomy were performed, along with resection of the right tracheal wall from the 1st to 4th tracheal rings, partial cricoid cartilage, part of the SCM and the right esophageal muscular layer. The tracheal defect measured approximately 3.5 × 2.5 cm, accounting for about 50% of the tracheal circumference. A right-sided SCM myoperiosteal flap was then prepared to repair the tracheal defect. Subsequently, a tracheostomy was performed through the cricothyroid membrane.
Fifteen days postoperatively, the drainage fluid from the right neck appeared brown-yellow and turbid, with a foul odor. The fibrolaryngoscope revealed necrosis at the tracheal repair site, characterized by blackened tissue and purulent discharge (Figure 1F). Emergency debridement confirmed necrosis of the SCM myoperiosteal flap. Necrotic tissue was excised, and a tracheocutaneostomy was performed at the site of the tracheal defect.
Two months following the surgery, the patient received I131 therapy. At the 8-month follow-up, the fibrolaryngoscope revealed the tracheocutaneous fistula was healing well and the trachea remained patent (Figure 1G and H). A tracheocutaneostomy closure was then performed. A pedicled platysma myocutaneous flap, measuring approximately 3.0 × 2.0 cm, was harvested from the right side of the tracheocutaneostomy (Figure 2A). The flap was rotated to the left, sutured to close the fistula, and reinforced with fascial coverage (Figure 2B and C). The incision was closed with proper alignment of the upper and lower edges (Figures 2D and E). After 2 weeks, a follow-up fibrolaryngoscope showed healthy granulation tissue at the closure site, with no signs of infection, collapse, or stenosis (Figure 2F).
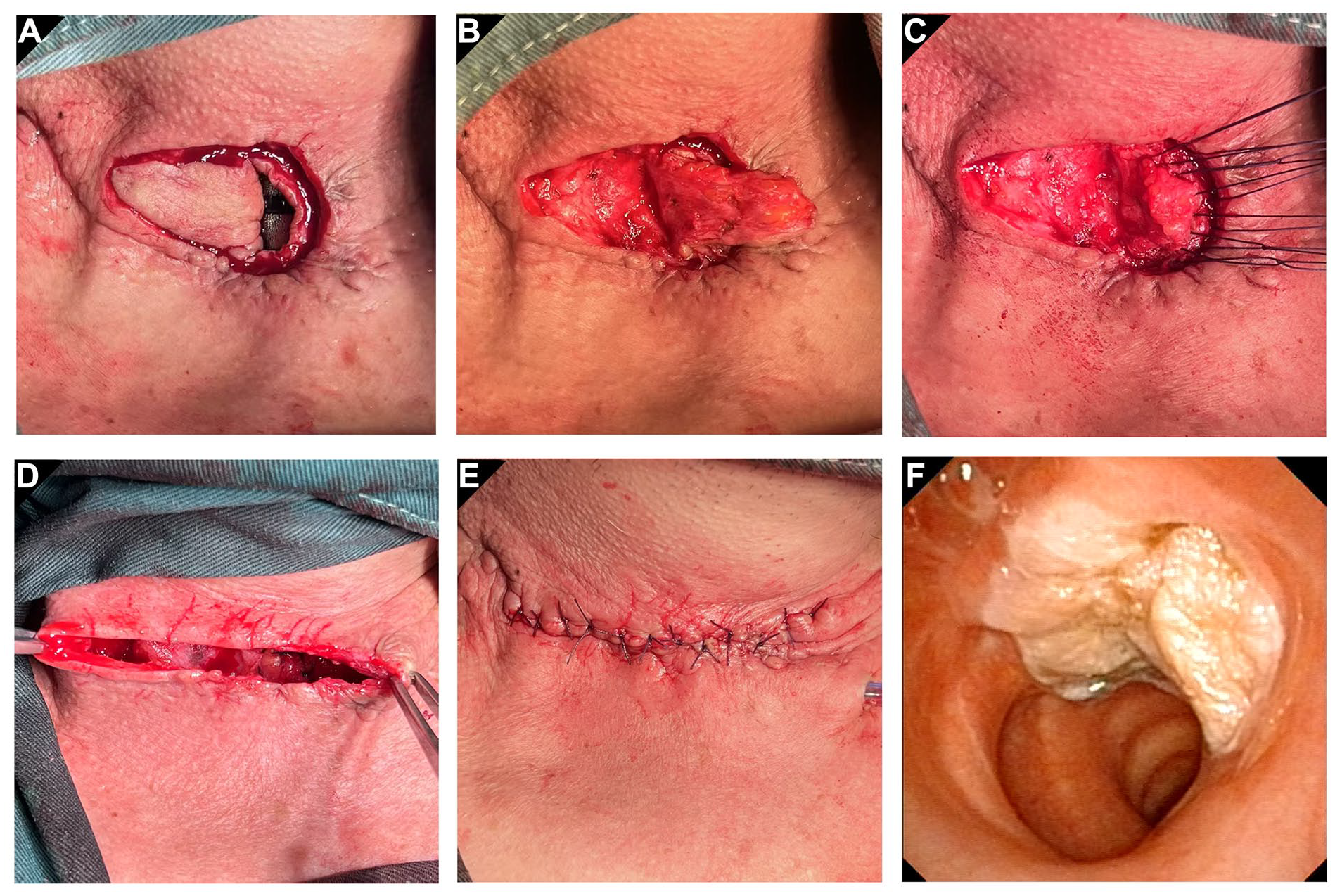
Closure of the tracheocutaneostomy using a pedicled platysma myocutaneous flap. (A) Creation of a platysma myocutaneous flap on the right side of the tracheocutaneostomy, with incision of the remaining edges. (B) Rotation of the flap to the left. (C) Suturing the tracheocutaneostomy closed with the platysma myocutaneous flap, reinforced with local fascial. (D) and (E) Proper alignment of the edges to close the incision. (F) The platysma myocutaneous flap showed the reconstructed tracheal wall without air leakage or stenosis, 2 weeks after tracheostomy closure.
