Abstract
Guillain–Barre syndrome (GBS) is the most prevalent and severe form of acute paralytic neuropathy, commonly triggered by infections and characterized by an abnormal autoimmune response. Reports of multispace deep fascial infection (DFI) in the head and neck complicated by GBS are exceedingly rare. We report a 69-year-old woman with DFI who developed postoperative limbs weakness. GBS was diagnosed after neurology consultation and cerebrospinal fluid (CSF) puncture examination. Then the patient was treated with intravenous immunoglobulin, as well as supportive care and rehabilitation exercise. After our treatment, the patient was discharged successfully. GBS is a rare complication after head and neck surgery. When progressive weakness occurs in patient of DFI, the possibility of GBS should be considered, and CSF and electrophysiological examinations should be performed as soon as possible. For patients with DFI complicated with GBS, prognosis depends on early diagnosis and treatment.
Keywords
Introduction
Guillain–Barre syndrome (GBS) is an immune-mediated disorder in which the peripheral nervous system is attacked by the body’s own immune system. Globally, the incidence of GBS is estimated at 1.12 per 100,000 annually. 1 The condition often follows a respiratory or gastrointestinal viral infection. 2 While the prognosis for most GBS patients is generally favorable, mortality rates range between 4% and 5%, and this rises to 15% to 30% in patients requiring mechanical ventilation. Moreover, approximately 20% of patients may experience long-term severe disabilities.3,4
In around 60% of cases, GBS is preceded by an infection. 5 Recently, there has been a growing body of literature documenting cases of GBS following surgeries, even in the absence of prior infection, with approximately 5% of GBS cases linked to surgical procedures. 6 GBS has been observed following orthopedic, gastrointestinal, neurosurgical, gynecologic, and thoracic surgeries.6,7 However, GBS occurring after neck surgery remains exceedingly rare, and to the best of our knowledge, the present case is the first to be documented in the literature.
Case Report
A 69-year-old female patient was admitted to our hospital presenting with a 10-day history of sore throat and cervical swelling. On physical examination, acute hyperemia and edema of the pharyngeal mucosa were noted, along with swelling of the left oropharyngeal wall, narrowing of the oropharyngeal cavity, and mucosal edema of the epiglottis. Swelling was also observed in the left submaxillary region and anterior neck, along with inspiratory trifossa sign. Enhanced computed tomography (CT) scans of the neck and chest revealed multiple abscesses in the submandibular, para-pharyngeal, retropharyngeal, carotid spaces, and the superior mediastinum (Figure 1a-d). Laboratory tests indicated a total leukocyte count of 9.43 × 109/L, a neutrophil percentage of 87.9%, elevated C-reactive protein (391.66 mg/L), and procalcitonin levels of 2.85 ng/mL. The patient had a history of hypertension and type 2 diabetes. An emergency surgical intervention involving extensive incision and drainage of the neck and superior mediastinum abscesses, along with a tracheotomy, was performed. Postoperatively, the patient was treated in the ICU with nasogastric tube placement, antibiotic therapy, ventilator support, and lavage of the abscess cavities. A follow-up CT scan showed a reduction in the size of the abscesses, and infection markers had improved. However, the patient developed a persistent fever, with a maximum recorded temperature exceeding 40°C. Following multidisciplinary treatment (MDT), the patient’s body temperature gradually normalized. One week postsurgery, the patient was transferred back to our department for further care.
