Abstract
Keywords
Introduction
Vertigo is the perception of motion without any movement and includes different definitions, such as spinning sensation, imbalance, and dizziness. Benign paroxysmal positional vertigo (BPPV) is one of the most common causes of vertigo. The overall lifetime prevalence is approximately 2.4%, which is more common in women. 1 The geriatric population experiences it 5 to 7 times more frequently than patients under 40.
Robert Bárány first described positional vertigo in 1921. 2 The term BPPV was introduced into the literature by Dix and Hallpike in 1952. 3 Otoliths, called otoconial debris, falling from the utricular macula into the semicircular canal are responsible for BPPV. Otoconial debris floating freely in the endolymph of the semicircular canals in the inner ear is called canalolithiasis, while calcium carbonate crystals adhered to the cupula are called cupulolithiasis. 4 It may occur due to age-related degeneration, head or general body trauma, ischemic events, surgical interventions, or it may develop spontaneously as an idiopathic condition.
Clinical symptoms of BPPV are characterized by short-term vertigo, nausea, imbalance, and positional nystagmus associated with changes in head position. 5 Diagnosis is based on the presence of these symptoms and observation of nystagmus during provocation maneuvers. After a latent period of a few seconds, the presence of fatigued nystagmus with rotational, horizontal, and torsional characteristics is essential for diagnosing BPPV. Depending on the character of the nystagmus, the affected ear and canal are identified, and canalith reposition maneuvers (CRM) are applied during treatment. Displacement of otoliths can be effectively treated with reposition maneuvers.
In our study, we determined the residual disease rates by performing a control test maneuver 1 week after CRM was performed on patients diagnosed with BPPV. We compared the rates according to canal involvement and aimed to reveal the effect of other risk factors on residual disease.
Materials and Methods
Patient Selection and Study Design
This study was designed and completed as a retrospective cohort study on patient data who applied to the otorhinolaryngology clinic of the tertiary Kastamonu Training and Research Hospital with complaints of dizziness and were diagnosed with BPPV between October 2021 and September 2024 (36 months). The otolaryngologist evaluated the dizziness-complaining patients with a detailed anamnesis and neuro-otological examination and referred those with a preliminary diagnosis of peripheral vertigo to the audiology unit. Three audiologists with at least 2 years of experience performed dynamic vestibular examinations on the patients.
The patient’s demographic information, affected side, age, body mass index (BMI), audiological evaluation and vestibular test results, Dizziness Handicap Index (DHI) scores, affected canal (posterior, lateral, anterior, bilateral), how long after the onset of the attack they applied to the outpatient clinic, repositioning maneuver results, and chronic disease associations were documented.
Patients under 18, patients with vertigo of central origin, and other types of peripheral vertigo other than BPPV (vestibular neuritis, sudden hearing loss, Meniere’s disease) were excluded from the study. Patients with vertebral pathology that restricted the CRM and patients diagnosed with BPPV after the diagnostic test maneuver but who did not want to have repositioning maneuvers were also excluded from the study. In addition, patients who did not come for their seventh-day follow-up and those with incomplete data were not included. Only the first application data of 2 patients who presented with recurrent BPPV in 3 years (one developed recurrent BPPV after 11 months and the other after 34 months) were included, and the data of the second attack were not used.
The study protocol was approved by the Kastamonu University Clinical Research Ethics Committee (2024-KAEK-63). When using patient data, the Helsinki reports were followed.
Dizziness Handicap Inventory
DHI is widely used as a reliable tool to assess the disability effects of vestibular system disease as perceived by the patient. 6 DHI consists of 25 self-assessment questions divided into physical, emotional, and functional subscales—total scores range from 0 to 100. The higher scores indicate a more excellent perception of dizziness-related disability. It is a reliable and valid tool used to assess the detrimental effects of dizziness on patients’ quality of life, including those with BPPV. We also filled out the questionnaire and recorded the data before starting dynamic diagnostic tests on our patients on their first visit.
Diagnostic Tests
All patients were applied to diagnostic Dix-Hallpike and head roll maneuvers. 3 According to patient information, if there was a triggering position for vertigo or nystagmus, the test was started from the healthy side. If the patient perceived no triggering regarding the position, the maneuver was started from the right side. Patients diagnosed with BPPV were given CRM and vestibular rehabilitation maneuvers to be applied at home after the test, and they were called for a checkup in the first week. Diagnostic tests were repeated in the control examination—the absence of nystagmus and vertigo after the successful maneuver. Unsuccessful maneuvers were classified as having residual disease. All patients who met the BPPV criteria for anterior semicircular canal were assessed with contrast-enhanced brain MRI to exclude intracranial pathologies.
