Abstract
Introduction
Over the last few decades, the management of locally advanced laryngeal cancer has shifted toward laryngeal preservation strategies to enhance patients’ quality of life without sacrificing oncologic outcomes. In many settings, this involves definitive radiation therapy (RT) or chemoradiation (chemoRT) and avoiding surgery.1-4
These recommendations are based on 2 randomized control trials, which confirmed equivalent oncologic outcomes when treating these patients via a nonsurgical approach.3,5 However, in some situations, advanced laryngeal cancers are less amenable to laryngeal preservation approaches, such as patients with a fixed larynx and poor swallowing at the time of diagnosis or for patients with bulky disease that may be less responsive to RT.
While oncologists commonly use National Comprehensive Cancer Network (NCCN) guidelines to develop cancer treatment plans, the guidelines for advanced-stage laryngeal cancer provide a variety of treatment pathways. For patients with T3-4N0-1 glottic and supraglottic laryngeal cancer, current NCCN guidelines recommend either definitive chemoRT or total laryngectomy with neck dissection (ND). 6
Adjuvant RT is generally recommended in cases of adverse features, but observation following surgery is a potential option in cases with no adverse features. Many practitioners recommend proceeding with an adjuvant chemoRT approach for these advanced laryngeal cancers, even in the absence of adverse features, particularly in patients with N1 disease.7-10
Currently, there are no prospective studies directly comparing the outcomes of patients with advanced T-stage laryngeal cancer undergoing laryngectomy with adjuvant radiation versus those without adjuvant radiation. In order to investigate this particular clinical scenario, we utilized the Surveillance, Epidemiology, and End Results (SEER) database for patients with advanced T-stage nonmetastatic laryngeal cancers (T3-4N0-1) to compare outcomes between those who underwent adjuvant radiation and those who underwent laryngectomy alone. Specifically, we sought to compare cancer-specific and overall survival outcomes in these 2 cohorts, stratified by nodal status and administration of adjuvant radiation treatment.
Methods
Study Population
The study population was extracted from the SEER 17 database of the US National Cancer Institute. This cancer registry covers an estimated 26.5% of the US population, consisting of records from Alaska Native Tumor Registry, Connecticut, Georgia, California, Hawaii, Iowa, Kentucky, Louisiana, New Mexico, New Jersey, Seattle-Puget Sound, and Utah. The study was exempt from the Institutional Review Board approval since SEER 17 is a publicly available database with no personal identifiers.
The study population included adult (≥18 years old) patients with T3-T4 and N0-N1 primary laryngeal squamous cell carcinoma (SCC) laryngeal cancer diagnosed between 2004 and 2019. Staging was defined by the patient contemporary American Joint Committee on Cancer (AJCC) sixth to eighth edition guidelines. Laryngeal cancer was defined as primary site International Classification of Diseases for Oncology (ICD-O-3) Histology codes: C32.0-Glottis, C32.1-Supraglottis, C32.2-Subglottis, C32.3-Laryngeal cartilage, C32.8-Overlapping lesion of larynx, and C32.9-Larynx, Not otherwise specified (NOS). Squamous cell carcinoma was defined as ICD-O-3 histological type codes 8050-8089. Only first primary, previously untreated SCCs were included. Only patients receiving total laryngectomy, total laryngectomy not otherwise specified, radical laryngectomy, and pharyngolaryngectomy were included. Patients receiving RT alone, chemotherapy, and RT prior to surgery were excluded. Other covariates included for analysis were: age, race, sex, marriage status, average county income, county rurality, time to treatment, year of diagnosis, and ND status. Cases with missing or unknown values for any included variables were excluded. The final cohort meeting the inclusion criteria consisted of 1820 patients.
Intervention and Outcome Assessment
The primary intervention examined in this study was treatment type, and patients were split into 2 cohorts: either surgery then RT, or surgery without RT. The primary outcome examined was cancer-specific survival, a net survival measure representing cancer survival in the absence of other causes of death. All cases were censored after 5 years (60 months) of follow-up.
Statistical Analysis
Baseline demographic and clinical data were analyzed using 2-sided Pearson’s chi-square tests for categorical variables, while paired T-tests, means, and standard deviations were calculated for continuous variables. Kaplan-Meier (KM) survival curves were constructed and stratified by nodal status. KM survival curves were compared across treatments using log-rank, Breslow, and Tarone-Ware tests. To analyze the association between treatment and cancer-specific survival, we performed uni- and multivariate Cox proportional hazards regressions stratified by ND status and adjusted for both the statistically relevant and all included covariates. The proportional hazards assumption was verified using log-minus-log plots and the time-dependent covariate test.
