Abstract
Facial neuromas are benign with a common clinical manifestation of gradual or sudden peripheral facial nerve paralysis. This study reports an atypical case of facial neuroma in the absence of facial nerve paralysis and discusses the diagnosis and pathophysiological features. A rare case of extensive facial neuroma histopathologically confirmed as a mixed growth of schwannoma and neurofibroma in a 51-year-old man was described. Analysis of the clinical manifestations and Computed Tomography/Magnetic Resonance Imaging (CT/MRI) characteristics was conducted, with documentation of the surgical intervention and associated complications. Intraoperative findings revealed complete destruction of the facial nerve structure, contrasting the patient’s preoperative lack of facial paralysis. The patient underwent complete resection of the tumor followed by end-to-side neurorrhaphy of the facial nerve to the hypoglossal nerve by means of great auricular nerve grafting. Postoperative pathology showed mixed growth of schwannoma and neurofibroma. Postoperative follow-up showed no significant recovery of facial paralysis. To our knowledge, this is the first report of an extensive facial neuroma with a mixture growth of schwannoma and neurofibroma without facial paralysis. The clinical manifestations of facial neuromas are diverse, necessitating a differential diagnosis of each case of temporal bone tumor.
Introduction
Facial neuromas are relatively rare and less common. These tumors can originate anywhere along the facial nerve, ranging from the cerebellopontine angle segment to the extratemporal segment, and tend to show a multisegmental distribution. 1 Pathologically, facial neuromas are mainly divided into 2 types: schwannoma and neurofibroma. The former are benign tumors occurring in the facial nerve fibrous membrane of Schwann cells, while the latter are tumors originating from the endoneurium within the facial nerve trunk, in which the tumors are not clearly demarcated from the nerve fibers. Most of the facial neuromas are benign, slow-growing, and with a clinical manifestation of slow or sudden unilateral peripheral facial nerve paralysis. The treatment is mainly based on surgical resection.
Here, we present a rare case of facial neuroma pathologically diagnosed as a mixed growth of schwannoma and neurofibroma. Although a complete destruction of the facial nerve structure was found intraoperatively, the patient’s preoperative neurological function was completely normal. The patient underwent complete resection of the tumor and end-to-side neurorrhaphy of the facial nerve to the hypoglossal nerve by means of great auricular nerve grafting.
Case
The patient in this report was a 51-year-old man who came to our outpatient department with symptoms of progressive hearing loss and high-frequency tinnitus in the left ear. The patient denied any otorrhea, earfullness, vertigo, headache, facial paralysis, numbness of the face and tongue, hyposmia, and hoarseness. During the last 6 months, he felt left ear pain which was getting progressively worse. Upon physical examination, notable findings included an elevation of the posterior wall of the bony segment of the left external auditory canal and an outward bulging of the tympanic membrane. No signs of peripheral facial paralysis.
Pure-tone audiometry of the left ear showed conductive hearing loss with a gap of between 40 to 60 dB and an As-curve for acoustic impedance. Auditory Brainstem Response (ABR) showed an air-conduction threshold of 90 dB and bone-conduction threshold of 50 dB in the left ear. The speech recognition threshold was 55 dB in the left ear and 25 dB in the right ear; speech recognition scores were100% in both ears. Examination of vestibular evoked myogenic potentials (VEMP) revealed that o-VEMP were unresponsive in both ears and c-VEMP were elicited bilaterally and symmetrically. Head impulse test did not show any abnormality, however caloric test showed a reduced left horizontal semicircular canal function without directional preponderance. The electroneurography of facial nerve showed normal.
CT shows an extensive long irregular mass in the left temporal bone. Enlargement of the facial nerve canal and stylomastoid foramen can be seen, with bone destruction of the genu, the tympanic, and the mastoid segments of facial nerve. The labyrinthine segment of the facial nerve is not involved. In addition, destruction in the mastoid region of the temporal bone as well as resorption and displacement of the auditory ossicles can be seen. There were no obvious abnormalities in the inner ear canal, the cochlea, vestibule, and semicircular canal (Figure 1). MRI showed abnormal signals in the left temporal bone, with moderately hyperintense signal in T2 weighted imaging (T2WI), hypointense signal in T1WI and moderate enhancement on contrast MRI, as well as scattered internal areas of small cystic changes (Figure 1).