Therapeutic Maneuvers
The patients diagnosed with posterior semi-circular canal (pSSC) canalolithiasis with the Dix-Hallpike test were subjected to modified Epley maneuvers, and the patients diagnosed with cupulolithiasis were subjected to Semont maneuvers.7,8 For home applications, Brand-Darrof exercises were explained for lateral canal BPPV canalolithiasis, the otoliths were repositioned with Barbecue, Vannucchi-Asprella, or Gufoni maneuvers.9-13 Since there was no specific reposition maneuver for lateral canal cupulolithiasis, the recommended treatment maneuver, habituation exercises, was applied.14,15 The Yacovino maneuver was applied to the anterior canal BPPV. 16 In patients with bilateral involvement, treatment was started in the severe ear, and CRM was applied to the other ear during the first week of evaluation.
After the treatment maneuver, the patient was advised to wait on the stretcher for 20 minutes and lie with the head elevated at 45° for the following 3 days. We recommended that the patients refrain from hyperextension of their heads for 3 days and avoid lying on the affected canal side due to position restrictions.
Statistical Analysis
Statistical analyses were performed using IBM SPSS, version 22.0 (IBM Corp, Armonk, NY, USA). Continuous variables were presented as mean ± standard deviation, and categorical variables were presented as frequency and percentage. The Kolmogorov-Smirnov test was used to examine the conformity of the data to normal distribution. An independent sample t-test or the Mann-Whitney U test was used for continuous variables in comparison with recovered patients and residual individuals, and the chi-squared (χ2) test was used for categorical variables. Normally-distributed values were given as the mean ± standard deviation, and non-normal values were given as the distribution median (min. and max.). Univariate analyses determined the relationship between age, gender, examination day, hypertension, diabetes mellitus, DHI, and residual BPPV. The omnibus test determined the compatibility of the tests. P < .05 was considered statistically significant.
Results
Two hundred one patients were included in the study: 91 (45.3%) male and 110 (54.7%) female. The mean age of the patients was 60.4 ± 13.8 (min = 23-max = 90) (Table 1). Right ear involvement was observed in 123 (61.19%) patients, whereas left ear involvement was observed in 69 (34.32%) patients. Nine (4.47%) patients had bilateral canal pathology. Posterior canal involvement was detected in 177 (88.5%) patients, lateral canal involvement in 16 (7.96%) patients, and anterior canal involvement in 8 (3.98%) patients (Table 2). Considering all patients, canalolithiasis was diagnosed in 130 (64.67%) patients and cupulolithiasis in 71 (35.32%) patients.
Comparison of Demographic Data and Comorbidities Between the Two Groups.

Abbreviations: BMI, body mass index; DM; diabetes mellitus; HT; hypertension; *: median, min-max.
Comparison of Vertigo Data between Both Groups.

Abbreviations: DHI, dizziness handicap inventory; SCC, semicircular canal.
In terms of systemic diseases, 78 patients (38.81%) had hypertension, 66 patients (32.83%) had diabetes, and 35 patients (17.41%) had coronary artery disease (CAD). Again, 66 patients (32.83%) were observed to have a BMI >30 (Table 1).
When we evaluated the first week after the maneuver regarding the residual disease, the residual disease was significantly higher in those with HT and DM (P = .029, .011, respectively). It was found that patients with high DHI scores had significantly-higher residual BPPV (P = .001). In residual disease, bilateral canal involvement (P = .001) and anterior canal involvement significantly differed from other canals (P = .001). Residual BPPV was significantly higher in those who applied to the otolaryngology outpatient clinic after the 72 hours of the onset of the vertigo attack than those who applied in the first 72 hours (P = .001) (Table 2).
There was no significant difference between the patients with successful maneuver and residual BPPV in age, gender, presence of obesity, presence of CAD, canalolithiasis, or cupololithiasis (Table 2). Similarly, there was no significant difference between the 2 groups in terms of patients’ betahistine use and previous history of otologic vertigo. Age and gender were not linked to residual disease in one-way analyses. However, at otolaryngology visits after 72 hours of the attack, HT, DM, and DHI scores were found to be significantly linked to residual BPPV (Tables 1 and 2; Figure 1).
