Abstract
Introduction
Hearing loss is one of the major contributors to global disability, affecting around 466 million people. This includes one-third of adults aged 65 and above, as reported by the World Health Organization in 2018. 1 Hearing loss is often associated with an increased prevalence of several health problems, 2 in addition to placing a burden on people and society. Studies indicate a clear connection between hearing impairment and cognitive decline in elderly people. 3 Therefore, effective intervention can have a substantial influence on their over quality of life. 4 Currently, no established therapy can restore pathological damage to the cochlea. As a result, the primary approach is to concentrate on prevention and rehabilitation. 5 Hearing aids (HA) are the first-line approach for addressing hearing loss. Nevertheless, people with severe to profound sensorineural hearing loss are unlikely to see major benefits. Cochlear implantation (CI) is the most appropriate option for such situations. 4 The global incidence of cases with CI has risen because of the expansion in the candidacy criteria to both children and adults who might benefit from it.6,7 Nowadays, bilateral cochlear implantation (BiCI) is now a recognized method for rehabilitating adult hearing. 8 It offers many advantages, including improved sound localization, increased sound detection in quiet settings, and better speech comprehension in noisy backgrounds.9-13
CI surgery is conventionally done under general anesthesia (GA). However, a minimally invasive safe option combined with local anesthesia (LA) and deep sedation is an alternative anesthesia technique. LA with sedation is used for many otological procedures such as mastoidectomy and tympanoplasty. However, its application for CI surgeries is limited. 14 Previous studies of CI under LA have demonstrated both the feasibility and safety of this option. Some advantages of this technique include improved intraoperative patient monitoring, cost reduction, and intraoperative sound perception testing compared to GA.15-18 Recent studies have focused on postoperative cognitive dysfunction (POCD) and have shown that GA poses a higher risk for the development of POCD compared to conscious sedation, thus decreasing the neurocognitive decline risk for older patients should be addressed in a multidisciplinary approach.19,20 Multiple studies have conducted comparisons between CI performed under GA and LA. These studies have shown that CI under local anesthesia with conscious sedation (LACS) significantly decreases both the duration of the operation and the length of hospital stay in comparison to CI performed under GA.16,17,21,22 Moreover, one study compared the cost of drugs used in GA and LA and found that drugs used in LA cost less when compared to GA. 16 Patient satisfaction following CI under LACS has been measured in some studies with a result of a positive patient experience during CI under LACS.22,23
The complications in CI were comparable between using LA and GA. The most frequently reported side effect of CI performed under LA is vertigo, then wound infection. Other complications including transient facial weakness, confusion, and tinnitus have been reported as well. However, there is no difference in their incidences when compared to GA. 24 Despite the comparable benefits gained among young and older populations (60 years old and above), only 5% of elderly patients who meet the candidacy of CI pursue surgery. This is due to various reasons including concerns regarding the increased possibility of complications, limited awareness, and difficult access to CI centers. 25
In the environmental aspect, the healthcare sector is responsible for global greenhouse gas (GHG) emissions ranging from 5.2% to 10%.26,27 Entrapment of (GHG) in the atmosphere is one of the major causes of global warming. Anesthetics have global warming potential up to 2,000 times greater than carbon dioxide. 28 Inhaled anesthetics contribute to 50% of emissions during the perioperative period. Studies recommend using total intravenous anesthesia (TIVA) or regional anesthesia when clinically appropriate due to their lower emissions capacity. 29 Thus, using LA with sedation in CI surgery could reduce these emissions.
Furthermore, an advantage of having CI surgery under LA is the possibility of fitting and activating the external device immediately after the surgery, resulting in an earlier hearing experience and rehabilitation. Therefore, we conducted this study to review the feasibility of simultaneous BiCI under LA with sedation in adults and to assess the surgical and audiological outcomes.
Materials and Methods
This is a retrospective chart review conducted on all adult patients who underwent simultaneous BiCI surgery under local and regional anesthesia with sedation at our tertiary CI center from 2018 to 2024. The inclusion criteria included adult patients (18 years or older) who were diagnosed with bilateral severe to profound sensorineural hearing loss, had minimal or no benefit from HA, and expressed a preference for the combined LA with sedation technique for simultaneous BiCI surgery. Exclusion criteria include patients with inner ear malformations, those who were lost to follow-up, and those with a history of adverse reactions to drugs involved in the LA procedure. Patients’ procedure who converted to GA will be excluded as well. Informed consent was obtained from the patients explaining the procedure and the type of anesthesia used with its advantages, disadvantages, and possible complications. Data were collected on related characteristics such as demographic data, onset of hearing loss, clinical and radiological evaluation, surgical steps and findings, postoperative recovery, and audiological outcomes. We assessed the feasibility of simultaneous BiCI surgery under this combined anesthesia technique through patients undergoing the surgery without complications and device activation outcomes postoperatively and compared the hearing experience with CI to HA through a questionnaire. The study protocol was reviewed and approved by the institutional review board (Reference No. 24/1167/IRB).
Anesthesia Protocol
General steps that were performed in all participating patients:
Premedication
These drugs were given 30 minutes preoperatively.
I.V. Glycopyrrolate: as a bolus injected slowly at a dose of 4 µg/kg
I.V. Dexmedetomidine: as a slow bolus injected over 10 minutes at a dose of 0.3 µg /kg
Intraoperative Monitoring
All patients were warmed with hot-air warming blankets and had ECG, NIBP, SPO2, EtCo2, and temperature probes attached in the operating room (OR). Bi-spectral index (BIS) and standard monitoring were used to detect and maintain deep sedation (a BIS of 70-60) throughout the procedure.
Sedation Protocol
Continuous infusion of 25 to 100 µg/kg/min of propofol was given. Followed by 50 µg/kg boluses to maintain a BIS of 60 to 70. After the desired BIS was achieved, Ketamine 50 mg was administered for analgesia via NMDA receptor saturation. Patients’ faces were exposed throughout the procedure (after cleaning and draping the operative site).