To further remove the effect of baseline imbalance in treatment cohorts, propensity score-matching (PSM) was used to compare overall survival for the different treatment groups. Propensity scores were computed using logistic regression with overall survival as the primary outcome, as a function of age, sex, race and ethnicity, T-stage, N-stage, primary site, year of diagnosis, and use of adjuvant RT. All statistical analyses, with exception to PSM, were performed using SPSS Statistics version 28 (SPSS Inc, an IBM Company, Chicago, IL, USA) with statistical significance defined by a P-value <.05. The PSM procedure and analysis were performed with RStudio (version 2022.12.0, Posit Software, PBC, Boston, MA, USA) using the MatchIt and Survminer packages.
Results
Patient Characteristics
We identified 1820 patients with T3-T4 and N0-N1 laryngeal SCC from 2004 to 2019 who met the inclusion and exclusion criteria (Figure 1). This cohort was 83.0% male and 17.0% female; 68.3% white, 17.9% black, 10.2% Hispanic, and 3.5% other races; and had an average age of 63 years. Regarding cancer stage, 34.5% was T3 and 65.5% T4; 81.5% was N0 and 18.5% was N1. 75.3% of patients received a ND, while 24.7% did not undergo ND or had an unknown ND status. Negative/unknown ND status was more frequent in patients not receiving adjuvant RT than those who did (30.8% vs 19.3%, P < .001). Patients who underwent ND had significantly higher odds of being N1 (OR 2.9, P < .05). When examined by nodal status, almost all N1 patients received ND (300/339), while 72% of N0 patients (1073/1481) received ND.
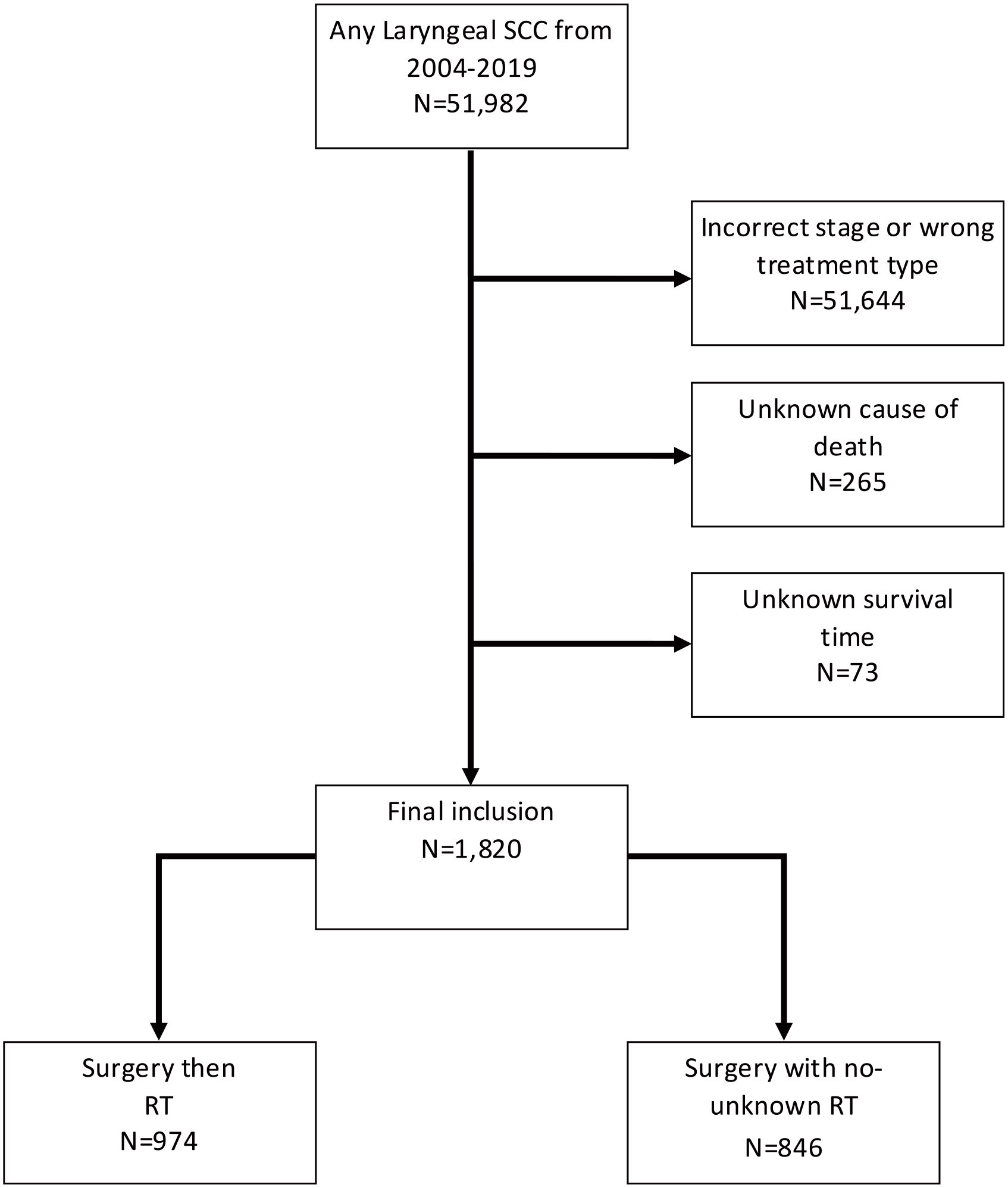
Diagram illustrating the flow of patients through the inclusion and exclusion criteria, and into the treatment groups.
Regarding treatment group, 53.5% (974) of patients received surgery followed by RT, while 47.5% (846) received surgery with no or unknown RT. Further breakdown of relevant demographic data and clinical diagnostic information are reported in Table 1. Although there were statistically significant differences between the treatment groups for certain characteristics, such as race and ethnicity, age, T stage, N stage, and neck dissection status, these were controlled for within the multivariate survival analysis.
Characteristics of Patients with T3/4N0/1 Laryngeal Cancer.
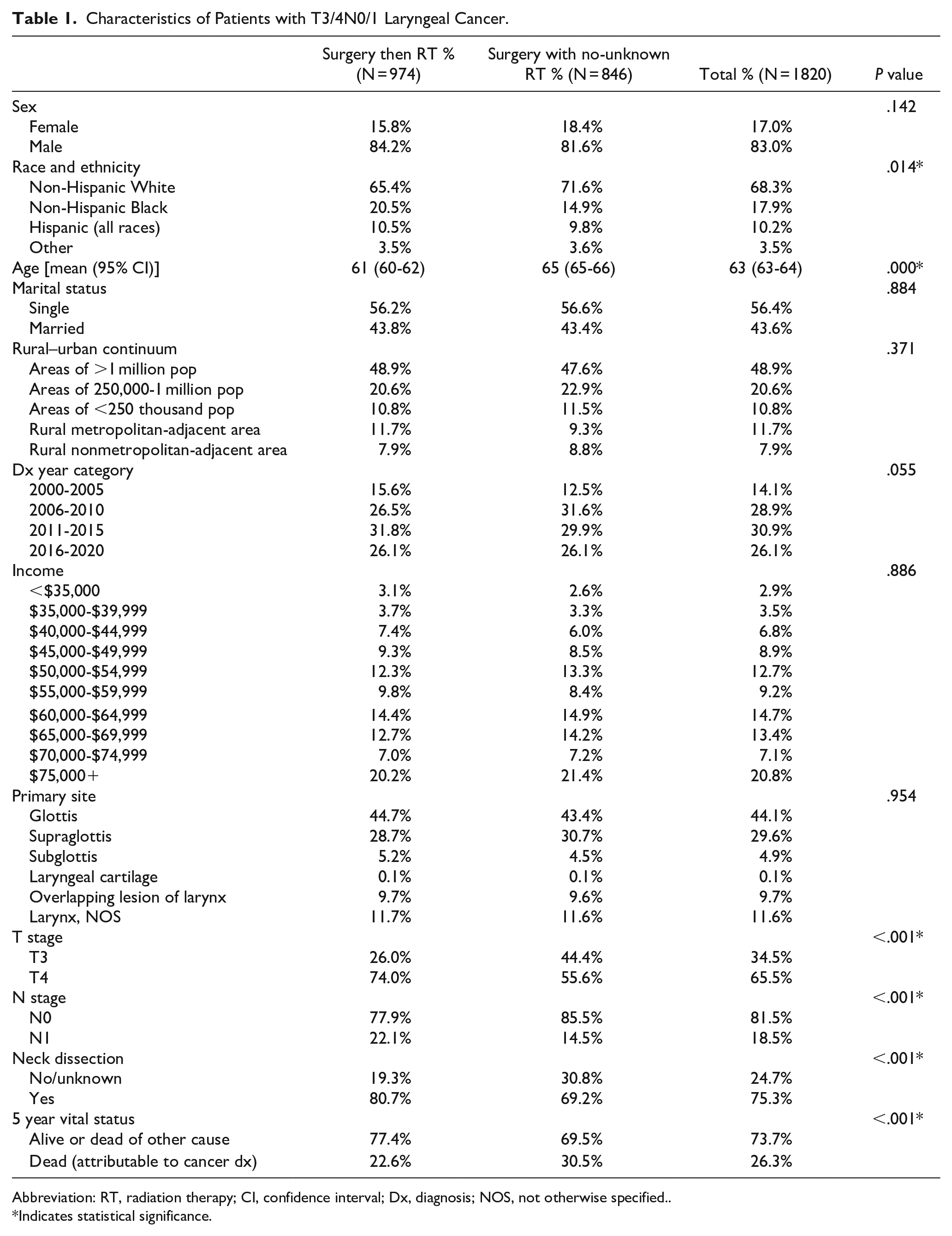
