Abstract
Nearly one third of nasopharyngeal carcinomas (NPCs) in the United States are associated with human papillomavirus (HPV). Surveillance for Epstein-Barr virus (EBV)-negative subtypes is customarily based solely on imaging and physical examinations. We present a case of HPV-positive NPC using serial circulating tumor HPV DNA (ctHPVDNA) as a biomarker of disease status. A 58-year-old Caucasian female initially presented with T1N1M0 EBV-negative p16-positive squamous cell carcinoma of the nasopharynx and was treated with concurrent chemoradiation. Regional nodal recurrence identified 7 months post-radiotherapy was treated with salvage left neck dissection. Surveillance was supplemented using a commercially available polymerase chain reaction (PCR)-based ctHPVDNA assay. Rising ctHPVDNA levels at 9 and 10 months post salvage surgery prompted positron emission tomography (PET). Biopsy confirmed recurrence in avid right hilar and paraoesophageal lymph nodes. Following definitive radiotherapy to the involved nodes and concurrent pembrolizumab, posttreatment ctHPVDNA decreased to baseline, but then increased after 6 cycles of pembrolizumab. Follow-up PET found left mediastinal recurrence outside of the prior treatment field, which was treated with concurrent chemoradiation with cetuximab. Again, ctHPVDNA level dropped to baseline but increased 3 months postradiation. PET scan showed a left lung nodule, and the patient received systemic therapy. Plasma ctHPVDNA monitoring correlated well with disease activity in our patient with HPV-positive NPC. ctHPVDNA detected disease earlier than standard surveillance methods and allowed for earlier intervention. Larger studies are needed to validate the utility of HPV biomarker surveillance in NPC.
Introduction
In endemic areas of the world, Epstein-Barr virus (EBV) is commonly present within malignant cells and implicated in nasopharyngeal carcinogenesis. In nonendemic regions, about a third of nasopharyngeal carcinomas (NPCs) harbor human papillomavirus (HPV) rather than EBV.1-3 Although routine posttreatment surveillance of EBV-associated NPC includes imaging and monitoring of EBV viral load, EBV-negative NPC is typically surveilled by physical examination and imaging alone. 4
Emerging evidence suggests that noninvasive, blood-based monitoring of tumor biomarkers can be highly predictive of cancer recurrence, such as in HPV-positive oropharyngeal squamous cell carcinoma (OPSCC).5-12 To our knowledge, published reports of biomarker surveillance for HPV-positive NPC have been extremely limited, with previous studies including a prospective study using HPV circulating tumor DNA (ctDNA) to diagnose and monitor a cohort of 4 NPC patients, 13 a retrospective case series of 2 NPC patients, 14 and a case report. 15 We herein report the case of a patient whose recurrence of HPV-positive, EBV-negative NPC was detected by plasma HPV ctDNA monitoring prior to development of clinical symptoms or imaging findings representative of disease recurrence. A review of pertinent literature supports further investigation of this strategy for early prediction of relapse.
Case Presentation
A 58-year-old Caucasian female never-smoker with no significant past medical history presented with T1N1M0 (American Joint Committee on Cancer eighth edition) EBV-negative (tissue and blood), p16-positive (tissue and blood) nonkeratinizing squamous cell carcinoma of the nasopharynx. 16 Initial nodal involvement on pretreatment imaging included left levels II and III. She was treated with intensity modulated radiotherapy (IMRT) to a total dose of 70 Gy in 35 fractions with concurrent weekly cisplatin chemotherapy. Posttreatment positron emission tomography (PET) with computed tomography (CT) 3 months after treatment showed questionable uptake in a nasopharyngeal mass and left cervical lymph nodes. An active surveillance approach was taken and repeat PET/CT 7 months after treatment revealed resolution of the nasopharyngeal mass; however, an enlarging hypermetabolic left cervical node at levels IIb and IV were noted (Supplemental Figure 1). A fine needle biopsy of a left cervical lymph node confirmed persistent disease or recurrence of her NPC after primary therapy. After discussion at our institutional multidisciplinary tumor board, salvage left neck dissection of levels II to V without adjuvant therapy was performed and revealed tumor consistent with recurrence in 12 of 54 lymph nodes. The largest node measured 2.4 cm, and extracapsular extension was present in multiple nodes at level IIA, IIB, III, and IV. Tumor tissue was positive for p16 by immunohistochemistry and EBV was negative based on EBER in situ hybridization. Furthermore, HPV positivity was confirmed via tumor tissue modified viral HPV16 DNA (TTMV-HPV16 DNA) detection in blood. Next-generation sequencing revealed a PTEN inactivating variant, low-level expression of PD-L1 RNA, low tumor mutation burden, microsatellite stable, and low score on the immune signature gene expression algorithm.