Airway Management
A nasal cannula supplied 2 to 3 L/min oxygen to spontaneously breathing patients. End-tidal Co2 (EtCo2) monitoring was achieved using an EtCo2 line attached to the nasal cannula in a way that did not disturb the patient. In case of airway obstruction of any degree (absent EtCo2 wave, noisy breathing, or abnormal breathing pattern requiring chin lift or jaw thrust), the nasopharyngeal airway was used. If an open airway is still not possible, a flexible laryngeal mask airway of suitable size was used to keep it patent. Equipment for endotracheal intubation was kept on hand for emergencies.
Local Anesthesia
Three to 5 minutes following ketamine injection, and with a stable BIS reading of 60 to 70 throughout that time, a local anesthetic block was carried out.
It included the following 2 techniques:
Ultrasound superficial cervical plexus block: To block the greater auricular and lesser occipital nerves
Located on the posterior border of the sternocleidomastoid muscle (SCM), at the C6 level, and superficial to the middle scalene muscle. The skin was sterilized, and the transducer was positioned on the side of the neck, above the SCM at its midway. After identifying the SCM, the transducer was shifted until the posterior edge was centered on the screen, finding the brachial plexus or interscalene groove between the anterior and middle scalene muscles. The cervical plexus appears as a tiny cluster of honeycomb-shaped hypoechoic nodules underneath the interscalene groove. Then, the needle was inserted and placed adjacent to the plexus. In-plane approach was used from the lateral side of the neck. Following negative aspiration, 1 to 2 mL of Bupivacaine 0.25% was injected to confirm the proper injection site. The remainder of the Bupivacaine (5-15 mL) was administered to envelop the plexus. Figure 1 shows an ultrasound superficial cervical plexus block.
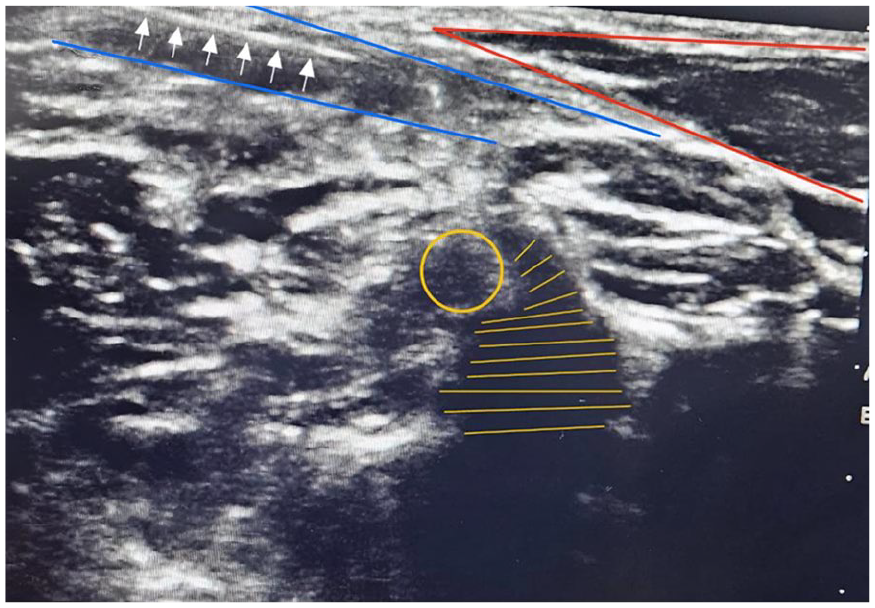
Ultrasound superficial cervical plexus block. Red lines: sternocleidomastoid muscle. White arrows: needle shaft. Blue lines: superficial (upper) and deep (lower) layers of cervical fascia. Yellow: C4 vertebra (horizontal lines) and C4 nerve root (circular).
Local anesthetic infiltration
Lidocaine 1% with 1:100,000 adrenaline was injected in the external ear canal beneath the superior and posterior wall skin and in the retro-auricular area from the top border of the auricle to the mastoid tip, but the facial nerve was not anesthetized. Another 50 mg of ketamine was given before starting the block in the other ear. At the end of the surgery, propofol infusion was stopped, as the BIS reading rose toward 100 (fully awake).
CI Surgery
Patients were positioned supine. Facial nerve monitoring on both sides using 2 channels in the midline, without any shaving. Prepping and draping of both surgical sites are carried out to keep both ears sterile. The surgical technique is consistent for all patients. About a 3 cm incision, located 1 cm posterior to the auricular sulcus was made. The palva flap was elevated anteriorly, followed by standard mastoidectomy. A posterior tympanotomy was performed and electrode insertion through the round-window approach was performed for all patients. Closure involved the use of 3.0 Vicryl sutures for the subcutaneous layer and 5.0 monocrystals for skin closure in the subcuticular technique. Cochlear impedance and electrical evoked compound action potential measurements were conducted. The patient was awakened by the anesthesia team and transferred to the recovery area. The fitting of an external device was done in the recovery area in accordance with the patient’s tolerance and preferences.
Audiological Assessment
As part of standard clinical practice, all audiological evaluations were performed before and after surgery. The aim of this was to assess patients’ eligibility for CI and to monitor their post-implantation auditory performance. Preoperative evaluation included unaided and aided (using the HA) pure tone audiometry (PTA), and speech audiometry. The speech audiometry included speech reception threshold (SRT) and word recognition score (WRS). Aided speech perception will be assessed with the Arabic phonetically balanced word test in quiet in a free field at 65 dB SPL. 30
Postoperatively, aided PTA was measured in the sound field through loudspeakers at frequencies from 250 to 8000 Hz. Pure tone average (PTA4) was calculated according to the thresholds measured at 500, 1000, 2000, and 4000 Hz. Aided SRT and WRS were assessed. The SRT was measured using spondee words, whereas WRS was tested using phonetically balanced monosyllabic words. Impedance telemetry was run during each follow-up visit.
Statistical Analysis
The aided PTA, SRT, and WRS had been measured preoperative as well as most recently. Where the average follow-up time was 14.8 months. Also, cochlear impedance had been measured intraoperative, at switch-on, and at 3 consecutive follow-up visits. Descriptive statistics for patients’ demographics and baseline measures included mean, standard deviation, and range for quantitative data were applied, while for qualitative categorical variables; frequency and percentage were used. The paired samples Wilcoxon signed-rank test had been used to investigate the change in the most recent PTA, SRT, and WRS measures compared to the preoperative ones. While the repeated measures ANOVA approach was used for the differences between cochlear impedance repeated measures followed by Bonferroni-corrected multiple pairwise comparisons using paired T-test. Normality assumptions were checked using the Shapiro–Wilk test and a P-value ≤.05 was considered statistically significant. Analysis was performed using R software version 4.2.2 “Innocent and Trusting.”