Discussion
PTC is the most common type of thyroid malignancy, generally with a favorable prognosis. However, advanced thyroid carcinoma may invade adjacent organs such as the trachea, esophagus, larynx, and carotid artery. Among these organs, the trachea is the most commonly affected organ. 2 When a tumor invades the tracheal submucosa or extends into the tracheal lumen, full-thickness resection of the tracheal wall is necessary, and concurrent reconstruction of the tracheal defect is critical. 3 The SCM myoperiosteal flap, owing to its abundant blood supply, is a valuable option for repairing long-segment tracheal defects not exceeding 1/2 circumference.1,4 In the case presented, the tumor invaded the upper trachea and a portion of the cricoid cartilage, with the transverse tracheal defect involving less than half of the tracheal circumference, making it amenable to tracheal window resection and reconstruction using the SCM myoperiosteal flap.
In the current case, the patient experienced necrosis of the SCM myoperiosteal flap after the first surgery, which, along with tracheal wall collapse, represents one of the most severe complications of this surgical technique. 5 Intraoperative examination revealed extensive lymph node metastases in the right cervical Ⅱ to Ⅵ regions, with adhesion to the SCM. To achieve an R0 resection of the tumor, portions of the SCM, including its sternal end, were excised, leading to compromised local blood supply and subsequent flap necrosis. The patient also had a history of type 2 diabetes mellitus, with inadequate perioperative glycemic management, which was a critical factor contributing to poor flap healing. This case highlights that the successful application of the SCM clavicular periosteal flap heavily relies on an adequate blood supply. While ensuring complete tumor resection, it is essential to preserve the integrity of the SCM as much as possible. If there is a risk of poor blood supply, alternative tracheal reconstruction strategies, such as utilizing the contralateral SCM myoperiosteal flap or performing tracheal sleeve resection with end-to-end anastomosis, should be considered. Moreover, optimal management of underlying conditions, such as type 2 diabetes mellitus, should be given equal attention.
Given the risk of recurrent infection after local debridement and delayed wound healing, which could potentially compromise subsequent tumor treatments, we opted not to perform concurrent reconstruction of the tracheal defect. As the contralateral side had also undergone neck dissection during the previous surgery, there was a risk of adhesion in the surgical region, including the SCM and surrounding tissues. This could lead to poor blood supply and potential flap necrosis during the preparation of a new flap. For these reasons, we selected a platysma myocutaneous rotation flap to close the tracheocutaneostomy. Postoperative follow-up demonstrated that the platysma myocutaneous flap was a safe and effective option for tracheal reconstruction.
This case highlights the critical importance of preserving the integrity of the SCM to ensure adequate blood supply to the flap, thereby reducing the risk of flap necrosis and infection. A staged approach, involving initial debridement to control infection and the creation of a tracheocutaneostomy, followed by secondary reconstruction of the fistula using a platysma myocutaneous flap, proved to be an effective strategy for managing flap necrosis after SCM myoperiosteal flap reconstruction. Further studies with larger case series are needed to validate these findings.
Footnotes
Acknowledgements
Our gratitude is extended to all the colleagues who contributed to the execution of this research. Furthermore, we express our sincere thanks to the patient who consented to supply data for the study.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by CAMS Innovation Fund for Medical Sciences (CIFMS) (2023I2M-C&T-B-005).
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to Partsicipate
Written informed consent was obtained from the patient for the publication of this work.
Consent for Publication
Written informed consent was obtained from the patient for the publication of anonymized data and any accompanying images.
