Abstract
Preservation of residual hearing and vestibular function is a crucial factor in cochlear implantation (CI), especially in patients with residual low-frequency hearing thresholds. We report a case of a patient who underwent unilateral endoscope-assisted CI with a challenging surgical view following rigorous posterior tympanotomy. A 53-year-old male presented with left-sided intractable tinnitus due to sudden sensorineural hearing loss that had occurred 10 years prior. Due to the abnormal location of the round window (RW), which was far more posterior and inferior than usual and impeded insertion of the electrode using the conventional RW approach, endoscope-assisted CI was performed. Pure-tone audiometry at 3 months after CI revealed satisfactory hearing thresholds. Furthermore, there was alleviation of the left-sided tinnitus, which was indicated by a marked decrease in both the subjective visual analog scale loudness and Tinnitus Handicap Inventory scores. With proper indications, we strongly recommend applying the RW approach with endoscopic assistance over conventional bony cochleostomy for the preservation of low-frequency hearing thresholds in cases where RW visualization is insufficient following posterior tympanotomy.
Introduction
Preservation of residual hearing and vestibular function is a crucial factor in cochlear implantation (CI), especially in patients with residual low-frequency hearing thresholds. Electrode insertion can be achieved through a round window (RW) or bony cochleostomy 1 ; however, the RW approach is preferred since it does not require direct cochlear drilling and bone dust does not enter the scala tympani. 2 In addition, the introduction of novel electrode array designs, including slim perimodiolar or lateral wall electrodes, has further improved the RW approach using 2-handed surgery techniques. 3 Intracochlear trauma is associated with postoperative loss of residual hearing4,5; therefore, it is important to minimize cochlear damage using the RW approach in terms of both postoperative speech outcomes and preservation of residual hearing function. 6
After mastoidectomy, posterior tympanotomy is usually performed to visualize the RW within the surgical field of view. However, anatomical variations such as prominent posterosuperior bony overhanging may impede satisfactory exposure of the RW membrane (RWM) through the facial recess in posterior tympanotomy. Drilling the overhangs is a promising alternative, as demonstrated by an increase in the visibility of the RWM by 1.5 to 3 times compared with conventional posterior tympanotomy. However, excessive bone drilling increases the risk of damage to the bony annulus, facial nerve, and chorda tympani nerve.7-9
The St. Thomas Hospital (STH) classification is used to categorize RW exposure, with Type III denoting cases where RW visualization is the most challenging. 10 This article describes the case of a patient who underwent unilateral endoscope-assisted CI with a challenging surgical view following rigorous posterior tympanotomy.
Case Report
Given the retrospective nature of the chart review, this study was approved, and the written informed consent was waived by the Institutional Review Board of the Clinical Research Institute (IRB number: 2024-08-001). A 53-year-old male patient with left-sided hearing loss due to sudden sensorineural hearing loss (SSNHL) that had occurred 10 years prior visited an outpatient clinic with a complaint of severe left-sided tinnitus. He had attempted auditory rehabilitation using a left-sided hearing aid; however, this did not alleviate the symptoms. The initial audiogram revealed an average threshold of 80 dB HL with a speech discrimination score of 0%; however, the low-frequency thresholds were preserved (20 dB HL and 40 dB HL at 250 and 500 Hz, respectively; Figure 1). Preoperative temporal bone computed tomography (TBCT) revealed no other remarkable findings.