Abbreviation: RT, radiation therapy; CI, confidence interval; Dx, diagnosis; NOS, not otherwise specified..
Indicates statistical significance.
Survival Analyses
Kaplan-Meier survival analysis
For N0 patients, those who received adjuvant radiation had a significantly longer mean survival time than those who received surgery without RT [Surgery with RT: 51.62 months (95% CI 50.36-52.87 months), Surgery without RT: 46.07 months (95% CI 44.44-47.70)]. Similarly, for N1 patients, those who received adjuvant RT had a significantly longer mean survival time than those who received surgery without RT [Surgery with RT: 46.80 months (95% CI 44.08-49.52 months), Surgery without RT: 36.24 months (95% CI 31.60-40.89)]. Univariate survival analysis is summarized in Table 2. All 3 measured survival tests found the survival distributions for the interventions were statistically significantly different with improved survival for patients who received adjuvant RT (Figure 2).
Survival Analysis of Cancer-Specific Mortality of T3/4N0/1 Laryngeal Squamous Cell Carcinoma by Nodal Status and Treatment Type.
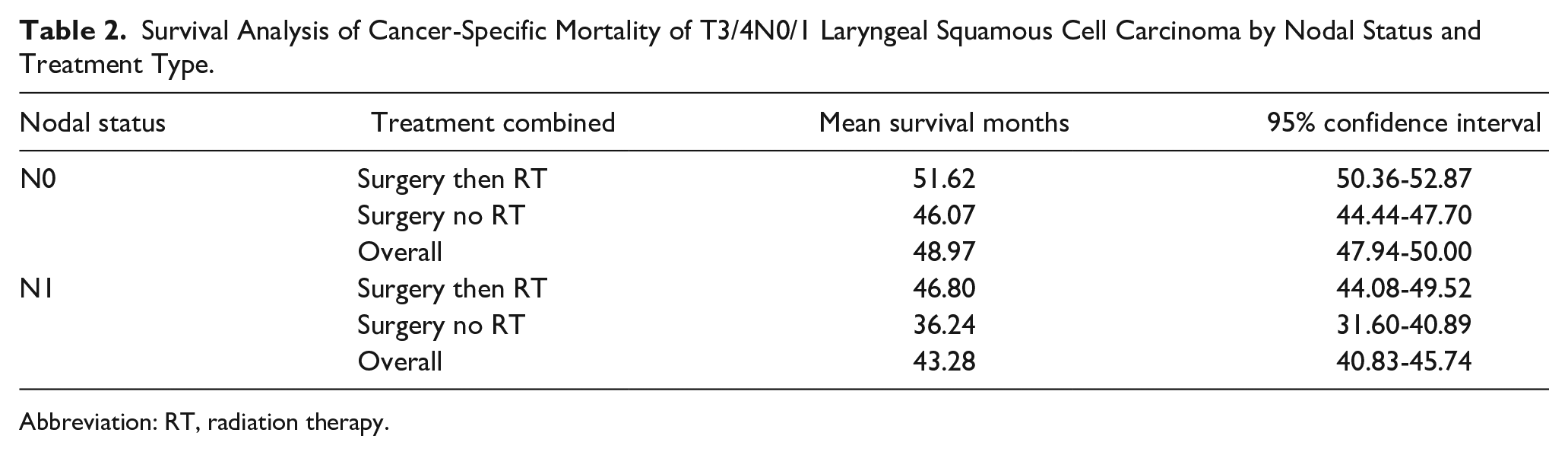
Abbreviation: RT, radiation therapy.
