Abstract
Keywords
Introduction
Mucociliary clearance and its function in the pathophysiology of sinusitis were first investigated by Messerklinger, whose work inspired the notion of sinus surgery. To better deliver topical medicines and increase mucociliary transport, functional endoscopic sinus surgery (FESS) aims to expand sinus ostia and restore appropriate aeration of the sinuses. Although the concept of FESS is simple, the diversity and severity of disorders treated in every FESS present unique obstacles to the operating surgeon.
FESS can alleviate the symptoms of chronic rhinosinusitis, improving the quality of life for individuals impacted by this common ailment. However, the possibility of serious complications is a significant downside. The proximity of critical anatomical structures like the brain, dura mater, orbit, internal carotid arteries, and skull base creates this risk. Several factors, including those associated with the anesthesia, the surgeon, and the disease itself, can increase the likelihood of complications during FESS. 1 Sinus surgery requires careful preoperative preparation to achieve the best potential outcomes and prevent problems. 2 Operating near delicate structures, such as the lamina papyracea and the base of the skull, while the patient is under general anesthetic increases the risk because the patient cannot provide feedback. The endoscope and tools are angled at an angle that increases the danger for right-handed and left-handed surgeons. Unfamiliarity with the anatomy and instruments caused by surgeon inexperience is another factor that increases the threat. Risks can also increase in cases of severe sinusitis, excessive bleeding, or revision procedures when typical sinonasal structures have previously been altered or obscured. While it is impossible to eliminate risk entirely with endoscopic sinus surgery (ESS) and avoid general anesthesia, several things can be done to make it safer. 3
New surgical instruments, imaging, simulation, and navigation have been developed and implemented in endoscopic sinus surgery at an ever-increasing rate since1970. 2
Methods
The literature review utilized Google and Google Scholar, PubMed, EBSCO, UpToDate, and Proquest Central at Kırıkkale University. The search was conducted using the terms “functional endoscopic sinus surgery,” “FESS,” “safety,” “image-guided,” and “complications” from 2000 to 2024. Review articles, randomized controlled trials, prospective studies, and retrospective research are all part of the data set pulled in by the search.
Anatomy and Physiology
It is necessary for the surgeon to be aware of the usual anatomy and of possible variations. The preoperative CT scan is useful in providing a road map for surgery. Any surgical procedure must not interfere with the functions of the nose.
Respiratory Region
In addition to warming, filtering, protecting, and eliminating gaseous waste, the respiratory region also humidifies. The most significant portion of the nasal passages is lined by respiratory epithelium and mucous cells. Warming to body temperature and approaching 100% humidity, air travels via the nasal cavity. Helping with this is the area’s neurovascular supply. Erectile tissue on the inferior turbinate and anterior septum controls blood volume, affecting the nasal airflow. Under typical circumstances, the superior cervical ganglia continuously activate this tissue to maintain a clear nasal cavity. 4
The nasal mucosa acts as a trap for particles that manage to pass the nasal vestibule. The mucociliary system aids in the removal of these particles. At a pace of 1 cm/minute, the ciliated pseudostratified columnar epithelium sweeps particles into the nasopharynx for further expulsion. 4
Nostril mucus acts as a barrier to prevent inhalation diseases. Immunoglobulin A, lysozymes, and lactoferrin are mucus components that protect the host.5-7
Olfactory Region
To achieve olfactory perception, air must be carried by orthonasal or retronasal airflow to the olfactory epithelium, which is situated at the top of the nasal cavity. When odorants get stuck in mucus, they bind to proteins that help concentrate and dissolve them. Before entering the brain, the particles are attached to cilia’s olfactory receptors, which send specific signals through the cribriform plate to synapse with neurons of the olfactory bulb. From there, signals are sent through the olfactory nerve into secondary neurons for higher processing.5-7
Nasal Vestibule
As one moves posteriorly via the anterior nares—also called the nostrils or the external nasal valve—the first place met is the nasal vestibule. Coarse hairs termed vibrissae cover the first half of the vestibule’s keratinized stratified squamous epithelium. These capture larger airborne particles.5-7
Lateral: fibrofatty alar tissue and the lateral crus of the lower lateral cartilage (LLC)