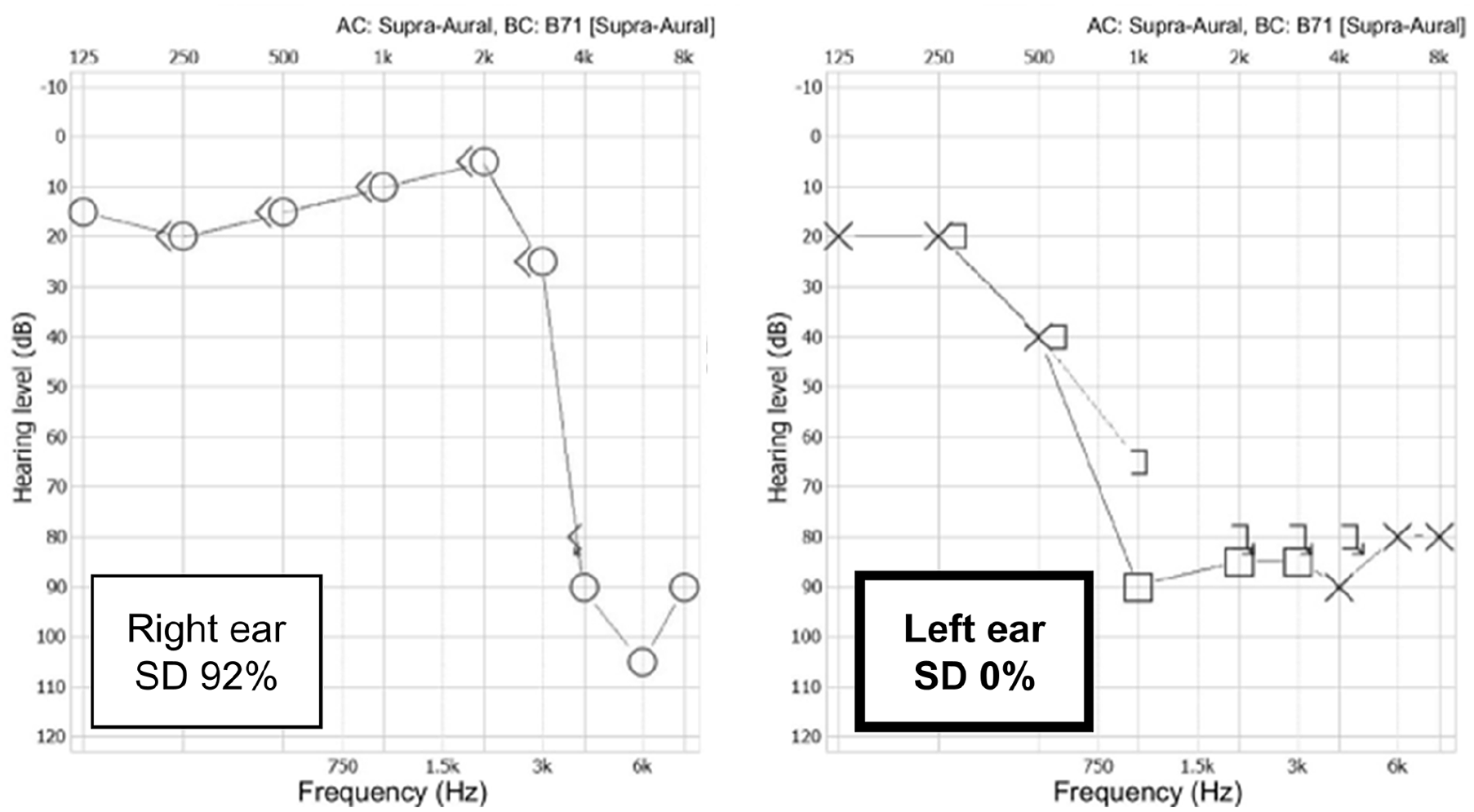
An initial preoperative audiogram following unrestored left-sided sudden sensorineural hearing loss revealed an average threshold of 80 dB HL, with a speech discrimination score of 0%. The low-frequency hearing thresholds were preserved, showing 20 dB HL and 40 dB HL at 250 and 500 Hz, respectively.
Cortical mastoidectomy was performed under general anesthesia following the protocol. However, after posterior tympanotomy, the RW could not be visualized even after modifying the microscopic view, tilting the angle of the surgical table, and drilling the facial nerve canal to the maximum level (Figure 2A). To avoid bony cochleostomy to preserve the low-frequency hearing, a 3 mm, 0°, 14 cm ear endoscope was inserted through the posterior tympanotomy site, which allowed identification of the RW. Specifically, it revealed that the RW was located far more posteriorly and inferiorly than usual (Figure 2B). After drilling the RW niche, the membrane was visualized and pierced using a sharp pick. Subsequently, a straight electrode array (MED-EL FLEX 24) was inserted using the endoscope-assisted RW approach without any complications (Figure 2C). The RW opening was sealed using soft tissue. Moreover, the surgery was completed after confirming that the auditory nerve response telemetry on each electrode channel yielded satisfactory electrically evoked compound action potentials.