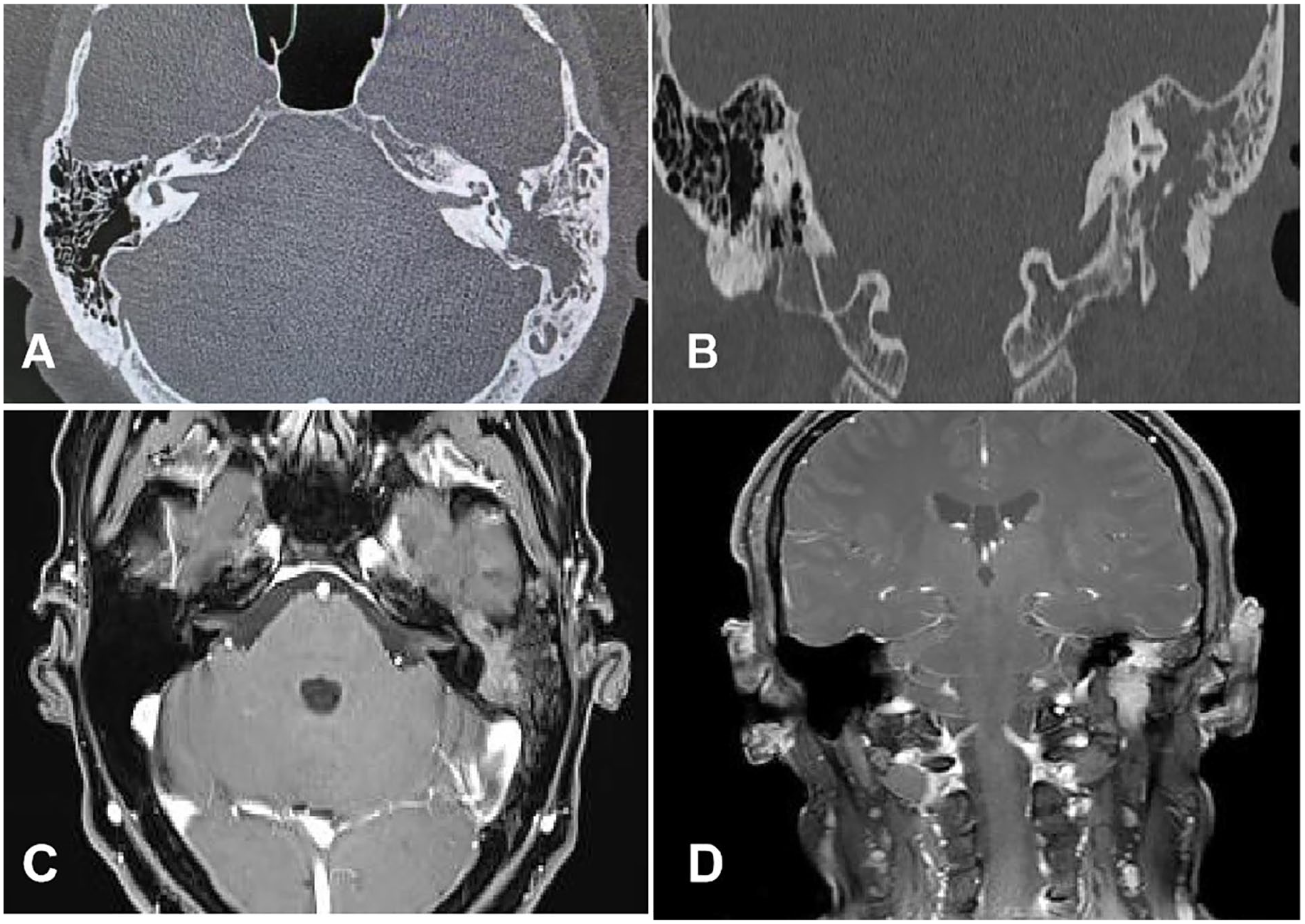
CT scan and MRI of the tumor. (A) Transverse section of CT scan; (B) coronal section of CT scan; (C, D) coronal and transverse sections of contrast-enhanced MRI scans.
Preoperative preparation was done based on the diagnosis of facial neuroma. A modified infratemporal fossa type A approach was used to perform a subtotal temporal bone resection. Intraoperatively, the tumor did not show any traces of the nerve bundles and was found to be growing in an expansive manner along the anatomical path of the facial nerve, from the geniculate ganglion to the outer segment of the temporal bone, with fish-like appearance and no obvious envelope (Figure 2). It had destroyed the tympanic bone, entered the tympanic cavity, and destroyed the stylomastoid foramen. After resection of all tissues along the anatomical path of the facial nerve, we identified the severed peripheral end of the facial nerve until intraoperative pathology showed normal nerve fibers. Due to the destruction of the geniculate ganglion, when we expose the safe border of the labyrinthine segment, the severed end of the labyrinthine segment was not structurally clear enough for end-to-end anastomosis. As a result, we performed an end-to-side anastomosis with the hypoglossal nerve via the great auricular nerve grafting (Figure 2). Postoperative pathology showed that the tumor was a mixed growth of facial nerve schwannoma and neurofibroma (Figure 2).
