Abstract
Keywords
Introduction
Cochlear implants (CIs) are a significant advancement for individuals with severe-to-profound sensorineural hearing loss, providing opportunities for auditory rehabilitation. 1 Despite their success, CI outcomes vary and may be influenced by the underlying pathology of hearing loss. This systematic review aimed to compare CI outcomes in patients with presynaptic and postsynaptic neuropathy, two distinct conditions with unique rehabilitation challenges. 2 Auditory neuropathy (AN) spectrum disorder is characterized by otoacoustic emissions and/or cochlear microphonics (CMs), along with absent or abnormal auditory brainstem responses. 3 AN can be classified based on lesion site: presynaptic (affecting inner hair cells or the synapse between inner hair cells and the auditory nerve) and postsynaptic (affecting spiral ganglion neurons or the auditory nerve beyond the hair cell-neuron synapse). Differentiating between pre- and postsynaptic neuropathy is crucial for CI efficacy, as the device relies on the integrity of the spiral ganglion neurons and the auditory nerve to transmit electrical stimuli effectively. 4
AN can be classified based on the anatomical site of dysfunction using audiological and electrophysiological assessments, including presynaptic, postsynaptic, and central auditory pathway disorders. Presynaptic AN, often caused by otoferlin gene mutations, involves deficits in inner hair cells or ribbon synapses.5–7 This condition results from decreased neurotransmitter (glutamate) release and delayed release timing, impairing the temporal coding of auditory signals.8,9 Postsynaptic AN includes abnormalities along the auditory nerve pathway, such as issues with unmyelinated dendrites of the auditory nerve, auditory ganglion cells, and their myelinated axons and dendrites. 3 Pathological processes may involve demyelination, reducing signal conduction velocity and causing dyssynchrony, or axonal degeneration, which decreases the auditory signal transmitted to the brainstem.7,8 Central AN pertains to lesions within the brainstem, often associated with cerebellopontine angle tumors such as vestibular schwannomas and meningiomas. 3
Genetic predisposition plays a crucial role in the etiology of AN and is linked to various gene mutations.10,11 This review does not aim to exhaustively investigate genetic factors; however, it includes an exploration of genetic influences to understand their interaction with CI outcomes. Recognizing the genetic landscape helps to appreciate the complex nature of AN and its management with CIs.12–14 The efficacy of cochlear implantation in presynaptic versus postsynaptic neuropathy remains an important area of study. Therefore, this review aimed to systematically examine the current evidence to compare the effectiveness of CIs in these two patient populations, thus providing insights that may inform clinical decision-making and guide future research.
Materials and Methods
Eligibility Criteria
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines 15 were followed to ensure transparency and completeness of reporting. Being a systematic review, ethical approval was not mandatory. The population, intervention, comparison, and outcome (PICO) framework was used to guide the research question and literature search strategy:
• Population: Patients diagnosed with AN, further stratified based on lesion site into presynaptic and postsynaptic neuropathy.
• Intervention: CIs.
• Comparison: Efficacy of CIs in presynaptic versus postsynaptic neuropathy to determine if lesion site influences outcomes.
• Outcome: Efficacy measures including auditory performance, speech perception, and quality of life.
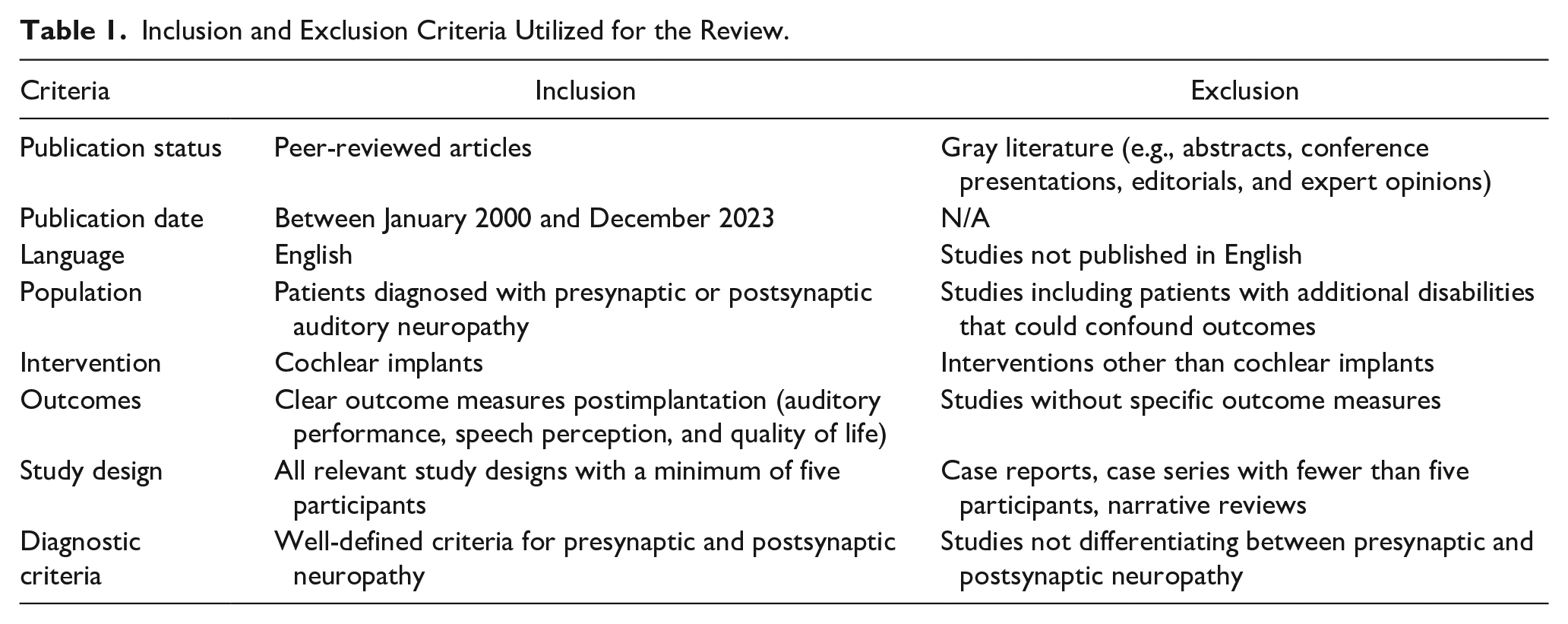
Table 1 shows the eligibility criteria for the different types of studies considered for this review.