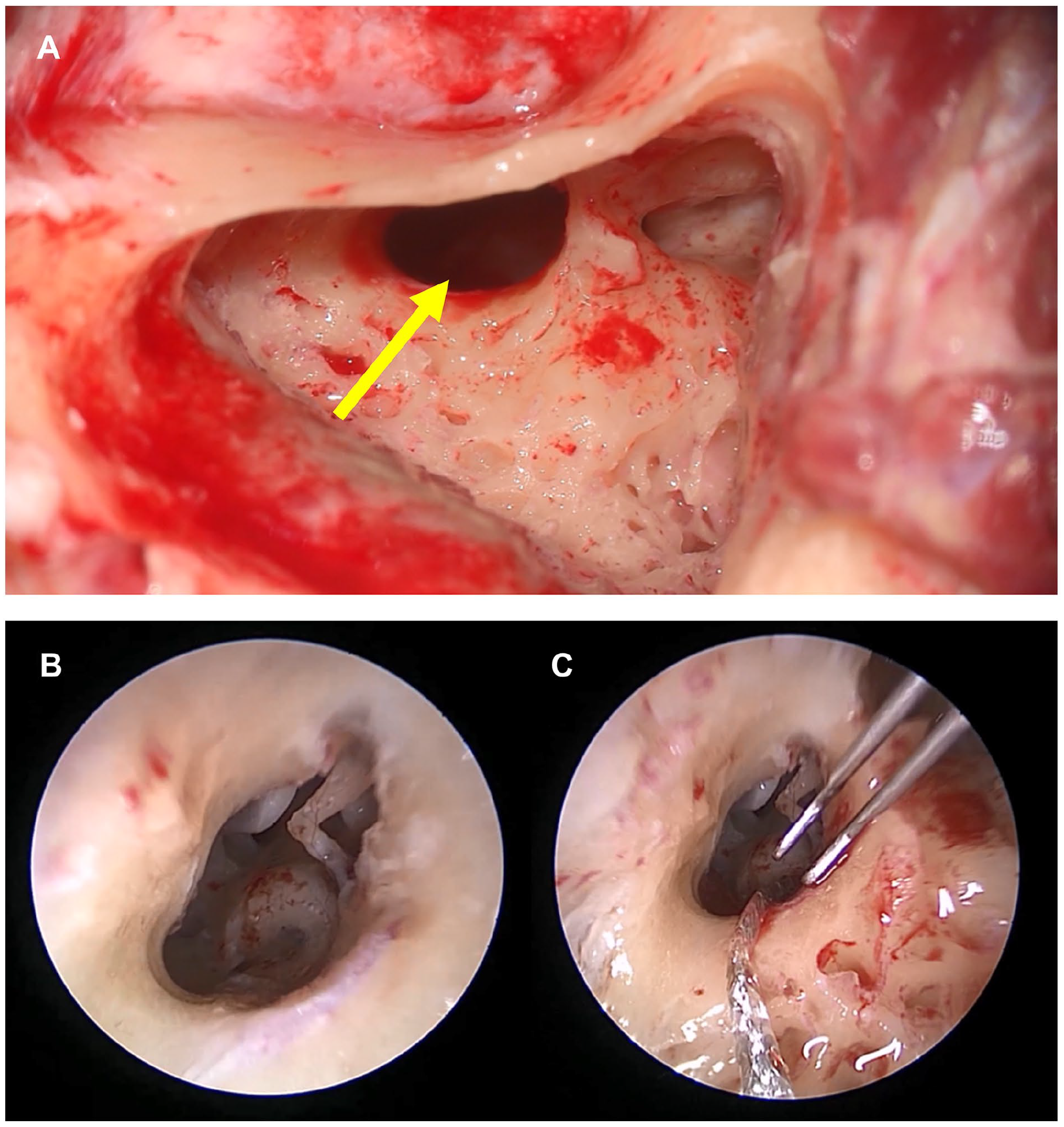
(A) A RW was not visualized after conventional mastoidectomy and posterior tympanotomy. Further drilling of the anterior bony canal of the facial nerve was implemented to the maximum level. However, even after modifying the microscopic view and tilting the angle of the surgical table, the RW niche was not visualized. The yellow arrow indicates the hidden RW niche. (B) After applying a 3 mm, 0°, and 14 cm ear endoscope through the posterior tympanotomy site, the RW was finally visualized and was found to be located more posteriorly and inferiorly than usual. (C) After drilling the RW niche, the RW membrane was visualized and pierced with a sharp pick. A straight electrode array (MED-EL FLEX 24) was successfully inserted using the endoscope-assisted RW approach without any complications.