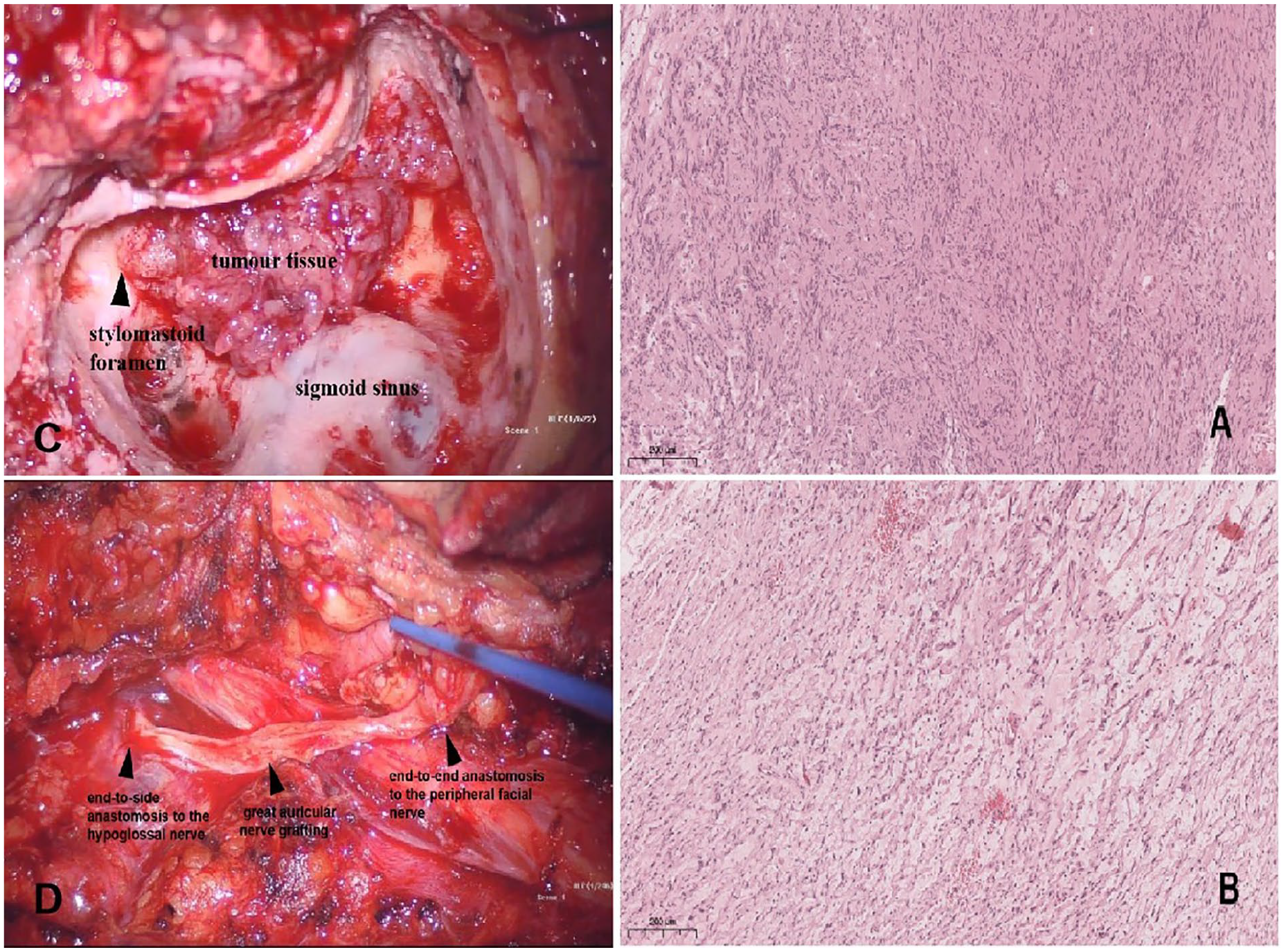
(A) Schwannoma component within tumor tissue. Tumor cells are shuttle-shaped and tightly arranged like fences. The nucleus is pointed at one end and blunt at the other. (B) Neurofibroma component within tumor tissue. Tumor cells are spindle-shaped, sparsely arranged, wavy, with indistinct cell borders and elongated nuclei. (C) Facial neuroma grew in an expansive manner along the anatomical path of the facial nerve. (D) Intraoperative demonstration of an end-to-side anastomosis of the great auricular nerve to the hypoglossal nerve and an end-to-end anastomosis to the peripheral facial nerve.