Inclusion and Exclusion Criteria Utilized for the Review.
Database Search Strategy
The search strategy for this review involved identifying relevant studies from seven databases: MEDLINE (via PubMed), Embase, Cochrane Central Register of Controlled Trials (CENTRAL), Web of Science, Scopus, CINAHL, and PsycINFO. The unique indexing system of each database was considered, using a combination of Medical Subject Headings and free-text terms with Boolean operators to improve search sensitivity and specificity. Search terms included those related to CIs, presynaptic neuropathy, postsynaptic neuropathy, and AN. Boolean operators “AND” and “OR” were used to combine search terms, where “AND” narrowed the search by combining concepts, and “OR” expanded it by including synonyms or related terms (Table 2).
Search Strings Utilized Across the Different Databases for this Review.

Abbreviation: CENTRAL, Cochrane central register of controlled trials.
Data Collection Process and Items Selected
Data extracted from each study included patient demographics, type of neuropathy (presynaptic or postsynaptic), genetic predisposition, cochlear implantation details (device type and surgical approach), and outcome measures. Outcome measures comprised auditory performance scores, speech recognition thresholds, and postimplantation quality of life assessments. Genetic predispositions, including specific gene mutations associated with AN, were noted when available; however, they were not the primary focus of this review. This information was collated to identify potential correlations between genetic factors and CI outcomes without detracting from the main comparison between presynaptic and postsynaptic neuropathies.
