Abstract
Chronic cough stands as a persistent and perplexing health challenge, characterized by its prolonged duration over 8 weeks and tenacious nature. 1 Globally, the prevalence of chronic cough ranges widely, with estimates suggesting a prevalence of 2% to 18% in the general population, varying by age, smoking status, and environmental factors. 2 Chronic cough carries a wide array of etiologies and have different terminologies in the literature, ranging from upper airway cough syndrome (UACS), irritable larynx syndrome, asthma including cough variant asthma, eosinophilic bronchitis, and gastroesophageal reflux syndrome. 3 Chronic cough in the realm of otolaryngology is often attributable to the reverberation of inflammation within sinonasal regions, which may incite afferent pathways through direct stimulation of cough receptors or indirectly by postnasal drip. 4 The inflammation serves as a central amplifier in the pathophysiology of cough, increasing the susceptibility of the cough reflex arc, which in turn perpetuates the chronicity of symptomatology. Current treatment modalities encompass a range of therapeutic options, from empirical treatment targeting potential causes to symptomatic therapy.
Intranasal corticosteroids (INCS) have emerged as a cornerstone in the management of sinonasal inflammation including allergic and nonallergic rhinitis, acute rhinosinusitis, and chronic rhinosinusitis with and without nasal polyposis, with their ostensible utility in chronic cough being attributed to their anti-inflammatory properties. 5 In fact, many guidelines and consensus statements suggest the use of INCS in the treatment of chronic cough secondary to UACS.6-9 However, it is unclear whether these recommendations are based on robust evidence with comparative observational studies or randomized controlled trials. This perspective aimed to assess the current landscape of INCS use for chronic cough, exploring the gap between clinical practice and the supporting evidence.
Despite an extensive and methodologically-robust systematic search across multiple databases and the application of comprehensive inclusion criteria aimed at capturing a wide range of studies, our review process has not yielded any studies meeting our predefined standards for eligibility (Figure 1 and APPENDIX 1). This lack of evidence underscores a crucial void in our clinical understanding and presents a stark contrast to the observed prevalence of chronic cough and its acknowledged impact on patient quality of life. The absence of eligible studies is particularly notable given the widespread recommendation of INCS in globally-used guidelines and consensus statements.6-9 Exploring the implications of an absence of evidence in the context of INCS use for chronic cough reveals a multifaceted issue within the literature and health care practice. While clinicians may be observing benefits in practice, this anecdotal evidence has not translated into structured research needed to inform evidence-based guidelines.
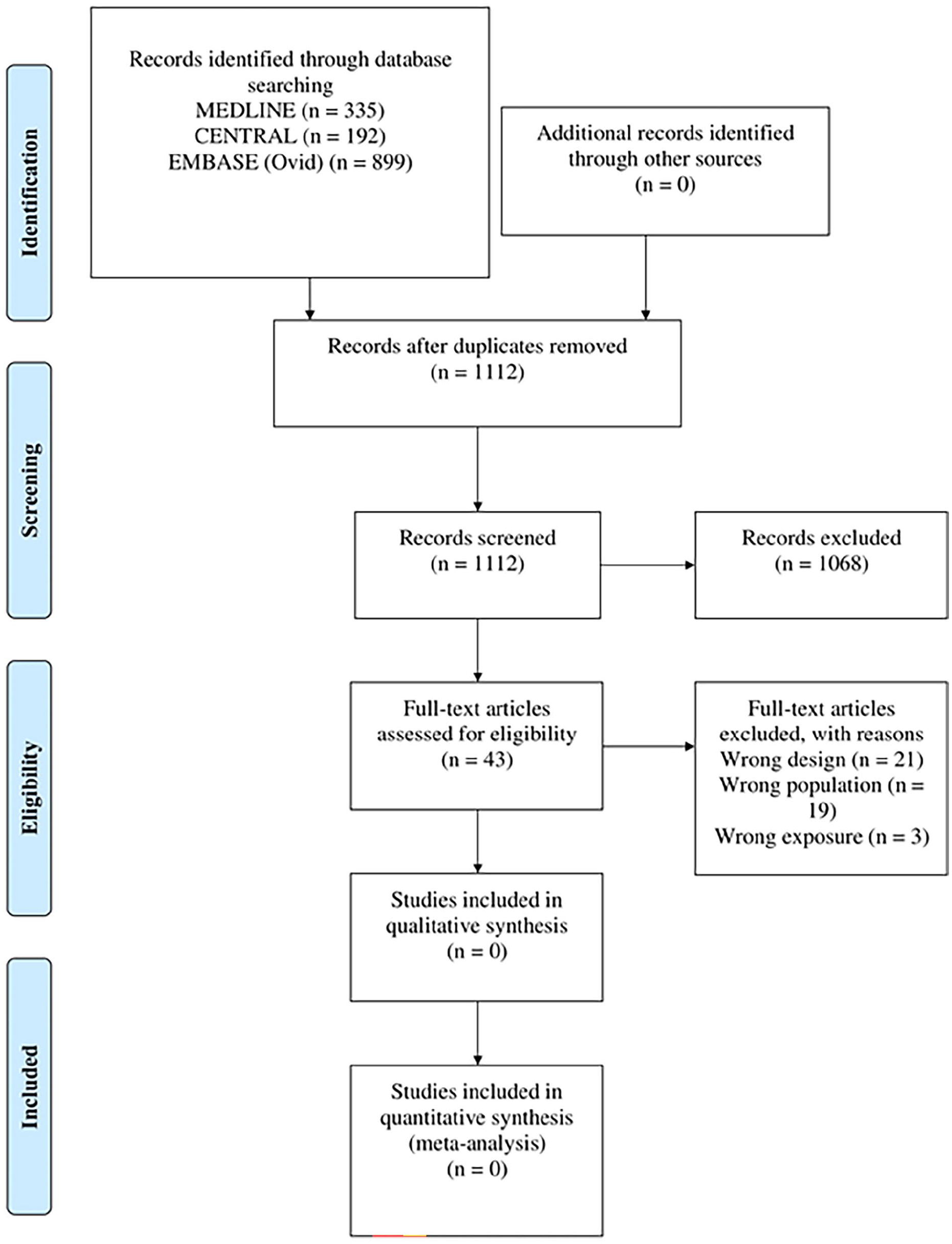
PRISMA flow diagram detailing the flow of records through the review process.
