Abstract
Myeloid sarcoma (MS) is a proliferation of immature myeloid cells that occurs extramedullary, often accompanying acute myeloid leukemia (AML). It typically presents in areas such as lymph nodes, skin, and the head and neck regions. The emergence of MS within the temporal bone is rare, and it can manifest through nonspecific symptoms. In this paper, we reported a case of A 47-year-old female presented with right-sided otalgia, otorrhea, tinnitus, reduced hearing, and ear fullness, and developed facial asymmetry. She was initially diagnosed with necrotizing otitis externa and was treated accordingly. Further investigations were conducted, leading to the diagnosis of MS. She began treatment with induction chemotherapy followed by consolidation therapy. And we reviewed the literature and included 14 MS cases with the same anatomical localization, discussing the primary presentation, immunohistochemistry, and treatment approaches of these patients. In conclusion, temporal bone MS should be considered in patients with a history of AML presenting with nonspecific symptoms. Diagnostic modalities for MS include computed tomography and magnetic resonance imaging scans. Systemic chemotherapy remains the preferred treatment approach. The complexity and diversity of temporal bone MS presentations necessitate further comprehensive research to enhance understanding and improve management strategies for this rare condition.
Introduction
Myeloid sarcoma (MS), also known as granulocytic sarcoma (GS) or chloroma, represents a tumor mass consisting of myeloid blasts with or without maturation, appears extramedullary in the context of various hematological malignancies (HM); such as acute myeloid leukemia (AML), chronic myeloid leukemia, chronic myelomonocytic leukemia, acute promyelocytic leukemia, myelodysplastic syndrome, and myeloproliferative neoplasms. 1 MS mostly occurs in combination with intramedullary AML (synchronous) and rarely in individuals with no history of HM (isolated). 2 The skin, gums, lymph nodes, soft tissues, periosteum, and bone are frequently-observed sites of involvement. 3 Pileri et al 4 reported that bone involvement accounted for 3.25% of patients with MS. Given this low percentage, MS involving the temporal bone is exceedingly rare in clinical practice. These invasive presentations may lead to the constriction of cranial nerves, giving rise to a range of neurological impairments, among them facial nerve paralysis, an occurrence seldom linked with concurrent cranial nerve issues such as ear pain, dizziness, and abducens nerve paralysis. 5 Additionally, there have been a small number of instances where primary and relapsed AML were misinterpreted as acute otitis externa (AOE), with an even smaller number manifesting as necrotizing otitis externa (NOE). 6 Here, we report a rare case of temporal MS with initial symptoms resembling NOE, and we conduct a review of similar cases that have reported MS with the same temporal localization.
Case Presentation
A 47-year-old female presented to the otorhinolaryngology department with complaints of right-sided otalgia, otorrhea, tinnitus, reduced hearing, and ear fullness persisting for the past 3 days. She also had 1 episode of vertigo and subsequent facial asymmetry 12 hours before admission. Otoscopic examination showed right external auditory canal (EAC) edema, obscuring the tympanic membrane. The left ear examination was normal. Rinne test was positive bilaterally, with the Weber test lateralizing to the right ear. She presented with right motor neuron facial nerve paralysis, House-Brackmann Grade V. The rest of the physical examination was unremarkable. Pure tone audiometry test revealed an air-bone gap of approximately 15 dB in the right ear, while the left ear showed normal results. Subsequently, she was diagnosed with NOE, and intravenous administration of antibiotics was initiated immediately alongside local administration of dexamethasone and ciprofloxacin drops.
Four weeks later, partial clinical improvement was noted; however, the facial paralysis remained unchanged. Upon detailed investigation, it was revealed that the patient had a history of AML, classified as M5 according to the French-American-British classification system, treated with complete chemotherapy leading to a remission of 5 years. She has no history of alcohol consumption or smoking. Recently, she underwent a colostomy procedure. Her current medication regimen includes bisoprolol 2.5 mg once daily, calcical 500 mg once daily, and vitamin D3 10,000 IU twice weekly.