Results
The study was carried out on 12 ears for 6 patients who underwent bilateral simultaneous cochlear Implants with LA, their mean age was 41.7 ± 16.0 years old. Among them, 66.7% were males and 33.3% were females. Four patients had hearing problems since childhood, and the remaining 2 were diagnosed with hearing loss in the adult age. The etiology of hearing loss was unknown for all of them; however, 1 patient had a positive family history of hearing loss. Most of the subjects (66.6%) had no comorbidities, only 1 had ischemic heart disease, while another had a history of cerebral aneurysm. All subjects met the criteria for CI surgery and radiological evaluations including high-resolution computer tomography and magnetic resonance imaging demonstrated normal findings in the majority of patients 83.3%, with only one finding of retrofenestral otospongiosis. No intraoperative complications were encountered in any patients, complete electrode insertion was successful in all of our patients. All patients woke up smoothly from sedation without any agitation or difficulties. Recovery was uneventful, with controllable pain without the need for narcotics. No complaint was reported in terms of dizziness, nausea, and vomiting. The surgical wounds were dry without any swelling in all patients. The external device was fitted and activated successfully in all patients immediately postoperatively in the recovery area, with none of them reporting any issues during the fitting. Patients’ experience was assessed and showed positive responses about undergoing surgery under LA and were satisfied with the same-day fitting. No balance problems were reported, only 2 patients experienced pain on the surgical site and controlled by analgesia. 83.3% of the patients would recommend others to have the CI surgery under LA. Responses for the patient’s experience are shown in Table 1.
Formed Questionnaire for Patient’s Experience.
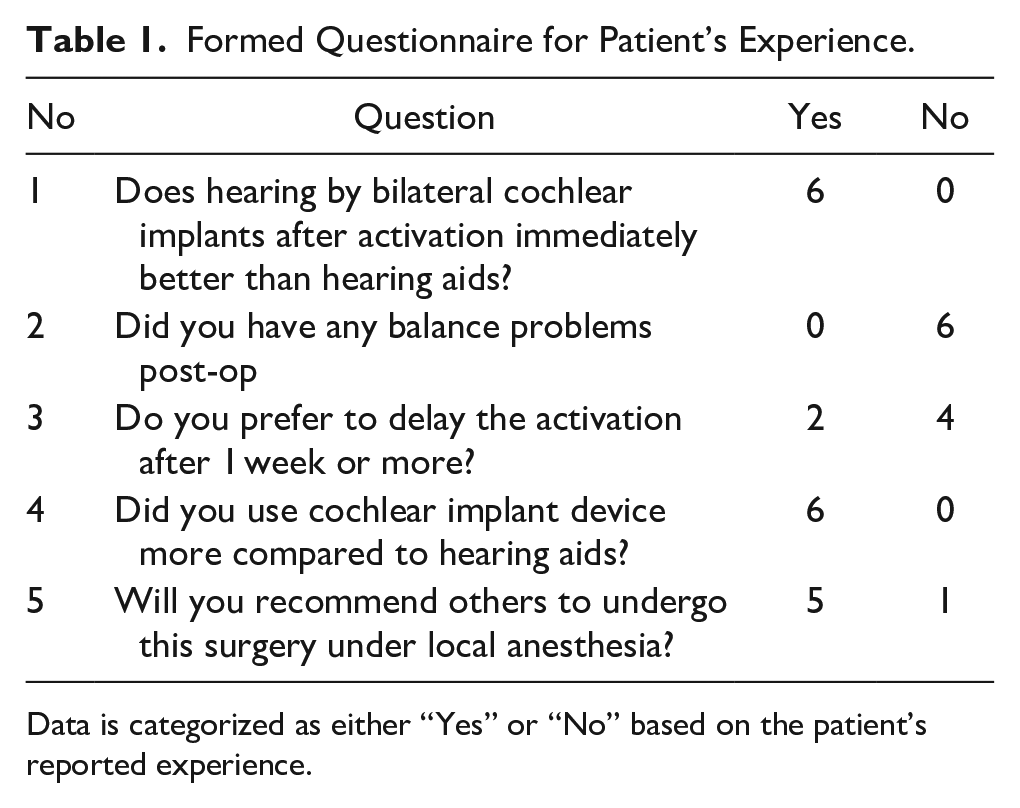
Data is categorized as either “Yes” or “No” based on the patient’s reported experience.
Their PTA4 preoperative aided measure was 60.9 ± 14.1, the average SRT was 49.4 ± 15.2 and the average WRS was 8.0% ± 9.4%. The average intraoperative cochlear impedance was 5.8 ± 2.1, 4.3 ± 1.3, and 4.4 ± 1.3 kΩ for apical, middle, and basal electrodes respectively. Patients’ demographics and baseline measures are shown in Table 2, while Table 3 shows the preoperative unaided PTA measures for our study population.
Descriptive Analysis for Patients’ Demographics and Baseline Measures.
