Abstract
NUT carcinoma is a rare, aggressive malignancy characterized by chromosomal rearrangements involving the NUTM1 gene. We present the case of a 47-year-old male with a history of hypertension, diagnosed with NUT carcinoma originating in the ethmoid sinus and accompanied by extensive neck metastases. The patient underwent a left modified radical neck dissection and endoscopic nasal tumor excision, followed by concurrent chemoradiotherapy. Pathological examination confirmed NUT carcinoma with metastatic cancer cells in 41 out of 72 lymph nodes of left neck, along with extranodal extension. The patient remained alive at the 4-month follow-up. This case highlights the importance of early identification and including NUT carcinoma in the differential diagnosis to optimize treatment outcomes.
Background
NUT carcinoma is a rare and highly aggressive malignancy characterized by chromosomal rearrangements involving the NUTM1 gene. 1 This poorly differentiated carcinoma can occur in various anatomical locations, with a strong predilection for midline structures, such as the head and neck. 2 Due to its rarity, nonspecific clinical presentation, and lack of proper diagnostic tools, NUT carcinoma is frequently misdiagnosed or diagnosed late, contributing to its poor prognosis. Currently, there is no standard treatment for NUT carcinoma. The primary treatment approach typically includes surgery, often combined with chemoradiotherapy. 3 The prognosis remains grim, with limited survival rates and a high risk of recurrence. Here, we report a case of NUT carcinoma originating from the ethmoid sinus, which presented with extensive neck metastasis and underwent surgical resection, followed by chemoradiotherapy. This case emphasizes the importance of considering NUT carcinoma in the differential diagnosis of poorly differentiated head and neck carcinomas to facilitate timely diagnosis and improve outcomes.
Case Presentation
A 47-year-old male patient with a history of hypertension presented with a painless left neck mass that had persisted for approximately three months. The patient initially sought evaluation at another regional hospital, where an incisional biopsy revealed metastatic poorly differentiated carcinoma in the left submandibular lymph nodes. Due to the findings, the patient was referred to our hospital for further management and underwent a comprehensive evaluation.
