Abstract
Introduction
The rhinologist is frequently confronted with the finding of mucosal contact (ie, contact between the septum and any turbinate) on endoscopic nasal exam. Determining the clinical significance of this finding can be challenging. In particular, the relationship between intranasal mucosal contact (MC) and headache is controversial. Mucosal contact headache (MCH) is a secondary headache disorder in the International Classification of Headache Disorders (ICHD-2), where it is described as “intermittent pain localized in the periorbital and medial canthal or temporozygomatic regions, associated with evidence of MC points by nasal endoscopy or computed tomography imaging.” 1 The International Headache Society does not recognize MC as a cause of migraine, but acknowledges that other disorders may cause migraine symptoms (see Table 1). In addition, several reports document dramatic improvement in migraine symptoms with surgical alleviation of MC.2-4 However, literature challenging the concept of MCH includes a CT study finding no association between headache and the presence of MC radiographically, 5 an experimental study on 10 volunteers noting the absence of significant pain or referred headache upon stimulation of various sites within the nasal cavity, 6 and a study comparing the prevalence of MC on diagnostic nasal endoscopy in patients with and without facial pain. 7 This last study by Abu-Bakra and Jones reported the prevalence of MC on endoscopy at 4% in a cohort of 973 patients and failed to find any correlation between MC and facial pain. However, there are noteworthy shortcomings of this study, including the lack of demographic data on the patient population, the relative absence of migraine in the population studied, the absence of data on the superior turbinate (ST), the exclusion of patients with septal deviation in the absence of rhinosinusitis, and the requirement of “blanching” of the nasal mucosa in the area of contact as an inclusion criterion. Several authors have reported MCH in patients that challenge these criteria,8-10 including the significance of the ST in MCH,2,10,11 which Abu-Bakra and Jones did not assess in their article.
ICHD-2 Diagnostic Criteria for Migraine Without Aura.

Abbreviation: ICHD-2, International Classification of Headache Disorders.
The purposes of this study are to determine the prevalence of MC using more relevant criteria, and to explore the correlation, if any, between MC and migraine.
Materials and Methods
This study was approved by the Commonwealth Health System-Wilkes-Barre General Hospital’s Ethics Committee/Investigational Review Board, Wilkes-Barre, PA.
Patient Selection
Patients in the primary author’s general otorhinolaryngology practice requiring upper airway endoscopy were evaluated. Inclusion criteria included any indication for upper airway endoscopy, such as symptoms of chronic rhinosinusitis and/or allergic rhinitis, headache/facial pain, dysphonia, dysphagia, and symptoms of laryngopharyngeal reflux. Exclusion criteria included history of previous endoscopic sinus surgery and/or sinonasal polyposis, acute viral upper respiratory infection within the past 30 days, and rhinitis medicamentosa. Three hundred fifty-five consecutive patients meeting these criteria constituted the study population.
Endoscopic Examination
Three hundred seven patients underwent rigid nasal endoscopy and 48 underwent flexible fiberoptic nasolaryngoscopy. All endoscopies were performed after topically decongesting and anesthetizing the nasal cavity with a solution of pseudoephedrine 1% and lidocaine hydrochloride 4% and allowing a wait time of 5 to 10 minutes. The spray was directed parallel to the floor of nose/inferior turbinate as well as superiorly toward the agger nasi. In some patients, inserting an oxymetazoline and lidocaine soaked cotton pledget between the septum and middle turbinate was necessary to achieve better visualization. All rigid endoscopies were performed initially with a Karl Storz 4 mm 30° telescope (Karl Storz Endoscopy-America, Inc., El Segundo, California, USA). All flexible endoscopies were performed with a Karl Storz 3.7 mm rhino-pharyngo-laryngoscope. When the STs could not be visualized by these methods, a Karl Storz 2.7 mm scope 30° scope was used, along with gentle lateralization of the middle turbinate with a Freer elevator, if necessary. All nasal endoscopic examinations were performed in the classic three-pass manner as described by Kennedy. 12 If the ST could not be seen in this manner, the scope was advanced just below and parallel to the nasal dorsum and turned upside down, so that the 30° lens was directed downward, and slid between the septum and agger nasi/middle turbinate.