The data were synthesized using a narrative review approach owing to the expected heterogeneity of the studies. This analysis aimed to determine whether CIs have a differential impact on auditory rehabilitation outcomes between patients with presynaptic and postsynaptic neuropathies.
Bias Assessment Protocol
Bias assessment of the studies included in this review was conducted using the ROBINS-I tool. 16 ROBINS-I considers several bias domains with risks ranging from low to critical, including the impact of genetic factors and intervention fidelity.
Certainty Bias
After appraising individual study risk of bias using ROBINS-I, the GRADE framework 17 was applied to assess the overall quality of evidence for each outcome. This assessment considered several factors, including study limitations (risk of bias), inconsistency of results, indirectness of evidence, imprecision of effect estimates, and the likelihood of publication bias. For each outcome, the studies were initially rated as having high-certainty evidence. The certainty was then downgraded by one or two levels if serious or very serious concerns were identified in any of the GRADE domains. For example, if studies exhibited a high risk of bias according to ROBINS-I, the certainty of the evidence would be downgraded. Certainty was also downgraded for inconsistency if there was substantial unexplained heterogeneity in the results, or for imprecision if confidence intervals were wide and crossed clinical importance thresholds.
Results
Study Selection Process
As detailed in Figure 1, the review initially identified 343 records from various databases, with no entries from registers. After removing 52 duplicates, 291 records were screened. Further, 59 records were excluded because of unavailable full texts, leaving 232 reports; of them, 36 could not be retrieved for evaluation. The