In our hospital, local examination confirmed multiple enlarged lymph nodes at levels II-III on the left side of the neck, with the largest measuring about 4 cm by 3 cm. Fiberscopy showed a granular lesion occupying the left middle meatus. A sinoscopic biopsy confirmed the lesion to be poorly differentiated squamous cell carcinoma. Imaging studies, including a head and neck CT scan, revealed a left ethmoid sinus tumor extending into the nasal cavity (Figure 1). Positron emission tomography-computed tomography (PET-CT) incidentally revealed a hypermetabolic lesion in the sigmoid colon, and subsequent colonoscopy confirmed the lesion to be adenocarcinoma. There was no evidence of distant metastasis based on chest X-ray and abdominal ultrasound.
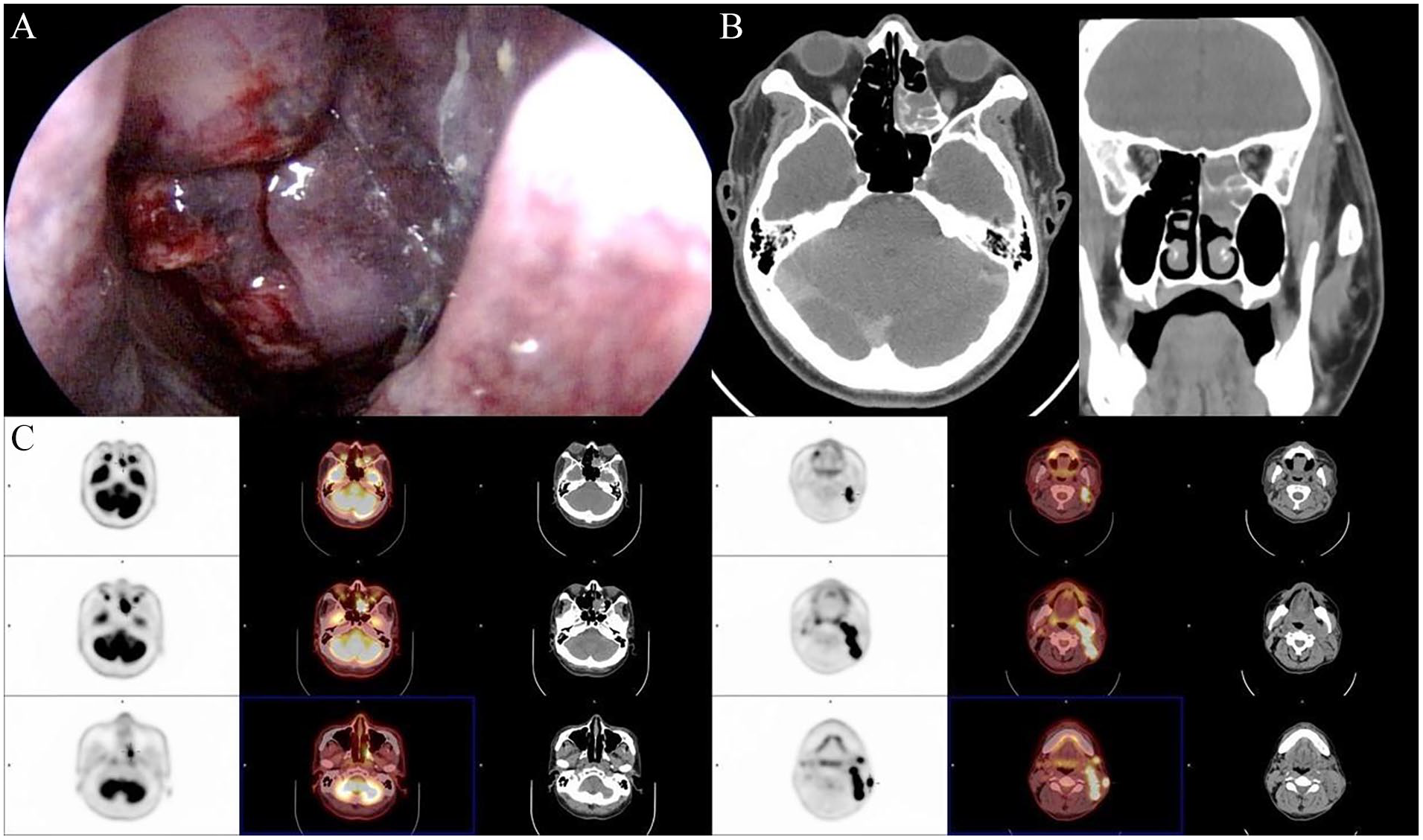
(A) Endoscopic view showing a granular, irregular, and erythematous lesion occupying the left middle meatus. (B) Axial and coronal computed tomography images reveal an expansile mass within the left posterior ethmoid sinus, extending into the nasal cavity. The mass is characterized by a heterogeneous appearance, with bony remodeling and destruction. (C) Serial PET-CT scans reveal intense fluorodeoxyglucose (FDG) uptake in the left ethmoid sinus (standardized uptake value (SUV) 15.88, delayed SUV 19.04), and increased uptake in lymph nodes in the left neck (level II-V, SUV up to 20.97), indicating an aggressive tumor with extensive ipsilateral neck metastasis.
The patient was admitted for definitive surgical treatment and underwent left modified radical neck dissection encompassing levels I to V, along with a left endoscopic medial maxillectomy and nasal tumor excision. Pathology confirmed the diagnosis of NUT carcinoma and examination of neck specimens revealed 72 lymph nodes, of which 41 were positive for metastatic cancer cells, with evidence of extranodal extension (Figure 2). The surgery proceeded without complications, and the patient was discharged in stable condition.
