Abstract
Introduction
Lymphadenopathy is a common complaint for children seeking care with a pediatric otolaryngologist, with a reported incidence as high as 45% to 90% in otherwise healthy children.1,2 A review from 2019 found that nearly 70% of children with lymphadenopathy had a nonspecific diagnosis and that 4% were due to granulomatous disease. 3 Among the top infectious etiologies of granulomatous disease are mycobacteria, and particularly nontuberculous mycobacteria (NTM). 4 NTM are acid-fast bacteria that are widely present in the environment, particularly in water and soil. 5 In immunocompetent children, typically under the age of 5, NTM often presents as unilateral cervical lymphadenopathy. 6 The incidence of NTM-induced cervical lymphadenopathy is rising in the United States 7 ; however, it is difficult to quantify precisely due to the prevalence of asymptomatic cases. Both healthcare providers and parents share an interest in determining the causative pathogen in these types of infections.
The diagnosis of cervical lymphadenitis is often made based only on the history and physical exam findings. Clinical presentations vary from asymptomatic cases to the presence of unilateral, nontender cervical lymphadenopathy present for weeks or months, with some patients exhibiting overlying violaceous skin color changes. If antibiotic therapy is prescribed for presumed bacterial infection, symptoms often fail to improve. 5 Definitive treatment may involve an excisional biopsy of the affected lymph node, which is both diagnostic and therapeutic. Other treatment options include observation, incision and drainage, and/or curettage.
Despite the rising incidence of NTM cervical lymphadenopathy, timely diagnosis is often challenging due to the difficulties in culturing mycobacterial organisms. 8 In cases with negative culture results, a presumptive diagnosis can be made based on histopathologic findings (eg, positive acid-fast bacilli stain), organism detection by polymerase chain reaction (PCR), or the presence of granulomatous characteristics. Additionally, it is important to thoroughly investigate and rule out alternative etiologies such as tuberculosis via blood testing and/or imaging.
There are few studies investigating how often a tissue culture from granulomatous cervical adenitis grows a specific organism resulting in a definitive diagnosis, 9 and even fewer conducted in the United States.6,10 We therefore sought to investigate the incidence of, and elucidate the challenges associated with, the identification of a specific organism in granulomatous cervical adenitis in pediatric patients in the Los Angeles area.
Patients and Methods
This retrospective descriptive study was approved by the Children’s Hospital Research Ethics Committee CHLA-21-00351 on November 19, 2021. All patient information was de-identified, and patient consent was not required. Patient data will not be shared with third parties. We conducted this retrospective chart review of patients at our high-volume tertiary care children’s hospital between January 2017 and November 2023. Children who were evaluated for nontender lymphadenopathy of the neck and subsequently underwent a surgical procedure, including excisional biopsy, incisional biopsy, curettage, or incision and drainage were considered for inclusion. Medical records were searched using relevant Current Procedural Terminology codes (Table 1), then individual charts reviewed for inclusion/exclusion criteria. Pathology, microbiology, and other laboratory reports were reviewed to document the presence of granulomatous cervical adenitis and the incidence of identification of a specific organism.
CPT Codes Used for Subject Identification.

Abbreviations: CPT, Current Procedural Terminology.
Data Collection and Analysis
Data collected from subjects’ medical records included age, sex, race, and insurance type. Insurance types were grouped into “public,” government-supported health insurance, and nongovernment supported healthcare including “private” and workplace plans. Demographics and clinical characteristics were summarized for the full cohort of patients with pathology/microbiology reports and across patients with (vs without) granulomatous cervical adenitis. Mean (SD) were used to describe continuous variables, and categorical variables were described as a frequency and percentage. Differences between groups were assessed via t-tests for continuous variables and Fisher’s exact tests for categorical variables. We defined statistical significance using a 2-sided P < .05. Data were captured and managed in the HIPAA-compliant web-based REDCap database,11,12 and all statistical analyses were conducted in STATA v 13.1 (StataCorp 4905 Lakeway Drive, College Station, Texas) and R Studio 4.2.2 (Posit, PBC).
