Abstract

Significance Statement
We report here a 35-year-old Caucasian female with 1 month history of a painless, sessile, non-ulcerated, and moveable lesion in the left border of the tongue. Histologically, the lesion was consistent with an intraneural pseudoperineuriomatous proliferation (IPP), a rare, usually solitary, and likely reactive process mimicking, for the presence of “pseudo-onion bulbs,” intraneural perineurioma. Pathologists, otolaryngologists, maxillofacial surgeons, and dentists have to be aware of it in order to avoid an inappropriate diagnosis.
Perineuriomas are uncommon and almost always benign peripheral nerve sheath tumors composed of perineural cells.1,2 Two main variants exist: extra-neural perineurioma (EP), which usually involves soft tissues and skin, and intraneural perineurioma (IP), which is typically restricted within the peripheral nerve boundaries. While based on different clinic-pathologic characteristics, different subtypes of EP (ie, soft tissue, sclerosing, and reticular) have been distinguished, IP is characterized by layers of concentric proliferation of perineural cells around nerve fibers.1,2 This change has been named “pseudo-onion bulbs” to be distinguished from the “true onion bulbs,” an architecturally similar proliferation involving Schwann cells that occurs in certain reactive and demyelinating condition.3,4 In the oral cavity, both EP and IP are rare.1-5
We describe a 35-years-old Caucasian female with a 1-month history of foreign body sensation for the presence of a painless lesion in the tongue. The patient, who denied smoking and alcoholic habits, therapies, and previous local surgery, associated the onset of the lesion to a chronic trauma caused by an orthodontic device. Her personal and family clinical histories were negative. Intraoral examination revealed a subcentimetric, sessile, non-ulcerated, and moveable lesion of soft consistency on the left border of the tongue. Neither orofacial asymmetric overgrowth (OFAO) nor cervical lymphadenopathies were clinically appreciated. Given the clinical suspicion of a benign mesenchymal tumor, an excisional biopsy was performed under local anesthesia. Histopathologic examination (Figure 1) revealed a well-circumscribed subepithelial lesion composed of concentric single- or doubled-layered spindle cells with ill-defined cytoplasmic borders distributed around axons within a