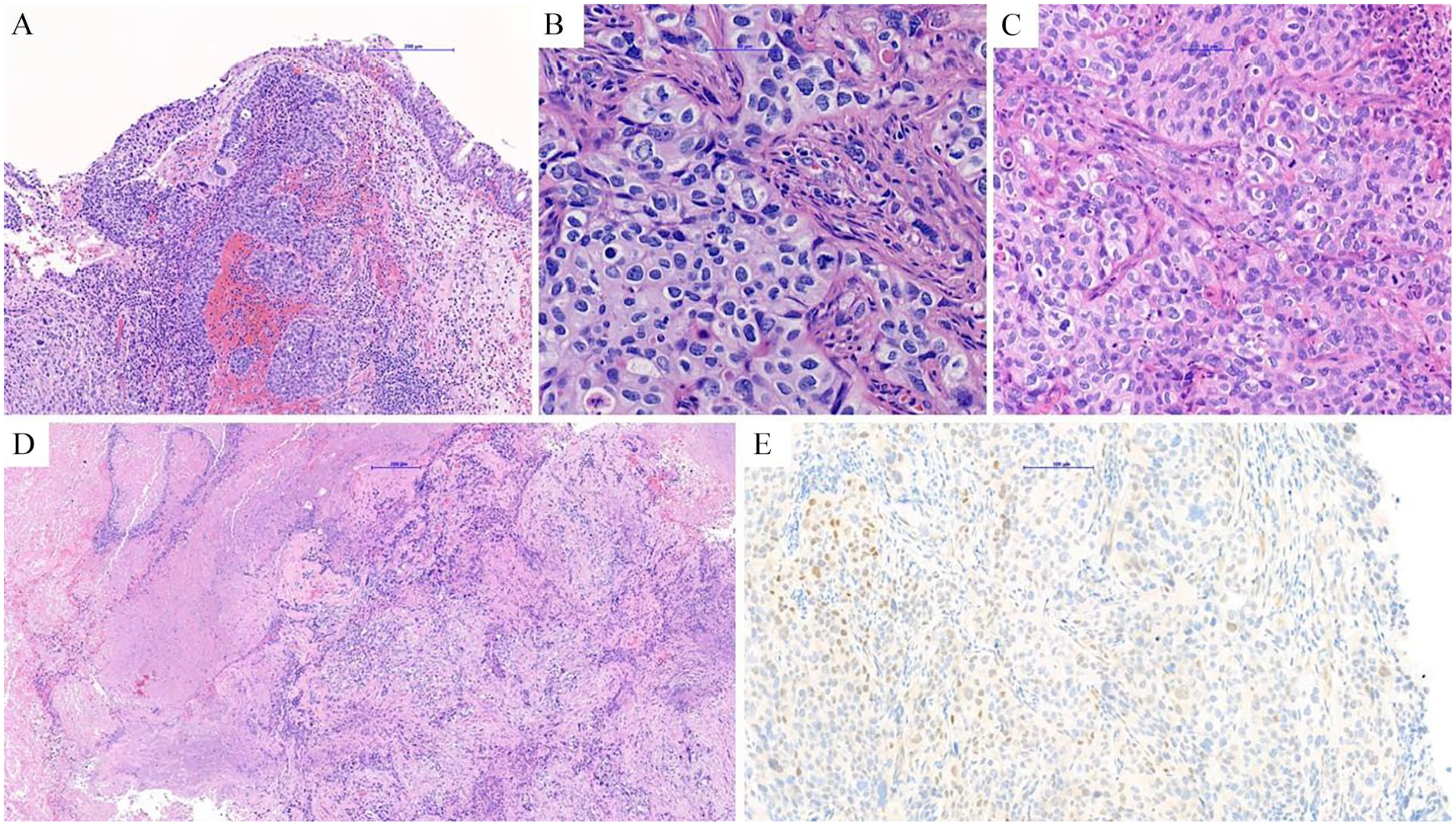
(A) Sections show sheets and nests of small to intermediate-sized undifferentiated neoplastic cells arising from respiratory epithelium. (B) and (C) The tumor cells have relative evenly sized nuclei with irregular outlines and vesicular chromatin. The cytoplasm varies from pale eosinophilic to basophilic. Nucleoli and brisk mitotic activity are discernible. Neutrophilic infiltration is also present. (D) Necrosis is also seen. (E) NUT stain: tumor cells show focal positive staining.
During the third week after surgery, a fiberscope examination revealed recurrence of the nasal tumor, and subsequently, the patient started concurrent chemoradiotherapy targeting both the left neck and tumor bed. Chemotherapy consisted of cisplatin 40 mg/m², while radiotherapy involved 6600 cGy in 33 fractions over 6 to 7 weeks. Subsequent clinical evaluations noted the shrinkage of the residual tumor. The patient remained alive and kept follow-up at the outpatient department for 4 months.
