Abstract
Keywords
Introduction
Septal deviation emerges as the predominant anatomical finding among patients seeking consultation at the Ear, Nose, and Throat (ENT) outpatient department (OPD) for nasal obstruction and its related symptoms.1,2 Surgical correction, that is, septoplasty 3 is among the most frequently performed procedures in otorhinolaryngology practices. 4 Septoplasty complications are common, with patients experiencing mild to moderate pain, bleeding, and mucosal swelling leading to nasal blockage, crusting, and excessive rhinorrhea in the early postoperative period, which can persist for a few days to weeks.5-7 Over the past several years, studies have suggested the potential use of steroids, 8 Non-steroidal-anti-inflammatory drugs (NSAIDs), 9 paracetamol, oral enzymes, 5 and some topical nasal sprays, 6 that is, saline or other, to relieve such symptoms. Normal saline has been adopted as the primary intervention for comprehensive recovery, whereas the utilization of steroids, oral enzymes, and NSAIDs exhibits global variation among medical practitioners and institutions, contingent on the specific characteristics and severity of the symptoms.5,6,8,9 However, these interventions have not garnered standard recognition for treatment owing to their undesirable side effects, delayed therapeutic onset, or inadequate symptom amelioration, resulting in their inefficacy to mitigate signs/symptoms of post-septoplasty or nasal surgeries. More scientific evidence is required for better outcomes in the early post-septoplasty era due to so much variation across treatments, each with its advantages and disadvantages.
Therefore, we aimed to evaluate the efficacy of 0.1% xylometazoline-hydrochloride nasal decongestant spray in post-septoplasty clinical nasal findings and symptomatic relief in comparison with physiologic 0.9% isotonic saline nasal spray as a therapeutic adjunct through a prospective-experimental study, based on the hypothesis that the incorporation of nasal decongestants into the post-septoplasty care regimen could yield significant advantages for patients’ recovery, due to their vasoconstrictive action.
Patients and Methods
Study Design, Site, and Participants
This bi-institutional, prospective, parallel, and triple-blinded experimental study was conducted in the ENT OPDs of Creek General Hospital (CGH) and United Hospital (UH), affiliated with United Medical and Dental College (UMDC), Karachi, Pakistan, from June 20, 2022, to June 20, 2023, after acquiring ethical approval. Patients aged 15 to 60 years undergoing septoplasty for symptomatic deviated nasal septum (DNS) belonging to UH and CGH were included in the study. Patients not from UH and CGH of age <15 years and >60 years, admitted for revision surgery or trauma or in which septoplasty was performed along with other nasal procedures like turbinate reduction or functional endoscopic sinus surgery or submucosal resection, history of acute or chronic rhinosinusitis and obstructive sleep apnea, perforated nasal septum, insufficient nasal valve, granulomatous condition of the nose and sinuses, pregnancy, craniofacial malformation (cleft palate or cleft lip), HIV or any bleeding diathesis condition, malignancy, allergic rhinitis, usage of antihistamines or anticoagulants, and history of allergies to allocated drugs were excluded from the study.
Sample Size, Sampling Technique, and Randomization
A total of 128 patients was sampled using the consecutive sampling technique. Patients were approached during their visit at the ENT OPD for checkup who were recommended septoplasty. Each eligible participants were assigned a number generated by excel that helps randomly and equally allocate them in 2 groups of treatment, ensuring no repetition occurred in any group. The randomization assisted in ensuring equivalence among both groups leading to generalizability free from any selection or participation bias. Group A was assigned 0.9% normal saline nasal spray as a placebo control group, and Group B was assigned 0.1% xylometazoline hydrochloride nasal spray as an intervention group. The randomization process and allocation of groups to respective treatments were executed by 2 authors, maintaining masking from participants, the statistician, and 2 additional doctors functioning as medication distributor and data collector. A box of identical nasal spray devices with an assigned number based on randomization was given by the authors to one of the doctors, whose role was to provide the patient with nasal spray from the box and instruct the patient for optimal usage, while the other doctor’s role was to collect data from the patients during follow-ups. All parties involved maintained mutual blindness regarding their roles, that is, no one was allowed to discuss neither reveal what duties they have in terms of being a part of study, a condition meticulously monitored by the authors of the study. The triple blinding helped to eliminate any observer, measurement, confirmation, and reporting biases.