Accordingly, a computed tomography (CT) scan of the temporal bone was carried out, demonstrating opacities in the right EAC, middle ear, and mastoid cavity, with soft tissue density present within. The left side was found to be normal (Figure 1). Following these findings, the decision to proceed with surgical intervention was made. During anesthesia, a biopsy was taken from the right EAC, followed by a right modified radical mastoidectomy procedure. Intraoperatively, inflammatory tissue was observed filling the mastoid air cells, the antrum, the attic, and the mesotympanum, covering the incus and malleus. Furthermore, a dehiscence was noted in the tympanic segment of the facial nerve canal, and granulation tissue was found covering the first third of the mastoid segment of the facial nerve. Biopsies were taken from the mastoid, middle ear, and ear canal for pathological examination, and immunohistochemistry (IHC) was performed, which showed monomorphic cell proliferation with positivity for CD99, myeloperoxidase (MPO), negativity for LCA, CD3, CD20, CD68, CD138, leading to the definitive diagnosis of MS. A postoperative Multislice CT with contrast of the head, neck, abdomen, and pelvis revealed no metastases, and the bone window analysis showed no lytic or sclerotic lesions. Later, the patient was admitted to the Department of Hematology. Upon admission, the patient’s vital signs were within normal limits. Clinical examination showed conjunctival pallor, indicating anemia. An external colostomy was also observed. Initial blood tests, detailed in Table 1, were conducted at admission, revealing absolute neutrophil count (ANC) at 1048 cells/µL, and platelet count of 135,000/µL. The peripheral blood smear showed no abnormal cells. The bone marrow aspiration exhibited morphologically-well-differentiated white blood cells (WBCs) and mild dysplasia, with blast cells comprising 1% to 2% of the differential (Figure 2). The integration of these findings suggested the patient had complete remission (CR). Based on these findings, the case was defined as extramedullary AML (EM-AML) without bone marrow involvement (BM-I). The patient commenced the induction chemotherapy regimen (5 + 2), which included administering cytarabine (200 mg/m2/day × 5) and doxorubicin (50 mg/m2/day × 2). She was subsequently followed up clinically and through laboratory evaluations with consecutive blood tests (Table 1). One month after induction and just prior to starting consolidation chemotherapy, the patient’s blood tests (Table 1), along with a bone marrow aspiration revealing a 3% blast count and the absence of immature WBCs in the peripheral blood smear, indicated CR. The patient was mapped out for a consolidation treatment plan that entailed 4 cycles of cytarabine at a dose of 2000 mg/m2, every 12 hours across 3 days. Two months after starting therapy, the patient was in a good health, and new blood tests were carried out (Table 1), with bone marrow aspiration (1% blast count), which confirmed CR. Subsequently, she received her second consolidation treatment, with the cytarabine dose being reduced to 1500 mg/m2, to balance treatment efficacy with patient tolerance and minimize potential side effects after the initial cycle confirmed CR. After the second cycle, the patient was hospitalized for monitoring. During this time, she experienced angina-characteristic chest pain. This was successfully treated with medication, and a subsequent echocardiogram revealed partial narrowing in her coronary arteries. The third and fourth chemotherapy cycles were scheduled at 6 week intervals, with radiotherapy (RT) set to begin 2 weeks after the fourth cycle. An elective catheterization was planned after the completion of RT.