remaining 196 reports underwent a detailed eligibility assessment. Exclusions were as follows: 42 did not meet PICO criteria, 35 were off-topic, 53 were individual case reports, 26 were scoping reviews, and 33 were literature reviews. Ultimately, seven studies met all criteria and were included in the review.18–24
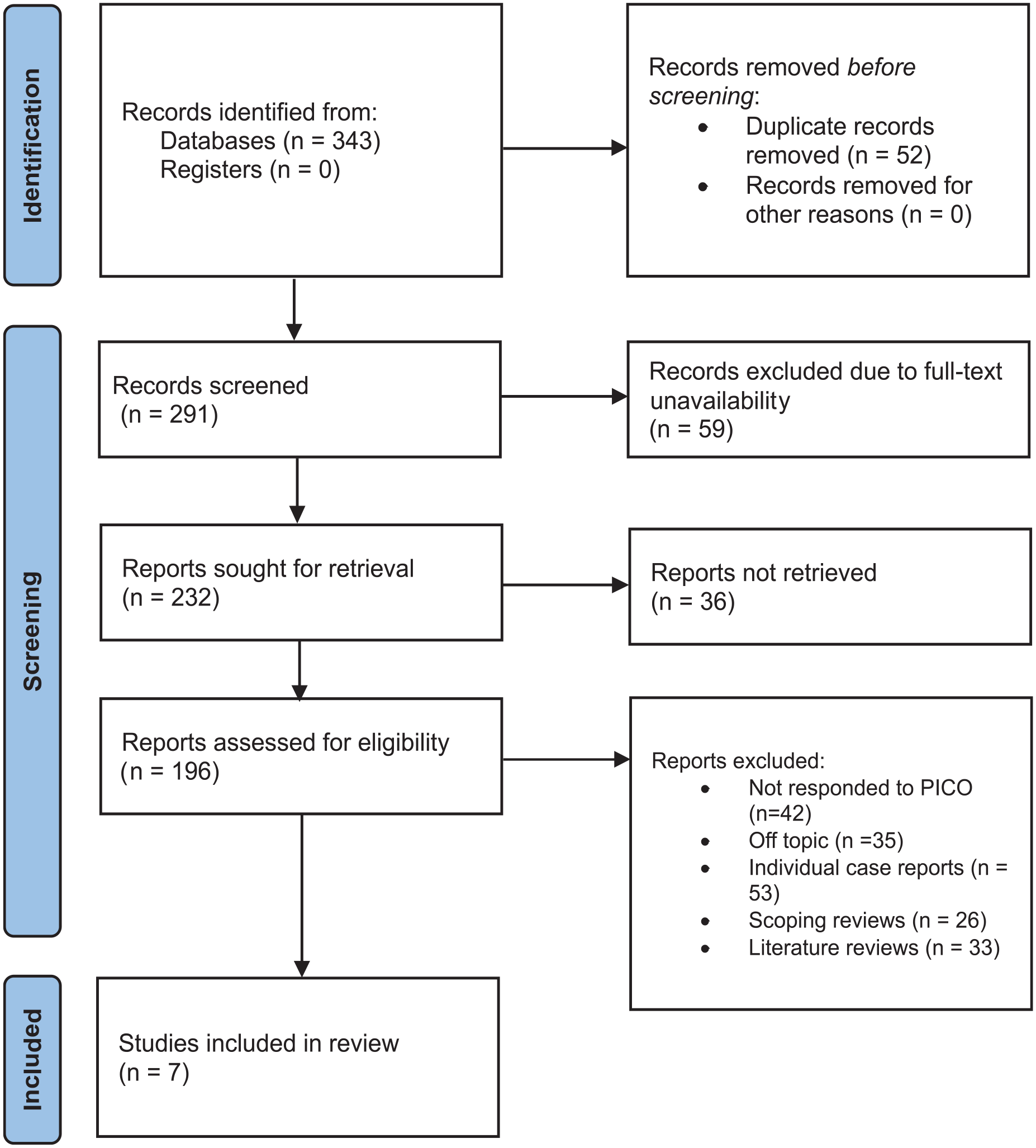
Article selection process representation of the review.
Observed Bias Across the Studies
As shown in Figure 2, the ROBINS-I tool indicated generally robust methodological quality, with most studies showing a low risk of bias in most domains. However, Lin et al., 18 Rodríguez-Ballesteros et al., 22 and Rosamaria et al. 23 faced moderate risks in specific domains related to confounding factors and the selection of reported results, indicating potential issues with group comparability or result reporting.
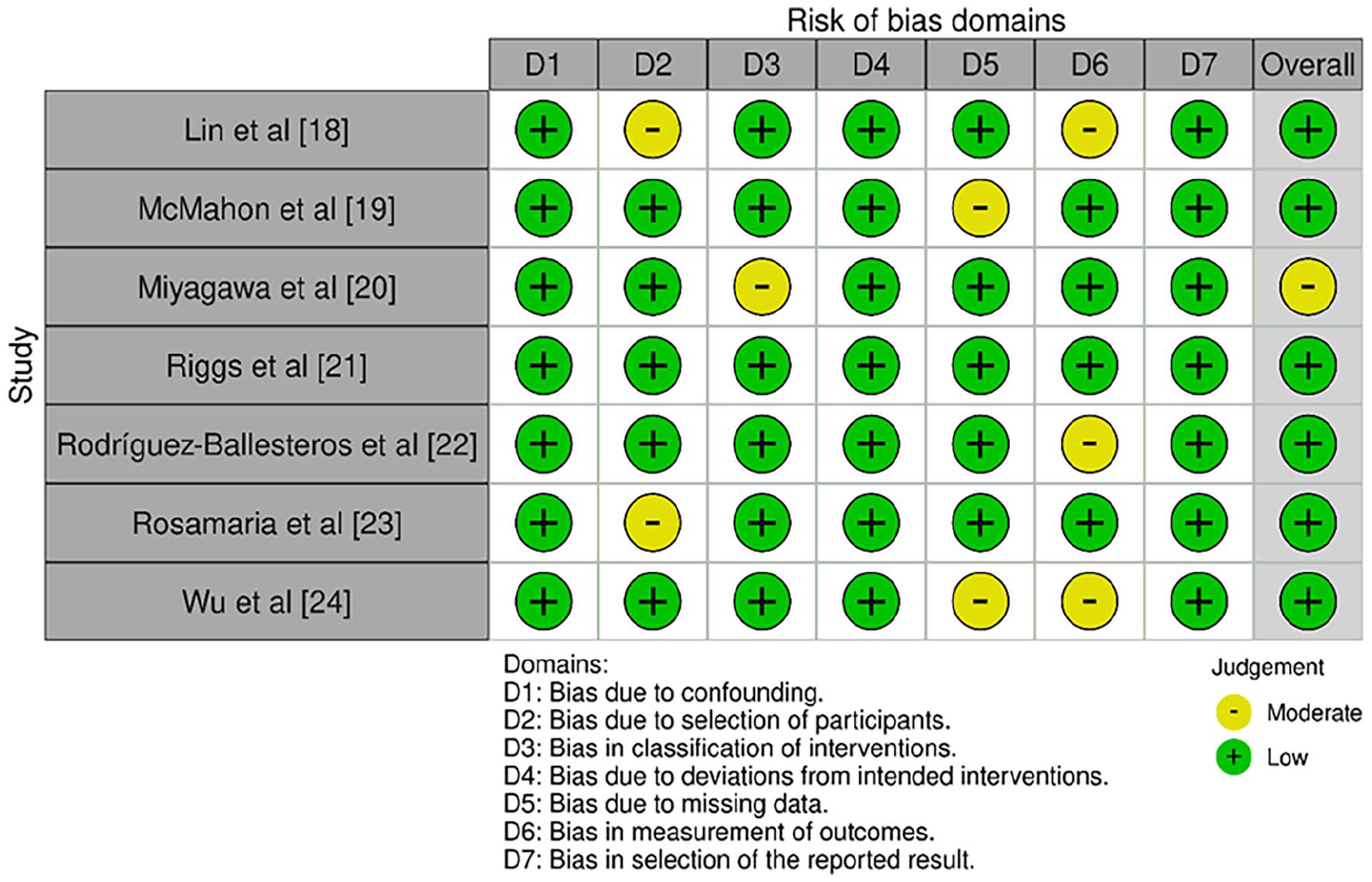
Bias assessment across the included studies using ROBINS-I tool.
Riggs et al. 21 achieved a low risk of bias across all domains, suggesting a particularly strong methodological design. McMahon et al. 19 and Wu et al. 24 also showed a low overall risk of bias, despite moderate concerns in one domain related to outcome measurement and result selection, respectively. These isolated moderate-risk instances did not substantially impact the overall conclusions.
The only study with an overall moderate risk of bias was Miyagawa et al., 20 with concerns primarily related to confounders, indicating potential issues in accounting for factors influencing outcomes.
Etiological Factors and Cohort Types Analyzed
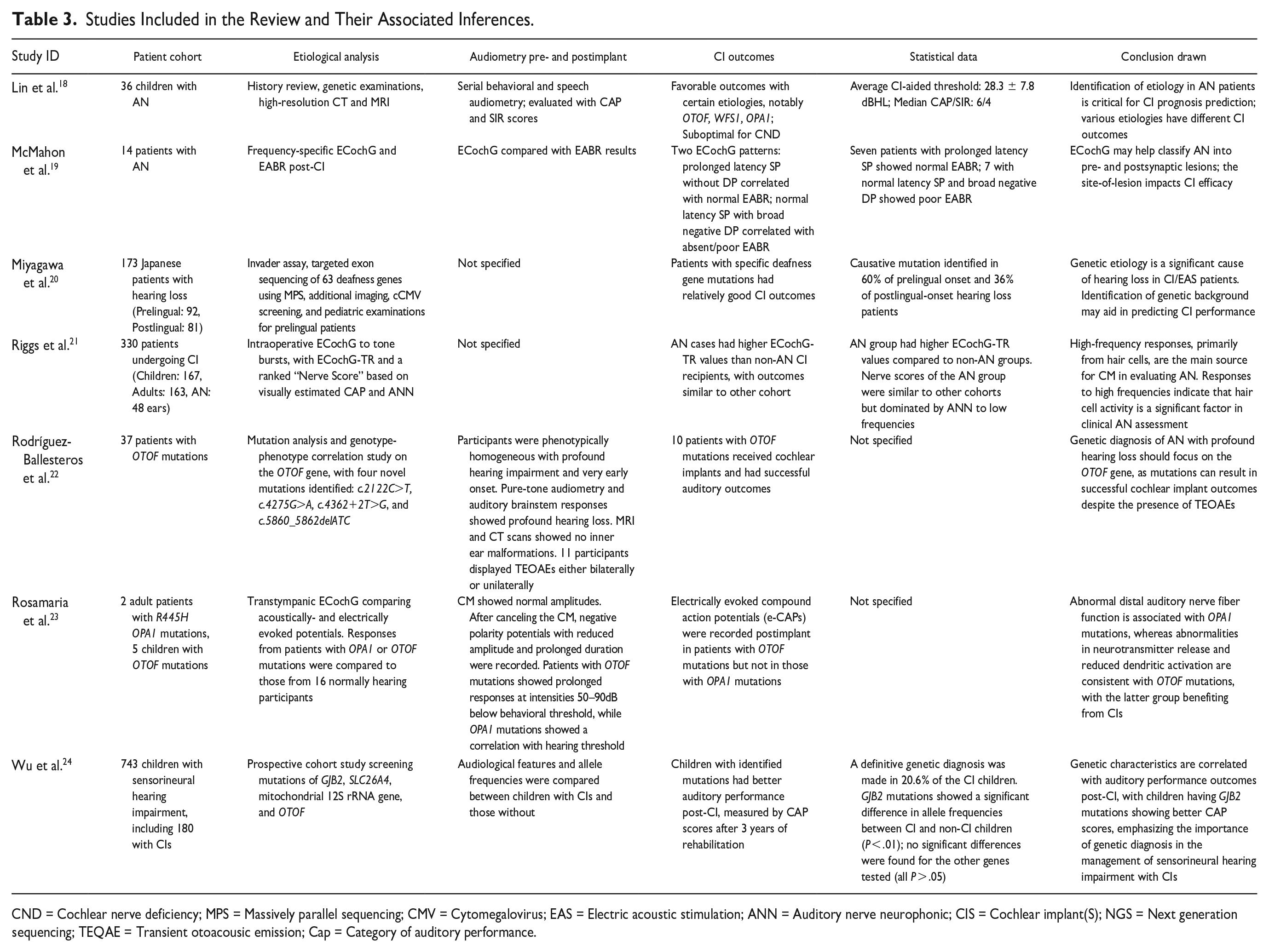
Table 3 presents observations from all included studies.18–24
Studies Included in the Review and Their Associated Inferences.