There were no postoperative complications such as facial nerve paralysis or dizziness. The postoperative cochlear view revealed that the electrode was well inserted through the RW and cochlear turns (Figure 3A). Pure-tone audiometry at 3 postoperative months revealed satisfactory hearing thresholds (Figure 3B). Pre- and postoperative subjective tinnitus loudness was evaluated using a visual analog scale (VAS; answering the question “how loud is your tinnitus?” on a 0 to 10 scale) and the Tinnitus Handicap Inventory (THI, 0-100 scale). At 3 months after implantation, the patient showed alleviation of the left-sided tinnitus, which was demonstrated by a marked decrease in both the subjective VAS loudness and THI scores (from 8 to 2 and from 74 to 0, respectively).
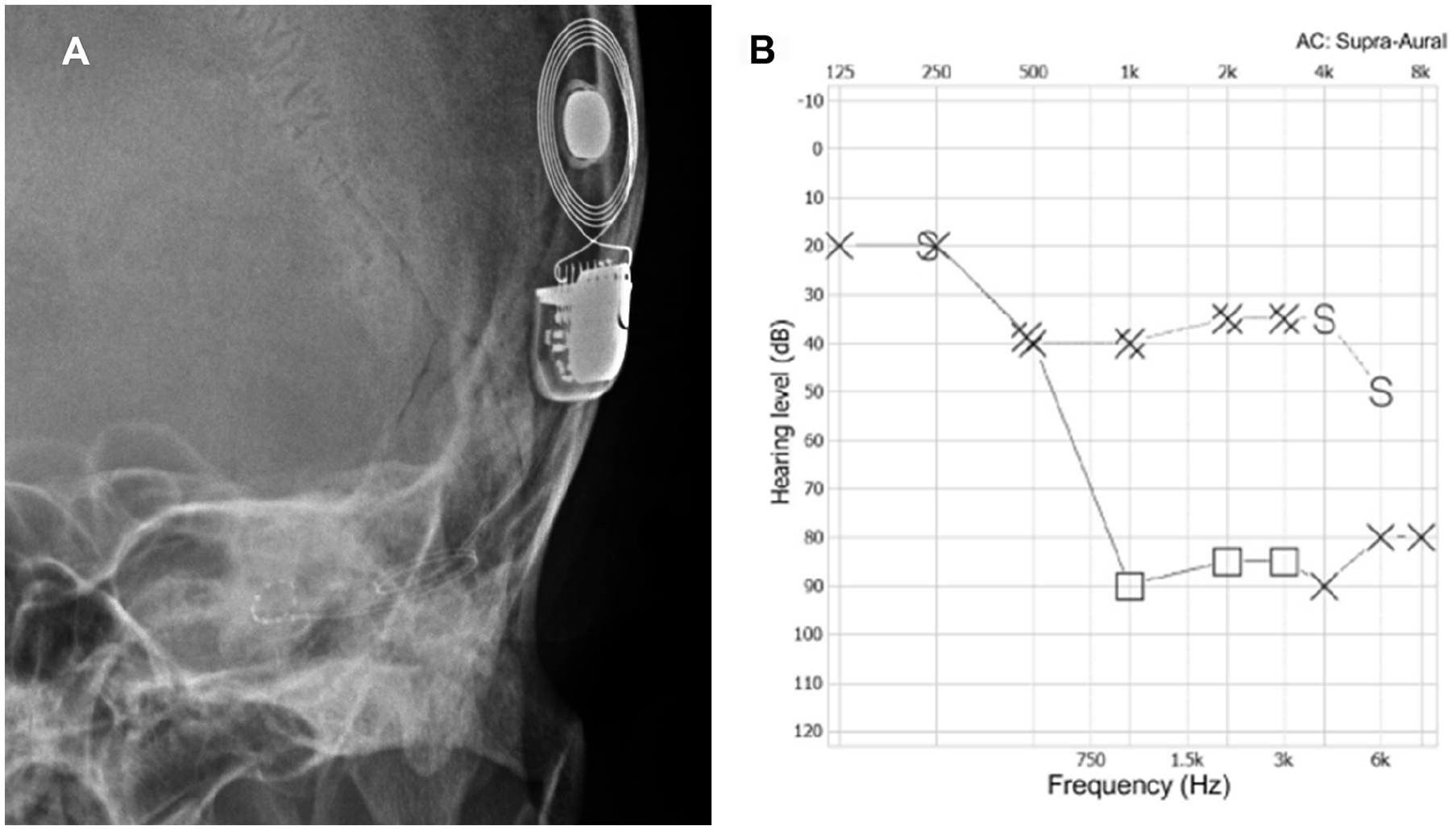
(A) A postoperative cochlear view indicated a well-inserted electrode through the RW and cochlea turns. (B) Pure tone audiometry at 3 postoperative months with the CI device inserted indicated satisfactory hearing thresholds.
Discussion
This study describes the protocol for addressing a challenging surgical view when preservation of low-frequency hearing is necessary, which is among the most important considerations in the management of electrode implantation. Several intra- or postoperative factors, including preservation of low-frequency hearing, improvement of speech perception, and protection of the facial nerve, should be considered during CI. Some patients with severe hearing impairment have preserved low-frequency hearing thresholds. 11 Preserving this residual hearing is essential for improving postoperative speech perception and tinnitus.12-14 The occurrence of tinnitus is considered to involve a neuroplastic response to auditory deprivation.15,16 Although the mechanism underlying the alleviation of tinnitus after CI remains unclear, acoustic masking provided by CI 17 or plastic changes in the central auditory system induced by continuous CI stimulation 18 are considered to be involved. Therefore, in patients with severe peripheral auditory deafferentation (ie, sensorineural hearing loss), reafferentation of the ascending auditory nervous system with CI and preservation of residual hearing may alleviate tinnitus.
