Abstract

Significance Statement
Carotid body tumors, also known as carotid body paragangliomas, are benign neuroendocrine tumors that arise from the paraganglion cells of the carotid body. These paragangliomas are highly vascularized and typically located at the carotid bifurcation with characteristic splaying of the internal and external carotid arteries. 1 Carotid body tumors are the most common type of paraganglioma seen in the head and neck area but represent approximately only 1% of all head and neck neoplasms, and they are seen bilaterally in up to 10% of cases. 2
Case Description
We present a case of a 47-year-old man who presented with fever and throat pain to the emergency department. Physical examination revealed increased bilateral neck fullness with a palpable left-sided neck mass. Laboratory evaluation was notable for elevated urine dopamine levels.
Contrast-enhanced computed tomography (CT) of the neck soft tissues demonstrated bilateral heterogeneously enhancing, space-occupying lesions centered within the carotid spaces at the level of the carotid bifurcation (Figure 1). Contrast-enhanced magnetic resonance imaging (MRI) of the neck further characterized and confirmed these avidly enhancing bilateral carotid body tumors (Figure 2). Magnetic resonance angiography of the neck showed no evidence of hemodynamically significant stenosis, vascular malformations, or aneurysmal dilation of the carotid arteries (Figure 3). Because these lesions were not amenable to surgical resection after further evaluation, management included 25 cycles of radiation therapy. Post-radiotherapy F18-fluorodeoxyglucose positron emission tomography with computed tomography (FDG-PET/CT) demonstrated no further growth of these radiotracer avid lesions. The patient reported a family history of possible paraganglioma, which prompted genetic testing and confirmed a succinate dehydrogenase complex subunit D (SDHD) mutation.
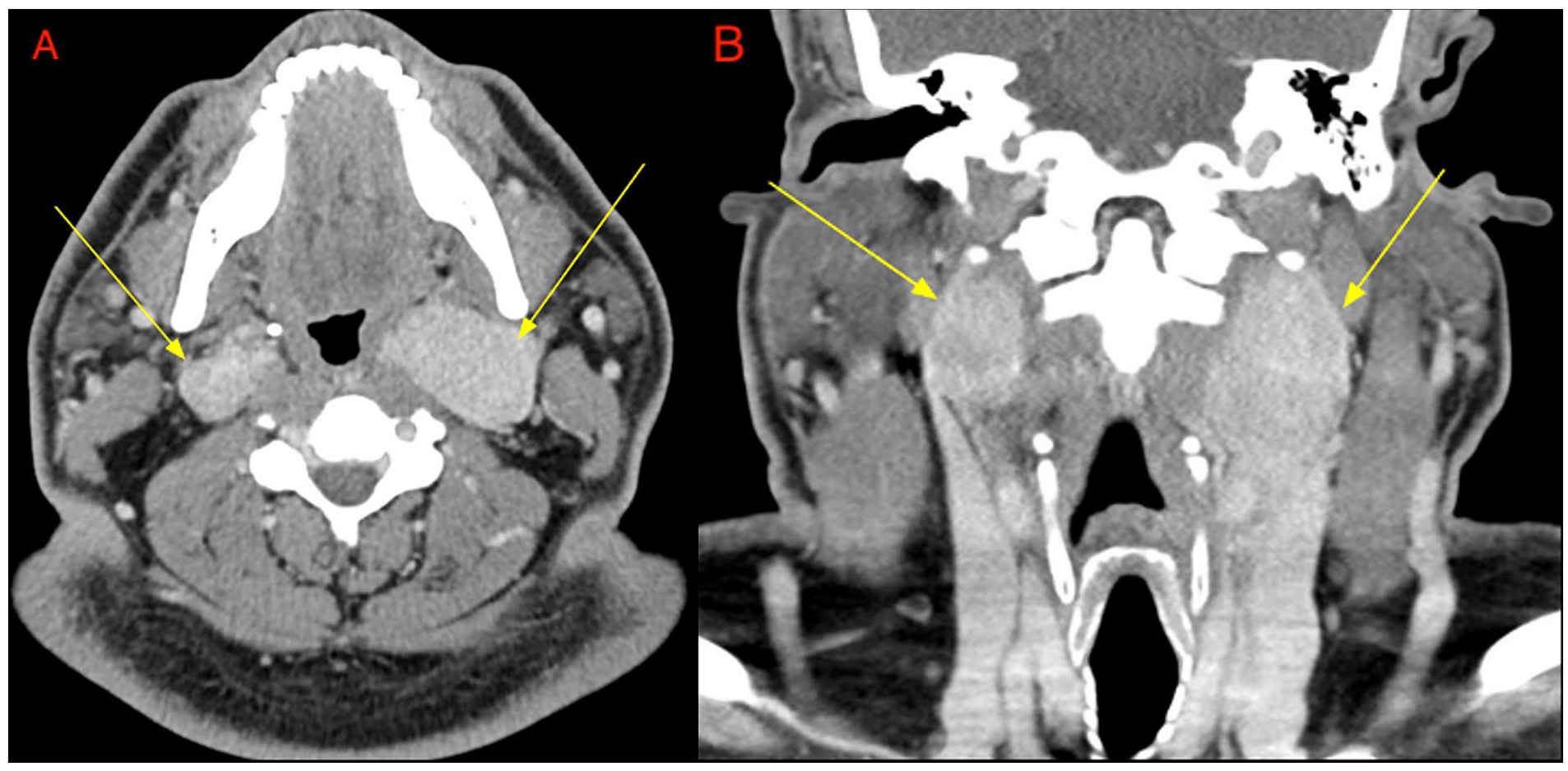
Contrast-enhanced CT of the neck soft tissues with axial (A) and coronal (B) reformats demonstrate bilateral heterogeneously enhancing, space-occupying lesions (yellow arrows) centered within the carotid spaces at the level of the carotid bifurcation. CT, computed tomography.
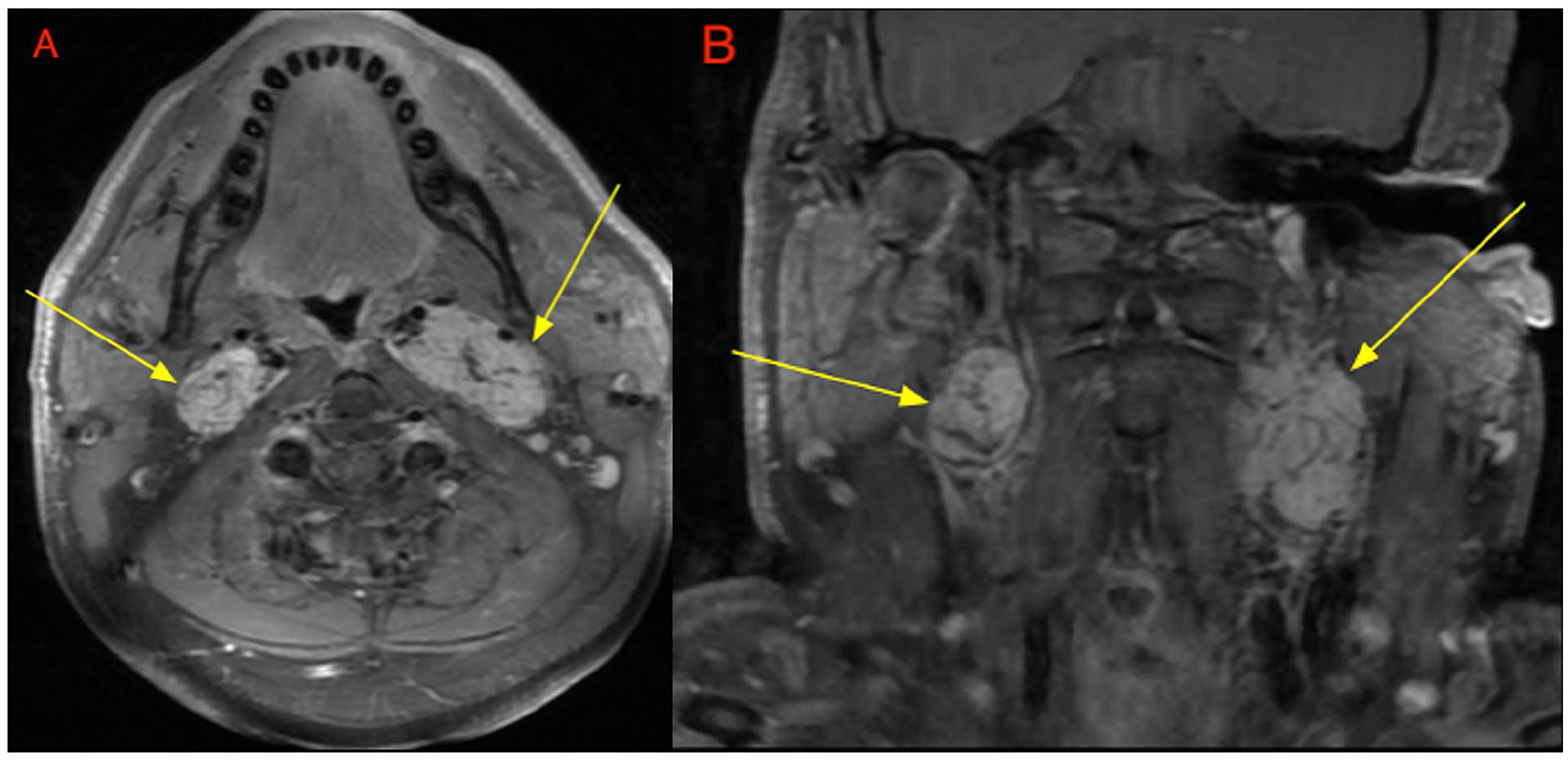
Contrast-enhanced MRI of the neck with axial (A) and coronal (B) sequences demonstrate T1 post-contrast avidly enhancing bilateral carotid space-occupying lesions (yellow arrows), splaying the carotid bifurcation. MRI, magnetic resonance imaging.
