Abstract
Introduction
Benign paroxysmal positional vertigo (BPPV) is a common vestibular disorder characterized by brief episodes of spinning sensation triggered by specific head movements. 1 There are 2 certain theories for the development of BPPV. The first one, cupulolithiasis theory, was described by Schuknecht. It is based on the attachment of otolithic debris to the cupula in crista ampullaris. Another one, canalithiasis theory, was proposed by Hall. This one is based on free-floating debris in the canal. It is worth noting that both of these theories highlight the presence of foreign particles in semicircular canal as the underlying mechanism of vertigo. 2 BPPV is the most prevalent cause of peripheral vertigo, affecting approximately 2.4% of the general population during their life. 3 BPPV significantly impacts patients’ quality of life, leading to functional limitations, anxiety, and impaired daily activities. 4
Uric acid (UA), a metabolic end product of purine metabolism, has been associated with various health conditions, including gout, cardiovascular diseases, and metabolic syndrome. 5 However, its link to BPPV remains controversial. Some studies suggest that the accumulation of purine crystal deposits, resulting from otoconial debris floating freely in the semicircular canals, could contribute to BPPV in individuals with gout. 6 Furthermore, heightened UA levels may stimulate the release of inflammatory mediators, leading to the generation of detrimental reactive oxygen species (ROS). 7 These ROS can harm blood vessels and disrupt blood flow. Consequently, the blockage of the vertebrobasilar arteries that supply the inner ear may contribute to peripheral vertigo syndromes like BPPV. 8 In contrast, some studies have mentioned an antioxidant role for UA, suggesting a protective role against inflammation and BPPV.9,10 Therefore, the exact role of UA in pathogenesis of BPPV is not fully understood. Also, existing research on the association between UA levels and BPPV has yielded conflicting results. Some studies report higher serum UA levels in BPPV patients compared to controls, while others find lower or no significant difference. 11 Thus, the purpose of our systematic review and meta-analysis is to assess the serum UA levels in BPPV patients compared to healthy controls.
Methods
Study Design
To address the main question of current research, a systematic review and meta-analysis was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline. 12 The research question was defined by applying the Population or Participants, Intervention, Comparison, Outcome (PICO) framework:
Population/Participants: Adult patients (>18 years old) with BPPV, which was confirmed by the Dix-Hallpike test or the supine roll test, who had a report of serum UA levels.
Intervention: Mean serum levels of UA in BPPV patients.
Comparison: Mean serum levels of UA in control group.
Outcome: Not applicable.
Search Strategy
An English language keyword search was conducted using Web of science, PubMed, Scopus, Google Scholar, Embase, Medline, and Cochrane Central Register or Control Trials up to November 2023. Additionally, the reference lists of reviewed articles were screened for additional studies, which could meet inclusion criteria. The search syntax was developed by a combination of relevant keywords and medical subheadings (MeSH) including: [“Benign Paroxysmal Positional Vertigo” OR “Vertigo”] AND [“Serum Uric Acid”]. The search process was conducted without any restrictions on language, country, or date for the inclusion of reports. If there was any article in languages other than English, it would be translated to English using Google Translate.
Eligibility Criteria
Only case-control studies that met our PICO criteria were eligible to include: comparing the mean level of serum UA between adult BPPV patients and control groups, of any sex, age, or ethnicity. All of the BPPV diagnosis should be confirmed by a positive result in the supine roll test or the Dix-Hallpike test. We excluded case reports, case series with less than 5 patients, conference abstracts, and reviews. Only systematic reviews were retained, and their references were manually checked.
Selection Process
Two authors screened the titles and abstracts independently based on predefined criteria. In case of any disagreement, another author was consulted to reach consensus. The full-texts of all abstracts that met the inclusion criteria and reference checking of systematic reviews were assessed independently by the same 2 authors.
Data Extraction
One author developed a data extraction sheet using Excel, which encompassed the characteristics of the study (year, country, and authors), individual’s demographic data, and serum levels of UA. Subsequently, a preliminary extraction was performed by 2 authors. The findings from this initial extraction were evaluated by 1 author, leading to revisions in the form to correct any possible previous errors.