CND = Cochlear nerve deficiency; MPS = Massively parallel sequencing; CMV = Cytomegalovirus; EAS = Electric acoustic stimulation; ANN = Auditory nerve neurophonic; CIS = Cochlear implant(S); NGS = Next generation sequencing; TEQAE = Transient otoacousic emission; Cap = Category of auditory performance.
Lin et al. 18 investigated a cohort of 36 children with AN, utilizing patient histories, genetic examinations, and advanced imaging (computed tomography and magnetic resonance imaging) to correlate clinical presentation with genetic and anatomical abnormalities.
McMahon et al. 19 investigated 14 participants with AN, using frequency-specific electrocochleography (ECochG) and electrically evoked auditory brainstem responses (EABR) post-cochlear implantation to characterize electrophysiological responses.
Miyagawa et al. 20 examined 173 Japanese patients with hearing loss, categorized by prelingual and postlingual onset. Their multifaceted etiological analysis included an invader assay, targeted exon sequencing of 63 deafness-associated genes using massive parallel sequencing, congenital cytomegalovirus screening, and pediatric examinations.
Riggs et al. 21 assessed 330 patients, including 48 with AN, undergoing CI. They used intraoperative ECochG responses to tone bursts and developed an ECochG-Tone Response (ECochG-TR) and a “Nerve Score” based on compound action potential (CAP) and auditory nerve neurophonic responses to evaluate auditory nerve integrity.
Rodríguez-Ballesteros et al. 22 investigated 37 participants with OTOF mutations through mutation analysis and genotype-phenotype correlation. They identified four novel OTOF mutations (c.2122C>T, c.4275G>A, c.4362+2T>G, and c.5860_5862delATC).
Rosamaria et al. 23 studied a genetically defined cohort of two adults with R445H OPA1 mutations and five children with OTOF mutations. They used transtympanic ECochG to compare acoustically and electrically evoked potentials with those from 16 participants with normal hearing.
Wu et al. 24 conducted a large-scale prospective study of 743 children with sensorineural hearing impairment, including 180 with CIs. They screened for mutations in genes like GJB2, SLC26A4, mitochondrial 12S rRNA gene, and OTOF to establish correlations between specific mutations and CI outcomes.
AN-Associated Outcomes Observed
Lin et al. 18 evaluated children with AN using audiometric tests and speech audiometry, focusing on categories of auditory performance (CAP) and speech intelligibility rating (SIR). Children with mutations in OTOF, WFS1, and OPA1 had favorable CI outcomes, whereas those with cochlear nerve deficiencies had suboptimal results. The average CI-aided thresholds were 28.3 ± 7.8 dBHL, with median CAP/SIR scores of 6/4, indicating a generally positive response to CI.
McMahon et al. 19 explored the relationship between ECochG and EABR post-CI. The study identified two ECochG patterns: one with prolonged latency summating potential (SP) correlating with normal EABR, and another with normal latency SP but broad negative detectable action potential (DP) correlating with absent or poor EABR responses. Seven participants with prolonged latency SP showed normal EABR, whereas seven with normal latency SP and broad negative DP showed poor EABR.
Miyagawa et al. 20 found that patients with mutations in specific deafness genes had good CI outcomes. A causative mutation was observed in 60% of patients with prelingual-onset hearing loss and 36% of those with postlingual-onset hearing loss.
Riggs et al. 21 found that patients with AN undergoing CI had higher ECochG-TR values than non-AN CI recipients. Despite this, nerve scores indicated comparable outcomes between AN and other cohorts.
Rodríguez-Ballesteros et al. 22 reported successful auditory outcomes in 10 patients with OTOF mutations. These patients had profound hearing loss with early onset. Pure-tone audiometry and auditory brainstem responses confirmed profound hearing loss and imaging studies showed no inner ear malformations. Eleven participants displayed transient evoked otoacoustic emissions, indicating preserved cochlear function.
Rosamaria et al. 23 studied patients with OPA1 and OTOF mutations, recording normal amplitude CMs and negative polarity potentials with reduced amplitudes and prolonged duration upon CMs cancellation. Electrically evoked CAPs were recorded postimplantation in patients with OTOF mutations but not in those with OPA1 mutations, suggesting differences in electrical response patterns between these genetic variants.