Study Procedure and Study Tool
All eligible participants underwent a comprehensive examination (history and physical examination) and an ENT examination (nasal obstruction evaluation). 10 The study was then preceded by gaining written informed written signed consent outlining the study’s goal and the participation scope, followed by surgery under either general or local anesthesia. An incision was made inside the nostril on one side of the septum, allowing access to the deviated tissue. Using specialized instruments, such as scissors or a microdebrider, the deviated cartilage and bone were carefully removed or repositioned to straighten the septum and improve nasal airflow. Following this, the septum was reshaped and reconstructed as necessary, and the incision inside the nostril was closed with dissolvable stitches. The surgery was performed by the same surgeon and anesthesiologist across the participating hospitals, ensuring procedural uniformity throughout the study. After surgery, the Nasal Obstruction Symptom Evaluation score was reevaluated and compared, and each patient was handed the assigned treatment by one of the data collectors for 1 week, twice daily, with a 12 hour interval between the 2 doses as a single spray per nostril, and was instructed on how to use it. Quilting sutures using vicryl rapide 3/0 through the septum were used to approximate the flap together and to prevent hematoma formation. Following the use of the assigned treatment, a modified self-administered, semi-structured instrument was constructed by authors with the help of the studies by Nanda et al 5 and Dağli et al 6 and was utilized by the second data collector (Supplemental Files 1 and 2) to evaluate demographic data, postoperative self-reported symptoms, postoperative clinical nasal findings, postoperative side effects, and patient satisfaction using a LIKERT scale through clinical evaluation related to the drug given for recovery were observed at the end of day 3 and day 7 of administration of the drug. The same antibiotics, analgesics, and nasal packing were considered at the end of day 7 among the patients in both groups in cases of absolute necessity, such as uncontrollable bleeding or various nasal findings. The study was conducted with complete confidentiality.
Primary and Secondary Outcome Measures
Primary outcomes measures: The patients were evaluated for postoperative septoplasty symptoms at each follow-up (Self-reported). (2) Nasal endoscopy was performed to evaluate post-septoplasty endoscopic findings at each follow-up objectively. (3) The patients were questioned and observed about any systemic or local side effects or complications from the use of drugs (Self-reported). Secondary outcome measures: (1) Patient satisfaction with the drug-assigned was assessed using the LIKERT Scale.
Statistical Analysis
The data were enrolled on an Excel sheet and then imported to SPSS version 23 developed and distributed by International Business Machines Corporation (IBM) for analysis. Descriptive statistics are presented for variables. Chi-square/Fisher’s exact test is applied to assess the relationship and difference between variables and assigned groups. A P value of <.05 is considered statistically significant.
Reporting of the Study
For this study’s reporting, the Consolidated Standards of Reporting Trials (CONSORT) guidelines are implemented (Supplemental File 3).
Ethics
The study was approved by the Institutional Review Board of the UMDC in Karachi (CGH/Ethics/Retro/2022/20/06/328).
Results
Study Sites and Patients
Between June 20, 2022, and June 20, 2023, 128 patients who were recommended septoplasty were screened on the basis on inclusion and exclusion at 2 distinct ENT-participating centers. One hundred twenty patients fulfilled the study’s eligibility requirements and were subsequently randomized. Before participation, all participants gave informed consent. The average time between evaluating patient, consent acquisition, and randomization for assigned intervention was 2 days prior to surgery. The primary analysis focused on a subset of 106 patients who received the assigned interventions, evenly distributed with 53 participants in each group (Figure 1). Among the 106 enrolled patients, 59 (55.7%) were males, while 47 (44.3%) were females, with ages ranging from 15 to 60 years. Most participants belonged to the younger and middle-aged categories, with a minor number of patients reporting suffering from hypertension and diabetes. There was no significant difference between control and interventional group patients in terms of sex, age, and comorbidities (Table 1).