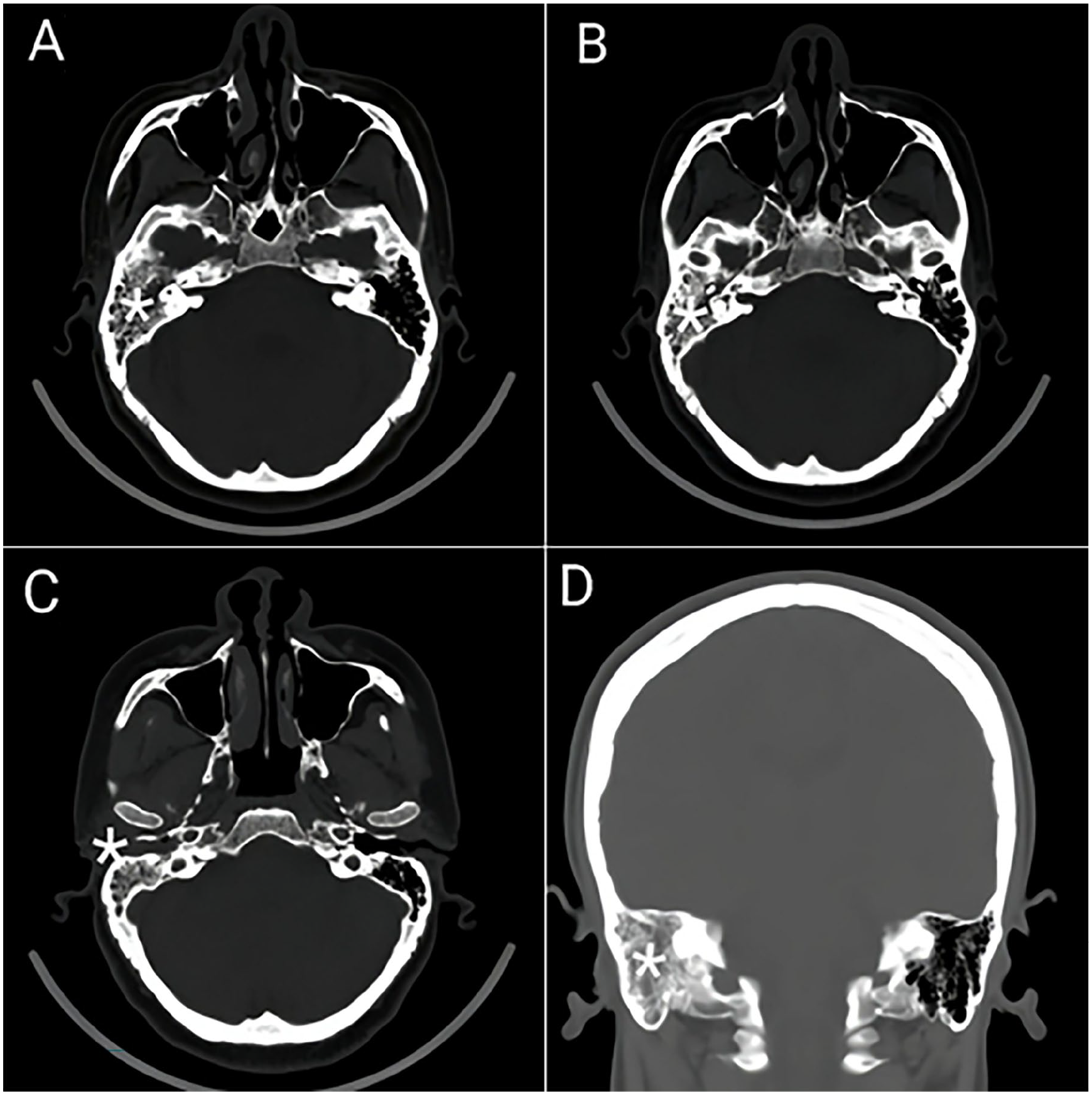
Axial CT images of the temporal bones (A-C). (A) Near-complete opacification within the right temporal bone and soft tissue filling the mastoid cavity (asterisk). (B) The extension of the opacification to the middle ear (asterisk). (C) Near-complete opacification involving the external auditory canal (asterisk). Coronal CT of the temporal bones (D). Soft tissue filling the right mastoid cavity (asterisk). CT, computed tomography.
Follow-Up Blood Tests.