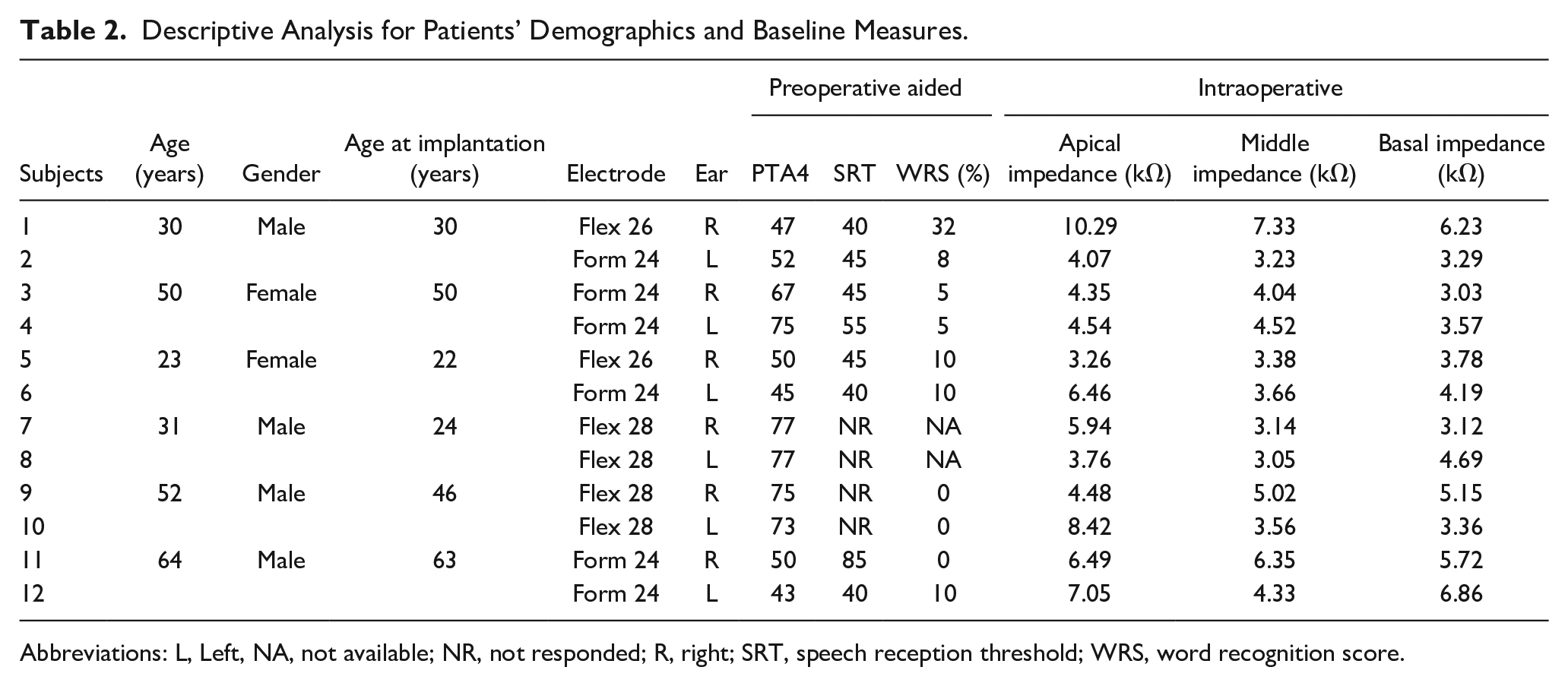
Abbreviations: L, Left, NA, not available; NR, not responded; R, right; SRT, speech reception threshold; WRS, word recognition score.
Preoperative Unaided PTA Measures Among Study Population.
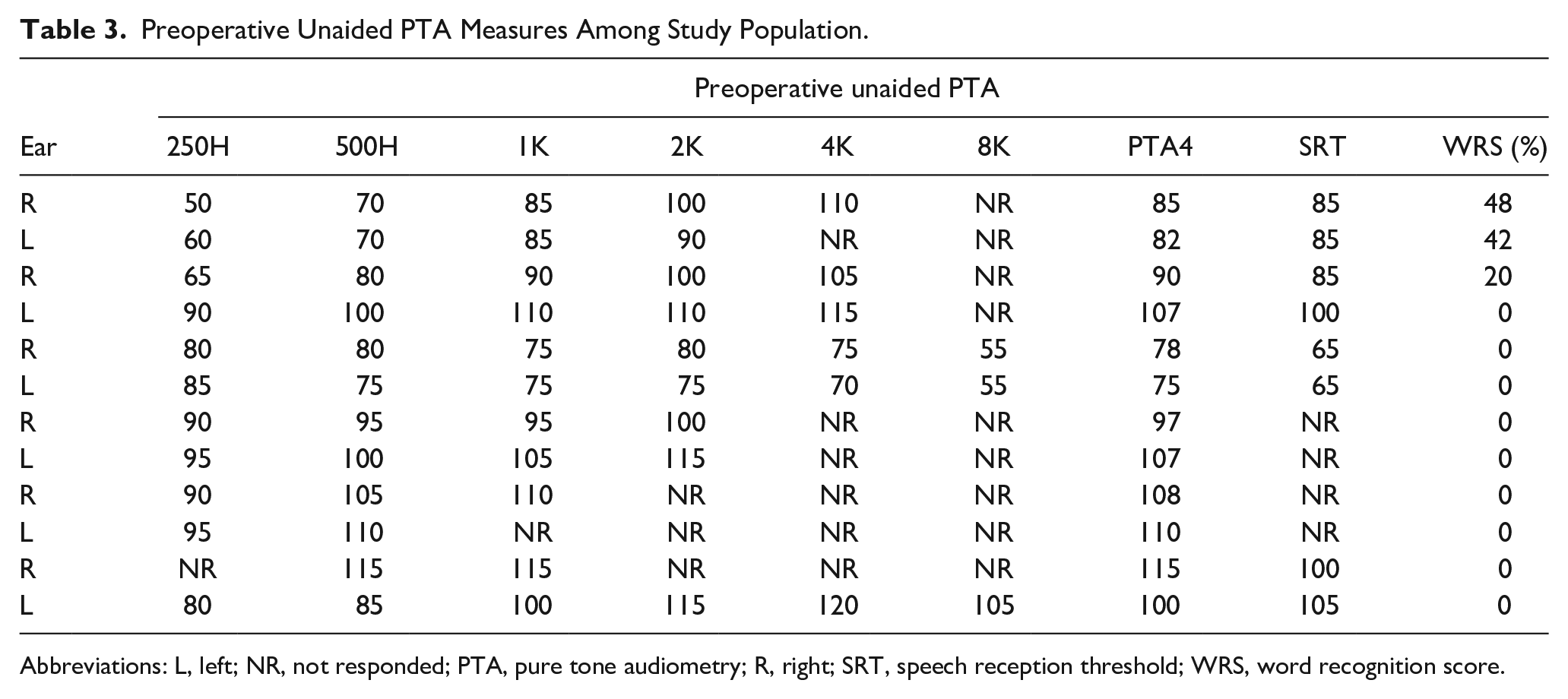
Abbreviations: L, left; NR, not responded; PTA, pure tone audiometry; R, right; SRT, speech reception threshold; WRS, word recognition score.