Thereafter, the patient was followed every 3 months using routine clinical examinations and imaging studies. During her first follow-up visit 3 months after surgery, whole body PET/CT and neck CT scan showed no evidence of disease. The patient advocated for additional surveillance given her history of recurrence. Five months after surgery, a commercially available digital droplet PCR test to quantify HPV DNA fragments (NavDx; Naveris) was performed with undetectable results. Follow-up clinical and imaging evaluations at 6 and 9 months from surgery continued to show no evidence of disease. However, at the 9 month postsurgical visit, a second HPV ctDNA test revealed 285 fragments of HPV16 DNA/mL of plasma (>7 fragments/mL is considered positive). A repeat assay at 10 months post-surgery was once again positive at 300 fragments/mL of plasma. With 2 consecutive positive HPV16 ctDNA results and no evidence of disease by clinical examination or conventional CT imaging, a PET/CT was performed (Figure 1) which revealed new hypermetabolic soft tissue densities correlating with a lymph node in the right hilum (0.7 cm) and a paraoesophageal lymph node anterior to the lower portion of the esophagus (1.3 cm) outside of the previously treated areas. Endoscopic ultrasound visualized a malignant appearing 2.3 cm lymph node at station 8L, and fine needle aspiration of this node confirmed p16+ carcinoma consistent with NPC metastasis. With no other known sites of active disease, the patient received definitive treatment (curative intent) of her oligometastatic disease with IMRT targeting the involved nodes to a total dose of 45 Gy in 15 fractions with concurrent and adjuvant pembrolizumab (6 cycles total; off-label use not approved for this indication).
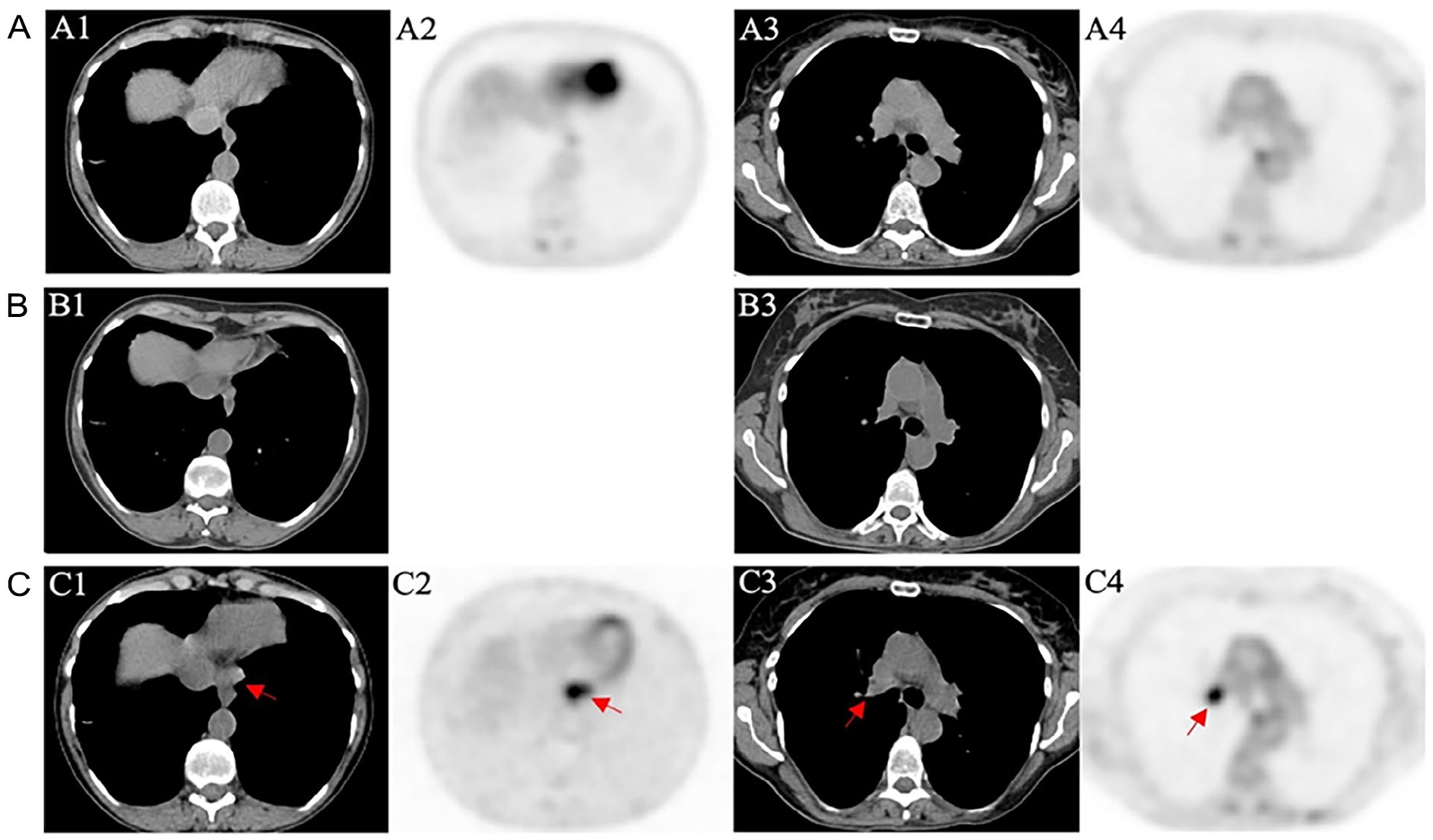