In our case, the chief complaint was left-sided tinnitus following SSNHL, which required the simultaneous recovery of the mid-to high-frequency hearing thresholds as well as preservation of the low-frequency hearing thresholds. To preserve low-frequency hearing, the RW approach is preferred over classical cochleostomy as a route for electrode insertion due to minimal intracochlear trauma as well as decreased fibrosis and ossification. This is because the RW approach uses the natural RW opening and does not create a separate opening for CI electrode insertion. 19 This “soft surgical technique” facilitates preservation of residual hearing following CI 20 as well as stronger electrical stimulation of cochlear nerve endings in the modiolus and better postoperative outcomes regarding speech perception, production, and language acquisition. 10
In some cases, visualization of the RW may remain difficult even after all possible surgical methods. As aforementioned, the STH classification is used to categorize the degree of RW exposure following the creation of a sufficient posterior tympanotomy site as a guide for the safe insertion of electrode arrays. 10 According to this classification, type I, II, and III RWMs indicate 100%, 0% to 100%, and 0% exposure, respectively. Type II can be subdivided into types IIa (50%-100% exposure) and IIb (0%-50% of exposure). Type III RWMs cannot be visualized even after all the best surgical efforts, which is similar to our case. Type III occurs when the RWM lies more posteriorly and closer to the facial nerve, which impedes the implementation of the RW approach. Accordingly, patients with type III RWM often require a bony cochleostomy for successful CI, which impedes the preservation of residual hearing. In such cases, using an endoscope can allow better surgical views, which facilitates guidance of the electrode using the RW approach rather than conventional bony cochleostomy.