Migraine Assessment
Headache history was obtained at the time of office evaluation. Patients were queried about their lifetime history of migraine by reading them the ICHD-2 definition of migraine (the ICHD-3 definition had not been published at the time of this study) and recording their verbal response to each item in the criteria. No distinction was made between migraine with aura and migraine without aura. Presence of allergy and/or mood disorder (anxiety, depression, etc.) was deduced retrospectively from the use of medications.
Mucosal Contact
MC was defined as any degree of contact between the septum and any turbinate. Data on whether this MC was due to septal anomalies (deviations, spurs) or turbinate variations (hypertrophy, concha bullosa) were considered irrelevant, since these anatomical variations may exist without MC. The turbinate(s) involved in MC with the septum was noted.
Data Acquisition and Analysis
Data were entered and analyzed in Excel format. Subsequently, the binary variable of migraine was assessed utilizing logistic regression incorporating the factors: age, race, gender, hypertension, use of tobacco, allergy, depression, MC, and body mass index. Multivariate regression was then performed using a backward stepwise approach to iteratively remove variables that do not significantly contribute to the model at a P < .100 cutoff. The alpha level was set at P < .05. The odds ratios of the regressions are reported. All statistical analyses were completed in R Studio version R 3.3.3 (GNU Operating System, Free Software Foundation, Boston, MA, USA).
Results
Demographics
Three hundred forty-nine of 355 (98.3%) patients were Caucasian, 3 (.85%) African American, 2 (.57%) Asian, and 1 (.03%) Hispanic, reflecting the demographics of the authors’ geographic area. There were 131 males (37%) and 224 females (63%) for a male:female ratio of 1:1.7. Age ranged from 15 to 93 with a median of 51.1 years.
Mucosal Contact
The overall prevalence of MC was 49.3% (175/355). This group was composed of 58 males and 117 females (male:female ratio of 1:2). Ages ranged from 15 to 90 with a median of 49.6 years. The largest group consisted of those with ST MC (n = 124; 34.9%), followed by those with inferior turbinate (IT) MC (n = 52; 14.6%), and middle turbinate (MT) MC (n = 50; 14.1%). Mucosal contact involving multiple turbinates was seen in 48 patients (13.5%; see Figure 1).
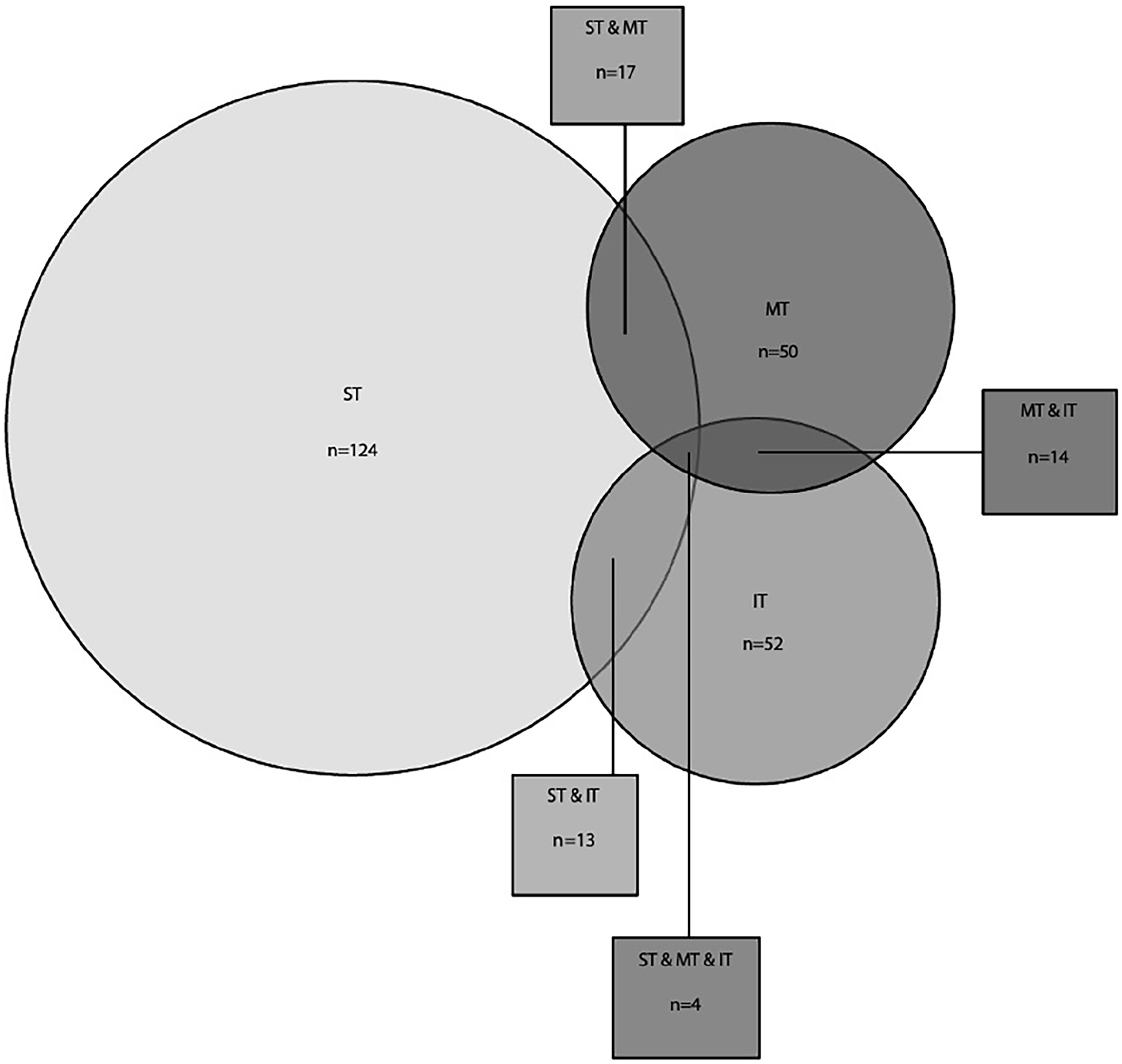