Quality Appraisal
All the included studies in the current systematic review were case-control studies. Two independent reviewers assessed quality of all studies using the Newcastle–Ottawa Scale (NOS) for case-control studies. This tool contains 8 questions to assess the 3 categories of selection, comparability, and exposure of cases with controls. 13 Selection category consists of 4 questions evaluating the definition of controls and their primary source of selection, the definition of cases, and their primary source of selection. Each question can get maximum one score. Comparability section includes one question studying the factors controlled between case and control groups. This section can get maximum 2 scores. Exposure section has 3 questions assessing non-response rate and method of evaluation of exposure in both case and control groups. This section can get maximum 3 scores. A total score of 6 or higher indicates high quality.
Statistical Analysis
The program listed the results of each study included in the meta-analysis: number of positive cases, total number of cases, and the standardized mean difference (SMD) with 95% CI. The total SMD with 95% CI was given for both of the fixed effects model and the random effects model. Cohran’s Q and I2 statistics were used to check the homogeneity. Forest plot was used to show the parameters, and Funnel plot was used to check the errors. High heterogeneity was defined as a significance level of P < .05 and I2 ≥ 50%. Calculations were added with MedCalc software version 20.02.
Results
Search and Screening Results and Studies Characteristics
Following the removal of 38 duplicate records, 237 papers were retrieved for further screening. Through the title and abstract screening stage, 209 records were excluded, and 28 records were included for full-text screening. Finally, 20 studies9,10,14-31 with 3967 individuals (n = 2081 cases and n = 1886 controls) were enrolled, which met the inclusion criteria with the appropriate report of outcomes of interest (Figure 1). Demographic information about these studies is presented as Table 1. All studies had case-control design with sample sizes ranging between 40 and 640 cases.
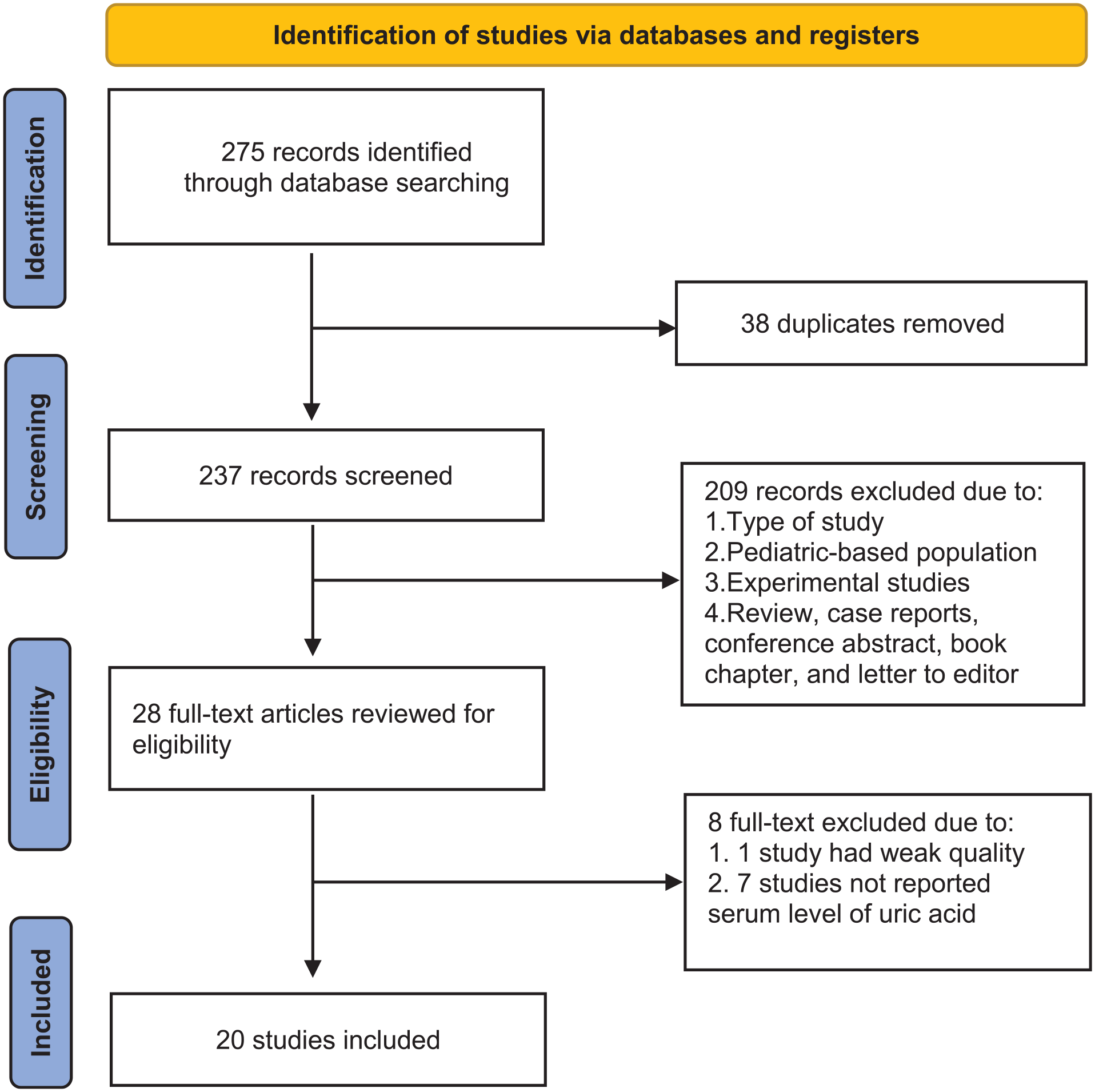
Flow diagram of the study selection process.
Baseline Characteristics of Included Studies.
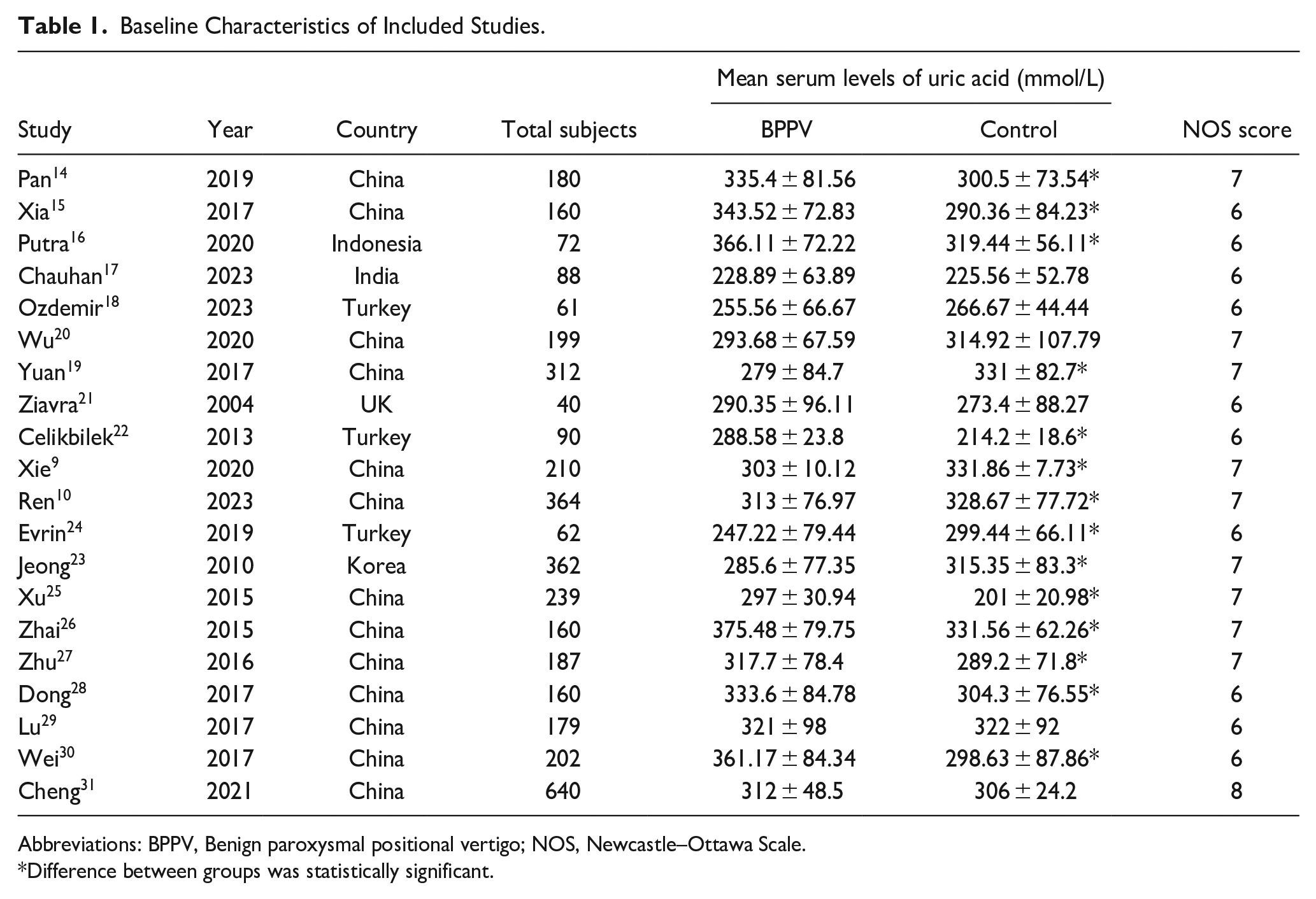
Abbreviations: BPPV, Benign paroxysmal positional vertigo; NOS, Newcastle–Ottawa Scale.
Difference between groups was statistically significant.
Quality Appraisal
Using the NOS scale for case-control studies, the quality of included studies was evaluated. 13 Most studies met sufficient quality levels for inclusion at score ≥6 (high quality; see Table 1). One study had fair quality (score = 5) based on NOS scale. Since it was aimed to reduce the risk of bias considerably, this study was excluded from the database.
Comparison of Serum UA Levels in BPPV and Controls