Wu et al. 24 compared audiological features and allele frequencies between children with and without identified mutations, finding that children with mutations had better auditory performance post-CI. A genetic diagnosis was made in 20.6% of children with CI. Significant differences in allele frequencies were noted for GJB2 mutations between CI and non-CI children (P < .01), whereas no significant differences were found for the other genes tested.
Genetic Inferences Observed
Table 4 shows the genetic inferences observed across the included papers. Lin et al. 18 reported that the genetic examination reclassified 18 patients, mostly with variants in the OTOF gene, and other rare gene variants such as WFS1 and OPA1, which are coupled with a deficiency of the cochlear nerve and indefinite etiologies. Their study has also reported excellent outcomes from CIS in patients with OTOF variants. Two different ECochG patterns were recorded in 14 AN patients by McMahon et al. 19 , and for those patients, genetic mutations were identified in the genes of OTOF and DIAPH1 associated with the disorder. The patients with prolonged latency SP had normal EABR, while the patients with normal latency SP and broad negative DP had poor EABR.
Observed Inferences Pertaining to Genetic Associations Across the Included Trials.

Abbreviations: AN, Auditory neuropathy; CI, cochlear implant; CIS = Cochlear implant(S); CAP, categories of auditory performance; CM, cochlear microphonic; DP, detectable action potential; EABR, evoked auditory brainstem responses; ECochG, electrocochleography; ECochG-TR, ECochG-tone response; MRI, magnetic resonance imaging; NGS = Next generation sequencing; SIR, speech intelligibility rating; SP, summating potential.
Miyagawa et al. 20 reported genetic testing results that reclassified 60% of prelingual and 36% of postlingual patients, identifying several deafness genes such as GJB2, SLC26A4, CDH23, and others for etiology. Specific deafness gene mutations accounted for good and rapid development of hearing behavior post-CIS. Among them, Riggs et al. 21 reported that AN cases had higher values of ECochG-TR than the non-AN CI recipients and also stated that OTOF gene mutations were related to AN. Further, this study indicated that in cases where there was a cochlear nerve deficiency, strong auditory nerve responses were generated. Rodríguez-Ballesteros et al. 22 identified 37 patients as AN based on OTOF mutations and found that 11 new mutations were identified. The study found that the patients with OTOF mutations had profound hearing impairment but successful auditory outcomes after CIS.
Rosamaria et al. 23 repositioned two adult patients with R445H OPA1 mutations and five children with OTOF mutations to AN and concluded that OPA1 and OTOF gene mutations were associated with AN. Abnormal cochlear potentials were revealed for patients with OTOF mutations, but their language development was almost normal within 1 year after CIS. Wu et al. 24 carried out genetic testing for the reclassification of 20.6% of the patients and reported that GJB2, SLC26A4, mitochondrial 12S rRNA gene, and OTOF genes were related to AN. Patients having GJB2 mutations were reported to perform better in CAP, after CIS.
Diagnostic Approaches Employed
• Audiological and Behavioral Tests: Behavioral audiometry, encompassing speech audiometry and pure-tone thresholds, was employed in studies such as Lin et al. 18 and Rodríguez-Ballesteros et al. 22 to assess the function of the auditory pathway and identify inconsistencies between the perception of speech and hearing threshold. These inconsistencies often presented themselves as early signs of AN. To complement these approaches, the auditory performance tests CAP and SIR were utilized to assess the functional impact of AN on the capacity for audition, offering a further critical tool for evaluating the efficacy of CI.
• Electrophysiological Testing: The use of electrophysiological methods, most specifically ECochG and electrically EABR, served to separate presynaptic and postsynaptic AN. Presynaptic AN had a prolongation of latency or SP in ECochG while the neural response was still normal in EABR. Postsynaptic AN, in contrast, showed abnormal or absent neural responses in EABR with normal CM potentials in ECochG. McMahon et al. 19 and Riggs et al. 21 used extensive uses of ECochG and EABR in defining AN’s integrity and confirming the presence of lesions, making such a differentiation between two varieties of AN possible with sharp precision.
• Imaging Studies: Imaging methods, namely CT and MRI, also played a role in delineating anatomical abnormalities contributing to AN. Lin et al. 18 and Riggs et al. 21 reported cochlear nerve aplasia and inner ear dysmorphia as significant contributors to postsynaptic AN. Imaging was crucial if the postsynaptic defect was suspected, helping to demonstrate cochlear nerve aplasia or hypoplasia, which plays a determinant role in CI outcome and prognosis.
Causes of Presynaptic and Postsynaptic AN Observed
• Presynaptic AN: The etiology of presynaptic AN was primarily due to genetic mutations and dysfunction at the inner hair cell level or its synapse with the auditory nerve. Most frequent causes were the mutations of the OTOF gene, encoding otoferlin, which lead to deep hearing loss without neural response recovery post-CI. Other associated genes were GJB2 (gap junction protein) and SLC26A4 (pendrin). Besides genetic etiology, cochlear dysfunction at the synaptic level was reported as one common feature of presynaptic AN. Congenital cytomegalovirus (CMV) infection also contributed to presynaptic AN, with viral damage affecting cochlear function, as noted in studies like Miyagawa et al. 20
• Postsynaptic AN: Postsynaptic AN was mainly due to structural abnormalities, such as hypoplasia or aplasia of the cochlear nerve, which were confirmed through imaging studies. These deficits disrupted the transmission of auditory signals from the cochlea to higher auditory centers. Genetic mutations, although less common, were also involved, such as mutations in OPA1, which led to mitochondrial dysfunction and damaged auditory nerve function, according to a report by Rosamaria et al. 23 At times, the postsynaptic AN was associated with more general neurodegenerative or neuropathic conditions that were affecting the auditory nerve pathways. The study indicates that there is no easy management for postsynaptic AN, mainly because of its complex etiology and the variable outcomes with CI.