Venn diagram showing number of patients with mucosal contact by turbinate level. IT, inferior turbinate, MT, middle turbinate, ST, superior turbinate.
Migraine
Migraine history was elicited in 113 subjects (31.8%). This group consisted of 25 males and 88 females for a male:female ratio of 1:3.5. Ages ranged from 17 to 89 with a median of 47.5 years.
Correlation Between MC and Migraine
History of migraine was elicited in 91 of the 175 patients with MC (52%). These consisted of 17 males and 74 females (ratio 1:4.4), ages 17 to 90 with a mean of 48.7 years. This includes 81 of 124 patients with ST MC (65.3%), 25 of 50 patients with MT MC (50%), 12 of 52 patients with IT MC (21.1%); 24 of 48 patients had multi-turbinate MC (50%). History of migraine was elicited in 22 of the 180 patients without MC (12.2%; see Figure 2). These consisted of 8 males and 14 females (ratio 1:1.75), ages 18 to 67 with a mean of 43.3 years. Mucosal contact was visualized in 91 of 113 patients with migraine (80.5%) and in 84 of 242 patients without migraine (34.7%). This included 81 of 113 patients with ST MC (71.7%), 25 of 113 patients with MT MC (22.1%), 12 of 113 with IT MC (10.6%); 24 patients had multi-turbinate MC (21.2%; see Figure 3).
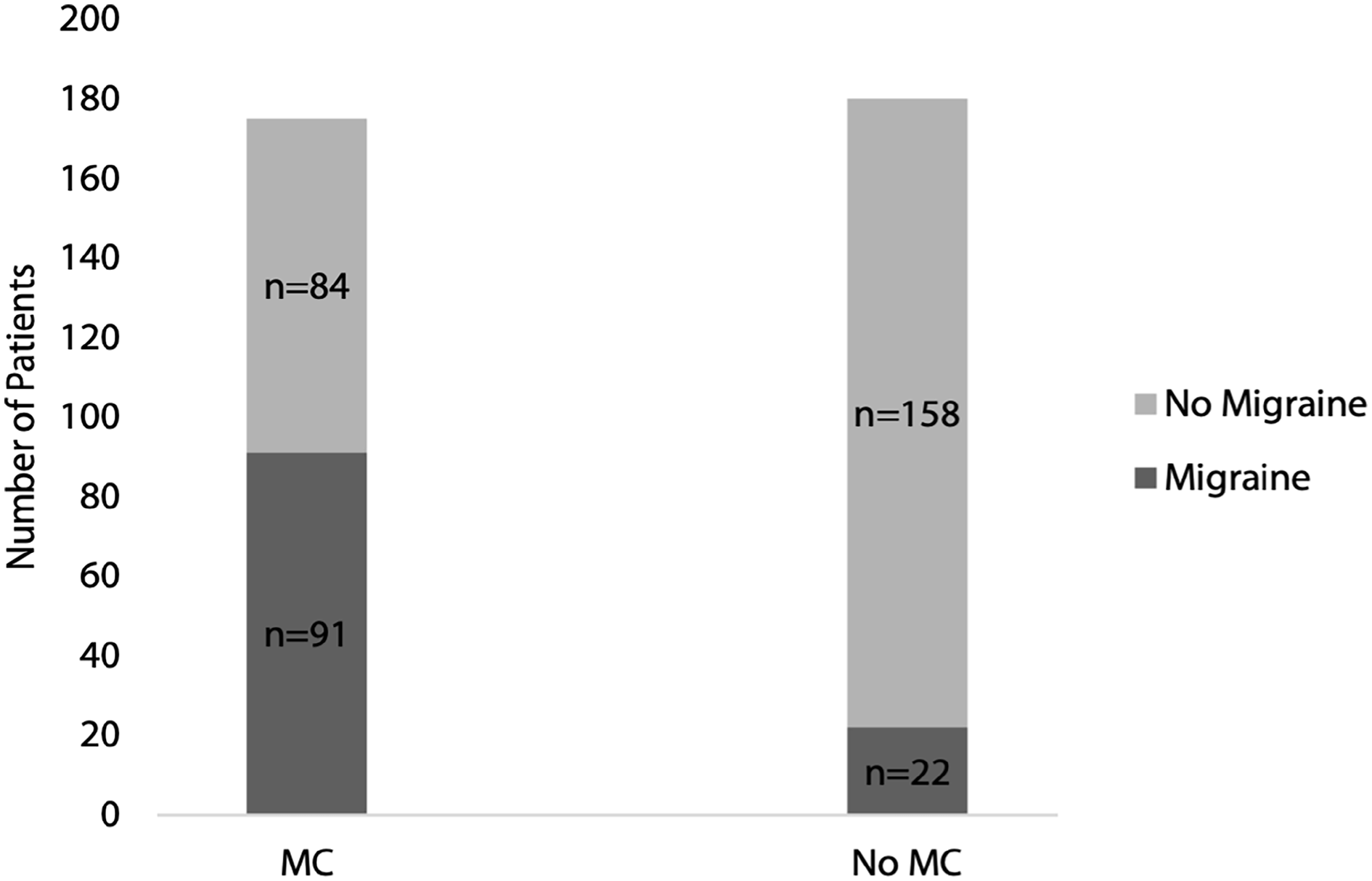
