Abstract
Introduction
Osteomas commonly affect the craniofacial region, particularly the temporal bone, jaw, and paranasal sinuses. They have been reported in various sections of the temporal bone, such as the mastoid region and internal auditory canal, with the external auditory canal (EAC) being the most frequent site.1-3 Unlike exostoses, EAC osteomas are true neoplasms, usually unilateral and solitary, and are not associated with cold water exposure. EAC osteomas can lead to stenosis, and impair epithelium migration and self-cleaning capability, thereby trapping keratinized epithelium and triggering the development of cholesteatoma.
The prevalence of cholesteatoma formation in congenital EAC stenosis ranges from 19% to 41%.4,5 However, due to the diverse etiology, the incidence of cholesteatoma coexisting with acquired EAC atresia/stenosis is <10%.6,7 There are few literature reports of EAC osteomas with cholesteatoma. Although EAC cholesteatoma (EACC) is a rare condition, it can result in bony erosion and anatomical disruption, leading to further complications. Our study aims to identify the risk of EACC development in patients with osteoma and proposes a stepwise approach to managing patients with EAC osteoma.
Patients and Methods
We conducted a retrospective review of patients diagnosed with EAC osteoma who underwent surgical intervention at the Chang Gung Memorial Hospital between January 1995 and December 2021. This study was approved by Chang Gung Medical Foundation Institutional Review Board (No. 202200219B0). Patients with congenital EAC anomalies and those who had undergone previous surgery resulting in postoperative EAC changes were excluded. Patients without high-resolution computed tomography (HRCT) of the temporal bone were also excluded.
The maximum diameter of the osteoma was measured in axial and coronal views on HRCT. We calculated the relative obstruction ratio caused by the EAC osteoma in the axial and coronal views of the temporal bone HRCT. After drawing a tangent line from the base of the osteoma, and a vertical line from the tangent line, to the top of the osteoma and the other side of the EAC, we computed the anteroposterior (AP) diameters of the osteoma and EAC in the axial view and the superoinferior (SI) diameters in the coronal view (Figures 1 and 2). The relative obstruction ratio was derived from the ratio of the largest AP and SI diameter of the osteoma to the diameter of the EAC at the same assessment point.
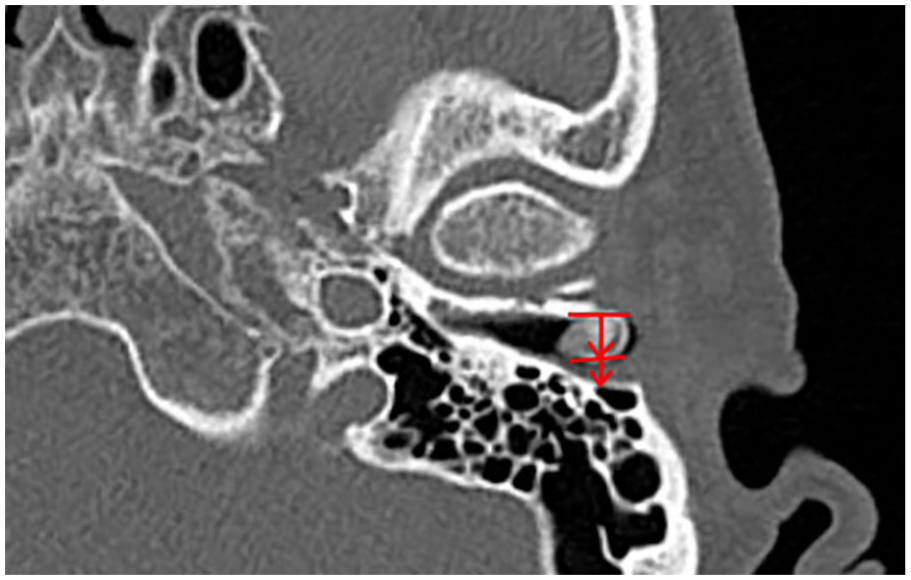
A tangent line was drawn from the base of EAC osteoma. A line vertical to the previous tangent line was then drawn to the top of EAC and the other side of EAC. The relative obstruction ratio caused by osteoma in anterior-posterior dimension was obtained from the ratio of diameters of osteoma to EAC. EAC, external auditory canal.