Medial: the medial crus of the LLC and the septal cartilage
Posterior: the Limen Naris
The Roof of the Nasal Cavity
Holes in the nasal roof mucosa allow the cribriform plate to pass through. The olfactory axons go through these holes. 5
In front: the nasal spine and nasal bone
Behind: the ethmoid’s cribriform plate and the sphenoid’s body
The Floor of the Nasal Cavity
In comparison to roof, the nasal cavity floor is wider. 5
On the front: the maxillary palatine process
On the back: the palatine bone’s horizontal plate
Nasal septum and inferior turbinate
When one enters the nasal cavity, the nasal septum and the inferior turbinate are the first structures observed. The quadrate cartilage is located anteriorly and extends dorsally to the perpendicular plate of the ethmoid bone and dorsally to the vomer, making up the nasal septum. 2
Because nasal septum deflections can considerably contribute to nasal obstruction and impair endoscopic visibility during surgery, preoperative identification of these abnormalities is crucial. The need for septoplasty in combination with FESS may be discussed with patients who have deflections in the septum. 8
The inferior turbinate follows the inferior lateral nasal wall back toward the nasopharynx. The inferior turbinates may swell up if there is a significant allergic component to the patient’s issues. Combining endoscopic sinus surgery with turbinate reduction may be the best option for these individuals. About 1 cm beyond the most anterior margin of the inferior turbinate lies the inferior meatus, where the nasolacrimal duct opens. 8
Middle turbinate
The middle turbinate is the next anatomical landmark met by the endoscope when it is inserted into the nasal cavity. When doing endoscopic sinus surgery, it is essential to locate the central turbinate. It has a medial–lateral component in the coronal plane and a posterior–anterior component in the sagittal plane. 8
The superior attachment of the middle turbinate is the cribriform plate of the skull base. Therefore, the middle turbinate requires particular caution when being manipulated. 8
The basal (or big) lamella is the horizontal part of the middle turbinate that separates the anterior and posterior ethmoid air cells. The crista ethmoidal, located directly in front of the sphenopalatine foramen, is the attachment site for the posterior and inferior portions of the middle turbinate to the lateral nasal wall. 8
Uncinate process
In endoscopic sinus surgery, the uncinate process is the following critical structure to locate. The infundibulum, or hiatus semilunaris, is anteriorly bound by an L-shaped lateral nasal wall bone. The ostiomeatal complex, in which the natural ostium of the maxillary sinus is found, is located in the infundibulum. 8
Patients suffering from sinus illness must have a patent ostiomeatal complex for their symptoms to improve. The uncinate process is attached to the lacrimal bone at its anterior end, and the ethmoidal process of the inferior turbinate at its inferior end. 8
Natural maxillary ostium
The natural maxillary ostium, seen once the uncinate process has been eliminated, is located about a third up the middle turbinate from its anterior margin. Situated above the inferior turbinate, it is roughly at the same level as the inferior border of the middle turbinate. 8
The mucociliary drainage from the maxillary sinus reaches its final destination at the bony ostium of the maxilla. The natural ostium must be incorporated into the surgically expanded maxillary antrostomy for the best possible outcomes. One of the most common causes of complications after FESS is neglecting to access the maxillary ostium during the endoscopic surgical antrostomy. 8
The maxillary sinus has a volume of around 14 to 15 ml. It is bounded superiorly by the inferior orbital wall, laterally by the nasal wall, and inferiorly by the alveolar section of the maxilla. 8
Bulla ethmoidalis
The ethmoid bulla, one of the most reliable anterior ethmoidal air cells, is the following structure to be encountered. The maxillary sinus ostium creates the posterior edge of the hiatus semilunaris and extends slightly beyond the orbital floor. 8
The bulla’s lateral margin is called the lamina papyracea. The ethmoid bulla may reach the base of the skull at its most superior point. Another possibility is the presence of a suprasellar recess above the bulla’s roof. This connection is made clear by thoroughly examining the patient’s CT scan before surgery. 8
Ethmoid sinus
The ethmoid sinus houses anywhere from 7 to 15 air cells. These air cells are bordered laterally by the lamina papyracea and superiorly by the skull base. There may be ethmoid cells in the area above the eye. After reviewing the patient’s CT scan, the surgeon knows these deviations. 8