Endoscopic ear surgery (EES) is a minimally invasive surgical technique for treating various middle ear diseases. 21 A crucial advantage of EES is its superior visualization and higher magnification compared with microscopy, which allows visualization of complex parts of the middle ear anatomy at a very high resolution. As in our case, structures that are difficult to observe using a conventional microscope can be better visualized using a straight or angled endoscope at various angles. There is limited evidence regarding the use of endoscopy for CI given that EES is limited by the fact that 2-handed surgery is impossible. Specifically, procedures that involve controlling perimodiolar electrodes require advance-off stylets, which should be manipulated delicately using both hands. Therefore, we instead used a straight lateral wall electrode to allow single-handed manipulation. Moreover, if the surgery had been performed without an endoscope, the posterior external acoustic canal wall would have been removed after canal wall-down mastoidectomy. This may yield various postoperative side effects due to unnecessary bone drilling and the wide mastoid space. Considering the adverse effects of an extensive surgical range and possible occurrence of facial nerve injury, endoscope-assisted CI via the RW approach should be considered in patients with poor RWM visibility when hearing preservation is required. However, it is important to note that the use of an endoscope during implantation foregoes the advantages of the 2-handed surgery technique and selection of electrode type (ie, straight or perimodiolar electrode array).
This study has several limitations. First, we did not consider factors such as the electrode insertion angle, depth, and length, which influence the degree of hearing preservation. 22 Second, the intraoperative surgical views would have been predictable if the anatomical relationships among the RW, facial recess, and facial nerve had been analyzed on preoperative TBCT scans. Several studies have measured the radiological angles for preoperative prediction of the residual hearing or visibility of the RW niche.22-26 For instance, among previously reported measurements, we have utilized a novel technique first described by Mandour et al. 27 When comparing the RW visibility of 6 different CI cases between postoperative documentation by 1 surgeon (J.D.L.) and preoperative predictions which were analyzed with the method by Mandour et al., all cases exhibited consistent descriptions (Supplemental Figure 1). Specifically, as expected, the TBCT image of our case showed intersecting lines, implying poor RW visibility. As such, the endoscope-assisted RW approach had to be applied to preserve low-frequency hearing thresholds, albeit of other conventional surgical indications (ie, bony cochleostomy). Resultantly, these preoperative considerations would facilitate anticipation of intraoperative difficulties, including challenging surgical views.
Conclusion
Taken together, with proper indications, we strongly recommend implementing the RW approach with endoscopic assistance over conventional bony cochleostomy to preserve low-frequency hearing thresholds when the RW cannot be sufficiently visualized after posterior tympanotomy.
Supplemental Material
sj-docx-1-ear-10.1177_01455613241285679 – Supplemental material for Cochlear Implantation in a Patient with Intractable Tinnitus: A Case Report of an Endoscope-Assisted Approach
Supplemental material, sj-docx-1-ear-10.1177_01455613241285679 for Cochlear Implantation in a Patient with Intractable Tinnitus: A Case Report of an Endoscope-Assisted Approach by Seung Jae Lee and Jong Dae Lee in Ear, Nose & Throat Journal
Footnotes
Author Contributions
Conceptualization: J.D.L.; data curation: S.J.L.; formal analysis: S.J.L.; investigation: S.J.L. and J.D.L.; supervision: J.D.L., visualization: S.J.L.; writing—original draft: S.J.L.; writing—review and editing: J.D.L.
Data Availability Statement
The data used to support the findings of the study are included within the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 2023 Inje University research grant (granted to S.J.L.) and Soonchunhyang University Research Fund (granted to J.D.L.).
Ethical Statement
This study was approved by the Institutional Review Board of the author’s institute (IRB number: 2024-08-001), and the requirement for informed consent from the patient was waived. The patient’s anonymity is preserved in the article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