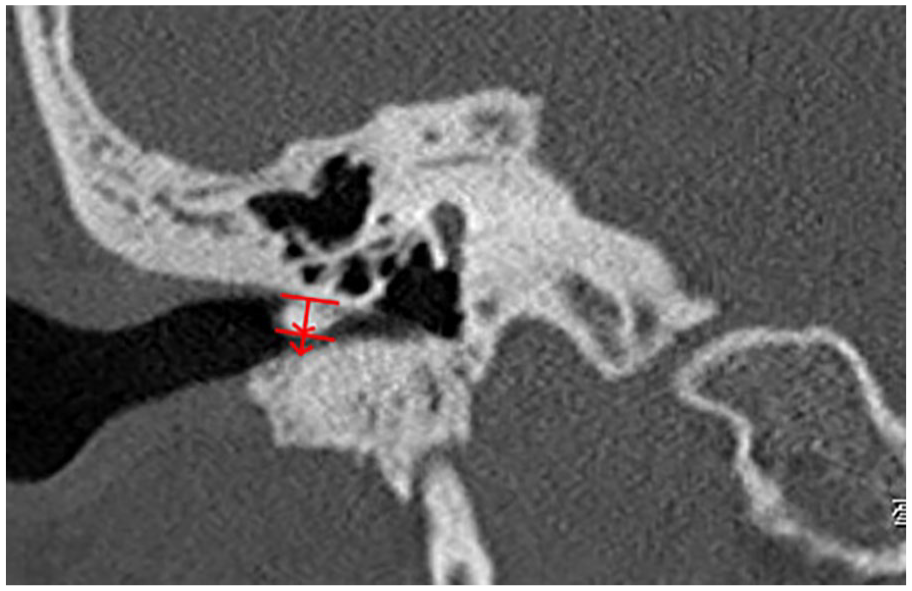
The relative obstruction ratio in superior-inferior dimension was calculated from the ratio of diameters of osteoma to EAC in coronal view. EAC, external auditory canal.
The location of the osteoma (medial or lateral) was determined using the midpoint of the osseous EAC. Prior to surgery, otoscopy was employed to identify pedicle formation (Figure 3). Pathologic examination was conducted on all patients to confirm the presence or absence of cholesteatoma formation. The patients were categorized into 2 groups based on the presence of EACC. Nonparametric variables, including sex, laterality, presentation symptoms, and EAC osteoma characteristics, were compared using Fisher’s exact test. The Mann-Whitney U test was utilized for the analysis of parametric data, such as age, maximum diameter of osteomas, and relative obstruction ratio. The data analysis was carried out using SPSS for Windows (version 20.0; IBM Corp), with statistical significance defined as P < .05.
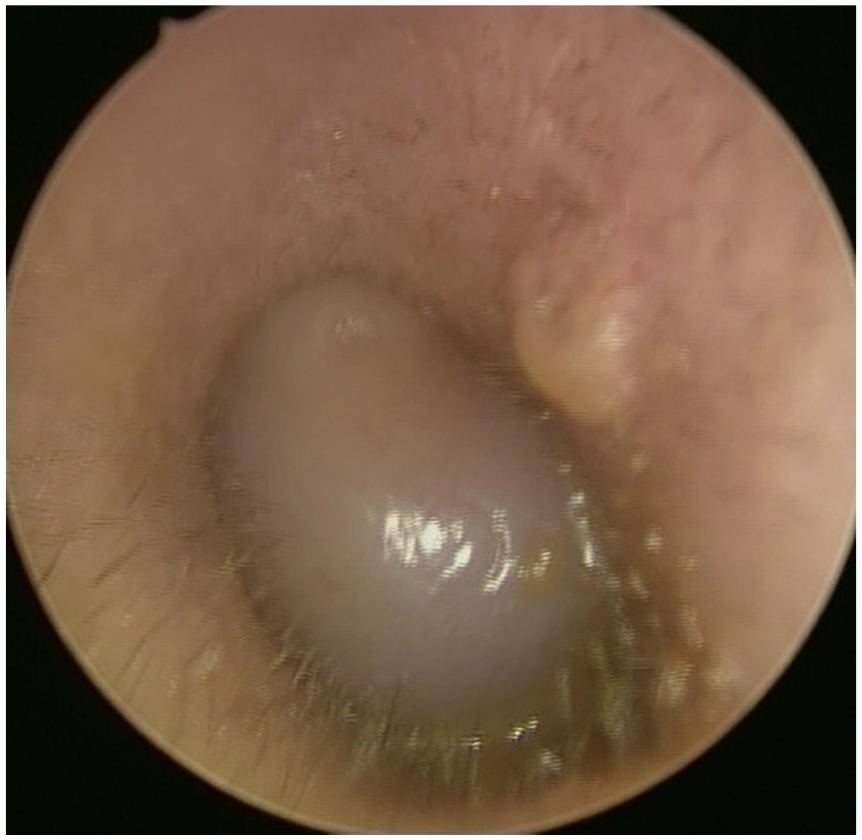
Osteoma without pedicle causes total obstruction of EAC and drum invisible. EAC, external auditory canal.