The anterior and posterior ethmoid cells are located on opposite sides of the basal lamella of the middle turbinate. The middle and superior meatuses get drainage from the ethmoid ridges, while the anterior ridges receive drainage from the inferior meatus. 8
Sphenoid Sinus
The sphenoid bone’s front surface may be seen once the posterior ethmoid cells have been exenterated. The sphenoid sinus is the most posterior paranasal sinus, located above the nose and below the back of the throat (nasopharynx). Located 30° from the horizontal, the sphenoid’s anterior face is around 7 cm from the nasal sill. 8
The sphenoid sinus is connected to several critical anatomical locations. At its most posterior and medial, the internal carotid artery leaves an imprint in the sphenoid sinus. Bone dehiscence occurs in around 7% of patients. 8
The roof of the sphenoid sinus is depressed anteriorly and superiorly due to the optic nerve and the bony covering it receives. The bone is dehiscent around the optic nerve in 4% of instances. Thus, opening the sphenoid sinus in a regulated fashion, usually at its natural ostium, is essential for a successful result. 8
The sphenoid sinus’s natural ostium can be found in various places. The ostium is typically found medial to the superior turbinate (60%), although it can also be found laterally (40%). 8
Frontal recess
The frontal recess is the opening via which drainage from the frontal sinus enters the nose. The ethmoid bulla typically marks the frontal sinuses’ back of the outflow channel. 8
The uncinate process or agger nasi cells (frontal anterior ethmoid air cells) border the frontal sinus outflow tract anteriorly. Outflow tract blockage and consequent frontal sinusitis can occur if any of these cells are more extensive or if scarring from surgery is present. The lamina papyracea often forms the medial wall of the frontal recess. 8
Variations in Nasal Cavity and Paranasal Sinuses
Turbinate variations
Pneumatization of the concha, or concha bullosa, is a prevalent variant of sinonasal morphology. The superior turbinate is pneumatized relatively infrequently compared to the middle turbinate, which is the more common site. Few articles discuss inferior concha bullosa concerning the inferior turbinate, and those that do tend to be case reports.9,10 When the ethmoidal process causes the osseous palate to become pneumatized, the consequence is concha bullosa. It ranges in size and can be either unilateral or bilateral. 10 It starts to show up about 7 and grows long after puberty ends.11,12 Patients with chronic sinusitis have the highest reported prevalence, which ranges from 15% to 80%. Based on its position, it has been categorized as either lamellar, bulbous, or extensive concha bullosa. Headaches, 11 associated mucosal contact, and a noticeable nasal blockage are some difficulties a big concha bullosa can produce if the pneumatization is substantial. Surgical correction may be necessary for bulbous and extensive forms. Mucopyocele, a rare consequence of fluid and pus filling the concha bullosa, is a nasal mass that needs to be distinguished from others. 12
Ethmoid cell variations
An “agger nasi” is a nasal mound. At the front superior part of the middle turbinate, agger nasi cells are the most anterior ethmoid cells. 10 The frontal recess can be narrowed, and frontal sinusitis can result from posterior pneumatization, typically found on both sides of the body.13,14 The agger nasi cells are best identified using CT images that have been reformatted in the sagittal and coronal planes. Their reported frequency varies between 10% and 98%. 15 One possible reason for the reduced frequency in pediatric patients could be that, due to the enlargement of the frontal sinus, agger nasi cells continue to form even after the ethmoid sinus has fully developed. The ability of these cells to provide endoscopic access to the frontal sinus is crucial. 16
In 1765, Albert von Haller first characterized the ethmoid cells that develop onto the orbit floor, also known as the roof of the maxillary sinus, next to and above the maxillary sinus ostium. 16 These cells are also called infraorbital cells. When they swell, they might narrow the back of the ethmoidal infundibulum and the opening of the maxillary sinus. Whether the Haller cells come from the cells at the front or the back of the ethmoid is debatable. A possible explanation for their varying prevalence—from 8% to 57%—is the lack of consensus on a single definition. The variance has been linked to recurrent maxillary sinusitis, a clinically relevant finding. This is because it narrows the infundibulum and ostium, negatively impacting maxillary sinus ventilation. 10 Furthermore, ethmoidectomy is more likely to result in orbital damage when Haller cells are present. 17