Laboratory Procedures
All specimens obtained from candidate procedures were sent to the clinical laboratory for further workup including culture and, if warranted, PCR. Specimens were inoculated to a variety of agar media for bacterial, fungal, and acid fast bacilli (AFB) culture. After incubation, isolates visualized on media were further identified by standard methods including macroscopic and microscopic morphology, matrix-assisted laser desorption/ionization-time of flight mass spectrometry (MALDI-TOF MS), and PCR.
Results
Of the 1538 charts reviewed, 163 patients underwent an inclusionary surgical procedure for lymphadenopathy (Figure 1), the majority being either excisional (n = 124) or incisional biopsy (n = 24), and the remainder curettage or incision and drainage. All 163 patients had pathology reports available for review. Twenty-five (15%) were confirmed to have granulomatous cervical adenitis based on histopathologic findings [eg, organism detection by AFB stain and PCR (n = 1), or the presence of granulomata (n = 24)]. The mean age of those with granulomatous cervical adenitis was 7.7 years (range 3.6 months-19 years), and 10 (40%) were female (Table 2). Nine of these 25 (36%) had an organism recovered by culture: Mycobacterium tuberculosis (MTB) (n = 3), Mycobacterium avium complex (MAC) (n = 1), Staphylococcus spp. (n = 2), Coccidioides spp. (n = 1), Streptococcus anginosus (n = 1), and Bartonella spp. (n = 1) (Table 2). There were no significant differences between those with and without granulomatous cervical adenitis with regards to sex, race, or insurance type (Table 2). However, those with granulomatous cervical adenitis tended to be younger (7.7 years vs 11.3 years, P = .04) (Table 2).
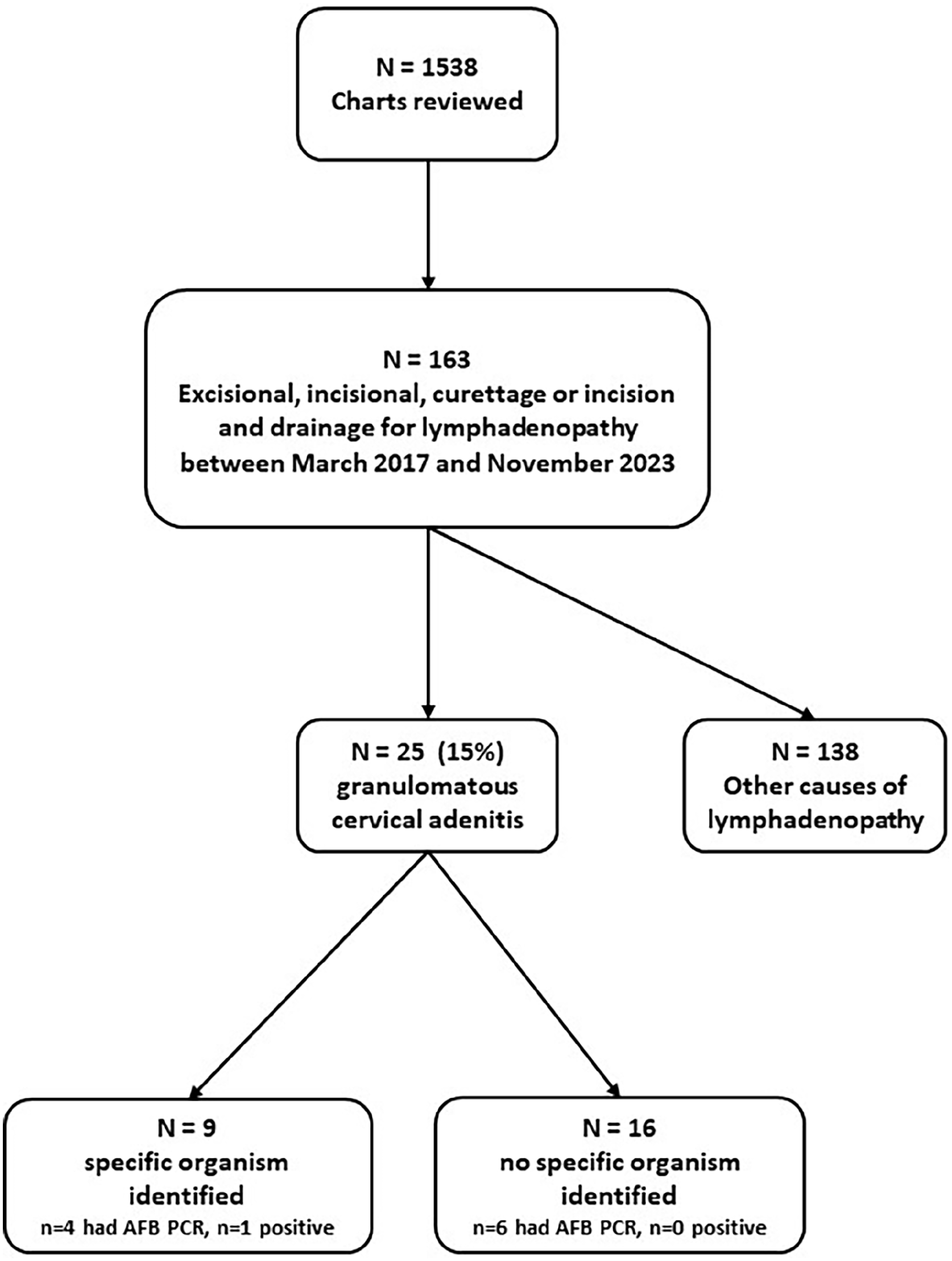
Flow chart of study participants. PCR, polymerase chain reaction.
Descriptive Characteristics Between Those With and Without Granulomatous Cervical Adenitis.
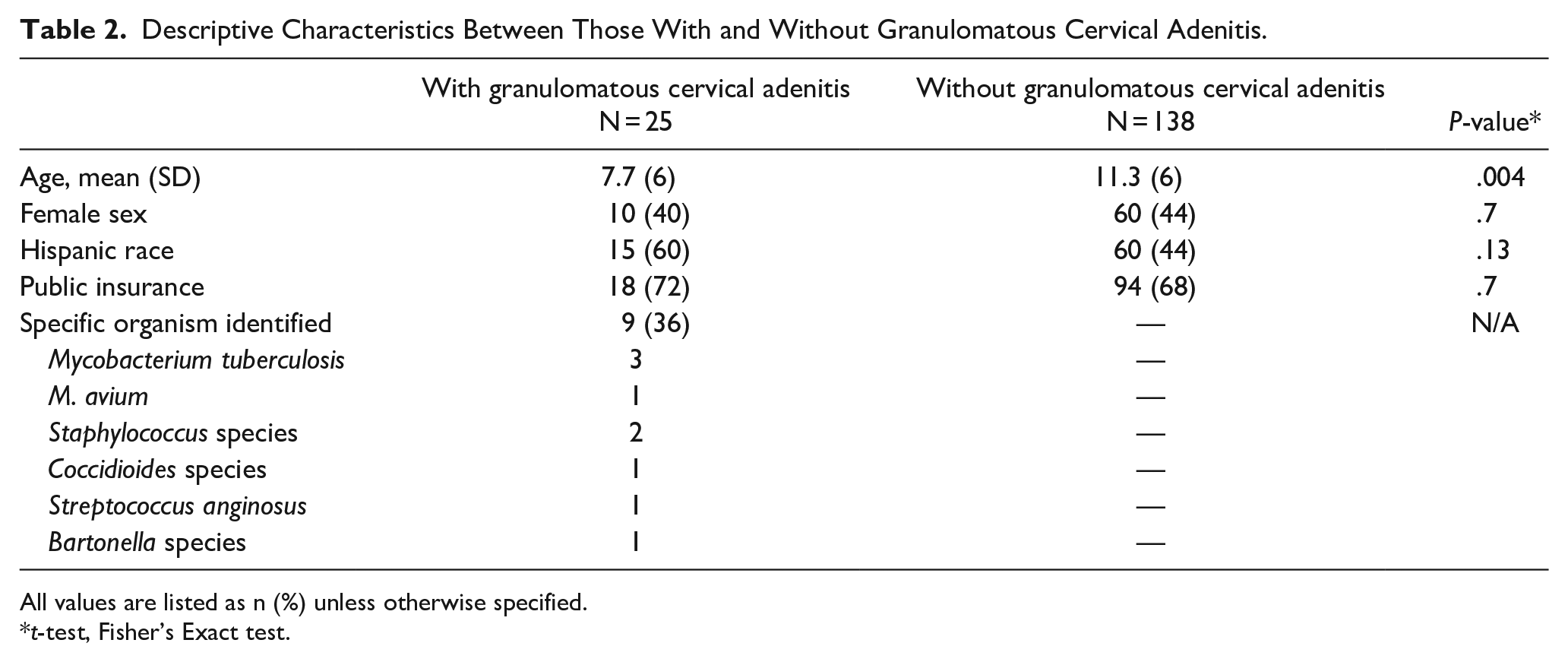
All values are listed as n (%) unless otherwise specified.
t-test, Fisher’s Exact test.