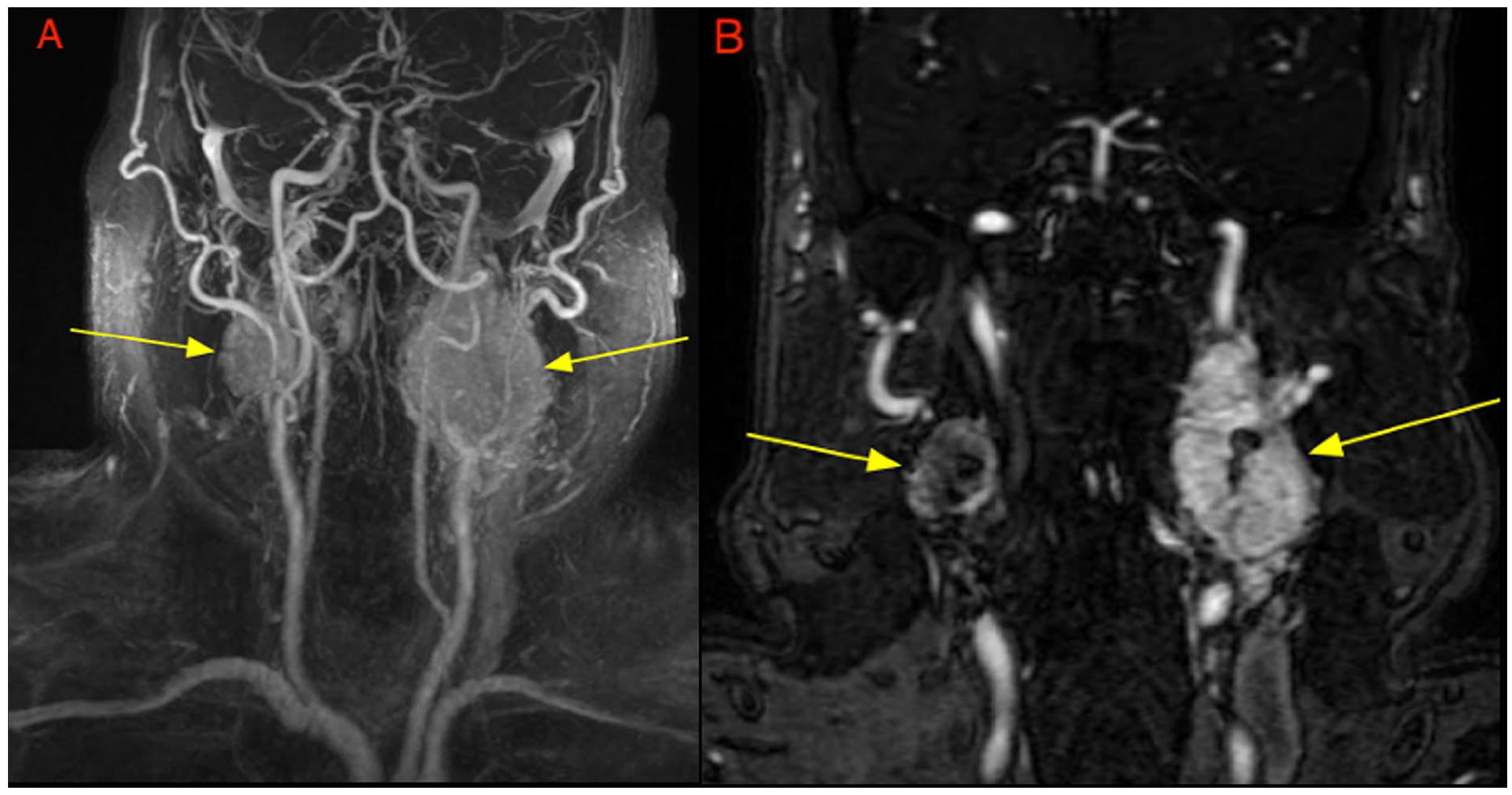
MRA of the neck with maximum intensity projection (A) and contrast-enhanced coronal sequences (B) demonstrate splaying of the carotid bifurcation bilaterally by the enhancing masses (yellow arrows) with the patent flow of the carotid arteries. MRA, magnetic resonance angiography.
Discussion
Carotid body paragangliomas can either arise sporadically or through genetic predispositions. 3 The succinate dehydrogenase (SDH) complex, which is responsible for converting succinate to fumarate in the citric acid cycle, includes 4 subunits: SDHA, SDHB, SDHC, and SDHD. Mutations in SDH genes have been linked to familial head and neck paragangliomas and pheochromocytomas. 3 Other genetic mutations related to paragangliomas include VHL (Von Hippel–Lindau tumor suppressor), NF-1 (neurofibromin), and RET (rearranged during transfection) proto-oncogenes. 3 Therefore, in cases of paragangliomas including those with carotid body involvement, multifocal origin, or concurrent pheochromocytoma, genetic analysis may help identify hereditary risk factors.4,5
Carotid body paragangliomas usually present as a slow-growing neck mass near the angle of the mandible and may uncommonly secrete catecholamines. On CT, the mass will typically have a soft tissue density with splaying of the internal carotid and external carotid arteries, along with rapid contrast enhancement due to its hypervascularity. On MRI, the lesion tends to be isointense relative to muscle on T1-weighted sequences and hyperintense on T2-weighted sequences. A characteristic “salt and pepper” appearance is typically described, which represents punctate areas of hemorrhage and flow voids, respectively. 6 Functional imaging can also be used as a tool in initial staging or posttreatment surveillance. 7 After diagnosis, definitive management involves surgical resection. Con-tinued imaging surveillance is necessary after surgery due to the risk of recurrence and metastasis (5%-10%). 3
Footnotes
Author Contributions
Jagan D. Gupta and Neel D. Gupta conceived the case topic and designed the paper methodology. Abdul R. Khan, Kelvin Li, and Mohammed M. Ansari conducted the data collection and performed the investigation and analysis. Abdul R. Khan, Mohammed M. Ansari, Kelvin Li, Jagan D. Gupta, Neel D. Gupta, Triet M. Do, and Enrique Palacios interpreted the results and contributed to the manuscript writing. All authors reviewed and approved the final manuscript, ensuring accuracy and integrity in the reported findings.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