Surveillance imaging of the chest at 3 (A), 9 (B), and 11 months (C) post-salvage neck dissection. PET/CT was performed at 3 (A) and 11 months (C); only CT was performed at 9 months (B). Columns 1 and 2 consist of images of the lower mediastinum while columns 3 and 4 correspond to the level of the hilar regions. Standard posttreatment PET/CT (A) showed no evidence of disease. CT imaging at 9 months (B) failed to detect signs of disease despite positive ctHPVDNA results at this time prompting the subsequent PET/CT (C), which demonstrated hypermetabolic masses in the mediastinum (red arrows in C1/C2) and right hilar region (red arrows in C3/C4) later confirmed by biopsy to be consistent with NPC recurrence. PET, positron emission tomography; CT, computed tomography; ctHPVDNA, circulating tumor human papillomavirus DNA.
HPV ctDNA measurement after completion of radiation therapy remained near pretreatment levels (298 fragments/mL of plasma) and then decreased to 227 fragments/mL of plasma following 3 cycles of pembrolizumab. At the end of 6 cycles of pembrolizumab, ctHPVDNA levels increased to 2913 fragments/mL of plasma (Figure 2). The latter result prompted another PET/CT which showed involved mediastinal lymph nodes (left upper paratracheal, pre-vascular, left hilar, and subcarinal) outside the prior radiation field. The patient was treated with concurrent cetuximab with IMRT to a total dose of 45 Gy to the new areas of disease approximately 16 months after her salvage neck dissection. Her HPV ctDNA level decreased but continued to be detectable (287 fragments/mL of plasma) 1 month after treatment completion. However, her circulating tumor HPV DNA (ctHPVDNA) levels increased significantly (6734 fragments/mL of plasma) after 3 months, prompting concern for ongoing disease, and PET/CT confirmed progressive disease in the left lower lobe of the lung. Given concern for metastatic disease, the patient was started on combination systemic therapy with carboplatin, paclitaxel, and pembrolizumab.