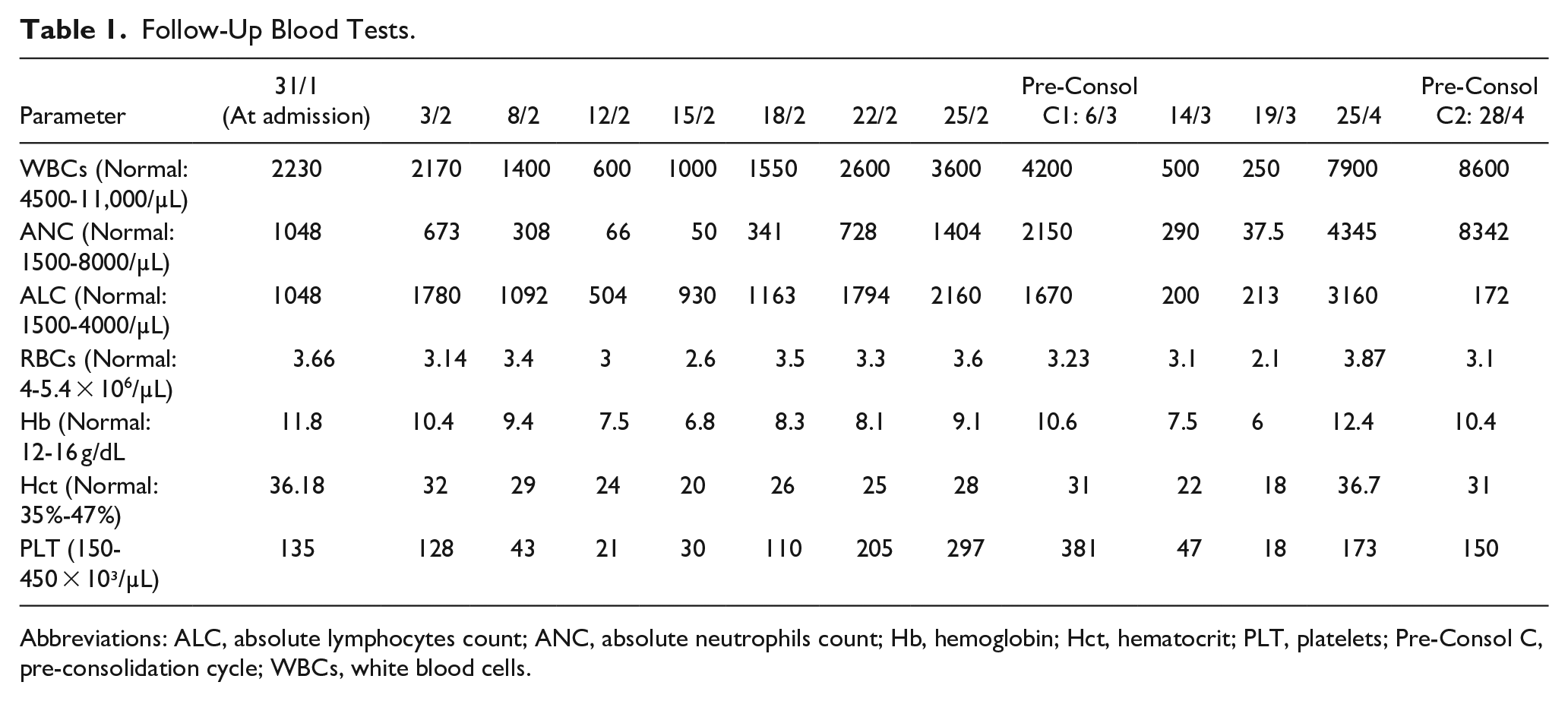
Abbreviations: ALC, absolute lymphocytes count; ANC, absolute neutrophils count; Hb, hemoglobin; Hct, hematocrit; PLT, platelets; Pre-Consol C, pre-consolidation cycle; WBCs, white blood cells.