Serum levels of UA were higher in BPPV groups than controls (SMD: 0.265, 95% CI: −0.163 to 0.693; see Table 2). This difference was not significant (P = .225). Due to high heterogeneity between 20 studies (I2 = 97.44%, P < .0001), the random effects model was used. There was no publication bias visualized by a symmetric Funnel plot (Figure 2), and supported by nonsignificant Eager’s test (P = .43) and Begg’s test (Kendal tau = .1579, P = .33).
SMD and SE for Included Studies.
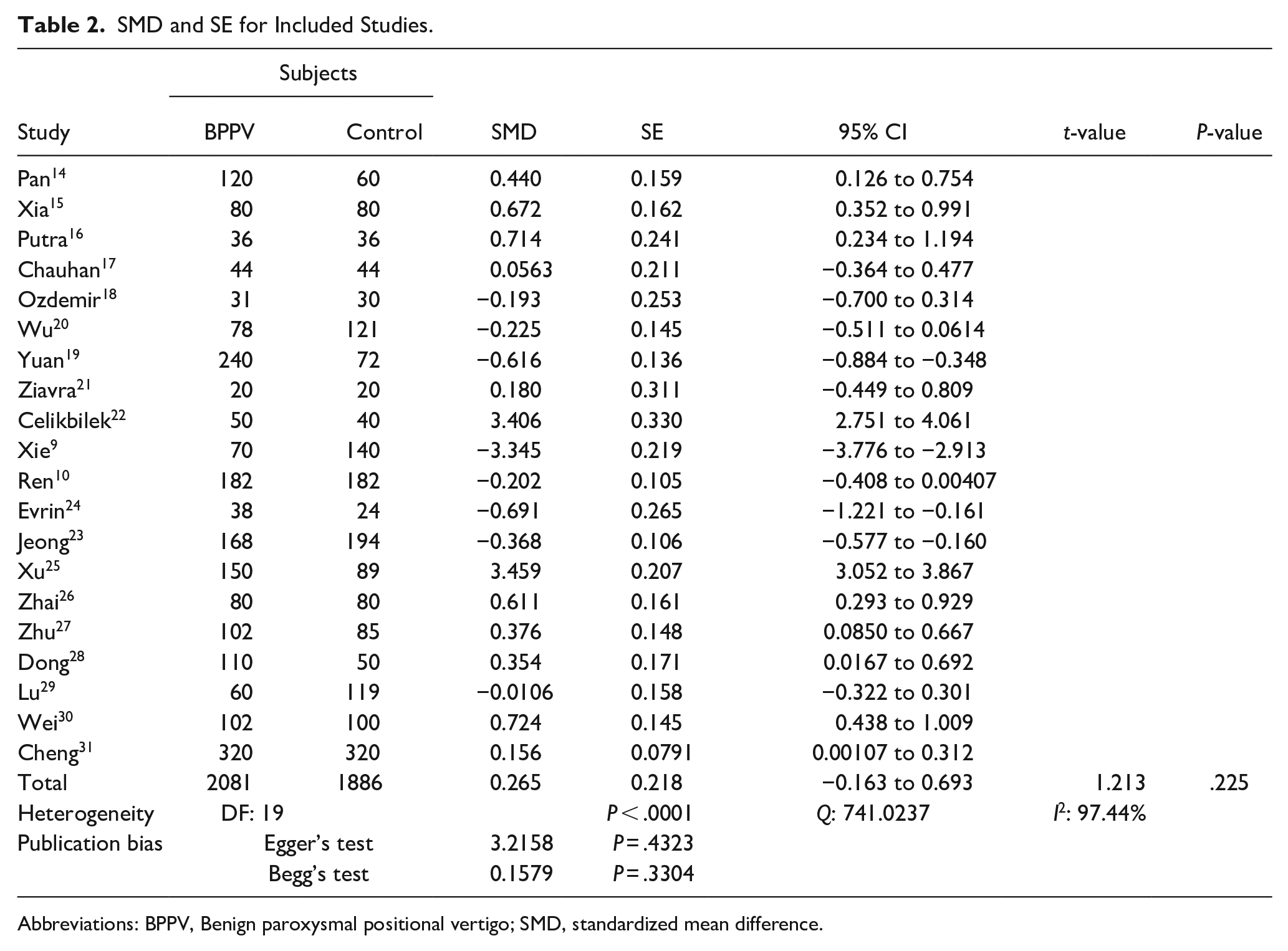
Abbreviations: BPPV, Benign paroxysmal positional vertigo; SMD, standardized mean difference.
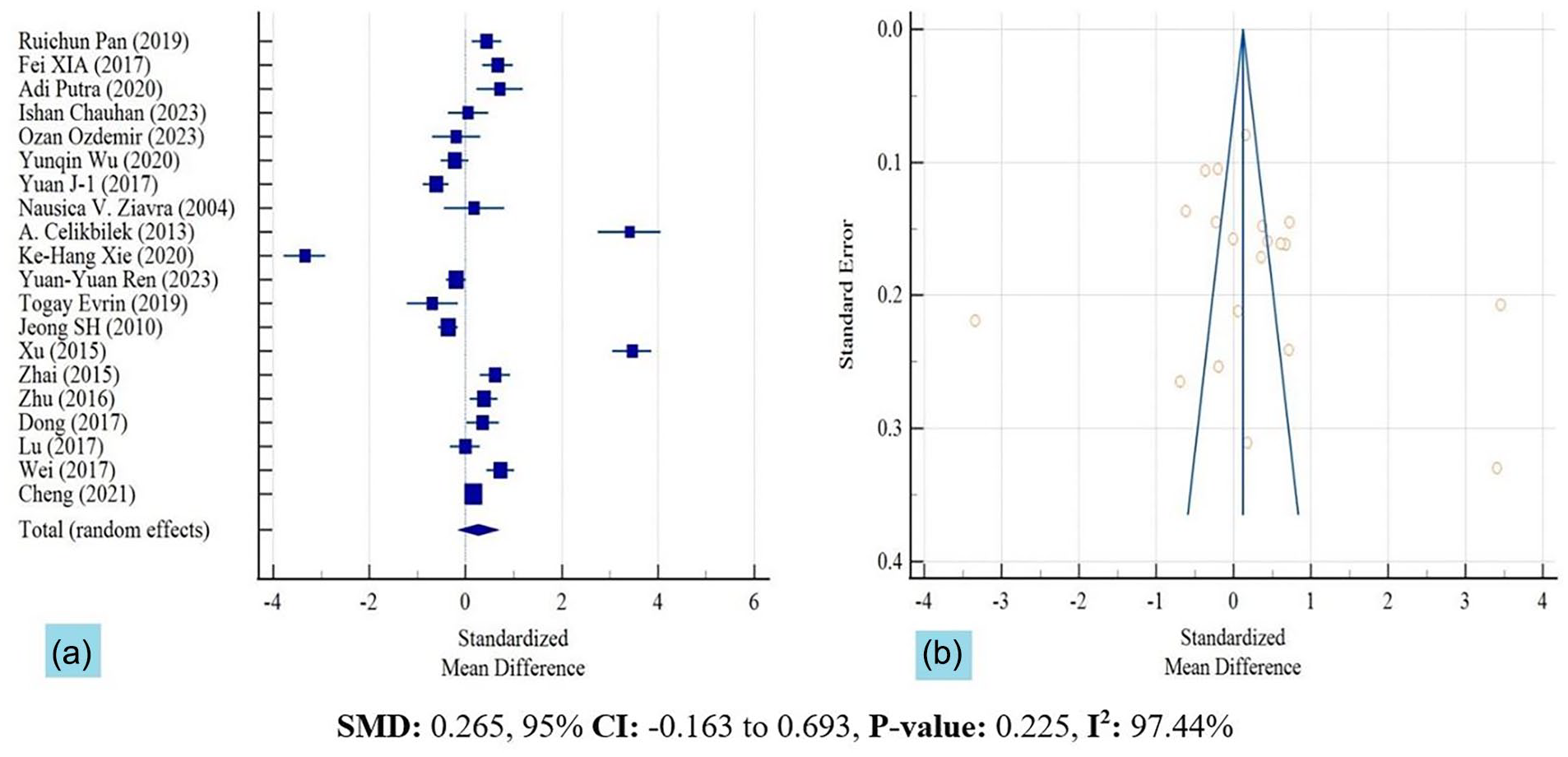
(a) Forest plot of serum levels of uric acid in the BPPV and control groups across the included studies, and (b) Funnel plot of SMD in serum levels of uric acid between the BPPV and control groups.
Discussion
In this systematic review and meta-analysis, we explored the relationship between serum UA and BPPV. By aggregating the SMDs from 20 case-control studies (comparing 2081 BPPV cases and 1886 healthy controls), we discovered no significant difference in serum level of UA between BPPV patients and healthy controls, with an SMD of 0.265 (95% CI −0.163 to 0.693, P = .225). This finding is in contrast with a meta-analysis by Yung et al in 2019 on 11 case-control studies that reported significantly elevated UA level in BPPV patients with a SMD of 0.78, (0.15, 1.41, P = .015). By inclusion of more studies (20 vs 11) with larger sample sizes (2081 vs 1162 cases), our analysis likely has a higher power to detect the true difference between studies. In addition, we found high heterogenity in results of published studies. Among the studies included in our analysis, 10 studies (50%) reported higher UA levels in BPPV patients,14-16,22,25-28,30,31 4 studies (20%) indicated lower UA levels in BPPV patients,9,19,23,24 while 6 studies (30%) found no significant difference in UA levels between BPPV patients and healthy controls.17,18,20,21,29,31 This diverse range of findings underscores the inconsistency in findings and complexity of the UA–BPPV relationship.