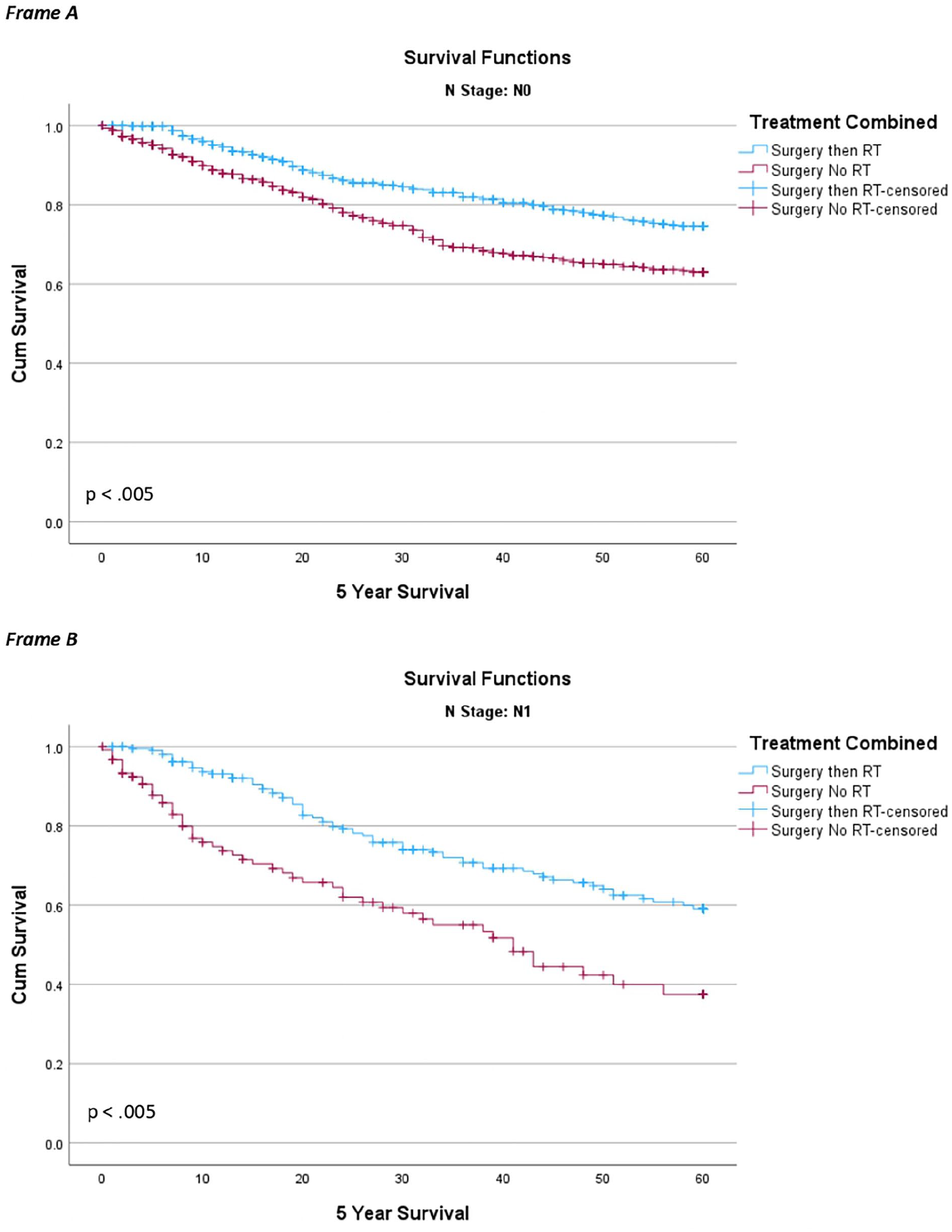
Kaplan-Meier 5-year survival curve of cancer-specific mortality by treatment type. (Frame A) N0 patients, (Frame B) N1 patients.