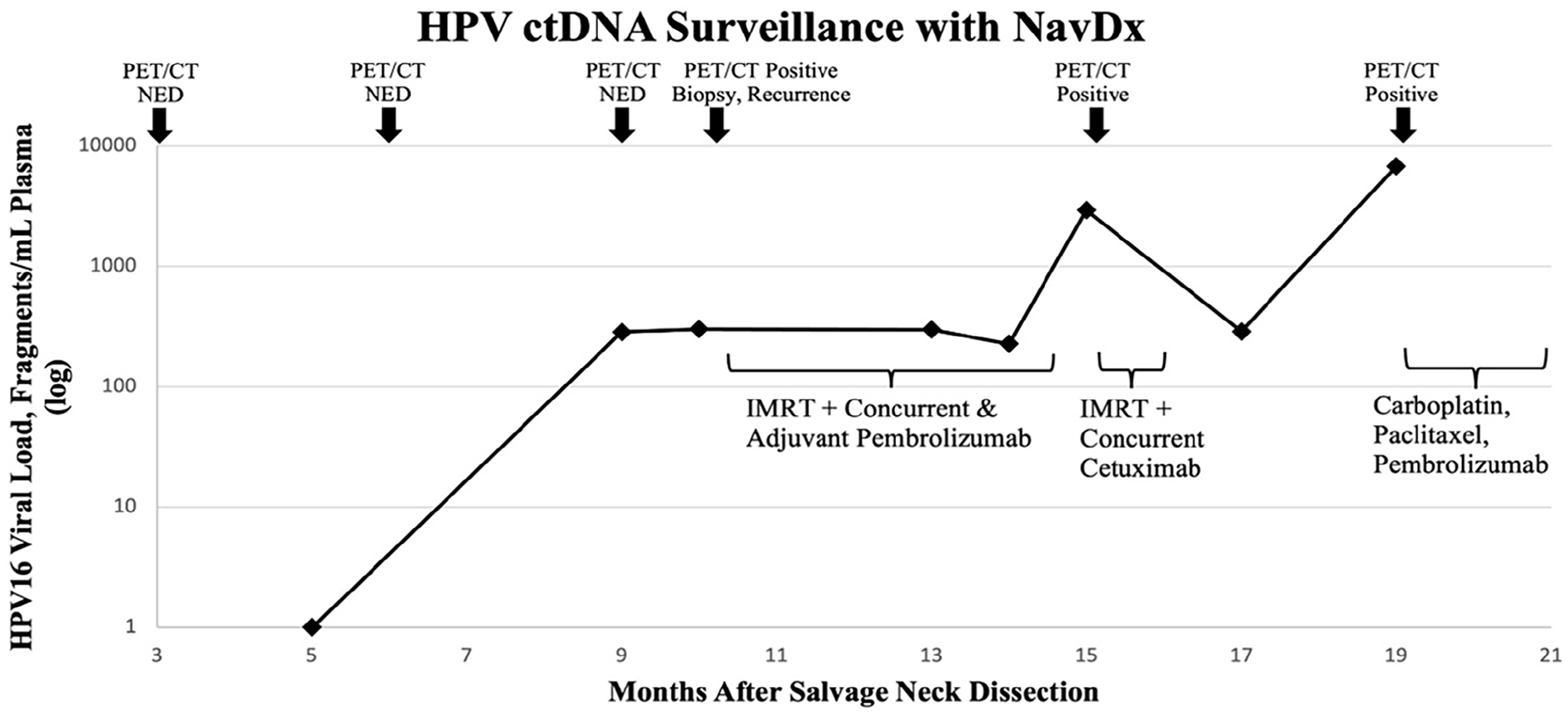
Graph of HPV ctDNA levels over time using a commercial blood-based assay that quantified viral DNA fragments in our patient. Rising levels predated and correlated well with positive findings on PET/CT and biopsy results. NED, no evidence of disease; ctDNA, circulating tumor DNA; PET, positron emission tomography; CT, computed tomography.
Discussion
Nearly one third of NPCs in the United States are positive for HPV, 17 yet practice guidelines do not currently recommend HPV testing in NPC. This is partly due to lack of evidence on how knowledge of HPV status might affect prognostication and improve management.2,3 With recent advances in ctDNA technology, there is potential for a sensitive yet noninvasive test to monitor HPV-related cancers across anatomic sites (cervical, anal, oropharyngeal, and nasopharyngeal).5,18-20 Here, we present a case in which recurrence of a patient’s HPV-positive NPC was detected by ctHPVDNA prior to the onset of symptoms or evidence of disease on standard clinical examinations or imaging studies. This suggests a role for serial ctHPVDNA measurements to monitor disease status. In our case, the patient’s ctHPVDNA levels tracked with burden of disease, and ctDNA monitoring provided a lead-time of at least 1 month. The findings suggest that plasma ctHPVDNA kinetics over time could help predict recurrence or at least trigger more intense surveillance and earlier intervention when relapse is confirmed.