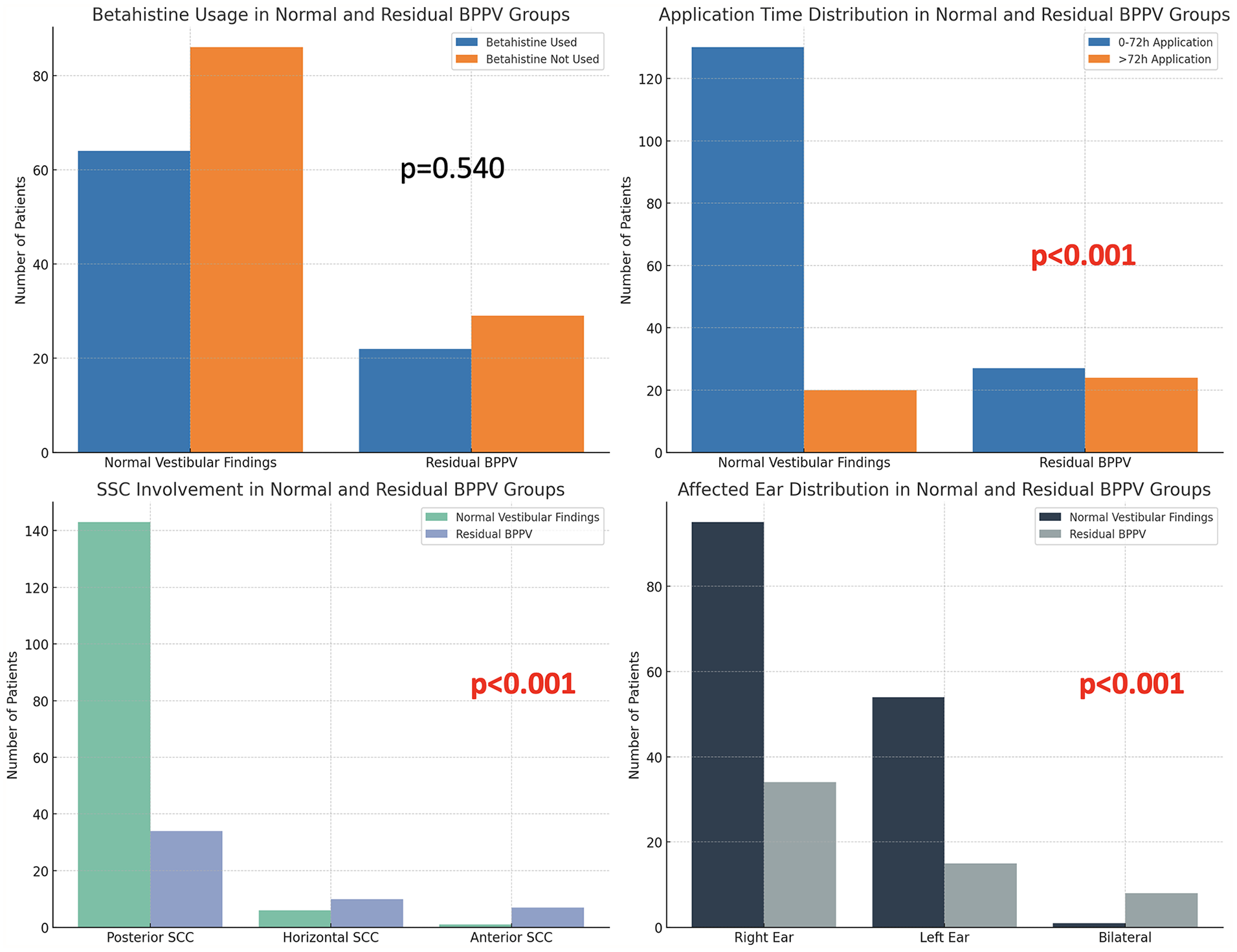
This graphic compares data from both groups according to betahistine usage, application time, affected ear side, and involved canal.
Discussion
BPPV is a disease that improves clinically with repositioning maneuvers and sometimes resolves spontaneously over time. In a systematic review and meta-analysis, the rate of residual disease was found to be very wide, from 30% to 70%. 17 Multiple factors affect the success of CRM. The study aimed to examine the residual disease rates and the factors that may affect them with the first week control tests of CRM applied to patients diagnosed with BPPV.