Results
We identified 43 patients diagnosed with EAC osteoma, including 33 (76.7%) males and 10 (23.3%) females. All osteomas were unilateral, with 24 (55.8%) appearing on the right side and 19 (44.2%) on the left. A total of 28 (65.1%) EAC osteomas were located in the medial EAC, while 15 (34.9%) were situated in the lateral EAC. The top 3 symptoms accompanying EAC osteomas were incidental finding of EAC mass, hearing impairment, and aural fullness. Other symptoms such as otorrhea, otalgia, tinnitus, headache, and ear itching were observed in a small proportion of patients. A total of 9 (20.9%) patients with EAC osteomas developed EACC.
The average age of patients without and with cholesteatoma was 32.6 ± 12.6 and 33.0 ± 13.5 years, respectively. In the group without cholesteatoma, there were 26 males and 8 females, whereas in the group with cholesteatoma, there were 7 males and 2 females. Of the EAC osteomas without cholesteatoma, 21 were located in the right EAC and the remaining 13 were in the left EAC. In the cholesteatoma group, 3 osteomas were on the right side and 6 were on the left side. There was no significant difference in age, gender, and side between these 2 groups (Table 1).
Demographics of Patients With EAC Osteomas.
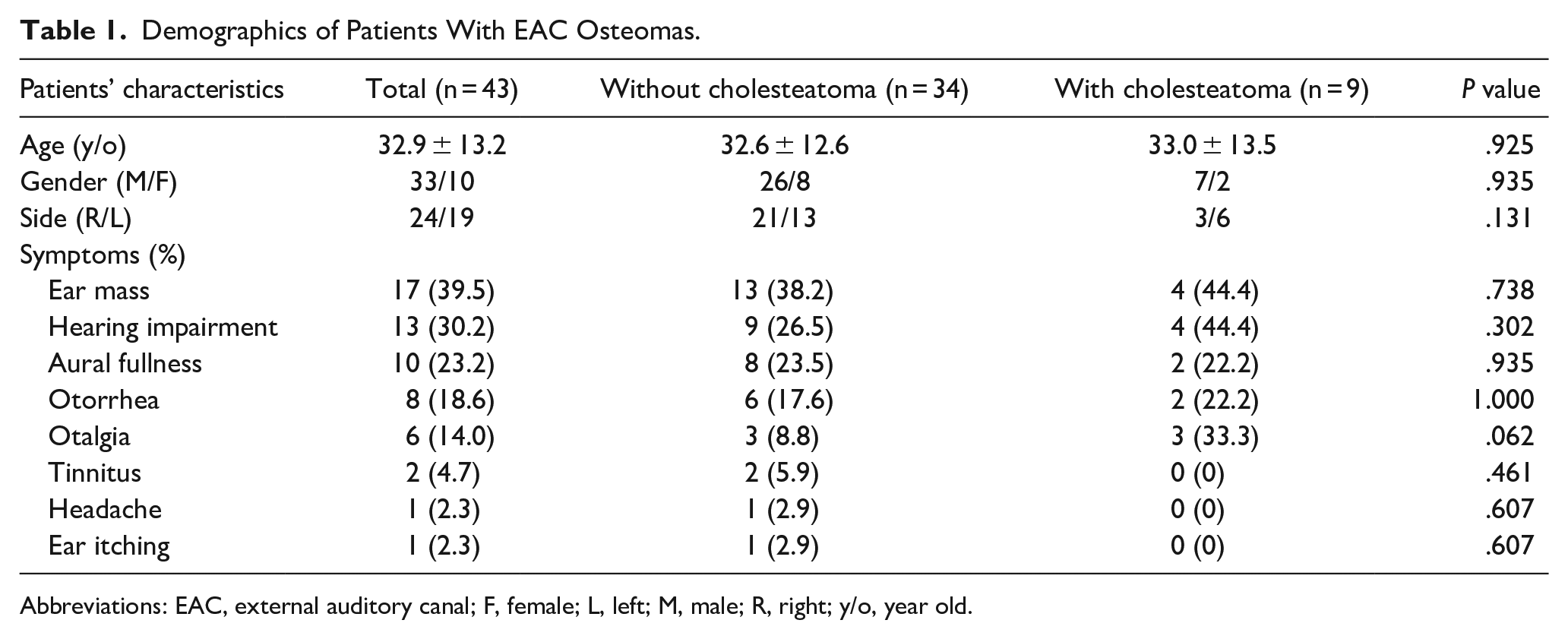
Abbreviations: EAC, external auditory canal; F, female; L, left; M, male; R, right; y/o, year old.