Preoperative Audiological/Electrophysiological Testing and Homogeneity
Preoperative audiological and electrophysiological testing were often different in the studies, with considerable heterogeneity among the methods used for assessment. Lin et al. 18 carried out serial behavioral and speech audiometry, while McMahon et al. 19 made use of frequency-specific ECochG and electrically EABR. Rodríguez-Ballesteros et al. 22 used pure-tone audiometry and auditory brainstem responses, while Rosamaria et al. 23 used CMs and transtympanic ECochG. Miyagawa et al. 20 focused on genetic testing rather than direct audiological tests, and Wu et al. 24 compared the audiological characteristics without mentioning electrophysiological testing. These different approaches showed that the preoperative assessments had no homogeneity due to the fact that studies covered all types of assessments-behavioral, electrophysiological, and genetic.
CI Outcome Evaluation
CI results were assessed through different research methodologies from the different studies. Lin et al. 18 reported on outcomes using CAP and SIR scores, in addition to CI-aided thresholds. McMahon et al. 19 associated outcomes with ECochG and EABR response patterns postimplantation, but Rodríguez-Ballesteros et al. 22 noted effective auditory performance without providing specific measures. Rosamaria et al. 23 assessed outcomes by electrically evoked compound action potentials (e-CAPs), while Miyagawa et al. 20 based their inference of outcomes on genetic correlations rather than standardized audiometric measures. Riggs et al. 21 evaluated their outcomes by intraoperative ECochG-TR and nerve scores, and Wu et al. 24 evaluated their auditory performance by CAP scores 3 years postimplantation. The timeline and measurements to assess outcomes were not uniform; many studies would state the time to follow up, for example, Wu et al. 24 while others did not.
GRADE Assessment Observations
As shown in Table 5, the studies reviewed indicate that understanding the etiology, particularly genetic factors and lesion sites, is essential for forecasting CI outcomes in patients with AN. The potential for bias was judged to be low to moderate, indicating careful study design and reporting. The studies displayed high uniformity and direct relevance to the clinical questions, with precise outcomes contributing to the reliability of the conclusions. No additional factors that could diminish the confidence in the evidence were identified, making the trustworthiness of the collected evidence moderate.
GRADE Observations Pertaining to the Included Studies.
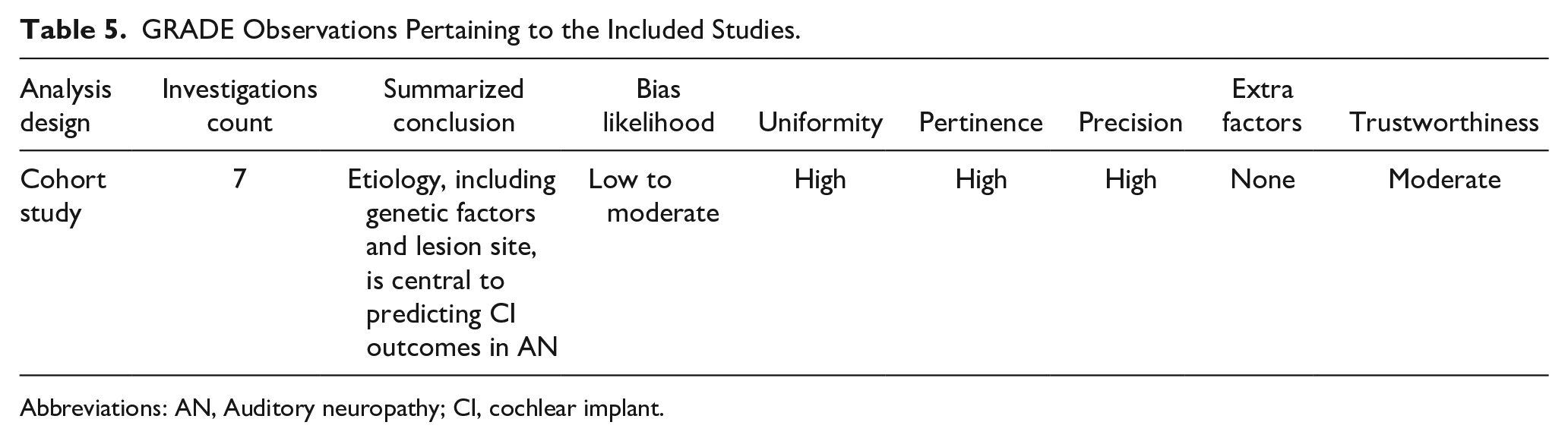
Abbreviations: AN, Auditory neuropathy; CI, cochlear implant.