Attached closely to the optic nerve, the Onodi cell—also called the sphenoethmoid air cell—is a posterior ethmoid cell pneumatized laterally and somewhat above the sphenoid sinus. There is some evidence that having an Onodi cell raises the likelihood of optic nerve and internal carotid artery injuries. For this reason, it may be beneficial to identify Onodi cells before surgery to reduce the possibility of this complication.10,18 To achieve successful pituitary surgery, it is necessary to remove these cells since they obstruct the view of the sellar floor’s edge and prevent a thorough examination and removal of lesions in the sellar, para sellar, and suprasellar compartments. 19 Onodi cell infections can also put pressure on the optic nerve, which can cause pain in the back of the eye. 12 Various definition criteria and the difficulties of cell evaluation on the coronal plane CT contribute to the reported frequency’s broad range, which can be anything from 2% to 50%. 20
Originating in the frontal recess, superior and laterally extending over the orbit are Supraorbital ethmoid cells (SOEC), which are anterior ethmoid cells. On the frontal bone, behind the frontal recess and sinus, these cells represent the pneumatization of the orbital plate. The usual location for their drainage is the side of the frontal recess. Fifteen per cent of adults have at least 1 SOEC, with 5% of frontal sinuses exhibiting several SOECs. Due to the high similarity between these cells and the frontal ostium, preoperative identification is crucial for endoscopic dissection. 10
SOECs can be misconstrued as many frontal sinuses or a septated sinus. 10 SOEC 21 was significantly associated with frontal sinus septation in the study of Comer et al. Their existence raises the possibility of orbital injury during endoscopic sinus surgery and has been linked to orbital proptosis. There is a higher chance of skull base injury due to cerebrospinal fluid (CSF) leakage and retraction of a lacerated anterior ethmoidal artery into the orbit if the supraorbital cell is not recognized during surgery using the anterior ethmoidal artery as a landmark. 22
Indications for Endoscopic Sinus Surgery
Common reasons for undergoing endoscopic sinus surgery 8 :
Recurring sinus infections
Nasal polyps
Antralchoanal polyps
Sinus mucoceles
Surgical removal of specific tumors
Repair of CSF leaks
Decompression of the orbit (eg, for Graves’ ophthalmopathy)
Decompression of the optic nerve
Dacryocystorhinostomy
Choanal atresia correction
Retrieval of foreign objects
Control of nosebleeds (epistaxis) 2
After failure of initial medicinal treatment for chronic rhinosinusitis (CRS) endoscopic sinus surgery is considered. The criteria for what is considered maximal medical care and the scheduling of surgery still need a broadly agreed consensus.23,24 Endoscopic sinus surgery is typically reserved for patients with proven rhinosinusitis who have not responded to medicinal therapy. This diagnosis is made after a detailed patient history and a comprehensive physical examination, which may include CT scans. 8
Nasal polyps may be untreatable with only medical intervention. Patients with nasal polyps and chronic rhinosinusitis still needed surgery after 12 weeks of therapy with fluticasone propionate nasal drops, according to research by Aukema et al. 25 Antrochoanal polyps are similarly treatable only by surgical excision. 8
After years of research into surgical options for CRS, both with and without polyposis, FESS has shown promise in enhancing patients’ quality of life. 26
Complicated acute rhinosinusitis (ARS) is managed in part by FESS. In many cases, Chandler’s categorization is used to classify the extracranial and intracranial ARS problems. Orbital cellulitis, subperiosteal abscess, orbital abscess (OA), and cavernous sinus thrombosis are the conditions that fall under this category, with OA being the most severe. If medical treatment fails to alleviate visual impairment or elevated intraocular pressure in cases of preseptal or orbital cellulitis, FESS may be considered. 23
Because the orbit and the ethmoid sinuses are so closely connected, endoscopic sinus surgery has allowed doctors to reach several orbital diseases transnasally without requiring skin incisions. The following conditions can necessitate an endonasal approach to the orbit: medial orbital wall fractures, benign sinonasal tumors invading the orbit medially, lesions of the extraconal medial orbital apex or space, posttraumatic optic neuropathy or Graves’ disease, and decompression of the orbit and optic canal. 27
ESS has also replaced other procedures for mucoceles, fungal sinusitis (invasive and non-invasive), silent sinus syndrome, pituitary tumors, CSF leaks, benign and malignant sinonasal tumors, lesions of the petrous apex, pterygomaxillary fossa, and ventral skull base lesions. Cases of nasal and paranasal space cancers, including those that reach the anterior skull base, can be treated by expanded sinus surgery. 28