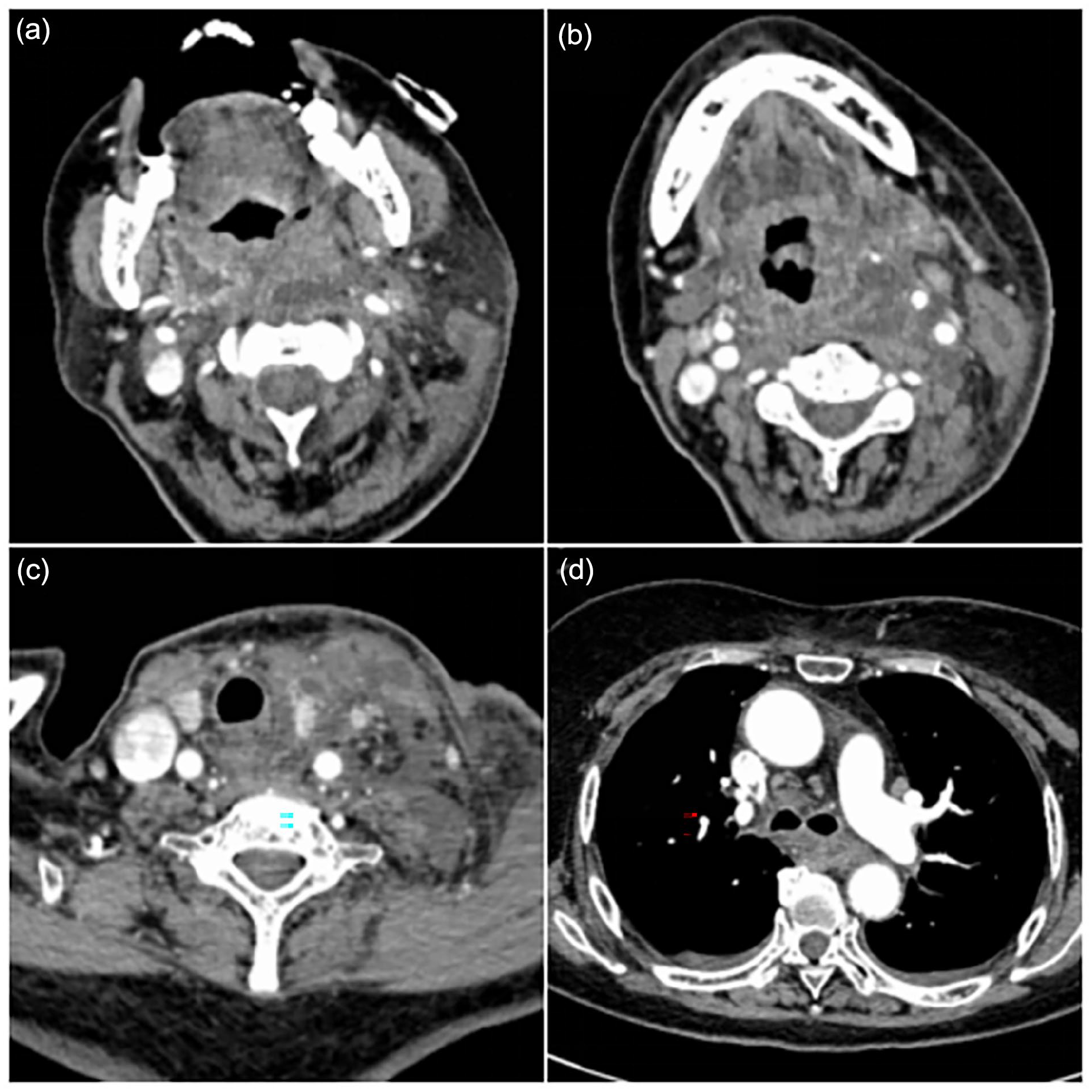
(a) Enhanced CT of oropharynx. (b) Enhanced CT of epiglottis. (c) Enhanced CT of thyroid. (d) Enhanced CT of superior mediastinum. CT, computed tomography.
Upon the patient’s return to our department, a detailed physical examination revealed profound weakness in all 4 limbs (muscle strength grade 0). Laboratory tests showed that the patient’s myoglobin levels exceeded 1000 ng/mL. Following rehydration, urinary alkalization, diuretic therapy, and myoglobin levels returned to normal; however, limb weakness persisted. Computed tomography angiography of the head revealed lacunar cerebral infarctions, while electromyography indicated peripheral nerve damage, primarily axonal in nature. A lumbar puncture was performed, and cerebrospinal fluid (CSF) analysis demonstrated albuminocytologic dissociation. These clinical findings, combined with the CSF and electrophysiological results, fulfilled the diagnostic criteria for GBS. The patient was treated with intravenous immunoglobulin (0.4 g/kg/day for 5 days). Following this treatment regimen, gradual improvement in muscle strength was observed, with strength improving to grade 3.
Despite this progress, the patient’s swallowing function did not recover, necessitating transfer to the rehabilitation department for intensive therapy. After 24 days of rehabilitation, the patient regained the ability to walk independently, and the nasogastric tube was successfully removed. Following a total treatment duration of 73 days, the patient made a full recovery and was discharged.