Bar graph showing presence or absence of migraine in patients with and without mucosal contact.
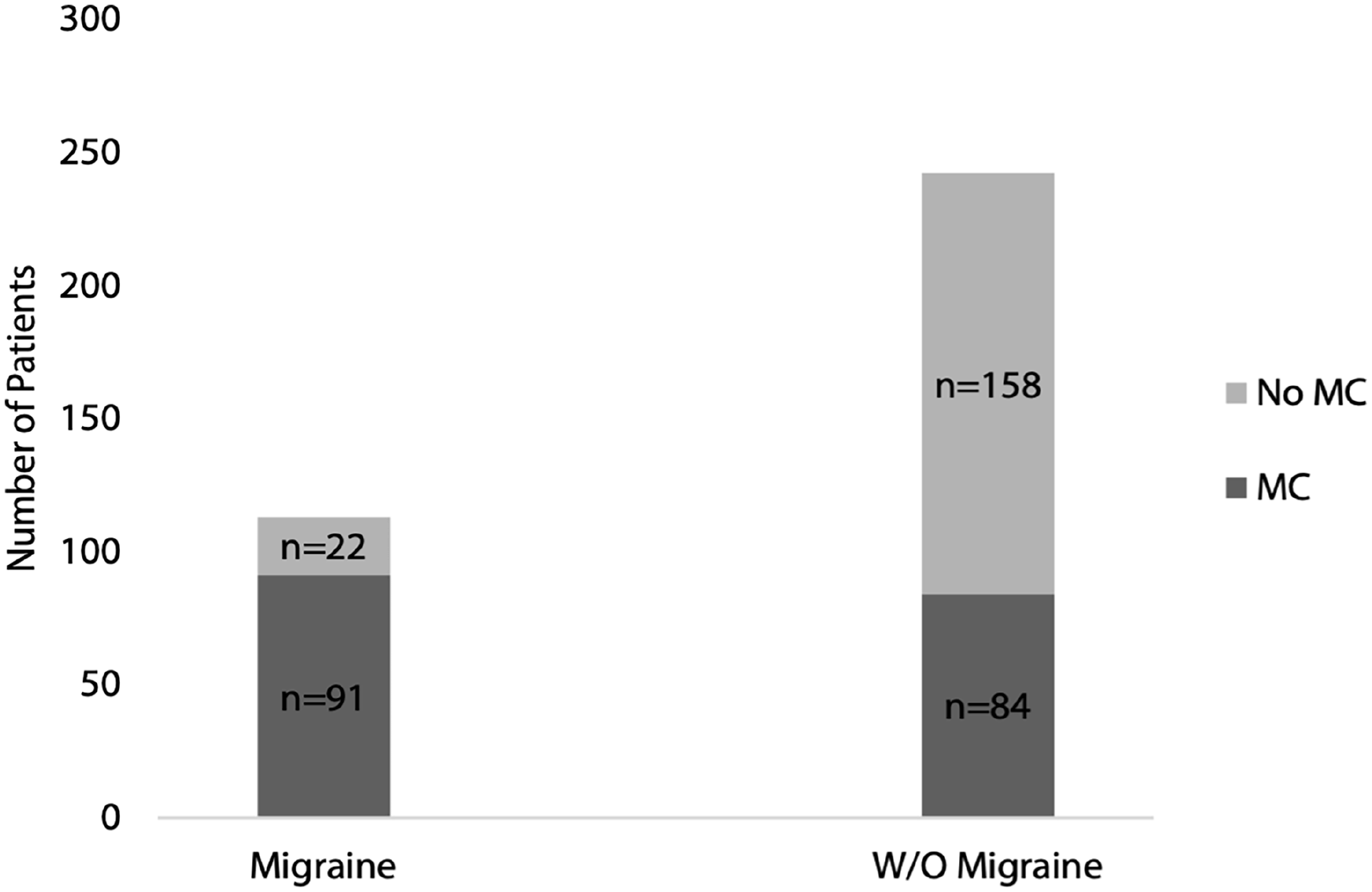
Bar graph showing presence or absence of mucosal contact in patients with and without migraine.
Statistical Analysis
Univariate analysis determined that MC and allergy were associated with increased odds of migraine (P < .001 and P < .009, respectively), while mean age and male sex were associated with decreased odds of migraine (P < .016 and P < .001, respectively; see Table 2). When accounting for the additive effects of different variables (multivariate regression), we found that MC, allergy, and female sex remained significantly associated with migraine (P < .001, P = .049, and P < .001, respectively), but age lost significance (P = .08). Univariate analysis isolating each turbinate showed both any MC at all (inclusive of ST, MT, or IT) and ST MC alone predict migraine (P < .001). MT MC alone approached statistical significance (P = .051). Multivariate analysis, however, shows ST and MT MC drive the prediction of migraine (P < .001 and P = .034, respectively), but IT MC is insignificant (P = .272; see Table 3).
Univariate and Multivariate Logistic Regression of Demographic and Comorbid Factors and Migraine.