Preparation of the Patient
The patient is positioned on the operating table, which is aligned with the television monitor. The head of the bed is raised to place the patient in a reverse Trendelenburg position. The endotracheal tube is secured at the left corner of the patient’s mouth. The patient’s eyes are either fully protected with a transparent cover or partially covered, leaving the medial portion accessible for the surgeon to monitor for signs of orbital hematoma. Both nasal cavities are initially packed with cotton soaked in oxymetazoline for decongestion. The patient is then draped. If navigation is utilized, preoperative imaging should be uploaded to the system, and the accuracy of the tracking system should be verified. 2
Techniques for ESS
Endoscopic Uncinectomy Procedures Using FESS Technology
Uncinectomy is sometimes the first step in FESS. If the uncinate process can be seen without touching the central turbinate first, then uncinectomy can be done without intermediate steps. To minimize mucosal irritation and the possibility of fracturing the central turbinate, medialization is performed carefully utilizing the curved section of the Freer elevator. 8
After that, a sickle knife or the pointed end of a Freer elevator can be used to make an incision and execute the uncinectomy. The incision should be made in the anterior part of the uncinate process since it is softer than the more rigid lacrimal bone, which houses the nasolacrimal duct. The free uncinate edge is then removed using Blakesley forceps. 8
Antrostomy and ethmoidectomy
The natural ostium of the maxillary sinus can be located once the uncinate process has been eliminated. At this point, the lamina papyracea can be palpated to check for dehiscence and to verify its placement over the eye that has been shielded. The standard placement of the ostium is one-third of the way back, at the level of the inferior margin of the middle turbinate. 8
The natural ostium is enlarged radially with a cutting device. It is debatable what the best diameter for a maxillary antrostomy should be, but 1 cm is usually sufficient for adequate outflow and postoperative monitoring in the office. Avoid puncturing the papyraceous lamina at all costs. 8
Anterior ethmoidectomy
The next step is to locate and then open the ethmoid bulla. The inner and medial aspects of the bulla can be opened with a J-shaped curette. After accessing the cell, the bony parts can be removed with true-cutting forceps or a microdebrider. Removing the whole lateral bulla improves visibility and allows for more precise dissection in the back. Once again, the lateral lamina papyracea should be protected at all costs. 8
Using a J curette, the remainder of the anterior ethmoid cells can be uncapped; a microdebrider or true cutting forceps can open the cells further. Before further opening of cells using powered instruments, a curette can be used to gain a tactile sense of the bone, determine its thickness, and confirm the correct orientation. Mucosal preservation leads to better postoperative outcomes. It is essential to minimize mucosal stripping wherever possible. 8
Clearing the anterior ethmoid cells to the skull base requires the surgeon to proceed cautiously as they near the ethmoid roof while referring back and forth between the endoscopic image and the preoperative CT scan. While image-guided or computer-assisted surgery can help a surgeon gauge the distance to the skull base, it cannot substitute an in-depth understanding of the human body. 8
The surgeon should always enter inferiorly and medially when progressing posteriorly to new air cells and then open laterally and superiorly once the more distal anatomy can be determined visually and feeling. When the basal lamella of the middle turbinate is reached, the anterior ethmoidectomy is finished. 8
Posterior ethmoidectomy
The first step in posterior ethmoidectomy is perforating the basal lamella immediately above and to the side where the middle turbinate vertical and horizontal segments meet. The inferior coronal part of the basal lamella and the posterior sagittal region of the middle turbinate must be protected. The integrity of the middle turbinate depends on this L-shaped strut being maintained. The microdebrider can then scrape away the superior and lateral sides of the basal lamella. 8
It is possible to remove more posterior ethmoid cells in a similar method, paying attention to the position of the skull base and lamina. The surgeon has to be aware that the skull base usually slopes downward at an inclination of around 30° from front to back. This means that the skull base is more dorsally positioned than frontally. The sphenoid is revisited for this dissection. 8
Manifest of Safer FESS is shown at Figure 1.