At the 2-month postoperative follow-up, the patient showed no recovery of facial muscle function but with normal static performance.
We have obtained written informed consent from the patient for the publication of this case report.
Discussion
Clinical manifestations of facial tumors are varied and symptomatic, with peripheral facial palsy being the most prevalent. Other symptoms include hearing loss, tinnitus, vertigo, hemifacial spasm, among others. In general, the onset of facial paralysis is associated with the tumors’ location, extent, and size. Tumors situated in the tympanic segment of facial nerve lead to nerve compression in the narrow facial nerve canal, thereby precipitating facial paralysis. Conversely, tumors in the mastoid segment, which occupy a larger area, tend to cause facial paralysis at a later stage. Due to the slow growth of the tumor, it is often misdiagnosed, and is often detected when the tumor grows to a large size or expands intracranially or extracranially, or is confirmed by the presence of facial paralysis on biopsy of a mass in the external auditory canal. In this case, the patient had predominantly symptoms of tinnitus and hearing loss without any manifestation of facial paralysis. The patient initially attributed these symptoms to age-related hearing loss and did not seek medical attention until the onset of ear stuffiness and pain, suggesting a potential insidious onset of facial neuroma, which necessitates serious consideration. For patients misdiagnosed with Bell’s palsy, the correctness of the diagnosis must be questioned if there is no recovery within 6 months after therapy. In patients with progressive facial paralysis without any other causes, the possibility of this disease should be considered, especially if the facial paralysis is accompanied by facial spasms or convulsions. In patients with vestibulocochlear symptoms, systematic audiological and vestibular function tests, as well as CT or MRI of the temporal bone, should be performed.
Facoini et al 2 reported 4 pathological types of facial neuromas: facial nerve schwannoma (FNS), facial nerve neurofibroma (FNN), hemangioma and meningioma, of which FNS is the most common. FNS have an eccentric growth of cells originating from ectodermal Schwann cells, while nerve fibers are relatively uninvolved, resulting in relatively large tumors and late onset of symptoms. The tumor cells are arranged in bundles under the microscope. FNS is usually encapsulated and usually first destroys the facial nerve canal or invades through the defective facial nerve canal to form a soft tissue mass. Clinical manifestations depend on the location of the tumor. Facial paralysis is common and can be gradual or acute. On CT scan, it shows segmental enlargement and local destruction of the facial nerve canal and soft tissue mass. On T1WI of MRI, FNS appears to be hypointense, and on T2WI, it appears hyperintense compared to the surrounding brain tissue.3-5 On contrast-enhanced MRI, small FNSs show strong homogenous enhancement but can have heterogeneous enhancement due to close compacted Antoni type A cells (hyperintense) and loosely packed Antoni type B cells (hypointense), and large FNS can show cystic degeneration.3-5
FNNs are relatively rare, predominantly affecting infants and children. 6 FNNs are either solitary or multiple, and in the case of multiple cases, they are called neurofibromatosis. The tumor originates from mesodermal tissues. Histologically, FNNs are distinguished by a plexiform or swirling pattern of fusiform cells featuring elongated nuclei, encapsulated by collagen, and lacking a distinct capsule. 7 Typically, the tumor extends along the long axis of the facial nerve within facial nerve canal, causing the facial nerve to thicken in a rod-like or spindle-shaped manner. The tumor is indistinctly demarcated from the nerve fibers, leading to early symptoms of facial paralysis. Chi et al 8 found that the FNN was confined to the enlarged facial nerve canal with the presence of intact epineurium and canal wall. Compared to FNS, FNN tends to involve multiple segments of the facial nerve, usually containing geniculate ganglion or tympanic segment. 9 There were no “skip lesions” (discontinuous involvement of facial nerve segments) in FNN, as was also true in Dai et al’s 10 report. But “skip lesions” were found in 20% to 29.2% cases with schwannomas.11,12 In other words, “skip lesions” may be an important exclusive point of FNNs from schwannomas. On T1WI of MRI, FNN appears to be isointense, and on T2WI, it appears to be hyperintense. Significant enhancement of FNN can be seen in the contrast-enhanced MRI.