Postoperative Audiological Assessment
The aided PTA4 showed a statistically significant decrease at the most recent visit compared to the preoperative one (decreased from 60.9 ± 14.1 preoperative to 29.2 ± 4.6 recently with an effect size = 32.0, P-value for paired Wilcoxon signed-rank test = .0025). Mean follow-up period was 59.3 weeks. SRT also showed a statistically significant decrease at the most recent visit compared to the preoperative one (decreased from 49.4 ± 15.2 preoperative to 27.9 ± 8.1 recently with an effect size = 19.17, P-value for paired Wilcoxon signed-rank test = .022), while the WRS showed a statistically significant increase at the most recent visit compared to the preoperative one (from 8.0% ± 9.4% preoperative to 63.5% ± 20.4% recently with an effect size = −51.0, P-value for paired Wilcoxon signed-rank test = .008). PTA, SRT, and WRS measures compared to the preoperative ones are shown in Table 4 and Figure 2. Moreover, the average CI usage data were retrospectively available from the data logging of our CI recipients, which showed an increase in device usage from 9.8 to 12.9 hours between the first visit and the last visit.
The Change in the Most Recent PTA, SRT, and WRS Measures Compared to the Preoperative Ones.
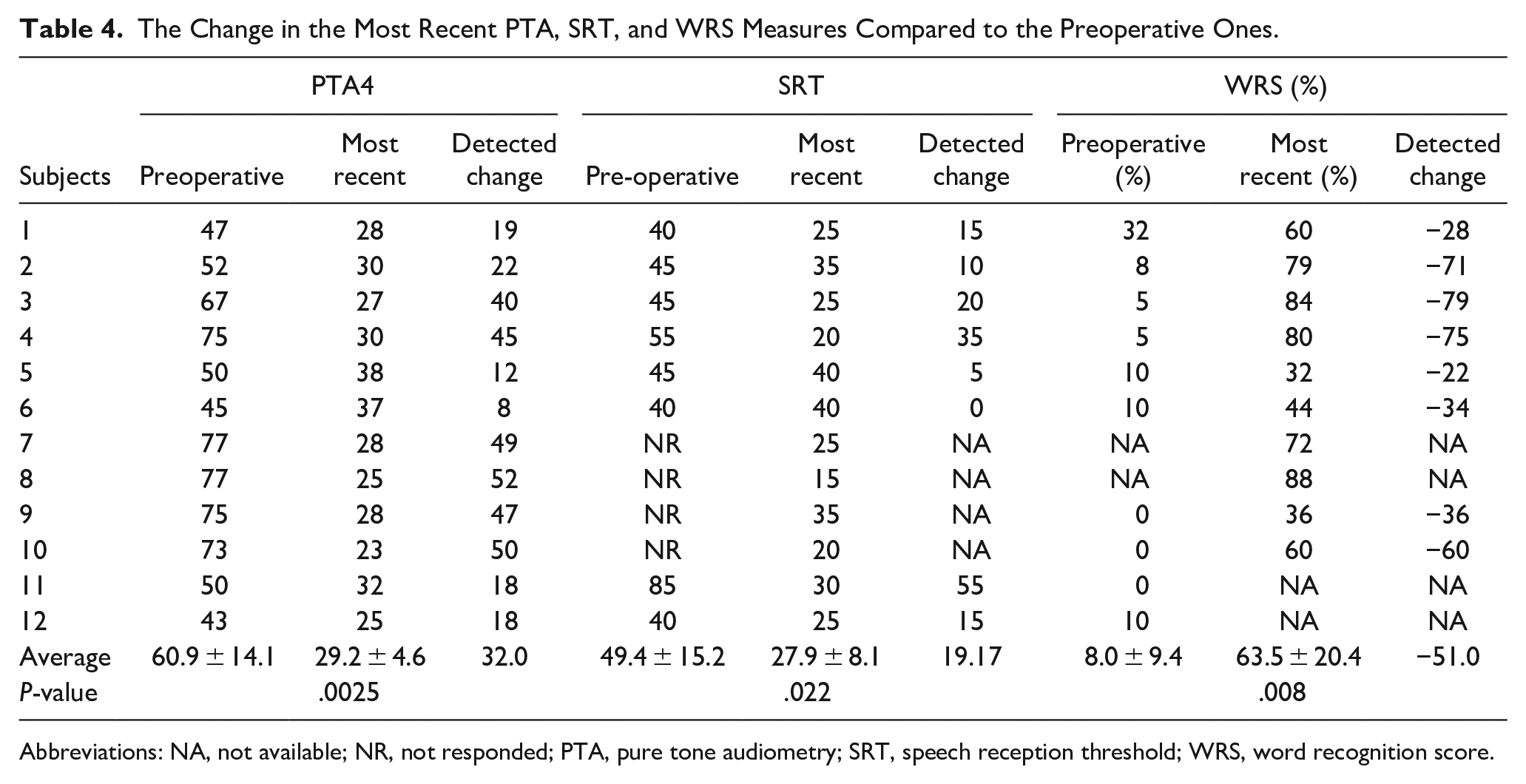
Abbreviations: NA, not available; NR, not responded; PTA, pure tone audiometry; SRT, speech reception threshold; WRS, word recognition score.