Song et al reported in their randomized controlled study that the most optimal reassessment time for BPPV is 1 day later. 18 Çelik et al suggested that residual disease should be investigated with a control Dix-Hallpike 20 minutes later in the same session for patients who cannot come for a follow-up the next day. 19 The literature agrees that routine checkups are generally performed in the first week, especially in the first month. In our clinical study routine, we determine that the most appropriate checkup period for patients is 1 week, and we continue our study routine accordingly. This research article was derived from the analysis of those data.
The relationship between systemic diseases and BPPV has been investigated, and the extent to which they affect has been tried to be revealed. Messina et al 20 also support the hypothesis of a vascular role in the etiopathogenesis and recurrence of BPPV. A recent observational study has shown that hypertension and diabetes are significantly associated with the risk of recurrent BPPV, with the risk increasing when both comorbidities are present simultaneously. 21 In our study, we also found the presence of both diseases to be significantly higher in terms of residual disease.
Several studies mention mechanisms for residual disease after CRM, and these studies also mention the effect of systemic diseases on recurrent diseases. Çetin et al 22 found that DM poses a risk for residual BPPV and reported that the presence of obesity has a negative effect on the recovery of residual disease. Our study found the presence of DM significant for residual disease, while obesity did not create a significant difference (P = .271). It may not be possible to perform CRM effectively in morbid obesity.
The first-line treatment for BPPV is nonpharmacological interventions such as CRM and vestibular rehabilitation. In a randomized controlled study conducted by Sayın et al, 23 they found that betahistine given in addition to vestibular rehabilitation exercises improved both visual analog scales and DHI. In their study, Erdur et al 24 found that adding betahistine to CRM did not contribute to DHI and anxiety symptom scores. In current study, we did not prescribe betahistine to any patient after establishing the diagnosis of BPPV. However, we did not stop the betahistine prescribed by other physicians (emergency room, family physician, or other clinic physicians) that they consulted before otolaryngology, and we recommended that they continue to use it as started. In comparing 115 patients who first applied to the ENT polyclinic and were followed up only with CRM and 86 patients who had previously started betahistine, no significant difference was observed in residual disease (P = .540).
Bilateral involvement is less common in BPPV and is associated with trauma. 25 The most common combination is both posterior canal canalolithiasis. In our study, all 9 patients with bilateral involvement had posterior canal BPPV. Ipsilateral multi-canal involvement was not observed in our patients. In bilateral involvement, the general clinical approach is to revise the severe side first and then the contralateral side. As an expected result, the rate of residual disease in bilateral cases was higher. Only 1 patient had complete recovery on both sides at the first week follow-up, and maneuvers were performed again for the other 8 patients, and controls were called again. In a 61-year-old male patient who we followed up with bilateral posterior BPPV canalolithiasis after a traffic accident in a vehicle, his complaints lasted for 5 weeks and then resolved. Bilateral involvement was significantly higher regarding residual disease than unilateral involvement (P < .001).
In patients with peripheral vertigo, recurrence of the disease may cause anxiety and panic attacks. 26 Recurrences can be seen in BPPV, and the frequency of BPPV development increases, especially after vestibular neuritis (VN). One study reported that patients who had VN had a 15% risk of developing BPPV over 10 years. 27 Our study included 21 patients with a history of otologic vertigo. No statistically-significant relationship was observed between the presence and absence of a history of otologic vertigo in terms of residual disease (P = .450).
DHI score is a useful tool for predicting BPPV and indicates its severity. 28 In our study, the DHI score (44.74 + 12.98) of the residual disease group was found to be significantly higher than the other group (33.16 + 9.66) (P < .001). DHI scores also increased as the feeling of dizziness caused by the disease increased. Although we cannot specify a definitive cutoff value, we recommend a control test after 1 week and repeat CRM if necessary for patients with a DHI score above 40 at admission.
Conclusion
In conclusion, since BPPV is more common in older patients and can cause morbidity after falls, it is essential to treat it effectively. Some characteristics of the first application can determine appropriate patients for a 1 week check. If we make an inference from our results, we recommend that patients with additional diseases such as HT, DM, bilateral involvement, anterior canal involvement, DHI >40, and patients presenting after 72 hours be called for a checkup 1 week later.
Footnotes
Acknowledgements
The author(s) wish to thank Sultan Keskin Demircan, MD, for his assistance. The authors would also like to acknowledge and thank the patients whose data contributed to this study’s results.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The Kastamonu University Clinical Research Ethics Committee approved the study, which was conducted according to the Declaration of Helsinki (No.: 2024-KAEK-63).