The relative obstruction ratio of EAC osteoma was 68.5% in osteomas without cholesteatoma and 74.9% in osteomas with cholesteatoma. The maximum diameter of osteomas with and without cholesteatoma was 12.67 ± 4.09 and 7.67 ± 3.27 mm, respectively. In the group without cholesteatoma, 21 osteomas had pedicles while the other 13 did not. In the cholesteatoma group, 2 osteomas had pedicles and 7 did not. The maximum diameter of osteomas with cholesteatoma was significantly larger than that of those without cholesteatoma (P < .001). The proportion of osteomas without pedicles was significantly higher in the group with cholesteatoma (P = .037). However, no difference was observed in the relative obstruction ratio between these 2 groups (Table 2).
Characteristics of EAC Osteomas in Patients With and Without Cholesteatoma.
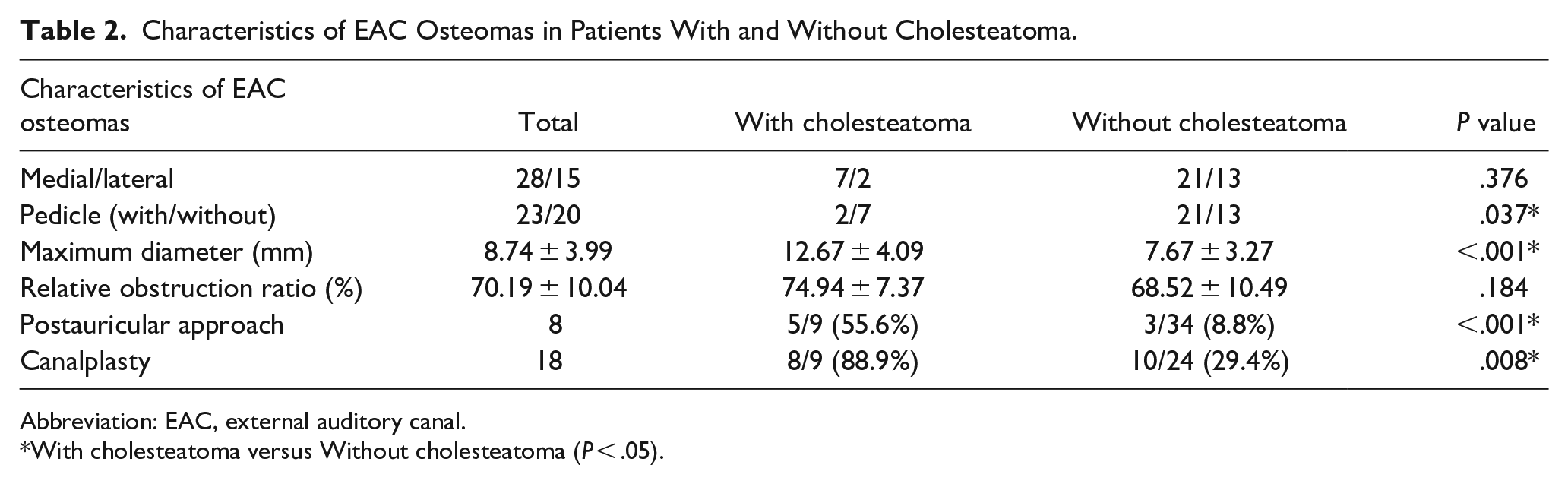
Abbreviation: EAC, external auditory canal.
With cholesteatoma versus Without cholesteatoma (P < .05).