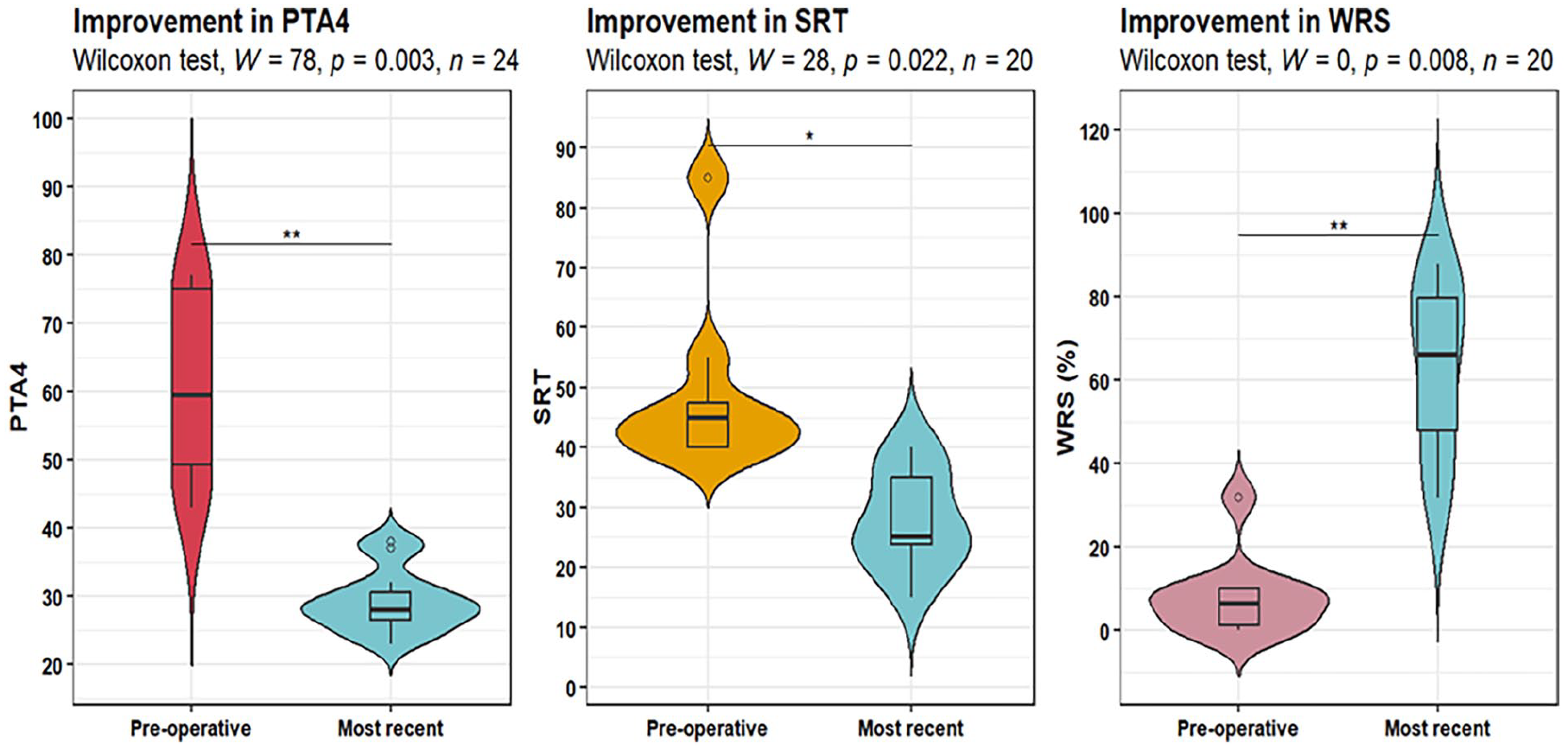
Improvement in the most recent PTA, SRT, and WRS measures compared to the preoperative ones.
Cochlear Impedance Measures
Impedance measures did not show any significant changes at the follow-up visits compared to the intraoperative ones neither for apical, middle nor basal electrodes. Only the second and the third follow-up visits showed statistically significant decreased impedance measures for the apical electrodes compared to the first visit (decreased from 7.2 ± 1.4 kΩ in the first visit to 6.8 ± 1.4 and 6.4 ± 1.8 kΩ in the second and third visits with effect sizes = 0.47 and 0.78 and P values = .035 and .014, respectively). The change in cochlear impedance measures is shown in Table 5 and Figures 3 to 5.
The Change in Cochlear Impedance Measures.
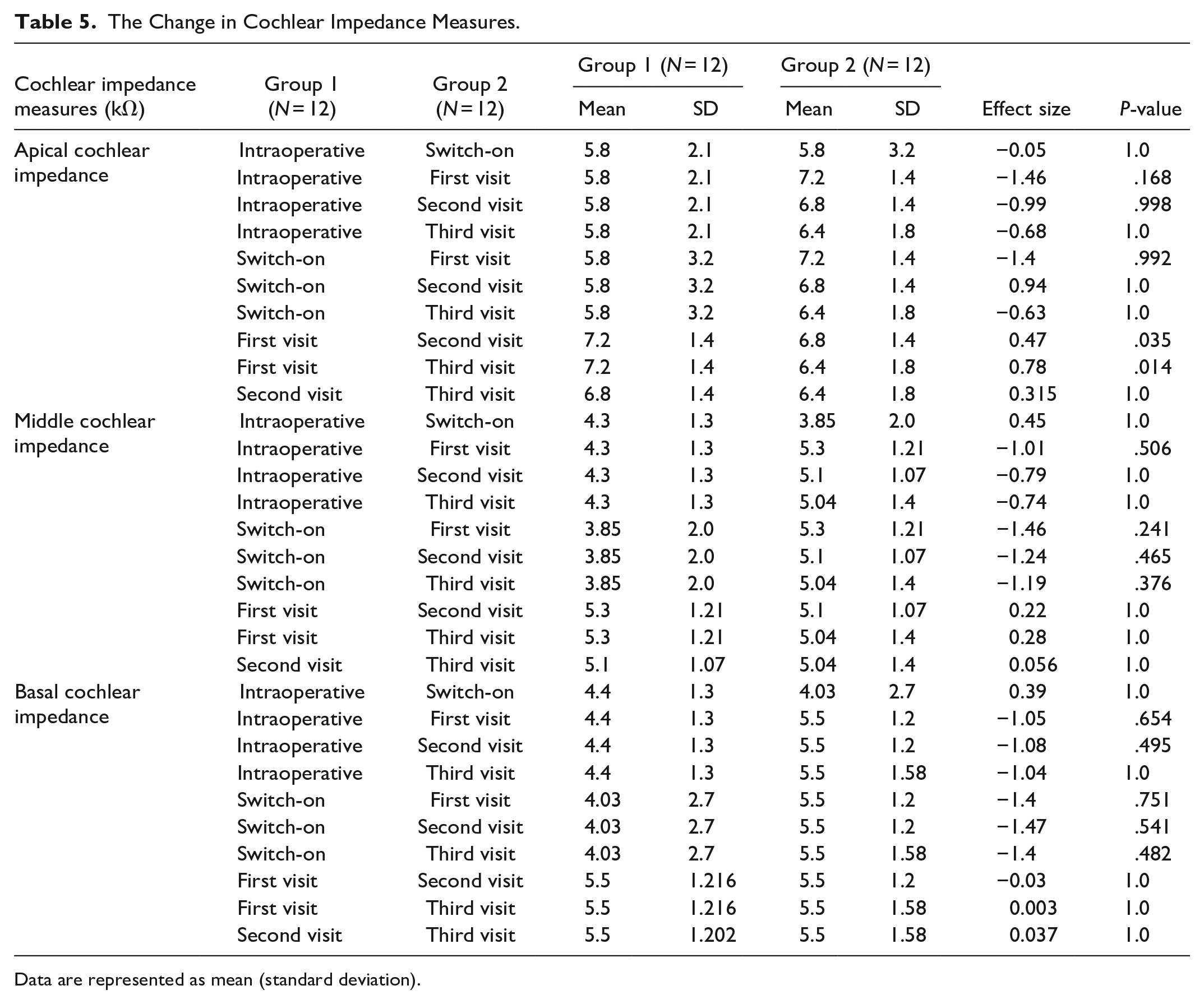
Data are represented as mean (standard deviation).