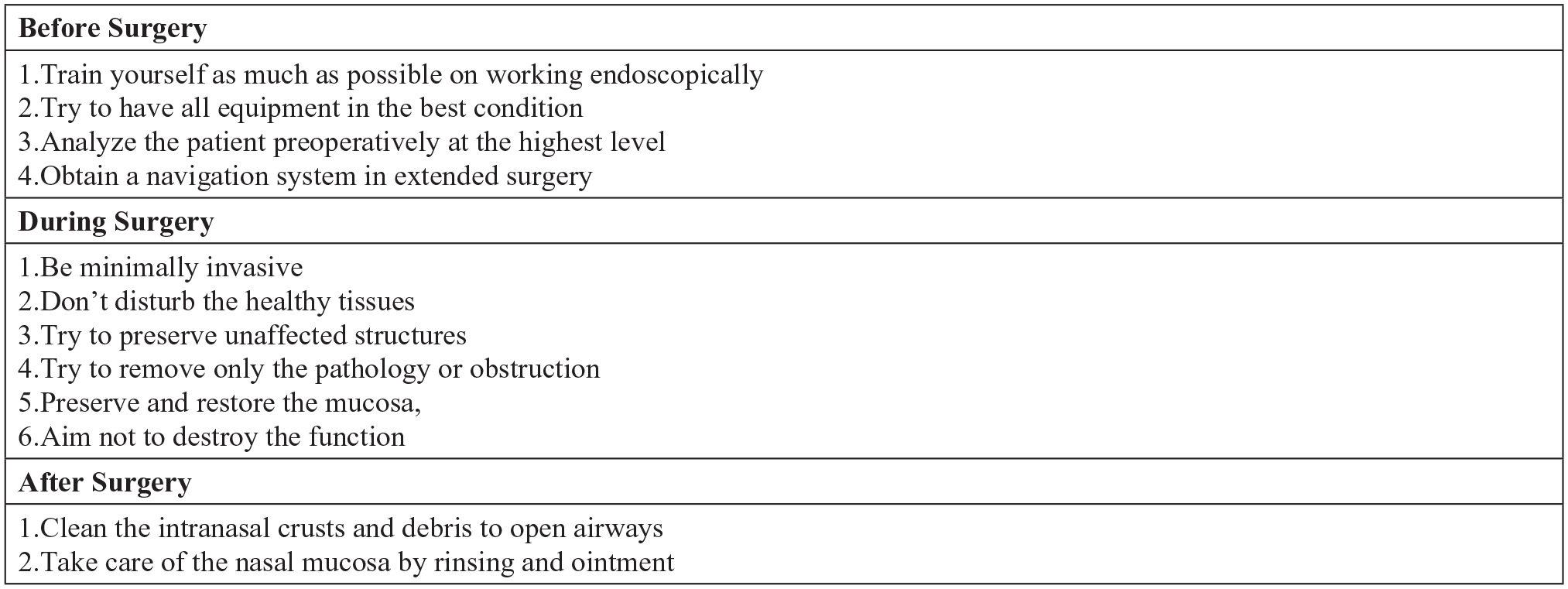
Manifest for safer FESS. FESS, functional endoscopic sinus surgery.