In terms of operative method, 5 (55.6%) patients with cholesteatoma underwent surgery via the postauricular approach, while only 3 (8.8%) patients without cholesteatoma had their osteoma removed through the same method. Canalplasty was performed concurrently in 8 (88.9%) patients with cholesteatoma and 10 (29.4%) patients without cholesteatoma. No recurrence of osteoma or cholesteatoma was noted in any of the patients during the 3 year follow-up period post surgery (Table 2).
Discussion
Osteomas are benign lesions typically characterized by focal, pedunculated bony overgrowth of the osseous EAC with normal overlying squamous epithelium. Microscopically, they consist of lamellated bone with surrounding fibrovascular channels. 8 Unlike EAC exostosis, osteomas are usually narrow-based, unilateral, and solitary, without any association with chronic cold-water exposure.
EACC are erosive lesions composed of exfoliated keratin with stratified squamous epithelium. This differs from keratosis obturans, which is an abnormal accumulation of desquamated keratin in the EAC without any erosive bony changes. EACC presents as a scalloping soft tissue mass, often appearing with bone fragments within and extending into nearby structures. 9
Multiple osteomas can occur simultaneously with colon polyps, intestinal polyposis, dermoid tumors, and epidermoid cysts, a condition known as Gardner syndrome. 10 When osteomas are solitary and unilateral, they can develop in the mastoid region and EAC. Some reports have noted EAC osteomas appearing frequently over the tympanosquamous suture.11,12 Symptoms of osteomas may include hearing impairment, aural fullness, otalgia, otorrhea, tinnitus, and even headaches. In our study, we had one case presenting with a headache as the chief complaint. A possible mechanism for the headache is that the tumor encroached on a branch of the auriculotemporal nerve, causing referred pain to the temporal region. 13
EAC osteomas with cholesteatoma are rarely reported in the literature. Notably, Ata et al reported a case that developed sinus thrombosis.14-19 Cholesteatoma formation may result in frequent ear or mastoid inflammation/infection and can even lead to an upper neck abscess or meningitis.15,19 In this study, we found no specific symptoms indicating the formation of cholesteatoma in cases of EAC osteoma. Osteomas can cause acquired EAC stenosis and impairment of epithelium migration and self-cleaning ability, which may subsequently lead to the trapping of keratinized epithelium and the potential development of cholesteatoma.
Cholesteatoma secondary to congenital aural stenosis (CAS) is well documented. Jahrsdoerfer et al defined CAS as an EAC with a diameter <4 mm. They adopted this criterion because cholesteatoma rarely developed when the diameter of the EAC exceeded 4 mm. 20 In a study by Casale et al, 18 (19.1%) of 94 ears with congenital EAC stenosis developed cholesteatoma. 4 In another study, Mazita et al observed cholesteatomas in 17 (41.4%) of 41 patients with canal anomalies. 5 However, due to the diverse etiology and rarity of acquired EAC atresia/stenosis, the true incidence of secondary cholesteatoma is difficult to determine. In a retrospective review of data collected over 27 years, by Becker and Tos, the incidence of secondary cholesteatoma resulting from acquired EAC atresia was 9%. 7 Meanwhile, Magliulo found that only 3 (7.3%) of 41 patients with acquired EAC atresia developed canal cholesteatoma. 6 In our study, the incidence of cholesteatoma in EAC osteoma was 20.9%, which is higher than the incidence of cholesteatoma in acquired EAC atresia. This may be because we only included EAC osteoma with surgical treatment in this study and thus overestimated the incidence of cholesteatoma.
Li et al discovered that stenotic EAC was larger in their cholesteatoma than noncholesteatoma group due to the erosive characteristics of cholesteatoma. After removing patients with cholesteatoma and defective bony canal form the analysis, no significant difference was found between the cholesteatoma and noncholesteatoma groups in the extent of EAC stenosis. 21 Portmann and Portmann proposed the “rule of the ratio V/S” (where V represents the volume of circulating air coming from outside, and S represents the surface area of the skin of the cavity). They suggested that normal epithelial skin could transform into a cholesteatoma matrix if this ratio was reduced. 22 Osteomas could occupy the space of the EAC and increase the surface area, thereby accelerating the development of cholesteatoma. In our study, we retrospectively reviewed only patients who received surgical intervention; therefore, the true incidence of acquired cholesteatoma caused by EAC osteoma is unknown and requires further investigation in a prospective, large-scale study.