Cox proportional hazards survival analysis
The results of uni- and multivariate analyses using Cox proportional hazards regression are summarized in Table 3. After adjusting for included covariates (listed above), N0 patients who received laryngectomy and who did not undergo adjuvant radiation had a 47% higher risk of cancer-specific death than patients receiving adjuvant RT (adj. HR 1.47, 95% CI 1.18-1.84). N1 patients who did not undergo adjuvant radiation had a 90% higher risk of cancer-specific death than patients receiving RT after surgery (adj. HR 1.90, 95% CI 1.27-2.84). These results were statistically significant (P < .05) even after adjusting for statistically relevant covariates and stratifying by nodal status. After adjusting for propensity scores, adjuvant RT carried a significant overall survival benefit over no adjuvant RT (HR 0.73, 95% CI 0.60-0.87; P < .001). This is plotted in Figure 3, demonstrating significant propensity score adjusted improvement in survival outcomes for total survival (A) and 5-year survival (B).
Adjusted Hazard Ratio for Survival by Treatment Type T3/4N0/1 Laryngeal Squamous Cell Carcinoma by Nodal Status and Treatment Type Using Cox Proportional Hazard Model.

Abbreviations: adj. HR, adjusted hazard ratio; CI, confidence interval; HR, hazard ratio.
Adjusted for age, T stage, and neck dissection status.
Adjusted for age, sex, race, marital status, rural-urban environment, year of diagnosis, income, T stage, neck dissection status, and diagnosis year.
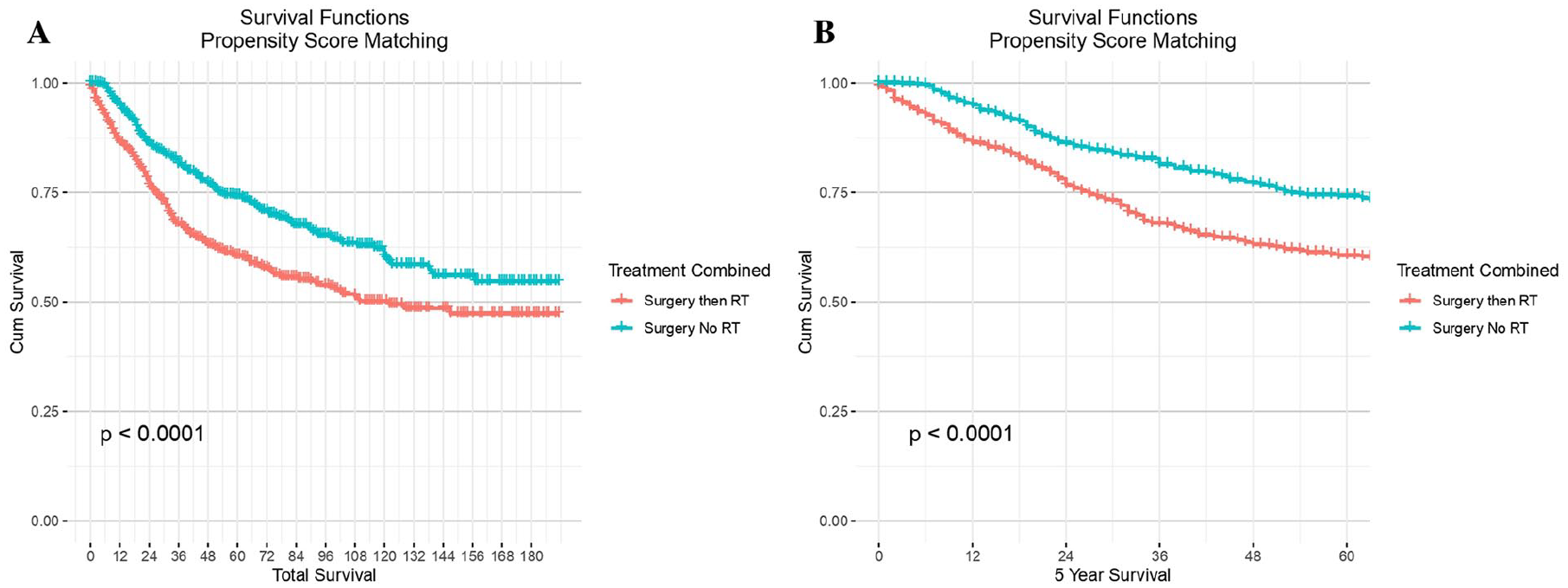
Kaplan-Meier curve of overall survival by treatment type following propensity score matching. (Frame A) Total survival, (Frame B) 5-year survival.