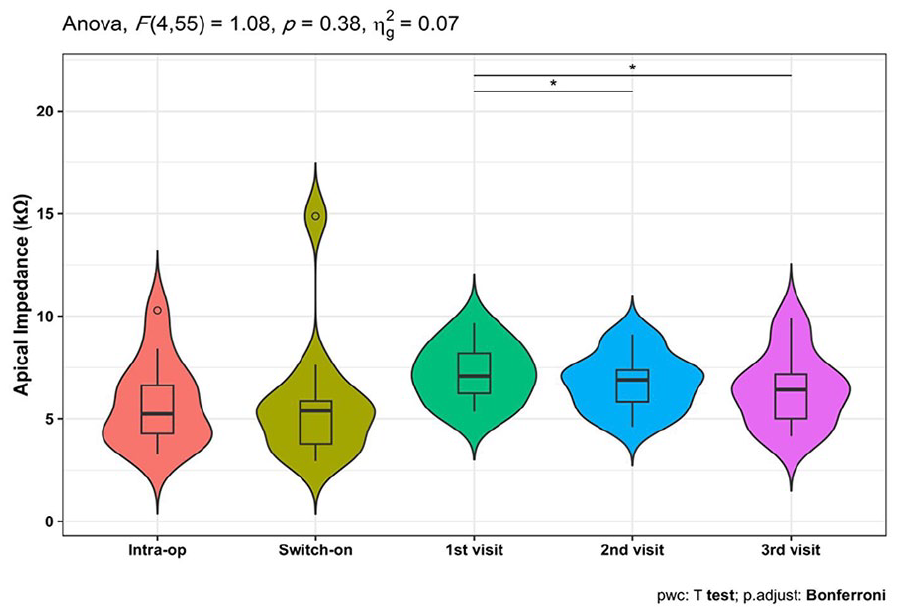
Improvement in apical Impedance repeated measures. * indicates significant differences.
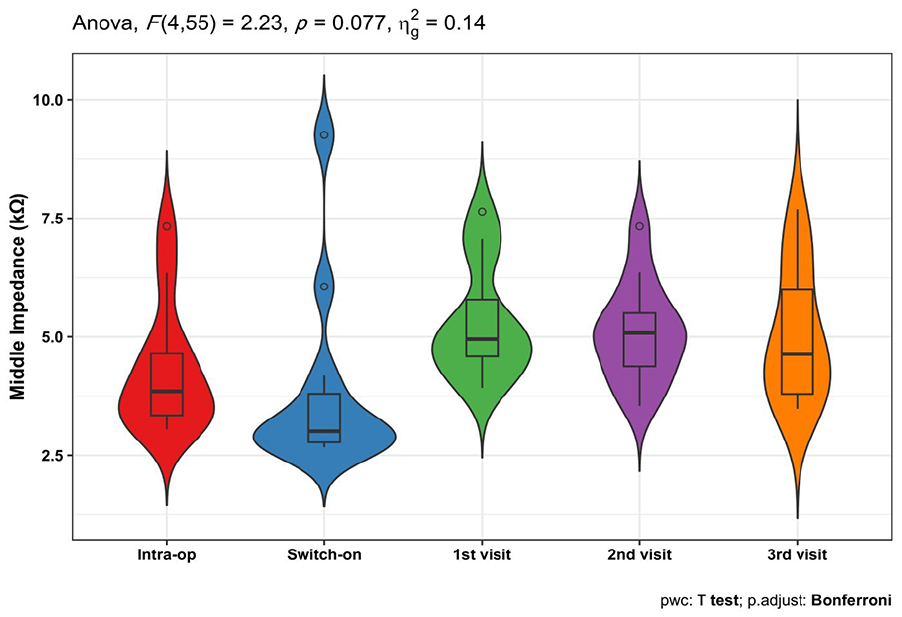
Improvement in middle impedance repeated measures.