collagenized stroma. Cytologic atypia/pleomorphism, mitosis, and necrosis were absent. The overlying epithelium appeared devoid of cytological atypia. Immunohistochemical analysis, performed as described previously,5-7 revealed that the spindle cells were immunoreactive for Epithelial Membrane Antigen (EMA) (Figure 2A) and that the perineurial sheath was absent. In addition, Neurofilament and S100 immunostains highlighted axons (Figure 2B) and demonstrated the scarcity of Schwann cells (Figure 2C), respectively. Proliferation index (Ki67) was less than 1%. Based on these findings, the diagnosis of IPP was done. The postoperative course was unremarkable, and recurrences were not appreciated for 1 year after surgery.
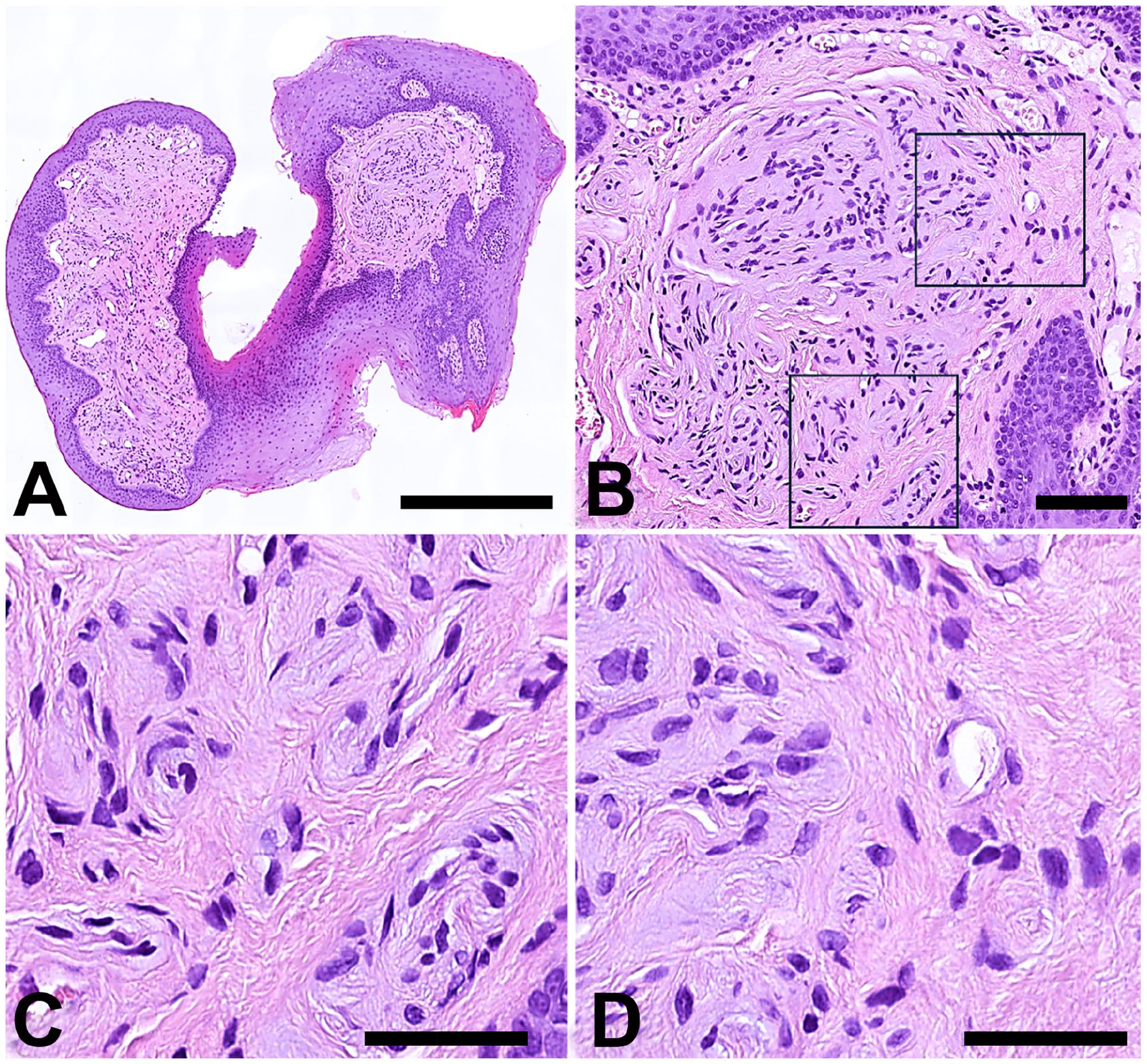
Low- and high-power views of the excised lesion are shown in (A) and (B-D), respectively. High magnification reveals that the lesion is composed of little nervous branches centred by axons and encircled by no more than 2 layers of spindle cells in a fibrotic background. Arrows point to the continuity of the fibrotic tissue around the “pseudo-onion bulbs” with the surrounding stroma due to the absence of perineurial cells. Panels (C) and (D) illustrate the inner and peripheral parts of the lesion delimited by squares in (B). Hematoxylin-eosin. Bars: 500 µm in A, 100 µm in B and 50 µm in C and D.
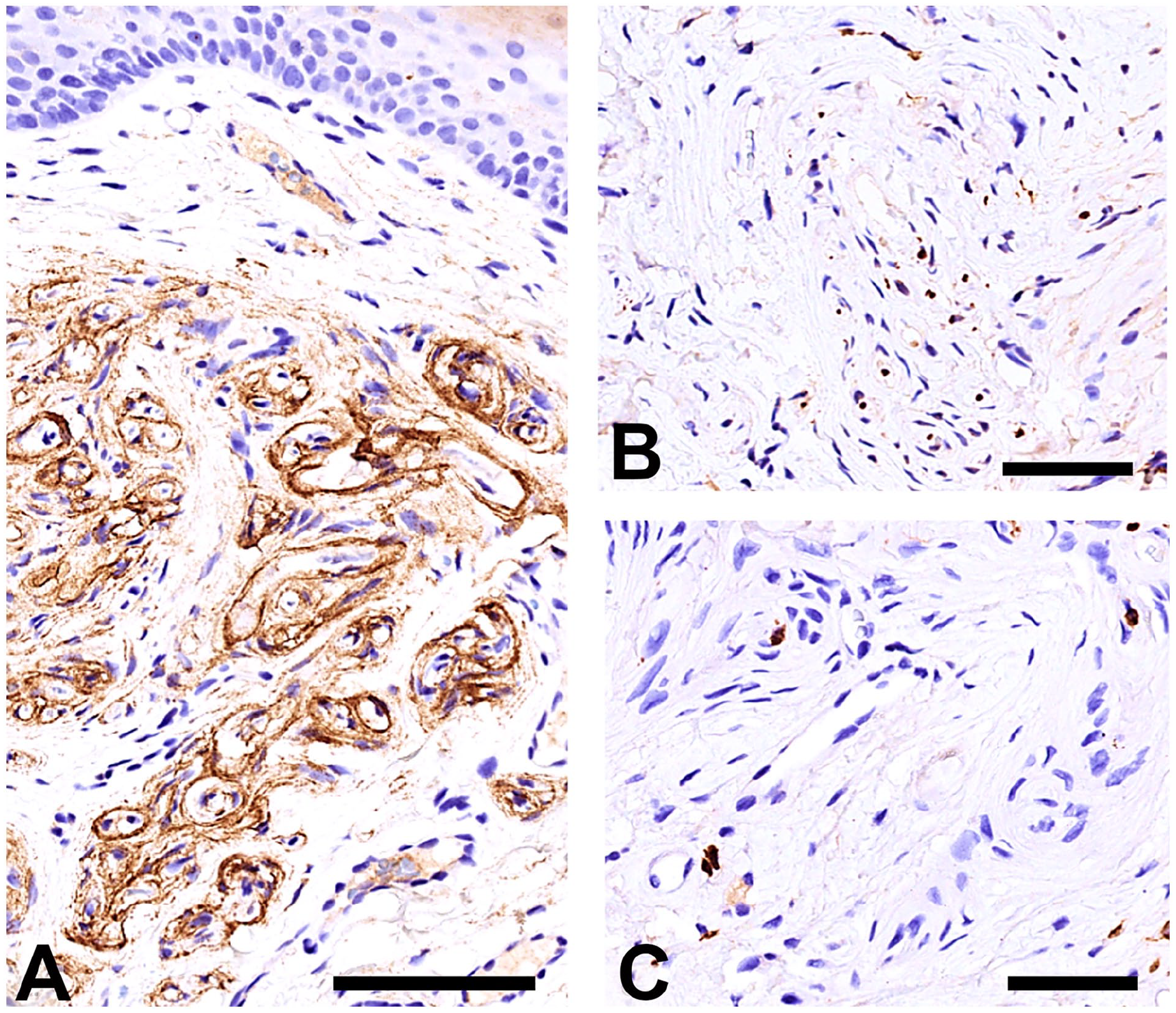
Immunohistochemical profile of the lesion. (A) EMA. (B) Neurofilaments (NF200). (C) S100. EMA confirms the absence of perineurial cells around the nerve and highlights the “pseudo-onion bulbs” (A), which are composed of no more than 2 perineurial cells. NF200 stains the preserved axons (B) and S100 demonstrates the scarcity of Schwann cells (C). 3,3ʹDiaminobenzidine (DAB) was used as chromogenic substrate. Bars: 100 µm in A and 50 µm in B and C.
IPP was identified in 2013 by Koutlas and Scheithauer 4 as an apparently reactive intraneural perineurial cell lesion occurring in the oral cavity that shared features (ie, “pseudo-onion bulbs”) with IP. Based on the clinical presentation, IPP is clinically commonly interpreted as either fibrous hyperplasia or, as in our case, benign mesenchymal tumor.4,8 In addition, as in our case, IPP has been reported more commonly in the tongue of adult females. 