Discussion
This retrospective study investigated the outcomes of 1820 patients with locally advanced squamous cell carcinoma of the larynx undergoing either surgery followed by RT or surgery alone. The study found the addition of adjuvant RT improved cancer-specific and overall survival in patients with N0 and N1 laryngeal carcinoma, regardless of age, T stage, ND status, and various sociodemographic factors. Adjuvant RT also showed significant benefit in terms of overall survival using the propensity score method to adjust and compare these background factors in the 2 groups. The risk of cancer-specific and overall death was shown to be higher in patients with surgery alone versus those who underwent adjuvant RT.
The current guidelines for managing locally advanced laryngeal carcinoma focus on preserving laryngeal function, emphasizing voice production, airway patency during breathing, and airway occlusion during swallowing, without compromising oncological control. 10 According to these guidelines, single-modality treatments such as transoral laser surgery or RT alone may suffice for early-stage disease, while locoregionally advanced disease generally requires either total laryngectomy with ND or definitive concurrent chemoradiation. 11 However, the indications for adjuvant RT for patients undergoing surgery with bulky laryngeal disease, but otherwise limited nodal disease and few pathologic risk factors are poorly defined.12-14
Kim et al evaluated the role of adjuvant RT for locally advanced, node-negative laryngeal cancer at a single tertiary referral center and concluded that there was no clear benefit of postadjuvant radiation therapy on overall survival (OS) or local control rate (LCR). 15 However, this study was limited by the asymmetric distribution of patient factors in the 2 groups, which was not adjusted for in the analysis. Additionally, the smaller study sample size (N = 60) may not have had the statistical power to detect the differences in survival found in the present analysis (N = 1820).
In a National Cancer Database cohort study of 1460 patients with pT3N0 and pT4aN0 laryngeal cancer, Graboyes et al found that postoperative RT versus close observation increased overall survival in surgically managed pT3aN0 and pT4aN0 laryngeal SCC on multivariable analysis; however, the survival benefit in pT3aN0 was not statistically significant (pT4aN0 aHR = 0.58, 95% CI = 0.46-0.73; pT3aN0 aHR = 0.88, 95% CI = 0.64-1.21). 16 Interestingly, the confidence interval for the pT3aN0 cohort was more than 2 times greater than the pT4aN0 cohort. They also found that nearly 50% of patients with pT4aN0 laryngeal SCC and negative surgical margins did not receive standard-of-care adjuvant RT, which mirrors the findings in our cohort, where only 60% of T4 tumors received adjuvant RT. The results of this study, which did not account for N1 disease, in combination with our findings, highlight the critical need for national provider education to improve RT utilization in locally advanced laryngeal SCC in the United States. While no international studies exist for comparison, process improvement strategies are warranted at the national level to increase adherence to NCCN guidelines.
Similarly, in a study of 369 patients with T3-4aN0 glottic laryngeal SCC with negative margins, Zhou et al observed no difference in the cancer-specific survival rates between patients who received postoperative RT and those who did not (62.5% vs 69.5%, P = .074). 17 However, this single-institution study did not report the distribution of clinicopathological factors, including clinical stage, between the 2 cohorts and was limited by its small sample size, wherein only 18.1% (N = 57) of the entire cohort underwent postoperative RT.
Contrary to these results, a meta-analysis was conducted in 2018 investigating the role of postoperative RT for patients with stages III-IV laryngeal carcinoma that included 7 published studies containing 2007 patients. 18 This study found that the addition of postoperative RT in this pooled dataset conferred significantly better survival rates [OS HR: 0.67 (95% CI 0.56-0.79); DFS HR: 0.72 (95% CI 0.53-0.99); LCR HR: 0.29 (95% CI 0.09-0.99)]. 18 Similar to the results of our study, this meta-analysis showed that postoperative RT improved the prognosis for both pT3 and pT4. These findings, when considered with the results of the present study, suggest that postoperative RT may increase 5-year survival in locally advanced laryngeal cancer and should be evaluated further.