The preoperative MRI of this patient showed hypointense signal on T1WI and moderately hyperintense signal on T2WI, with a relatively uniform signal intensity and moderate contrast enhancement. In light of the growth pattern of the mass along the facial nerve, as evidenced by CT and MRI, the author posited that the facial nerve origin was the more probable diagnosis, despite the absence of facial paralysis. The final pathological result of the surgery was a mixed growth of FNS and FNN. During surgery, we found the tumor to be unencapsulated, fish-like, without neural structures, and occupying the entire area from the geniculate ganglion to the extratemporal aspect of the temporal bone. It was indeed surprising to note the patient’s intact facial nerve function in the absence of discernible facial nerve structures, suggesting the presence of residual nerve fiber bundles that are sufficient to preserve facial nerve functionality. Nonetheless, the threshold for the number of residual bundles required to maintain nerve function remains to be elucidated, necessitating further investigative research. Unfortunately, in this person, we did not send all of the patient’s tumor tissue intraoperatively to identify the nerve structures.
Facial neuroma should be differentiated from acoustic neuroma, Bell’s palsy, trigeminal schwannoma, facial spasm, parotid adenoma, glomus jugulare tumor, and middle ear cholesteatoma. Given the absence of facial paralysis, which is a hallmark of facial neuroma, it was particularly imperative to differentiate this case from middle ear cholesteatoma. The author’s preoperative diagnosis of facial neuroma was based on the tumor’ growth along the path of the facial nerve. Therefore, we believe that although the clinical manifestations of facial neuromas may vary, radiographic analysis prior to surgery plays a crucial role.
The therapy for facial neuroma predominantly hinges on surgical intervention, with the guiding principle being the maximal preservation or restoration of facial nerve function, while ensuring complete tumor resection and preservation of hearing. Chi et al 8 reported that an attempt was made to search for facial nerve tissue along the facial nerve during surgery for FNS, but no facial nerve tissue was found in the postoperative pathological sections, whereas the facial nerve tissue wrapped by the tumor could be seen in the histopathological section of FNN, which suggests that the surgery for FNN is almost impossible to preserve the facial nerve tissue, but there is a possibility of preserving the facial nerve by separating the FNS. The tumor and the affected nerve should be excised with safe margins, with both distal and proximal nerve margins undergoing frozen section examination to confirm the absence of tumor remnants before nerve grafting. The resected nerve trunk is transplanted with the great auricular nerve or peroneal nerve to restore the continuity of the facial nerve. Peripheral nerves can be repaired through nerve anastomosis as appropriate, and end-to-end anastomosis is the most physiological and effective.
In this case, the patient underwent tumor resection by a modified infratemporal fossa type A approach, which revealed that the tumor appeared to originate from the mastoid segment, with the labyrinthine segment remaining unaffected. Due to the large extent of the tumor, we performed a total resection of the tumor despite the patient’s lack of preoperative facial paralysis. The challenge remained to determine the amount of tumor tissue that could be excised while still preserving the integrity of the facial nerve function. Finally, a facial nerve-sublingual nerve end-to-side anastomosis was conducted by means of auricular nerve grafting. Recovery of facial palsy was not evident at follow-up 2 months after surgery, so more follow-up time may be needed.
Conclusion
In this case, we report a rare case of extensive facial neuroma histopathologically confirmed as a concomitant growth of schwannoma and neurofibroma wherein the patient did not exhibit facial nerve paralysis. Notwithstanding the intraoperative observation of comprehensive facial nerve disruption, the patient’s neurological function prior to surgery remained entirely intact. The patient was subjected to a total excision of the neoplasm, followed by an end-to-side neurorrhaphy of the facial nerve to the hypoglossal nerve, using the great auricular nerve as a bridge.
Footnotes
Acknowledgements
None.
Author Contributions
Y.L. wrote the manuscript. L.Z. collected and analyzed the data. Z.H. proposed the concept and designed this study. J.Z. and Z.Y. collected the data. All authors read and approved the final manuscript.
Data Availability Statement
If more in-depth information is needed, please reach out to the corresponding author with a justified request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Key disciplines of Huadong Hospital (ZDXK2214).
Ethical Statement
Not applicable.
Informed Consent/Patient Consent
The patient has signed the informed consent.
Trial Registration Number
Not applicable.