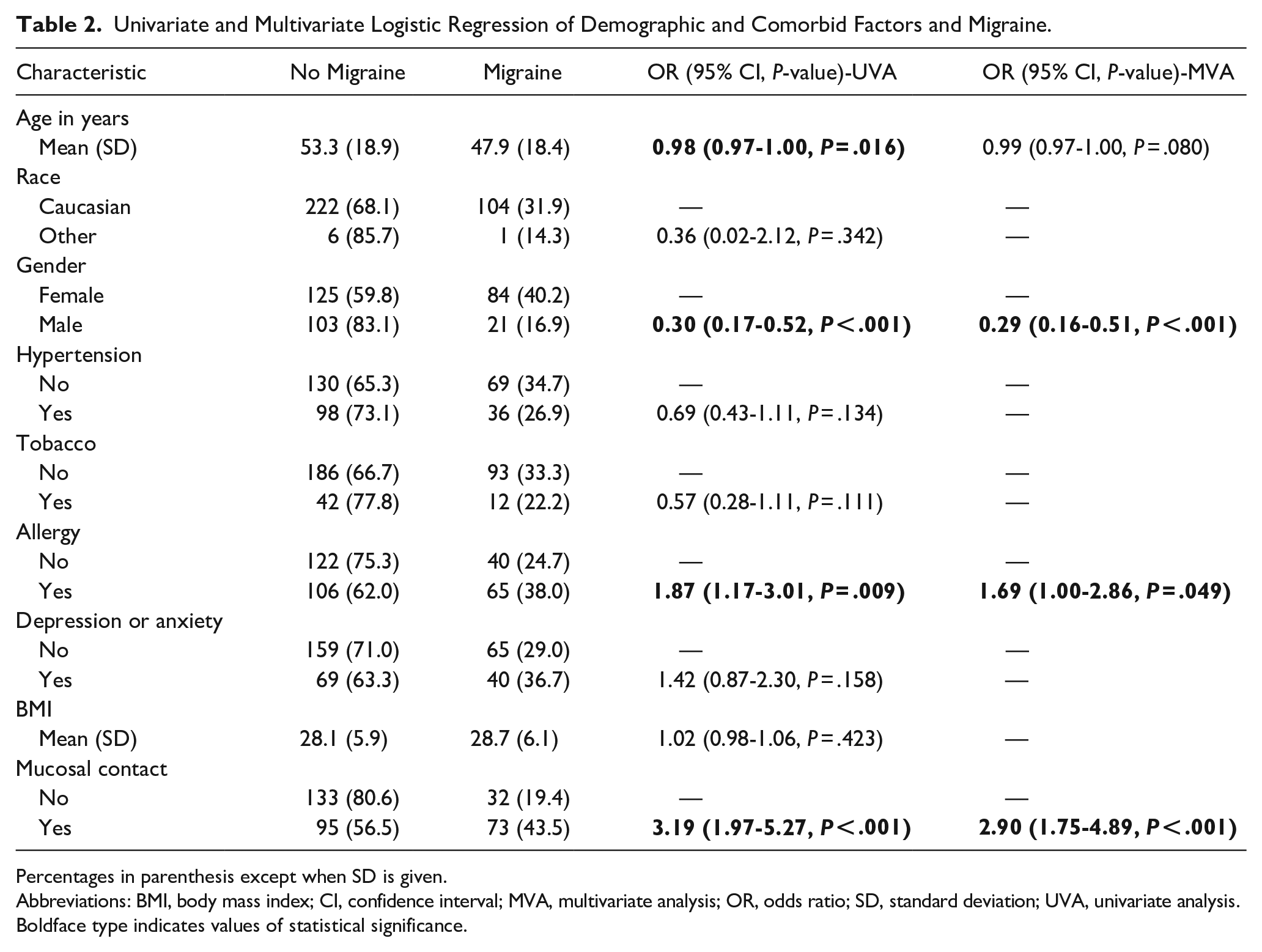
Percentages in parenthesis except when SD is given.
Abbreviations: BMI, body mass index; CI, confidence interval; MVA, multivariate analysis; OR, odds ratio; SD, standard deviation; UVA, univariate analysis.
Boldface type indicates values of statistical significance.
Univariate and Multivariate Logistic Regression of Mucosal Contact by Turbinate and Migraine.