Of the 16 patients with granulomatous cervical adenitis and no identified organism, the majority (62.5%) received minimal (oral antibiotics), or no treatment and symptoms resolved over time. Eleven patients were diagnosed based on pathologic findings, which were “suggestive of nontuberculosis mycobacterial infection” and their infections resolved with antibiotics (n = 5) or no treatment (n = 5) or were lost to follow up (n = 1) (Figure 2). Of the remaining 5, 1 had a positive AFB stain, but no organisms were ever identified, all MTB testing was negative, no treatment was prescribed for the presumptive NTM infection, and they were lost to follow up after 4 months with improving symptoms; 1 was diagnosed with periodic fever, aphthous ulcers, stomatitis, pharyngitis, adenitis syndrome and treated with cimetidine and prednisone, and the final 3 were lost to follow up (Figure 2).
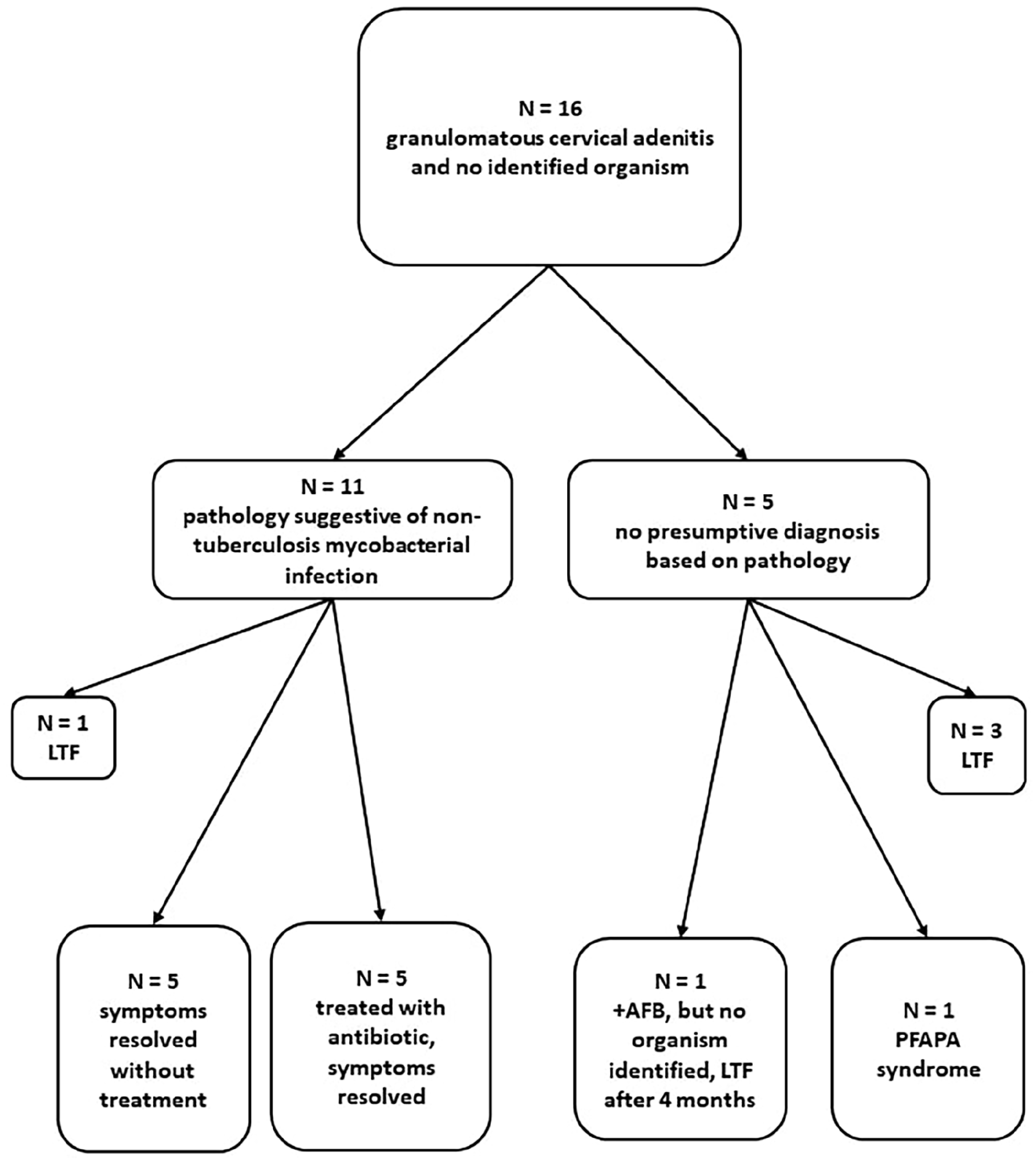
Flow chart of study participants with no identified organism. LTF, lost to follow up; PFAPA, periodic fever, aphthous ulcers, stomatitis, pharyngitis, adenitis.
Among the 138 study patients without granulomatous cervical adenitis, the causes of lymphadenopathy included mostly diagnoses of reactive lymph nodes/reactive lymphoid hyperplasia or Hodgkin’s lymphoma.
Discussion
In our study, we observed a relatively high proportion of granulomatous cervical adenitis as the cause of pediatric cervical lymphadenopathy compared with previous reports. There are few studies in the literature reporting on the incidence of NTM-induced granulomatous cervical adenitis and incidence of identification of a specific organism in pediatric patients. Al Kadah et al found only 6% of 251 cases with cervical lymphadenopathy in Germany were due to granulomatous lesions. 13 Similarly, Flyger et al 14 reported NTM accounted for only 7% of 121 cases of granulomatous inflammation in cervical lymph nodes over a 10-year period in Denmark. Other prevalent etiologies included sarcoidosis and tuberculosis. 14 One contributing factor for the discrepancy between our and previous studies is that our study focused solely on pediatric patients, who more commonly present with NTM cervical lymphadenitis, whereas many international studies included patients of all ages.