The studies that found lower level of UA in BPPV often refers to antioxidant role of UA in the pathogenesis of BPPV. The intricate link between BPPV and oxidative stress is underlined by the crucial role of calcium carbonate balance in the endolymph for normal otoconial function. The primary cause of BPPV symptoms is attributed to an increased release of calcium-rich material from the otoconia into the endolymph. Oxidative stress, which increases the migration of calcium from the endoplasmic reticulum into the cell, leading to the rupture of the mitochondrial outer membrane and apoptosis, is implicated in the close association between BPPV and calcium metabolism.18,32,33 In this regard, Ren et al investigated the role of UA and calcium homeostasis in BPPV and found low UA as a risk factor of BPPV. They proposed that oxidative stress disrupts the balance of calcium ions in the endolymph, impacting the behavior of otoconia. 10 Pan et al proposed an alternative explanation, identifying a higher prevalence of rs2074880 (T/G) polymorphisms in the calcium voltage-gated channel subunit alpha1 (CACNA1A) gene in BPPV patients. This suggests that the CACNA1A gene, which regulates calcium channels, may play a role in the pathogenesis of BPPV. They also found higher UA levels in serum of individuals with TT genotype mutation of rs2074880. 14 This finding provides another potential link between UA levels and BPPV, further complicating our understanding of this condition. It underscores the need for continued research to fully elucidate the complex interplay of genetic and biochemical factors in BPPV.
The role of oxidative stress and its association with UA levels has been thought to be related to the pathogenesis of BPPV. 33 Tsai et al discovered elevated levels of oxidative stress markers (such as superoxide dismutase, malondialdehyde, and C-reactive protein) in blood samples obtained from individuals diagnosed with BPPV. 34 Ozdemir et al found higher urinary markers of oxidative stress in patients with BPPV during episodes of vertigo. These urinary markers levels returned to normal following treatment (repositioning maneuver). 18 Xie et al explored the role of major intravascular antioxidants, including UA, albumin, bilirubin, and creatinine, and found lower level of all 4 markers in BPPV patients. However, their subgroup analysis only showed significant lower UA levels in females and not in males suggesting an association between weaker antioxidant capacity in women and occurrence of BPPV. 9
Many studies correlate BPPV to higher serum UA levels with different pathophysiologic mechanisms, including inflammatory response and vascular damage. Celikbilek et al reported an increase in serum UA levels in patients suffering from BPPV with a noted surge in UA during episodes of vertigo and a trend toward normalization once the attack subsides, indicating that UA level is primarily associated with the occurrence of a BPPV attack itself, rather than being a component of cardiometabolic conditions. In their multiple logistic regression model, serum UA was independently linked to BPPV (P < .001). It was demonstrated that for each unit (mg/dL) increase in serum UA, there was a 3.35-fold increase in the likelihood of BPPV risk. 22 Elevated levels of serum UA may trigger an inflammatory response in otoconia’s supporting gelatinous matrix through an activated immunopathological mechanism. 35 Both animal and in vitro studies have suggested that UA could enhance inflammatory mediators known to cause vascular damage. This could occur either through the generation of ROS leading to endothelial dysfunction or through the secretion of endothelin-1.8,36 The reversal of these effects by allopurinol treatment further backs up this inflammatory theory. 37
Although some underlying mechanisms are mentioned for the possible association of the changes of serum UA (whether low or high) with BPPV, the current meta-analysis did not find a significant correlation between them. It is suggested that the changes of serum UA do not probably affect or cause BPPV as recently, it has been proposed that most cases of BPPV are likely related to migraine attacks on the inner ear causing a fluid shift in the inner ear and displacement of otoconia.38,39 It means that migraine and not biologic markers such as UA are probably the potential etiologic factors in BPPV.
This study was not without limitations. Most of the studies were from Asia and only one study was from Europe and no study was found in other parts of the world, which limits the generalization of our findings. In addition, the quality of most studies was not satisfactory. More studies with larger sample sizes from different parts of the world are needed to explore the true pathophysiologic role of UA in BPPV.
Conclusion
In conclusion, while many studies correlate BPPV with higher or lower serum UA levels, our analysis found no significant difference in serum level of UA between BPPV patients and healthy controls. It means that serum level of UA (whether low or high) is not likely the underlying factor of development of BPPV. However, continued research to fully understand the complex interplay of genetic and biochemical factors in BPPV is needed.
Footnotes
Acknowledgements
We are thankful to Professor Hamid R. Djalilian who helped us considerably to enhance the quality of the current review.
Author Contributions
Data Availability Statement
Data for this systemic review were collected from the cited articles as included in the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