Since the nasopharynx and oropharynx are adjacent anatomic structures, a major question that arises is whether these tumors are artificially divided and in fact have similar disease biology when HPV is an underlying driver of the disease. Indeed, histopathology is similar across HPV-positive cancers and they tend to be more differentiated with more keratinization and fewer lymphocytic infiltrates than nonvirally mediated head and neck cancers. 21
Current guidelines for follow-up of squamous cell carcinoma of the head and neck call for affected patients to be followed by physical examinations. 4 However, large studies of patients with HPV-associated OPSCC have shown that clinical surveillance alone is of limited utility. 22 In addition to clinical examinations, PET/CT is the preferred imaging modality at 12 weeks after the completion of definitive chemoradiation to assess treatment response and to identify active disease throughout the body. 4 There are no consensus guidelines for imaging surveillance in asymptomatic patients beyond 6 months, and institutional practices vary. 4 A recent retrospective review on the use of PET/CT for surveillance showed clinically occult head and neck cancer in 9% (15 of 175) at 12 months, and 4% (3 of 77) at 24 months; however, long-term surveillance did not affect disease-free-survival or overall survival, and HPV status was not examined. 23
Our case illustrates the potential for a noninvasive blood test such as plasma ctHPVDNA level to be an effective tool to monitor tumor burden in patients with HPV-positive NPC. 24 The paradigm builds on decades of evidence showing that plasma EBV DNA viral load aids in risk stratification and can detect disease recurrence earlier than by clinical or imaging examinations.25-29 Blood testing also presents an opportunity to reduce financial toxicity through greater cost efficiency. A recent analysis estimated the cost of 3 distinct surveillance scenarios and found combining ctDNA testing with physical examinations and nasopharyngoscopy every 3 months for 2 years was substantially cheaper than surveillance strategies that followed traditional guidelines. 23
Recently, HPV DNA monitoring has been explored in HPV-positive OPSCC, and multiple studies utilizing PCR or sequencing assays have demonstrated that fluctuations in plasma HPV DNA can assist in gauging treatment response in early to advanced disease.6-12,29,30 One study used the same commercial ctHPVDNA assay (NavDx) that was applied in the current NPC case to show that OPSCC patients with rapid ctHPVDNA clearance have superior outcomes following completion of chemoradiation therapy. Conversely, patients with poor HPV ctDNA clearance kinetics have an increased risk for recurrent disease. 30 In a subsequent prospective trial, the NavDx assay was an effective tool in cancer surveillance with a positive predictive value of 94% and a negative predictive value of 100% for detecting disease recurrence after treatment. 5 Extrapolating from these data and supplementing with evidence in the current case report, it seems timely and important to study such an assay for its value in monitoring HPV-positive NPC patients so that earlier intervention might be considered compared to current surveillance strategies.
Supplemental Material
sj-docx-1-ear-10.1177_01455613241278756 – Supplemental material for Plasma Circulating HPV DNA Surveillance in a Patient With Nasopharyngeal Cancer
Supplemental material, sj-docx-1-ear-10.1177_01455613241278756 for Plasma Circulating HPV DNA Surveillance in a Patient With Nasopharyngeal Cancer by Siddharth Sheth, Tyler Walburn, Jason Tasoulas, Samip Patel, Ankit Agarwal and Margaret L. Gulley in Ear, Nose & Throat Journal
Footnotes
Data Availability Statement
Not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Sheth reported personal fees from Naveris, Medscape, and research funding from AstraZeneca, Merck, Inovio, Exelixis, Regeneron. Dr Sheth belongs to the speaker’s bureau of Exelixis. No other disclosures were reported.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statements
Not applicable.
Patient Consent
The investigators obtained consent from the patient to describe her case and clinical details in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