Surgical management of clinically negative neck lymph nodes (N0) remains an area of ongoing study. 19 Patients with early-stage (T1-T2) glottic lesions are perceived to have a lower risk for occult nodal metastasis and, thus, do not require prophylactic ND. By contrast, patients with locally advanced (T3-T4) glottic lesions or any (T1-T4) supraglottic primary site have a relatively higher chance of occult nodal disease and have traditionally undergone ND20,21,22. In our analysis, nearly a quarter of the total cohort did not have NDs at the time of laryngectomy. Of note, negative/unknown ND status was more frequently observed in patients not receiving adjuvant RT than for patients who did (30.8% vs 19.3%, P < .001). The reasons for not undergoing ND are not readily available in the database and does suggest potential provider selection biases warranting further study to determine need for educational outreach to surgical providers to optimize dissemination of guideline recommendations supporting ND. Regardless, the results from our analysis suggest that adjuvant RT appears to confer a survival benefit regardless of ND and nodal status. We, therefore, advocate for ND in surgically managed advanced laryngeal carcinoma.
Limitations of this study include those inherent to a retrospective study and registry design, which limit the generalizability of our results. These include inaccuracies in cancer staging, missing adjuvant chemotherapy and patient history data, including smoking status, and incomplete clinical data, such as site of locoregional failure, and radiation planning information. As well, the unavailability of comprehensive pathology report information limits the ability to assess the impact of specific postsurgical risk factors on patient outcomes. Regarding treatment data, the SEER database has high specificity for patients who received treatment but only moderate sensitivity. Thus, if the data indicate that a patient received treatment, it is highly likely that they did, but if the data show that a patient did not receive RT, it is uncertain whether the patient truly did not receive it or if the registry missed it. As a result, the treatment variables used were labeled as either “yes” or “no/unknown” to reflect this limitation. Nevertheless, even if some patients who received treatment were classified in the “no/unknown” category, it would only reduce the probability of detecting significant differences between groups.
The study is also limited by the inability to adjust for selection bias in which patients received adjuvant RT. Patients who were not offered adjuvant RT may differ from those who were not offered adjuvant RT, such as a higher rate of worse pretreatment laryngeal function in the latter group. Patients who do not receive adjuvant RT may be more likely to have worse social support and follow-up, which can adversely affect survival outcomes and other metrics 23 . Additionally, studies have shown SEER registry data to underestimate rates of radiotherapy receipt in certain disease subsites. 24 Further, registry studies assessing adjuvant RT are susceptible to immortal time bias, which overestimates the effect size of adjuvant therapies as patients must survive postoperatively to start and finish radiation. 25 This is further compounded by the fact that registries do not contain data on specific postoperative RT timing intervals, another quality indicator in head and neck cancer treatment. Future studies should prospectively evaluate survival outcomes to mitigate these biases and confirm our findings.
While the incidence of laryngeal cancer has decreased in the United States, mortality has not decreased at a similar rate, resulting in an increased case-fatality rate overall.26,27 Therefore, improving treatment decision-making is vital. We hope this study increases awareness of the beneficial role of adjuvant RT in advanced laryngeal cancer to potentially curb this trend and improve patient outcomes in the future.
Conclusions
The present investigation is the first to directly examine primary surgery’s survival outcomes and observation versus adjuvant RT in patients with T3-4N0-1 laryngeal cancer. This national database analysis provides additional evidence suggesting that postoperative RT decreases the risk of cancer-specific and overall death in patients with T3-4N0-1 laryngeal carcinoma regardless of demographic features, laryngeal subregion, or pathologic stage. Although these findings support the practice of adjuvant radiation in this patient population, providers must also communicate the expected risks of toxicity associated with RT, which should be weighed against potential survival benefits. Need for optimal personalized treatment is emphasized by the fact that survival outcomes for advanced-stage laryngeal cancer have not improved over the past several decades, leading to increased national case-fatality rates. Better dissemination and implementation of vetted multidisciplinary treatment pathways will be critical to serving this need.
Footnotes
Acknowledgements
The authors acknowledge The UTHSC Center for Cancer Research for its invaluable support throughout the entirety of this project.
Data Sharing
Research data are stored in an institutional repository and will be shared upon request to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