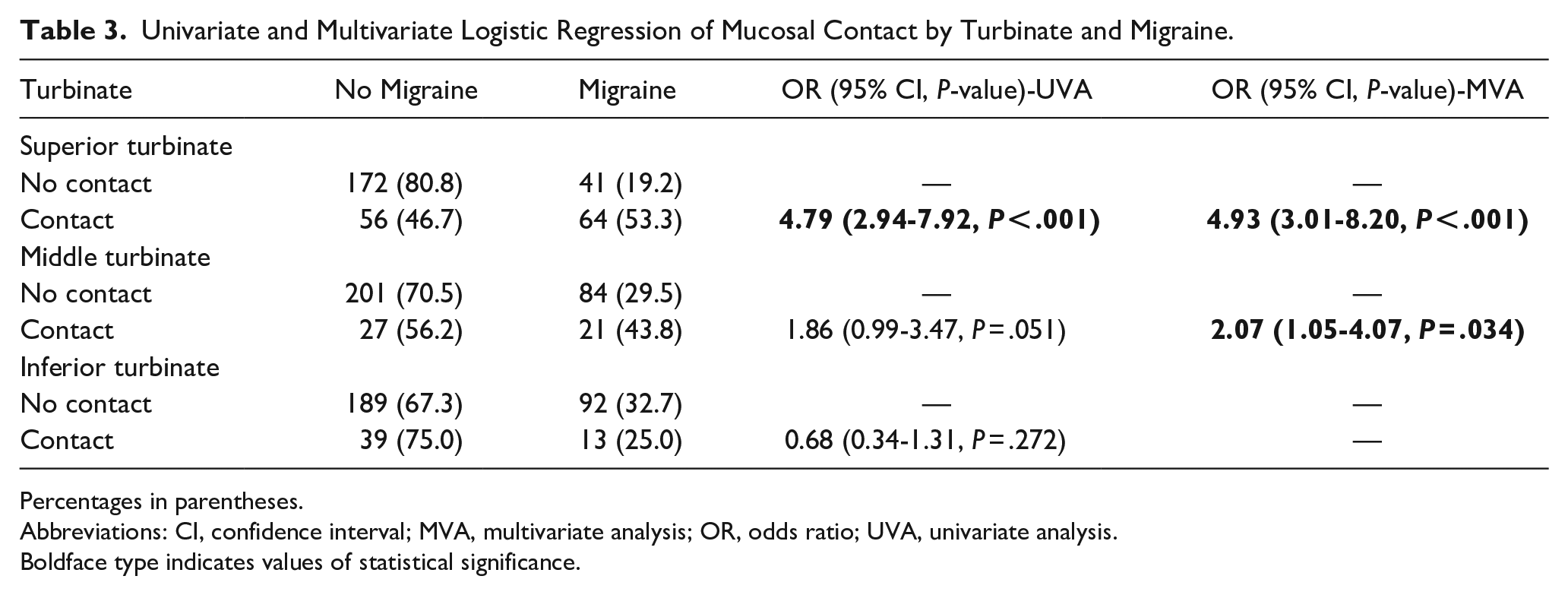
Percentages in parentheses.
Abbreviations: CI, confidence interval; MVA, multivariate analysis; OR, odds ratio; UVA, univariate analysis.
Boldface type indicates values of statistical significance.
Discussion
The term “rhinogenic headache” has been used to describe any headache caused by septal-turbinate MC.13-15 This term is technically inaccurate, because it literally means “headache that generates a nose.” The term “rhinopathic headache” 8 may be more appropriate. Most of the literature describing this phenomenon lacks details about headache diagnosis, that is, IHS criteria and definition of the type of headache being treated were seldom reported. Although tension-type headache is more common, it is rarely troublesome enough for patients to seek medical care. Migraine, however, results in greater disability, poorer quality of life, and greater utilization of medical services. For these reasons, for this study we decided to limit our investigations into the relationship between MC and headache to migraine.
We found septoturbinal MC to be much more common than previously reported. However, the turbinate most commonly involved was the ST, which can be extremely difficult to visualize endoscopically. Without specific attention directed toward the ST, MC in this region could easily be missed. This may help explain the drastic difference in the prevalence of MC in our study compared to that of Abu-Bakra and Jones, who did not report on the ST. Different definitions of MC may also have played a role in our disparate findings. We used the consensus definition from several articles, including the 1 article that catalogued criteria for MCH. 9 In contrast, the inclusion criteria of “blanching” of mucosa to indicate clinically significant MC, and the exclusion criteria of septal deviation in the absence of rhinosinusitis, used by Abu-Bakra and Jones, 7 seem at best overly restrictive. The latter criterion in particular eliminates the very subjects which most studies on MCH seek to investigate.
We found MC in general was associated with migraine. Specifically, ST and MT MC are responsible for this correlation, since the association of IT MC with migraine was insignificant. This might be expected due to the neuroanatomy of the nose and trigminovascular system. The IT is innervated by the second division of the trigeminal nerve (V2). V2 has no direct connection to intracranial structures, unlike V1, which innervates not only the upper regions of the nasal cavity (superior and middle turbinates), but also the intracranial blood vessels and the meninges, structures implicated in migraine symptoms. We speculate that the mechanism by which MC may cause migraine is physical compression of the sensitive mucosa between 2 bony surfaces (the turbinate bone and the bone of the abutting septum) in the region innervated by V1. Such mechanical compression of ST mucosa has been found to cause histopathologic changes that include epithelial denudation, laminal ischemia, and nerve hypertrophy. 16 We theorize that these changes initiate both anterograde conduction of sensory nerve impulse from this region back to the brain, as well as retrograde transmission of impulses to other peripheral branches of V1 innervating intracranial blood vessels and the meninges. The central processes that occur in migraine, such as dilation of intracranial blood vessels, spreading cortical depression, and neurogenic inflammation of the meninges, 17 mediated by these intracranial V1 terminals, may thus originate from this pathologic mucosal compression occurring in the nasal cavity.