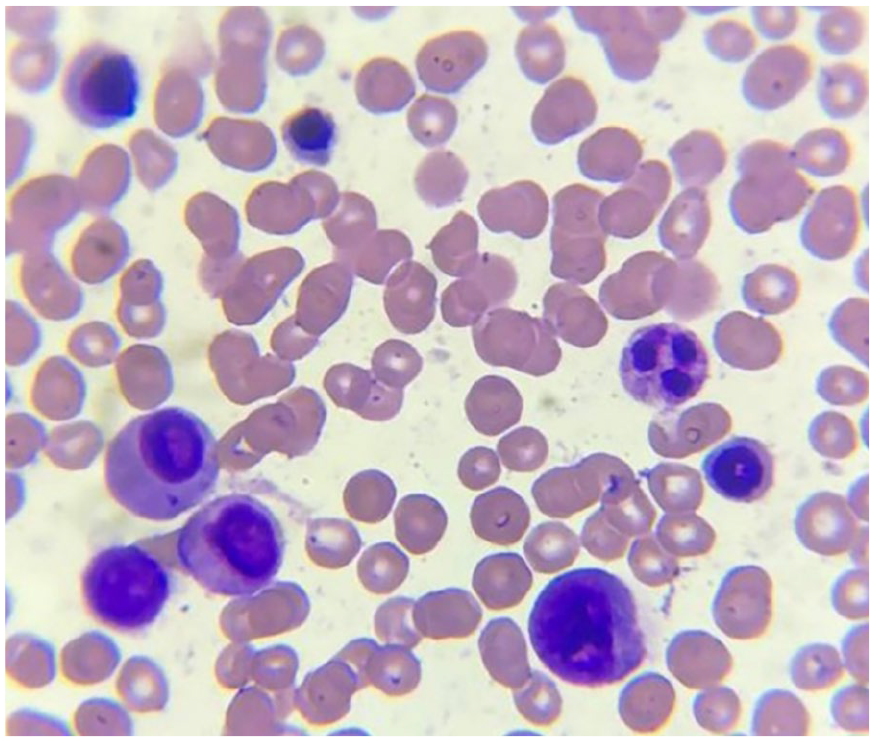
Bone marrow aspirate: 1% to 2% blast cells, well-differentiated white blood cells, mild dysplasia.
Discussion and Conclusion
MS or GS, also known as chloroma, consists of immature granulocytic cells, presenting an extramedullary manifestation commonly associated with AML. 7 The development of MS appears to be influenced by a complex interplay of factors. The bone marrow microenvironment and its communication signals, including cell-to-cell and cell-to-matrix interactions, likely play a role. Additionally, the specific activity patterns of chemokine receptors on leukemic cells may guide their movement and influence the sites of MS development. While potential roles for CD56 and CD11b expression is suggested, their direct involvement remains unclear and requires further investigation. 2 MS can arise in various body sites, including soft tissues, bones, lymph nodes, peritoneum, and the gastrointestinal system. 7 According to Pileri et al, 4 bone involvement was present in 3.25% of patients with MS, making temporal bone MS exceptionally uncommon in clinical settings. We conducted a comprehensive review of the MEDLINE and Google Scholar databases, focusing on literature published between 2015 and 2024, which led us to include 14 cases with temporal MS (Table 2). Our selection criteria included only freely available full-text English studies with human subjects. Paid non-English studies, as well as non-human studies, were excluded.
Summary of Reported Cases of Myeloid Sarcoma in the Temporal Bone.
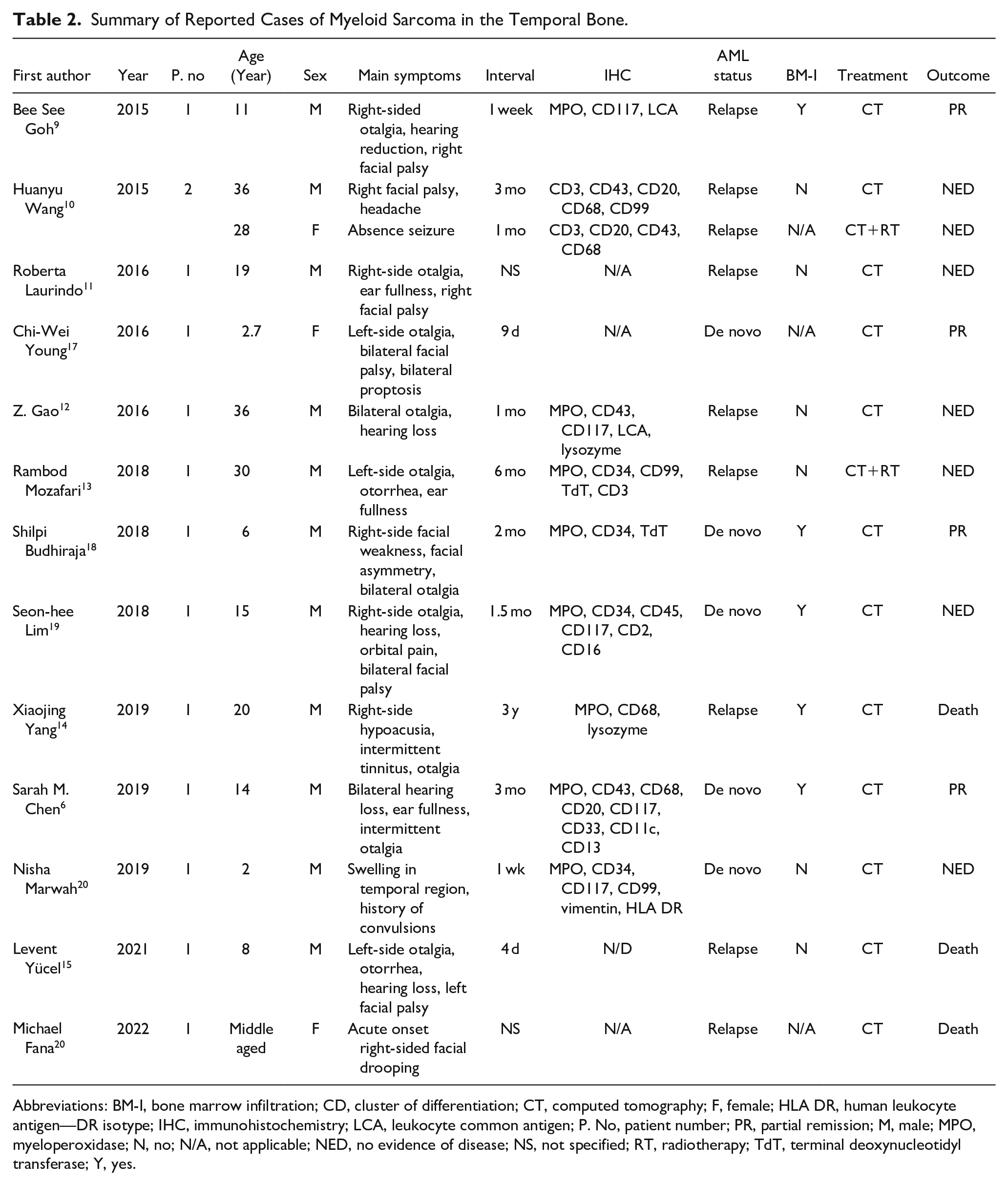
Abbreviations: BM-I, bone marrow infiltration; CD, cluster of differentiation; CT, computed tomography; F, female; HLA DR, human leukocyte antigen—DR isotype; IHC, immunohistochemistry; LCA, leukocyte common antigen; P. No, patient number; PR, partial remission; M, male; MPO, myeloperoxidase; N, no; N/A, not applicable; NED, no evidence of disease; NS, not specified; RT, radiotherapy; TdT, terminal deoxynucleotidyl transferase; Y, yes.
MS occurs in approximately 2% to 8% of patients with AML. It can serve as the primary indicator of relapse in individuals previously treated for AML and currently in remission. 8 Among the 14 patients, 9 had a history of AML.9-16 Of these, 2 patients had concurrent BM-I alongside EM-AML,9,14 while 5 patients only exhibited EM-AML without BM-I.10-15 In 2 cases, a definitive diagnosis for BM-I was not established.10,16