Complications and Ways to Avoid Them
Endoscopic sinus surgery complication rates are challenging to pinpoint because of the vast variation in how often problems are reported. FESS riskiness is seldom compared to alternative surgical procedures in prospective, randomized studies. FESS has a complication rate of 0% to 1.5% for significant problems and 1.12% to 20.8% for minor issues. Powered instrumentation may enhance the severity of the problems rather than the number of occurrences. 29
Orbital Injury
The maxillary and ethmoid sinuses flank the orbit on its inferior and medial sides. Staying low and locating and medializing the uncinate off the orbital wall are crucial steps in a successful uncinectomy. Identifying the inferior orbital wall as a marker and locating the ostium while doing a maxillary antrostomy is also vital. During an ethmoidectomy, the lamina papyracea is carefully examined on the CT scan to detect any dehiscence that might cause a breach of the orbital wall. Dehiscence of the optic nerve and carotid artery should also be evaluated, as should the presence of sphenoethmoidal cells. If bleeding develops from a break in the lamina papyracea, the periorbita should be checked, and ophthalmology should be contacted intraoperatively to take a reading of intraocular pressure. If it is, mannitol and intravenous dexamethasone should be given in addition to eye massage. Anesthesia patients with intraocular pressures above 40 mm Hg and awake patients with retro-orbital discomfort, cherry red macula, or a Marcus-Gunn pupil require more severe treatment, such as canthotomy or cantholysis. 30 The bleeding vessel must be located and staunched once the canthotomy has been done. Prompt treatment of orbital hematomas is essential to prevent optic nerve compression and ischemia. 2
Skull Base Injury/Cerebrospinal Fluid Leaks
Skull base slope, sinus height, skull thickness, and the presence of dehiscence may all be evaluated with great accuracy using a preoperative CT scan. Because the length of the lateral lamella varies from patient to patient and side to side, the Keros classification helps gauge both. 2
Keros Classification 1 :
Keros type I: 1 to 3 mm deep
Keros type II: 4 to 7 mm deep
Keros type III: 8 to 16 mm deep 2
The likelihood of sustaining a skull base injury increases in proportion to the depth of the lamella (Higher Keros type). Most injuries occur at the cribriform plate and lateral lamella. 2
Both intraoperatively and postoperatively, CSF leaks can be detected. 2
Intraoperative detection of CSF leakage necessitates immediate localization of the leaking structure. The danger of infection increases and hospital stays are longer when investigation is delayed. The area should first be cleaned. Overlay autogenous mucosal grafts are utilized to close defects less than 2 mm. The middle turbinate can be used as a composite graft (bone and mucosa) for lesions between 2 and 6 mm in size; for defects more significant than 6 mm, a multilayered restoration is done using cartilage or bone and fibrin glue.31,32 Depending on the severity and location of the defect, allogenic materials, and vascularized flaps may also be employed. 33 The Valsalva maneuver and subsequent rise in intracranial pressure can be reduced by thorough extubation after the surgery. 2
A CT scan is done to rule out cerebral hemorrhage and pneumocephalus. 2
Beta transferrin, intrathecal fluorescein, and imaging can be used to identify a CSF leak. 2
Epistaxis
Normal postoperative bleeding lasts only a few days, but severe bleeding requires immediate medical attention. Initial steps in treating severe bleeding include stabilizing the patient and protecting the airway. Any anticoagulants that have been restarted postoperatively should be temporarily stopped. The nose needs examining to find the source of the bleeding. Topical vasoconstrictors, resorbable or non-resorbable packing, cautery (silver nitrate or electrocautery), and other methods are available for treating epistaxis. Surgical artery ligation or endovascular embolization may be explored if the bleeding is severe and cannot be stopped by other means. 34 The surgeon needs an in-depth understanding of how the nose’s blood vessels work. In extreme cases of posterior epistaxis, the sphenopalatine artery can be ligated where it enters the nasal cavity through the sphenopalatine foramen at the rear end of the middle turbinate. 2
Recurrence of the disease
Although for patients with persistent sinus infections, FESS has emerged as the gold standard in treatment,2,35-39 there is usually recurrence or persistence of disease after FESS for CRS as surgery is very rarely curative (Fokkens et al., 2020). 40 Recirculation of mucus through the accessory ostium can occur if a maxillary antrostomy is not performed correctly, widening the natural ostium. 2
When the middle turbinates become more lateralized, it blocks the sinuses from draining properly. Nasal blockage after sinus surgery can also be caused by synechiae, which can develop if the nose is not properly cleaned and cared for after surgery. Possible reasons for recurrent rhinosinusitis include (1) excessive removal of mucosa, (2) bone exposure, and (3) failure to remove bony septations. 2