Discussion
GBS is widely regarded as an autoimmune disorder, 8 though its precise etiology remains elusive. While the majority of GBS cases are postinfectious, other factors, such as vaccination, 9 surgery, 10 trauma, 11 and kidney transplantation, 4 have been reported as triggers. Campylobacter jejuni is the most frequently implicated pathogen, but other agents, including Epstein–Barr virus, cytomegalovirus, hepatitis E virus, Mycoplasma pneumoniae, Haemophilus influenzae, influenza A virus, and Zika virus, have also been associated with GBS. In this case, the etiology could be attributed to both the underlying infection and the surgical intervention. However, cultures from the patient’s abscesses revealed Streptococcus constellatus, an uncommon pathogen in GBS. Given this finding, we postulate that the development of GBS in this patient was primarily postoperative in origin. The persistent high fever observed during the patient’s ICU stay may have been an exaggerated immune response. The ICU team noted limb weakness following the discontinuation of mechanical ventilation but initially attributed it to ICU-acquired weakness or rhabdomyolysis secondary to severe infection, delaying further diagnostic evaluation.
While surgery may elevate the risk of developing GBS, the underlying pathological mechanisms remain unclear. The prevailing hypotheses suggest an immune-mediated process triggered by perioperative infection or stress. One of the more straightforward theories posits a nonspecific mechanism, where surgical stress induces a systemic immune response that targets the myelin sheath of peripheral nerves. 12 In this particular case, the patient’s type 2 diabetes, severe infection, and surgical shock likely contributed to a state of immunosuppression. A preceding viral infection may have further activated the immune system, culminating in an exaggerated response following surgical stress.
Clinically, GBS typically manifests with progressive weakness, dysphagia, reduced or absent deep tendon reflexes, sensory disturbances, and in severe cases, respiratory failure. Patients often present with symptoms of hand and foot weakness, and then gradually develop closer to the trunk. It can also show symptoms such as difficulty raising head and reduced expression. When it involves the innervated bulbar nerve muscles, there is difficulty in eating or drinking water, and in severe cases, it involves the respiratory muscles, resulting in difficulty in breathing and weakness. Some patients also have sensory nerve symptoms, such as reduced perception of painful stimuli or touch, and appear to feel like wearing gloves and socks. Some patients show symptoms of autonomic nervous dysfunction, such as flushed face, blood pressure changes, and other symptoms. Although the clinical manifestations of GSB are varied, the diagnostic criteria can be as follows, as outlined in the 2014 guidelines, include (1) progressive weakness in the limbs, often accompanied by are flexia or reduced tendon reflexes; (2) characteristic findings on electromyography; (3) albuminocytologic dissociation in CSF analysis; and (4) exclusion of other potential neuropathic causes.
Numerous studies and extensive clinical experience have demonstrated the efficacy of intravenous immunoglobulin (IVIG) and plasma exchange (PE) in treating patients with GBS. Both IVIG and PE are considered standard treatments and should be administered as early as possible, 13 ideally before irreversible axonal damage occurs, as was the case with our patient. Emerging immunotherapies, targeting the specific pathological processes of GBS, offer promising new treatment avenues. When combined with conventional therapies, these novel approaches could improve outcomes in patients who do not respond adequately to traditional treatments. 14 However, many of these therapies have only been tested in animal models, and the complement inhibitor eculizumab is still undergoing clinical trials.15,16
Although the overall prognosis for GBS is generally favorable, mortality remains around 5%, with some patients left with residual neurological dysfunction. Poor prognostic factors include advanced age, antecedent C. jejuni infection, the need for mechanical ventilation, and the axonal subtype of GBS. 13 In this case, our patient responded well to immunoglobulin therapy and experienced no lasting functional impairment.
Conclusion
In conclusion, this case highlights the potential for GBS to develop following multispace deep fascial infections (DFI) in the head and neck. When patients with DFI present with progressive weakness, GBS should be considered in the differential diagnosis. Early diagnosis is critical, as GBS can be life-threatening, and timely CSF and electrophysiological evaluations are essential for confirming the diagnosis. IVIG and PE remain the most effective treatments and should be initiated promptly. Patients with DFI complicated by GBS often face prolonged treatment courses, poorer outcomes, and higher healthcare costs. Therefore, a MDT approach is vital, and thorough communication with the patient’s family is crucial for optimal care.
Footnotes
Authors’ Note
All authors agree that the receipt of letters, reprint requests, and proofs will be sent by Wanjie Luo (15183506612,
Data Availability Statement
All relevant data are within the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The research has obtained written informed consent from the patient and was approved by the Ethics Committee of the Zigong Fourth People's Hospital.