MS may present simultaneously with AML in 16% to 36% of cases, or develop post-AML diagnosis in up to half of cases. Furthermore, MS may precede the onset of AML by several months or even years in about 26% of instances 8 ; this type is referred to as isolated, primary, or nonleukemic MS. 7 Five patients had no previous HM history6,17-20; among them, one had EM-AML without BM-I, 20 leading to a diagnosis of isolated MS, 3 patients had concurrent BM-I,6,18,19 whereas in one case, no specific tests to determine BM-I were carried out. 17 Compression signs, severe pain, and abnormal bleeding are common symptoms seen with myeloid tumors, which vary based on their size and location. 8 When MS affects the temporal bone, symptoms become nonspecific and can be mistaken for conditions such as AOE or NOE, so t is crucial to include MS in the list of potential diagnoses when malignancy is strongly suspected. 6 Across 14 patients, the most frequent symptoms were otalgia,6,9,11-15,17-19 facial asymmetry,9-11,15-19 hearing decrease and hearing loss.6,9,12,15,19 Additionally, ear fullness,6,11,13 otorrhea,13,15 and a range of less frequent and atypical symptoms such as headaches, absence seizures, 10 orbital pain and proptosis, 19 due to MS orbital involvement, tinnitus, 14 convulsions, and temporal swelling. 20 Our patient had a wide range of symptoms, varying from right-sided otalgia, otorrhea, tinnitus, reduced hearing, and ear fullness to vertigo and facial asymmetry. The clinical manifestations led to a diagnosis of NOE, a condition predominantly observed in patients above the age of 60, especially those with compromised immune systems attributable to diabetes, a history of cancer, chemotherapy treatments, or HIV infection. 21 Characterized by its aggressive infection of the ear canal with potential to invade adjacent bone structures, NOE typically presents with otalgia, hearing loss, malodorous otorrhea, and proliferative lesions within the ear canal. 22 Neoplasms particularly can mimic NOE clinically; thus, histopathological examination of granulation tissue biopsies is essential in all patients. Additionally, CT imaging can effectively delineate lesion boundaries and assist in ruling out other differential diagnoses. 23 At the outset, due to noncompliance from the patient, the option of conducting an EAC biopsy with local anesthesia was not feasible. Furthermore, the considerable risks associated with general anesthesia warranted a temporary deferment of this approach. In NOE management, surgery may be needed to remove dead tissue and bone if the infection persists. 