The relatively high percentage of migraine patients in this study population (31.8%) can be explained in part by the inclusion criteria we used, that is, history of headache was an indication for nasal endoscopy. We used history of migraine and not active migraine in our study, since the literature recognizes active and inactive states of migraine, 18 and since 5 lifetime episodes of migraine satisfy the ICHD-2 and ICHD-3 definitions. These parameters undoubtedly “cast a wider net” and so would be expected to capture more of the migraine population for the purposes of this study.
We found the presence of allergy to be associated with migraine. A recent review 19 found that most studies reported a significant reciprocal relationship between allergy/atopic disorders and migraine. We urge caution in interpreting our results, however, due to the method we used to assess allergy: a retrospective chart review noting use of allergy medications, not actually allergy test results. Inappropriate use of allergy medications is very common, so it is possible we overestimated the number of truly atopic individuals.
Not surprisingly, we found advanced age and male sex to be negatively associated with migraine, consistent with the known demographics of migraine in many other studies.
Studies finding that most patients with self-reported “sinus headache” actually have migraine may be misinterpreted as evidence against the concept of MCH.20,21 However, these investigations have dealt almost exclusively with sinus inflammatory disease as the presumed cause of headache, not MC, and have not entertained the idea that migraine itself may be a MC-related phenomenon in some patients. In other words, MC may be “another disorder” 1 in the IHS definition of migraine to which headache symptomatology otherwise indistinguishable from migraine may be attributed. Even those authors who have conducted sinus CT studies in migraine sufferers have not investigated the possible role of MC in these populations.21,22
We acknowledge several shortcomings to this study. First and foremost, as an observational study, we cannot conclude that MC causes migraine, only that it is associated with migraine. Interventional studies will be needed to determine causation. Our patient population demonstrated limited racial diversity. Researchers have identified that significant gaps in migraine diagnosis and care can affect various racial and ethnic groups. 23 The mean age of our headache cohort is older than that is typically reported in migraine studies. This is likely due to the fact that we made no distinction between active and inactive migraine. Since we included patients without active migraine, recall bias could certainly introduce inaccuracies into our data. Also, since we did not distinguish between active and inactive migraine, we did not employ the lidocaine test 8 in an attempt to confirm the causative nature of MC in generating migraine. The overwhelming majority of patients did not present with headache at the time of office evaluation, thereby making the lidocaine test inappropriate. Future studies attempting to verify the causal link between MC and active migraine by utilizing the lidocaine test would be welcomed. Certain covariables associated with migraine, such as chronic pain conditions and patent foramen ovale, were not evaluated in our study. Lastly, we did distinguish between migraine with or without aura. These facts necessitate using caution in extrapolating our results to all migraine populations.
Conclusion
We found septoturbinal MC in over 49% of subjects, a much higher prevalence than previously reported. Attention to the ST and using more clinically relevant inclusion criteria were likely responsible for this. Mucosal contact was most frequently witnessed at the ST level. We have found that intranasal MC, particularly between the ST and septum, is associated with an increase in the prevalence of migraine. Further investigations into the role MC plays in migraine should be conducted. These investigations would ideally include controlled clinical trials investigating the efficacy of ST lateralization or resection on migraine outcomes.
Footnotes
Acknowledgements
The primary author would like to thank Donald C. Lanza, MD, for his encouragement in pursuing this research and for identifying the inaccuracy of the term “rhinogenic headache,” Kevin Musto, MD, and Daniel Ghezzi, PhD, for their assistance in data collection and analysis.
Author Contributions
D.M.C.: study design, data acquisition, interpretation and analysis, manuscript composition and preparation, final approval. A.K.: data acquisition and analysis. S.A.: statistical analysis, drafts, and revision of the manuscript. J.W.: acquisition of data, draft, and revisions of the manuscript. B.K.: statistical analysis, draft, and revisions of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dean M. Clerico is a paid consultant with Aerin Medical. The other authors state no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