The presence of a pedicle in the osteoma may further reduce the extent of stenosis, which could be challenging to detect by HRCT. Cholesteatoma could lead to a defective bony canal wall and induce inflammatory changes in the EAC. These factors could complicate determination of the degree of EAC obstruction on HRCT. However, HRCT could indicate EAC erosion or mastoid invasion by cholesteatoma. Before surgery, surgeons can estimate the size of osteomas from images. Our study found that larger osteomas were associated with greater cholesteatoma formation. However, the extent of EAC stenosis did not influence cholesteatoma formation.
In our study, the diameter of the osteoma was significantly larger in cases with cholesteatoma. This increased size may make presurgical examination of the eardrum more challenging. Fisher and McManus suggested that osteoma surgery should only be considered after conservative treatment has failed, with a focus on ensuring adequate access, preservation of meatal skin, and protection of the tympanic membrane. Postsurgical complications can include canal stenosis, temporomandibular joint prolapse, sensorineural hearing loss, persistent deep bony lip, and tympanic membrane perforation. Safe landmark identification and comprehensive exposure are recommended to avoid such complications. 23
In other studies, patients with EAC osteoma were treated using an endoscopic transcanal method. Small, pedunculated osteomas were removed via a direct transcanal approach, while broad-based osteomas without an obvious stalk attached to the EAC were removed transcanally using a skin flap. 24 Naim et al categorized different stages of EACC and proposed a stepwise approach for its surgical treatment. The transcanal approach is recommended for epithelial hyperplasia of the canal, while the endaural approach is used for periostitis and a defective bony canal. A postauricular approach, followed by canal wall down, is suggested for lesions causing erosion of adjacent anatomical structures. 25
Shin et al proposed another staging method that emphasizes the findings of temporal bone CT. Stage I indicates EACC limited to the EAC, and Stage II denotes lesion invasion into the tympanic membrane and middle ear. When the EACC creates a defect in the EAC and involves the air cells in the mastoid bone or beyond the temporal bone, the lesions are defined as Stage III and IV, respectively. EACC can be managed through local care, canaloplasty, tympanoplasty, mastoidectomy, or more invasive methods such as middle cranial fossa and transzygomatic approaches. The goal is to preserve normal EAC skin and surgically remove the cholesteatoma and necrotic bone tissue to prevent disease progression and bone destruction. 26 To the best of our knowledge, no literature discusses the treatment of EAC osteoma with cholesteatoma. In this study, we propose an individualized treatment algorithm based on patients’ symptoms, the size of the osteoma, and the presence and extent of cholesteatoma formation.
Preoperative HRCT is crucial for detecting cholesteatoma, and otoscopy can determine if the eardrum is visible after obstruction by osteoma. If the eardrum is well protected during surgery and no cholesteatoma formation is present, the osteoma could be removed by fracturing the pedicle using an endoscopic or transcanal approach, or by elevating the EAC skin flap and then removing the osteoma using a chisel, depending on whether a pedicle exists. If the eardrum is completely obstructed by osteoma, two-handed surgery via an endaural or postauricular approach is more suitable to protect the skin flap and eardrum. If cholesteatoma has formed, the extent of invasion should be examined using HRCT. When the cholesteatoma is confined to the EAC, an endoscopic/transcanal approach could be used for complete removal of both the osteoma and cholesteatoma. If the cholesteatoma invades areas beyond the EAC, such as the tympanic membrane, middle ear, or mastoid cavity, tympanoplasty and mastoidectomy should be considered to completely remove the cholesteatoma and osteoma (Figure 4). In our study, it was found that more than half of EAC osteoma with cholesteatoma had been removed by the postauricular approach. A total of 88.9% of patients with EAC osteoma and cholesteatoma also underwent canalplasty. This is because osteomas with cholesteatoma are usually large and invade the EAC. The bimanual postauricualar approach can safely remove the lesion and re-epithelialize the EAC. If cholesteatoma is present, enlarging the ear canal and clearing it completely is critical to preventing recurrence.