4 Even though typically solitary, multiple IPP have been reported in association with OFAO.8,9 Pathologic analysis is needed for the diagnosis. The most relevant differential diagnosis is IP, compared with which IPP is characterized by “pseudo-onion bulbs” containing no more than 2 layers of perineurial cell and by an intrafascicular collagen-enriched matrix that, as the result of disruption/absence of the perineurial sheath, may appear in continuity with that of the surrounding stroma. 4 Moreover, in contrast to IP, IPP more commonly comprises part of another lesion (eg, fibrous hyperplasia). 4 Immunohistochemical stains for EMA, S100, and NF are needed to identify the different components of the lesion. While EMA [either Glut-1 or Claudin-1 if EMA is negative (1-4,10)] highlights the perineurial cells and reveals discontinuity/absence of the perineural sheath, NF and S100 demonstrate the preservation of axons and reduction in Schwann cells, respectively.4,10
The pathogenesis of IPP is not known. However, a non-open chronic compression injury, as that likely occurring in our patient due to the orthodontic device, has been suggested.4,8 It may lead to nerve ischemia with focal loss of perineurial cells. This view is supported by the evidence that IPP-like lesions have been reproduced in experimental models of peripheral nerve ischemia 11 and that the loss of the perineurial barrier may cause differentiation of endo-neural fibroblasts into perineurial cells through the secretion of Desert Hedgehog by Schwann cells.12,13 Interestingly, in some patients with multiple IPPs associated with OFAO, mosaic PIK3CA variants have been identified8,14, proving that, at least in this context, IPPs are genetically distinct from both EP and IP.1-3,9,14,15
In conclusion, we have described the clinic-pathologic features of a rare lesion involving the oral cavity. We feel that pathologists, otolaryngologists, maxillofacial surgeons, and dentists have to be aware of IPP in order to avoid an inappropriate diagnosis, both when solitary and likely post-traumatic as also when multiple and associated with OFAO.
Footnotes
Data Availability
All available data have been reported in the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
All the clinic-pathologic investigations detailed in the manuscript have been conducted in accordance with the Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed Consent
Written informed consent for publication of data and images was obtained from the patient.