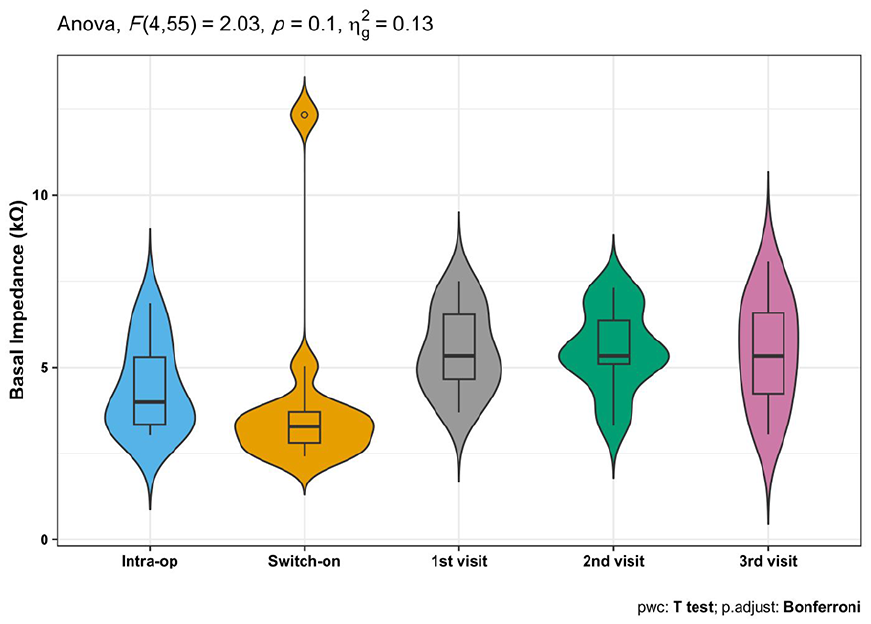
Improvement in basal impedance repeated measures.
Discussion
This study showed that LA with sedation in bilateral simultaneous CI in adults is feasible without major surgical complications and could be a better option for those who are unfit for GA. CI surgery under LA requires patient cooperation, so patient understanding of the procedure and how the anesthesia is performed is crucial.14,17 Our cases underwent CI surgery without complications, with full electrode insertion in all patients and no postoperative complications. Similar results have been reported elsewhere in the literature, demonstrating that CI surgery under LA is feasible and a good alternative for those unfit for GA. 14 In another study of 20 patients who underwent CI under conscious sedation compared to GA, they found that conscious sedation with LA for CI is safe and can treat hearing loss in older patients who are too ill for GA or worried about its effects on their cognitive and medical health. 16 Furthermore, a previous study reviewed 21 CI surgeries under LA, 1 patient had bilateral sequential CI, no significant intraoperative complications were observed, no conversion to GA was reported, and all surgeries achieved full insertion. 21 Likewise, all our patients completed the surgery under LA without the conversion to GA, However, we were prepared to turn the cases into GA. This was intended to be the management in case any of the following happened; airway obstruction if not responding to simple airway maneuvering (chin lift or jaw thrust) or applying a nasal airway, patient failing to reach the required depth of sedation. In addition, the literature included a prior report by Connors et al on a case that required conversion from LA to GA because the patient was unable to cooperate and was having hallucinations. 4 Another study by Aldhafeeri et al reported a case that required conversion to GA after the patient experienced vomiting during electrode insertion. 31 Notably, while the CI surgeries in previous reports were performed unilaterally, this current study provides evidence regarding the feasibility of simultaneous BiCI under LA with sedation.
To our knowledge, our cohort is considered the largest series conducted on simultaneous BiCI under local and regional anesthesia with sedation and could be the first to perform same-day fitting bilaterally in this approach. Our patients had no adverse events and activated the device bilaterally in the recovery area. Only 2 bilateral simultaneous CI under LA cases are documented. Abrar et al reported a 46-year-old blind man could not undergo GA for surgery due to cardiac comorbidities. LA was used for BiCI surgery. They returned home the next day with minor balance issues. However, activation was done 4 weeks postoperative. 32 Where our patients had the advantage of starting hearing rehabilitation on the same-day post-implantation. Another report by Vaid et al for a 28-year-old woman with multidrug-resistant tuberculosis underwent simultaneous BiCI under LA. She had nausea and vomiting during electrode insertion, but her postoperative period was uneventful, and she was discharged the next day. 33 In contrast, to the patients in this cohort who did not complain of any episode of vomiting intraoperatively nor pain that needed narcotics, this could be attributed to our atraumatic surgical approach or anesthesia protocol.
Positive responses were pointed out by our patients about their simultaneous BiCI surgery under LA. They were satisfied with the same-day hearing experience postoperatively. In our center, we advocate an early fitting in CI surgery, our patients were fitted and activated immediately postoperatively and reported that hearing experience by BiCI was better than HA. They did not report any balance problems or pain during the postoperative period. However, only 2 patients experienced mild surgical site pain during external device fitting, which was controlled by analgesia. These findings align with a study by Hamerschmidt et al that no pain was reported in their patients who underwent CI surgery under LA. 14 In contrast to ours, the previous study reported that dizziness, nausea, and vomiting occurred in the LA group. It was less than GA but not significantly different. 14 In the Pateron et al study, only 53% of patients would recommend CI under LACS. 17 but in the current study (83.3%) would. Our smaller sample size and lower average age may explain this difference.
Apical, middle, and basal electrode impedance did not change significantly between visits and intraoperative ones. Interestingly, only apical electrode impedance decreased significantly between first, the second, and third follow-ups, possibly due to the apex being the least traumatic during electrode insertion or the smaller diameter decreasing impedance faster. After follow-ups, PTA, SRT, and WRS improved significantly. These findings support other studies that found CI under LA improved audiological and speech outcomes.18,34 Additionally, the 2 case reports also had positive outcomes.32,33 Thus, our findings support this technique’s benefits.
These findings can shed light on future studies, especially in older patients unfit for GA. Because GA has a higher risk for cognitive decline, LA could be a safer alternative option.19,20 In addition, hearing restorative devices including CI and HA can reduce dementia risk by 19%.35,36 Although studies have shown the safety and efficacy of unilateral CI under LA,14,16,17 our study shows that simultaneous BiCI under LA with same-day fitting is feasible and improves hearing recovery. Our limitations include its retrospective research methodology and limited sample size. This research supports our surgical and anesthetic strategy and may improve CI surgery perioperative treatment in selected patients. Finally, this method needs more research with larger samples to compare it to others.
Conclusion
The feasibility of bilateral simultaneous cochlear implants under LA in adults is achievable, through a multidisciplinary approach between the patient, surgeon, and anesthesia teams. This approach is a potential alternative option for some patients and could optimize their hearing rehabilitation. This method needs further research in comparison to other techniques.
Footnotes
Acknowledgements
None.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Medical-Bioethics Research Ethics Committee (REC) at King Saud University, Riyadh (Ref. No. 24/1167/IRB).