Discussion
NUT carcinoma is a rare and highly aggressive malignancy characterized by chromosomal rearrangements involving the NUTM1 gene. 1 This poorly differentiated carcinoma is characterized by its rapid progression, and its diagnosis is often challenging due to a lack of specific clinical or histopathological features, as well as the absence of adequate diagnostic tools. The disease was first described in 1991 in two cases involving mediastinal tumors with a t(15;19) translocation.4,5 Initially thought to primarily affect midline structures in younger patients, NUT carcinoma has since been found in a variety of anatomic locations. Studies also report cases in nonmidline structures like the liver, salivary gland, and bone, further demonstrating its diverse presentation. 6 This led to its reclassification from NUT midline carcinoma to simply NUT carcinoma in 2015. 2
The hallmark of NUT carcinoma is the rearrangement of the NUTM1 gene, located on chromosome 15q14, with various fusion partners. 7 The most common translocation partner is BRD4, located on chromosome 19p13, forming a BRD4-NUT fusion oncogene in approximately 70% of cases. 8 Other fusion partners include BRD3, NSD3, ZNF532, and ZNF592, among others. 9 The resulting fusion proteins disrupt normal cell differentiation and promote oncogenesis through bromodomain and extraterminal domain (BET) proteins, which contribute to the dysregulation of cell growth and differentiation. 10
NUT carcinoma is exceedingly rare, with only few case reports in the literature to date. 11 The exact prevalence is not well known, but recent studies estimate an incidence rate of approximately 0.41 per million children per year in certain populations. 2 It is believed to be underdiagnosed due to low awareness and the lack of specific diagnostic markers. NUT carcinoma can affect individuals of any age, from newborns to the elderly, with a median age of around 30 years. 12 There is no significant sex predilection, as studies show an approximately equal distribution between males and females. 7
The most common presentation of NUT carcinoma involves rapidly growing masses in midline structures, such as the thorax, head, and neck. In the head and neck region, the most common sites of NUT carcinoma are the sinonasal tract, salivary glands, and nasopharynx. 13 Many patients present with advanced disease, characterized by significant local invasion and frequent metastasis. Due to the aggressive nature of the disease, most patients are diagnosed at an advanced stage. 3
Diagnosis of NUT carcinoma requires immunohistochemical staining with a NUT-specific monoclonal antibody, which is highly sensitive and specific for this tumor type. Additionally, fluorescence in situ hybridization or reverse transcription-polymerase chain reaction can be used to confirm the presence of NUTM1 rearrangements.1,14 Histologically, NUT carcinoma consists of poorly differentiated cells with frequent squamous differentiation, typically showing monomorphic, discohesive epithelioid cells with abrupt keratinization. 1 It can be challenging to distinguish from other poorly differentiated carcinomas without the aid of molecular diagnostics.
There are no standardized treatment protocols for NUT carcinoma. Current management typically involves a combination of surgery, radiotherapy, and chemotherapy. 15 Surgical resection is often attempted, but the tumor’s aggressive nature and propensity for early metastasis limit its efficacy. Radiotherapy and chemotherapy can improve short-term survival but do not typically result in long-term remission. 3 BET inhibitors, which target the oncogenic BRD-NUT fusion protein, are currently being studied as potential targeted therapies, though they are not yet part of standard care. 16
The prognosis for NUT carcinoma is poor, with a median survival of 6 to 9 months and a 5-year survival rate of less than 10%. 3 A prognostic model suggests that nonthoracic primary tumors with non-BRD4-NUT fusions are associated with improved survival, emphasizing the need for subtype-specific management strategies. 17 Early diagnosis and aggressive management may improve outcomes in some cases, but most patients succumb to the disease within a year of diagnosis due to rapid progression and resistance to conventional therapies. 3
Future research efforts are focused on improving the understanding of the molecular mechanisms underlying NUT carcinoma and developing targeted therapies, such as BET inhibitors, that can more effectively combat the disease. 11 BET inhibitors, such as RO6870810, have shown promise in preclinical models by disrupting BRD4-NUT fusion-mediated oncogenic transcription 18 However, resistance mechanisms, including upregulation of compensatory pathways, remain a significant challenge. Additionally, studies have identified the role of chromatin remodeling complexes in mediating resistance, highlighting potential co-targeting strategies that may enhance the efficacy of BET inhibitors. 19 Furthermore, histone deacetylase inhibitors, when used in combination with BET inhibitors, have shown synergistic effects in suppressing tumor growth in preclinical models by modulating chromatin accessibility and transcriptional regulation. 20 Immunotherapy is another promising avenue, and clinical trials investigating combinations of targeted therapies and immunotherapies are currently underway. 21 Enhanced awareness among clinicians and pathologists is crucial to improving early diagnosis and referral for appropriate treatment.
Conclusion
In conclusion, it is essential to include NUT carcinoma in the differential diagnosis of poorly differentiated carcinomas, particularly those involving midline structures, to ensure timely and accurate diagnosis and to improve patient outcomes.
Footnotes
Acknowledgements
This manuscript was revised in English by ChatGPT-4.
Data Availability Statement
The data supporting this case report is included within the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Our institution does not require ethical approval for reporting individual case
Informed Consent
Written informed consent was obtained from the patient for his anonymized information to be published in this article.
Trial Registration Number/Date
This study wasn’t included in a clinical trial.
Grant Number
This study did not receive any specific funding or grants from public, commercial, or not-for-profit sectors.