Published guidelines provide a comprehensive view of the management strategies for chronic cough. The World Allergy Organization-Allergic Rhinitis and its Impact on Asthma (WAO-ARIA) consensus on chronic cough recommends the use of INCS primarily for conditions such as AR where they have shown benefits. The guidelines highlight the necessity of tailored management strategies, especially in light of recent updates during the COVID-19 pandemic. 6 The revised Korean Cough Guidelines similarly endorse INCS for cough associated with AR, reflecting consistent observations in clinical practice. 7 The American College of Clinical Pharmacy evidence-based clinical practice guidelines by Pratter recommends INCS as part of the treatment regimen for chronic UACS secondary to rhinosinus diseases, previously referred to as postnasal drip syndrome. 8 Guidelines published in the American Family Physician also support the use of INCS for cough related to upper airway conditions, aligning with other guidelines’ recommendations. 9 These guidelines essentially recommend the same approach, despite the scarcity of robust evidence on the management of stand-alone chronic cough. Furthermore, the discrepancy might be indicative of research challenges specific to the field, such as difficulties in standardizing patient groups or measured outcomes for chronic cough, given its diverse etiologies and manifestations, as well as securing funding for studies on already widely-used treatments. 10
The dearth of robust evidence on the therapeutic role of INCS in chronic cough management has significant ramifications for clinical practice. Clinicians are often faced with the challenge of making treatment decisions in the absence of high-quality evidence, relying instead on clinical experience or extrapolations from studies with different focuses. This scenario is not unique to chronic cough; indeed, the broader field of cough has been highlighted as one where clinical practice often outpaces the evidence base, leaving practitioners to make decisions based on consensus guidelines that may lack rigorous scientific corroboration. The potential consequences of such a practice include variability in treatment outcomes and patient care, an increased likelihood of trial-and-error approaches, and a possible increase in the use of treatments with unproven efficacy. By delineating the current state of the literature, or rather the absence thereof, this perspective acts as an impetus for future studies. It underscores the need for well-designed clinical trials to evaluate the therapeutic potential of INCS in chronic cough—a field where such data are not just a scholarly void but a clinical necessity.
Footnotes
Appendix 1
Acknowledgements
None.
Author Contributions
S.B.: Literature search, data extraction, and writing and drafting the manuscript; D.J.L.: Data extraction, writing and drafting the manuscript, and critical revision; T.C.: Critical revision; O.K.: Data extraction; T.K.: Literature search; C.S.: Critical revision; R.J.L.: Conception and design and ritical revision; Y.C.: Conception and design, critical revision, and supervision.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability Statement
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
Not applicable.