22 However, the patient’s poor response to treatment, coupled with her AML-M5 history in further evaluation, demonstrated underlying problem and necessitated surgical intervention under general anesthesia to remove necrotic and inflammatory tissues and take EAC biopsies for histopathology and IHC, which came positive to MPO and CD99, establishing MS diagnosis. In diagnosing MS, IHC is considered a crucial test for confirmation; common positive markers include CD68/ KP1, MPO, CD 117, CD 99, CD 68/PG-M1, lysozyme, CD34, TdT, CD56, CD61, CD30, glycophorin, and CD4. For myeloid differentiation tumors, CD13, CD33, CD117, and MPO are prevalent markers, while in monoblastic differentiation tumors, CD14, CD163, and CD11c are commonly used. Adding B- and T-lineage markers like CD20, CD 45RO, CD79a, and CD3 to exclude other differential diagnoses. 8 In 11 patients, IHC was performed, and was detailed in 106,9,10,12-14,18-20; the most common markers, listed in order, were as follows: MPO (80%), CD117 (50%), CD34, CD43, CD68 (40% each) CD3, CD20, CD99 (30% each), LCA, TdT, lysozyme (20% each), CD2 CD11c, CD13, CD16, CD33, CD45 vimentin, HLA-DR (10% each). Recent studies indicate a misdiagnosis rate for MS ranging from 25% to 47%, 7 with common alternative diagnoses including lymphoma, undifferentiated cancer, malignant melanoma, extramedullary hematopoiesis, and inflammation during the initial stages. 8 Misdiagnosis in these cases often resulted from inadequate assessment of MS lesion immunophenotyping. 2
CT and magnetic resonance imaging (MRI) scans are commonly utilized for tumor localization and are valuable in differentiating MS from other conditions such as hemorrhage or abscesses. 8 Depending on the anatomical sites involved, MRI is more sensitive for assessing central nervous system, spinal, and musculoskeletal lesions, while CT scans are ideal for evaluating soft tissue.² A single-center study has elucidated that 18Fluoro-deoxy-glucose positron emission tomography/computed tomography (18F-FDG-PET/CT) is instrumental in identifying extramedullary disease in patients with histologically-confirmed EM-AML. This imaging modality is recommended for use at the point of diagnosis, prior to the commencement of treatment, for early assessments, as well as during CR, to appraise the efficacy of the therapy administered. 24 Nevertheless, financial constraints prevented the performance of both MRI and PET CT scans. A full-body CT scan of the patient showed no abnormalities, ruling out other MS locations. It is recommended to conduct bone marrow biopsy or aspiration to eliminate the possibility of other HM. 7 We performed bone marrow aspiration, which came unremarkable with no evidence of AML or any other HM. The choice of treatment is contingent upon the disease’s localization, whether it occurs at the primary diagnosis or during a relapse, the performance status, and the age of the patient. 7
Duminuco et al 25 documented that in eligible patients with EM-AML, intensive systemic chemotherapy based on anthracycline and cytarabine remains the preferred treatment approach, whether or not BM-I is present.