Image-Guided/Endoscopic Sinus Surgery for Safe FESS
In image-guided surgery (IGS), surgeons employ preoperative imaging data to pinpoint the exact position of a surgical tool concerning surrounding anatomical structures in real time. Even though it was initially designed for use in neurosurgery, FESS has quickly become one of the most popular applications of the technique. Diseased sinus tissue is removed, the sinus mucosa is preserved, sinus airflow is restored, inflammation is decreased, and mucosal epithelial and ciliary clearance is restored.2,35-39
Since the introduction of the endoscopic method in the mid-1980s, image-guided surgery has become one of the most critical developments in endoscopic sinus surgery. The sinuses can be dissected in real time as the surgeon watches an anatomical dissection on a computer display. A computer, monitor, tracking system, surgical tools, and data transfer gear are used in all IGS systems. The instrument’s location may be determined in real time thanks to the tracking system, which can be electromagnetic or optical. 36
With the confidence that the essential landmarks remain safe, even complex anatomical relationships may be comprehended and treated. These treatments have high one-time costs but low per-case expenses. Choosing between an optical and electromagnetic system is secondary to adopting computer technology. Both techniques are frequently used because of the high quality of the anatomical data they produce.41,42
Avoiding skin incisions, FESS has also been used for transnasal access to orbital pathologies like decompression of the orbit and optic canal in Graves’ disease or posttraumatic optic neuropathy, lesions of the extraconal medial orbital apex or space, benign sinonasal tumors that have invaded the orbit medially, and fractures of the medial orbital wall. Mucoceles, invasive and noninvasive fungal sinusitis, silent sinus syndrome, pituitary tumors, CSF leaks, and lesions of the ventral skull base, petrous apex, or pterygomaxillary fossa are all conditions for which FESS has become routine therapy. 2
FESS 43 carries a risk of major postoperative problems because of the sinuses’ proximity to vital anatomical structures such as the nasolacrimal duct, orbit, optic nerve, cavernous sinus, internal carotid artery, skull base, and lip. Nasolacrimal duct and lacrimal sac injuries, orbital wall injuries, vision loss, cerebrospinal fluid rhinorrhea, and bleeding problems are all possible outcomes. 38 Hassanin et al 44 found that the incidence of adverse events related to IGS device use was less than 3%, with CSF leakage, tissue damage, and nervous system injury being the most common. Imprecision was the most often reported device failure. 44 According to research by Orozco et al, 45 navigational errors might increase the risk of CSF leakage and navigational abortion.
When both an otolaryngologist and a neurosurgeon are working on a patient, having the ability to merge MRI and CT images into a single navigational aid is a huge time saver. While CT scans are superior at outlining the skeletal architecture of sinonasal cavities for the otolaryngologist’s nasal approach, MRI guidance with improved cerebral soft-tissue delineation will be necessary for the neurosurgeon to proceed with the combined treatment. This is especially important when dealing with issues in the pituitary gland or the front of the skull base. 46 The image guidance system may create excellent soft tissue and bone detail in a single hybrid picture. 35
A CT scan is the first step in IGS. The author’s preferred method of acquiring CT scans is a helical, 2-mm-thick axial CT scan performed with a custom-made headgear that features in-built metallic fiducial landmarking. The surgical headgear was made to register images of the patient’s anatomy automatically. The imaging data set is downloaded onto a computer in the operation room. Before the operation, the photos are loaded into the IGS system, and quality and correctness are double-verified. 35
Conclusion
Inflammatory and infectious sinus illnesses are the most prevalent indications for FESS. The 4 most common methods for FESS are endoscopic uncinectomy, maxillary antral ostomy/ethmoidectomy, anterior ethmoidectomy, and posterior ethmoidectomy. Although the concept of FESS is simple, the diversity and severity of disorders treated present unique obstacles to the surgeon who is operating close to important structures such as the eyes and brain. Sinus surgery outcomes can be improved and problems avoided with careful preoperative preparation
Footnotes
Acknowledgements
None.
Author Contributions
Authors were equally contributed to the planning, literature survey, and writing the manuscript.
Availability of Data and Materials
All data for this manuscript is presented in the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Committee Approval
There is no need for ethics committee approval because this is a review paper.
Informed Consent
There is no need to take informed consent, because this is a review paper.