Regarding the identification of organisms in granulomatous-positive lesions, our study only found 1 NTM case (MAC) out of 2 culture-positive cases. This finding is comparatively lower than what is reported in studies conducted in other endemic areas of the United States. 15 Hazra et al 6 reported that 60% of 19 children evaluated for lymphadenitis at Boston Children’s Hospital had positive NTM cultures, with 8 organisms identified (7 MAC and 1 Mycobacterium intracellulare). Similarly, Suskind et al identified 9 positive cases from samples of 81 children evaluated for lymphadenopathy at Children’s Hospital in Philadelphia, 10 and Venturini reported a 17.6% rate of NTM infections in children with lymphadenopathy in Italy. 16 Recovery of Mycobacterium species by culture is notoriously challenging due to their fastidious growth requirements and slow growth rate, but this alone does not explain the relatively low yield in our study. Possible explanations could include the types and number of samples that were available for culture, potential delays in culture set up as it was sent to a reference laboratory for testing, and patient exposure to antimicrobial agents that may impact the recovery of the organisms. However, in a study of 60 children in Seattle, Choi et al were able to identify a causative organism in 82% of children with lymphadenitis (not restricted to granulomatous disease), with specialized and disease-specific PCR primers. 17
Of the 3 patients in our study who were diagnosed with M. tuberculosis, 1 was a 3-month-old admitted from the emergency department (ED) with more than 1 mass and later found to have disseminated tuberculosis. The remaining 2 (one 17-month-old and one 17-year-old) presented in a very similar manner to those with no identified species (submandibular/neck swelling with no other notable symptoms) and were not confirmed to have M. tuberculosis until 1 to 2 months later when the AFB culture finally returned a positive result. Interestingly, the one 16-year-old subject with Bartonella spp., presented with the most concerning symptoms of night fevers and weight loss, was admitted from the ED and scheduled for biopsy by the Oncology Department. It is interesting to note that 4 out of 9 positive species identified were organisms related to acute bacterial adenitis. This may reflect the presence of a secondary bacterial superinfection of a granulomatous lymph node.
Given the difficulty in culturing mycobacterial organisms, physicians should be prepared to make a diagnosis of granulomatous cervical adenitis based on the clinical history and physical examination alone and generate an appropriate medical and/or surgical treatment plan. In those children who have suspected NTM cervical lymphadenitis, the International Pediatric Otolaryngology Group published consensus guidelines in 2023. 18 Their recommended diagnosis and management plans included diagnostic testing such as peripheral blood smear to determine the presence of malignancy, a tuberculin skin test in those patients with no TB or BCG vaccine exposure, certain serologic studies such as Bartonella, and neck imaging. 18 However, even among this expert group, they noted “significant practice variability” in initial tests and management options based on patient, location, and institutional factors. 18 Physicians should educate patients and their parents that a specific pathogen is not likely to be identified in those children with a diagnosis of granulomatous cervical adenitis. Nevertheless, parents should find comfort in knowing that the treatment plan remains unchanged irrespective of the finding of a conclusive diagnosis or identification of a specific causative organism. Additionally, it is important to acknowledge that parents may raise concerns about malignancy, and in such instances, providing reassurance and education is important.
Our study was limited by the retrospective nature and, at times, relying on incomplete or inconsistent pathology reports. Additionally, this study involved a limited number of participants with disease of the lymph nodes only, which can impact the generalizability, statistical power, and therefore the ability to draw specific conclusions about NTM or granulomatous disease more widely. Future studies can contribute to a better understanding of NTM-induced granulomatous disease in cervical lymph nodes by including a larger sample size, perhaps via a multi-center collaboration and by implementing quality control measures, particularly for laboratory techniques to improve the accuracy and reliability of pathology reports.
Conclusion
Despite the availability of a number of ancillary tests, our data demonstrate that the identification of a specific pathogen in cases of granulomatous cervical lymphadenitis is rare. NTM cervical lymphadenitis is relatively common in certain pediatric populations; however, the incidence of identification of a specific organism in our local population is low. As a result, physicians should be prepared to rely primarily on the history and physical exam findings to determine a working diagnosis as well as a medical and/or surgical treatment plan.
Footnotes
Acknowledgements
The authors would like to acknowledge and thank the patients whose data contributed to the results of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed the receipt of the following financial support for the research, authorship, and/or publication of this article: UL1TR001855 and UL1TR000130 from the National Center for Advancing Translational Science (NCATS) of the U.S. National Institutes of Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