Discussion
The collective findings from these studies underscore the critical role that etiology plays in predicting CI outcomes for individuals with AN and genetic mutations. Despite varying focal points and methodologies, these studies provide insights into the factors predicting CI success. The results on the influence of etiological factors on CI outcomes are consistent, although the degree of similarity varies. Lin et al. 18 emphasized the critical nature of identifying etiology in patients with AN, whereas Riggs et al. 21 highlighted the relevance of hair cell activity in clinical AN assessment. Both studies agree that understanding the underlying cause of hearing loss is crucial for anticipating CI success. McMahon et al. 19 and Rosamaria et al. 23 focused on electrophysiological diagnostics to elucidate the pathophysiology of hearing loss. Miyagawa et al. 20 and Wu et al. 24 have highlighted the role of genetics. Wu et al. quantified the correlation between genetic characteristics, specifically GJB2 mutations, and improved auditory performance post-CI, underscoring the prognostic value of genetic testing in CI management. Rodríguez-Ballesteros et al. 22 and Rosamaria et al. 23 identified OTOF mutations as beneficial for CI outcomes, suggesting that favorable genetic underpinnings can enhance CI efficacy. Collectively, these studies provide a multifaceted view of the complex interplay between genetics, electrophysiology, and clinical assessment in determining the efficacy of CIs.
Many studies have evaluated the potential benefits of cochlear implantation for individuals with AN. Initially, AN was considered a potential contraindication for CI, 8 but subsequent studies have documented successful outcomes.25,26 Some studies have reported suboptimal CI outcomes in individuals with AN compared with typical CI candidates, though these individuals often experience auditory enhancement postimplantation.27,28 However, results have varied, leading to inconsistent conclusions about CI efficacy in patients with AN.29,30 A limitation of these studies is the lack of precise classification of the lesion site within the auditory pathway, leading to heterogeneous study populations and potentially confounding outcomes.
Complications in assessing AN are exacerbated by the decline of OAEs over time in 20–30% of affected individuals, not limited to mutations in the OTOF gene 31 but also observed in other genetic forms of auditory synaptopathy and neuropathy.15,31 This decline compromises the diagnostic utility of OAEs, particularly in non-congenital or adult-onset cases. CMs have been proposed as an alternative diagnostic tool but are not widely used in screening protocols. This finding highlights the growing importance of genetic testing in evaluating candidates for CI.
Further investigation is needed to understand the implications of genetic mutations in the auditory cortex. Genes such as DFNB59, CACNA1D, and KCNQ4, which affect the midbrain and auditory cortex, 32 and genes such as PCDH15 and CDH23, essential for hair cell tip link formation, are necessary for interneuronal development in the auditory cortex. 33 Further studies should explore whether mutations in these genes influence CI outcomes through their impact on central auditory pathways.
Currently, insights from studies on CIs and AN do not fundamentally alter clinical practices for CI surgeons. Nevertheless, as CI technology and molecular treatments advance, understanding lesion locations within the auditory system will become increasingly critical. CI surgeons may integrate knowledge of genetic and other lesions affecting the auditory nerve into patient consultations regarding anticipated CI outcomes. Despite ongoing research, CI remains the recommended intervention for patients with AN mutations because of the overall positive auditory improvements observed. Genetic testing should be included in evaluating deafness, especially in pediatric cases, following contemporary guidelines. 34 A comprehensive multigene panel is necessary to identify mutations affecting the auditory nerve because relevant genes are often excluded from single-gene tests. 35 Clinicians should recognize that the lesion site is one of multiple factors influencing CI outcomes, as evidenced by considerable variability observed even among siblings sharing the same AN-related mutation.36,37
Raza et al. 38 conducted a literature review to identify pre- and postoperative predictors of CI success in children with AN. Their findings align with the objectives of our study that compared the CI efficacy in presynaptic versus postsynaptic neuropathy, emphasizing the importance of prognostic indicators. Shearer et al. 9 focused on the genetic aspects of AN, advocating for a molecular classification (synaptopathy vs. neuropathy), which resonates with our systematic review goals. They found that individuals with primarily presynaptic lesions have better CI outcomes, consistent with our findings.
Chaudhry et al. 39 reviewed outcomes following CI in patients with postsynaptic AN, providing a narrative synthesis of available literature. Their findings of generally positive post-CI outcomes in postsynaptic AN cases offer specific insight into the subset of patients with AN covered in our study. Rance et al. 3 reviewed the clinical and pathophysiological features of AN, distinguishing between presynaptic, postsynaptic, and central neural pathway disorders. Their diagnostic criteria were fundamental to our systematic review, supporting our goal of determining how the lesion site influences CI outcomes.
Conclusion
AN etiology critically impacts implant success. Retrospective analysis showed that CI performance depends on accurately characterizing neuropathy as either presynaptic or postsynaptic. Genetic etiologies, particularly mutations affecting synaptic transmission, have significant prognostic value. Patients with presynaptic mutations, such as those in the OTOF gene, generally show better outcomes post-CI than those with postsynaptic pathologies. This finding emphasizes the need for genetic profiling in preoperative evaluations to predict CI efficacy.
Electrophysiological assessments are crucial for distinguishing between pre- and postsynaptic lesions. These diagnostic measures provide insight into the functional status of the auditory pathway, thereby facilitating a more accurate prognosis of CI outcomes. The convergence of genetic and electrophysiological data strengthens clinical approaches, enabling a more tailored selection of candidates likely to benefit from cochlear implantation.
Footnotes
Authors’ Note
This work has not been presented previously.
Data Availability
The data used to support the findings of this study are included within the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting systemic review.
Statement of Informed Consent
As this study is a systematic review of previously published literature, informed consent is not required.