A retrospective study involving 51 patients with MS demonstrated that initiating first-line allogeneic hematopoietic stem cell transplantation (allo-HSCT) early in the progression of the disease is a viable treatment option. 26 All patients received chemotherapy, and just 2 received subsequent local RT.10,13
Consolidation therapy for MS, regardless of concomitant AML, should follow the standard approach used for AML. Higher risk patients, based on cytogenetic and molecular profiles, should undergo allo-HSCT preferably in first remission, while consolidation chemotherapy is recommended for lower risk or unfit-for-allo-HSCT patients. 2
Bakst et al 27 propose rRT as a consolidation treatment for isolated MS following chemotherapy.
Because of the concerns about potential recurrence due to the delay in RT appointment, we started directly with the consolidation chemotherapy, which involved administering 4 cycles of cytarabine, which is the selected regimen according to ELN recommendations for younger patients (18-60/65 years). 28
Anthracyclines, commonly used in AML treatment, have a range of side effects. Short-term effects include systemic symptoms such as fever, asthenia (79.5%), and sleep disorders, along with gastrointestinal issues (vomiting, nausea, mucositis, etc) and dermatological toxicity (nail changes, alopecia). 29 Long-term use can lead to chemotherapy-induced cardiotoxicity, with the risk of heart failure increasing with dosage (3%-5% at 400 mg/m² to 48% at 700 mg/m²). Patients under 5 or over 65, those with prior chest radiation, pre-existing heart disease, or known cardiovascular risks are at higher risk. 30
Cytarabine can cause severe side effects such as myelosuppression, leading to pancytopenia. Other effects include irritation at the injection site, digestive issues, and skin inflammation. Rare but serious effects include severe allergic reactions and neurological toxicity. Heart-related issues such as chest pain and pericarditis can also occur. 31
A recent cohort study on 1583 patients with AML found similar event-free survival (EFS) times between clinically-diagnosed EM-AML patients and non EM-AML patients (7.1 and 8.4 months, respectively), but for 38 histologically-confirmed EM-AML patients the EFS time was significantly decreased (3.6 months). Overall survival rates for clinically- and histologically-diagnosed EM-AML patients were significantly lower than non-EM-AML patients (14, 8.7, and 26.2 months, respectively). 32
Among the 14 patients, 7 had no evidence of disease,10-13,19,20 4 showed partial response,6,9,17,18 and 3 died after therapy.14-16 Yang et al 14 documented bone marrow suppression followed by a deadly infection and septic shock as the cause of death. Patients undergoing cytarabine-dependent therapy necessitate consistent monitoring through liver function tests, complete blood count, differential WBC count, platelet count, serum creatinine, blood urea nitrogen, and serum uric acid. 31
Following treatment, patients with MS undergo routine physical examinations and peripheral blood tests to monitor ongoing remission status. Marrow reassessment will be conducted if recurrence is detected. Posttreatment imaging is considered for patients with MS, but additional scans are not typically conducted if initial tests reveal no abnormalities. 27
In conclusion, although MS has various common locations, temporal bone MS—despite its rarity—should be considered in patients with an AML history who present with otalgia, facial palsy, or hearing decrease as the most common symptoms, as per our review. More comprehensive and extensive reviews should be conducted to enhance the understanding of this rare presentation of MS.
Finally, a multidisciplinary approach is key in managing temporal bone MS. Otolaryngologists detect early symptoms and manage surgical aspects, pathologists confirm diagnosis through biopsy analysis, while hematologists guide chemotherapy and monitor treatment response. This collaborative approach ensures accurate diagnosis and optimal disease management.
Footnotes
Acknowledgements
None.
Authors’ Contributions
ON: conceptualization, investigation, methodology, project administration, writing—original draft, visualization, and writing—review & editing. MA, ML, and AA: data curation, resources, writing—original draft, and writing—review & editing. AS and AY: supervision, validation, and final approval of the manuscript.
Consent
Written informed consent was obtained from the patient for publication of this report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request. Patient data has been de-identified to protect patient privacy and confidentiality.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