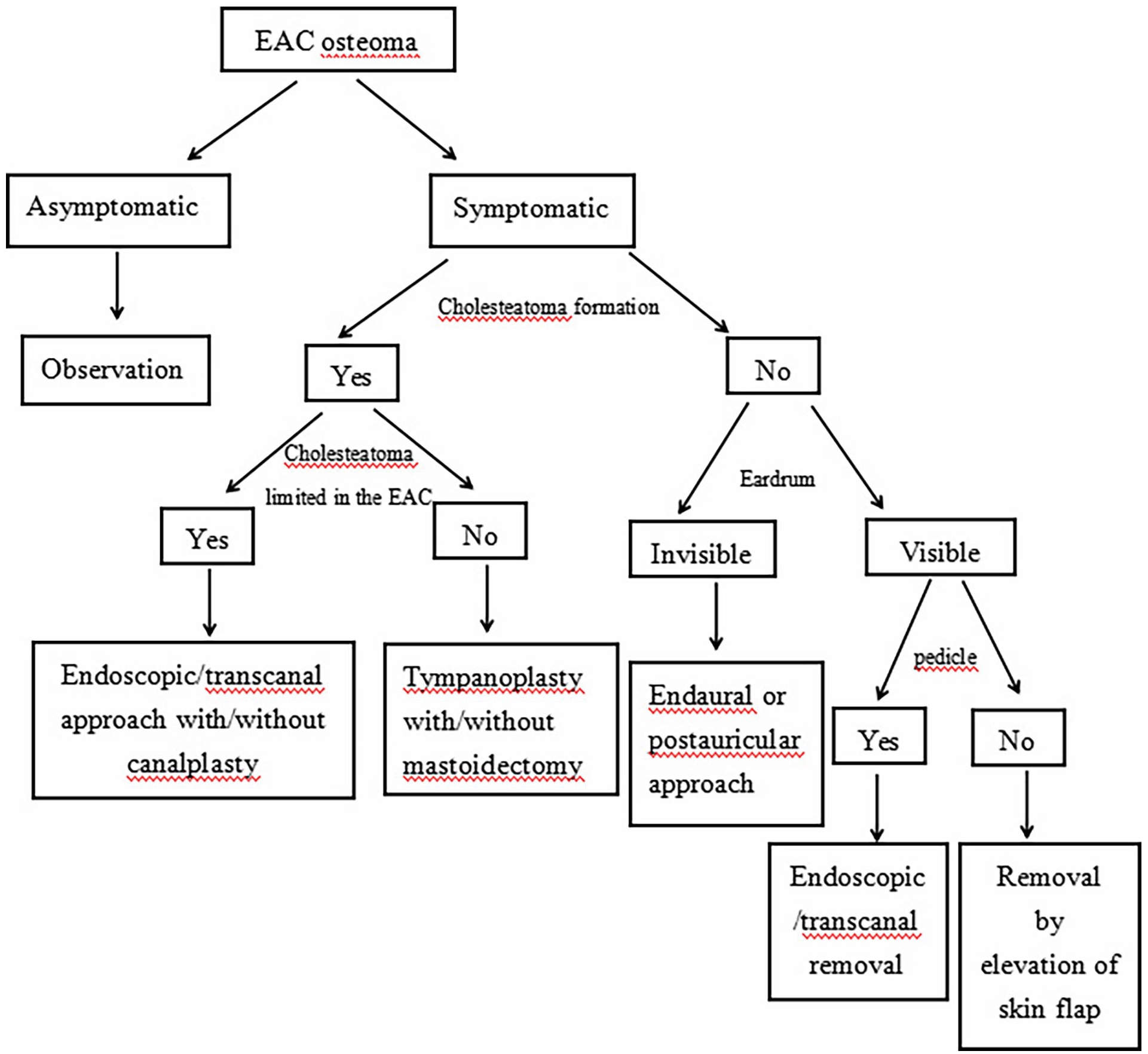
The treatment algorithm of EAC osteoma. EAC, external auditory canal.
Conclusion
Our findings indicate that larger osteomas are more likely to develop cholesteatoma, while the formation of a pedicle may reduce the occurrence of cholesteatoma. For asymptomatic patients, regular monitoring of EAC osteoma is advisable. In symptomatic patients, preoperative evaluation, including HRCT and otoscopy, is vital for assessing the extent of the osteoma and the potential coexistence of cholesteatoma. These factors are critical for preoperative consultations and surgical planning. The aim of surgical intervention should be removal of the osteoma, eradication of cholesteatoma, and widening of the EAC to prevent recurrence.
Footnotes
Acknowledgements
The authors would like to note that there are no acknowledgments for this article.
Data Availability
The data that support the findings of this study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
The report was prepared in accordance with the ethical standards of the institutional ethics committee and with the 1964 Helsinki Declaration.
Informed Consent
Written informed consent was not obtained, as this was a retrospective study.