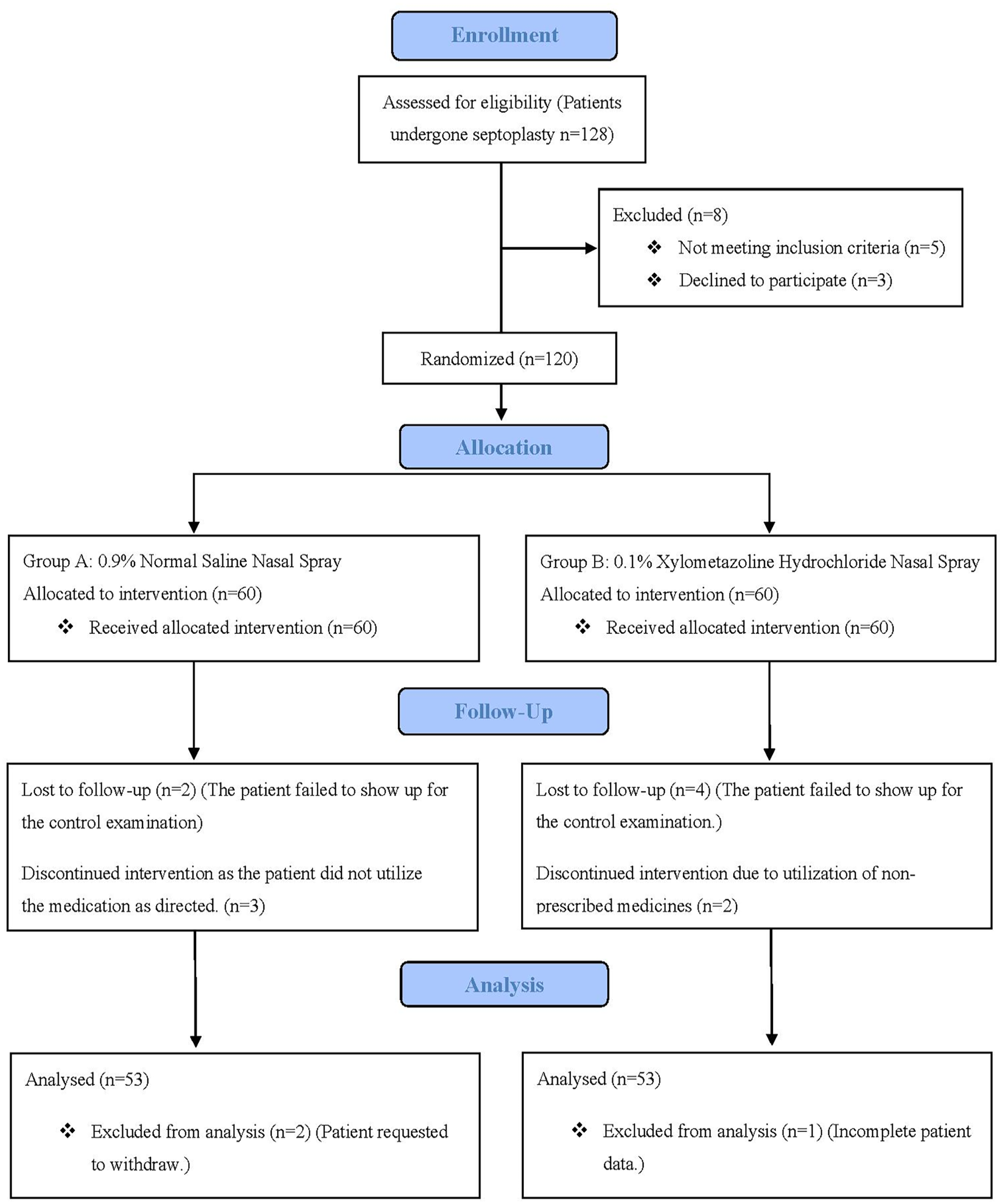
Comprehensive flowchart: Participant enrollment, randomization, allocation, follow-up, and analysis.
Demographic Data of Patients (N = 106).
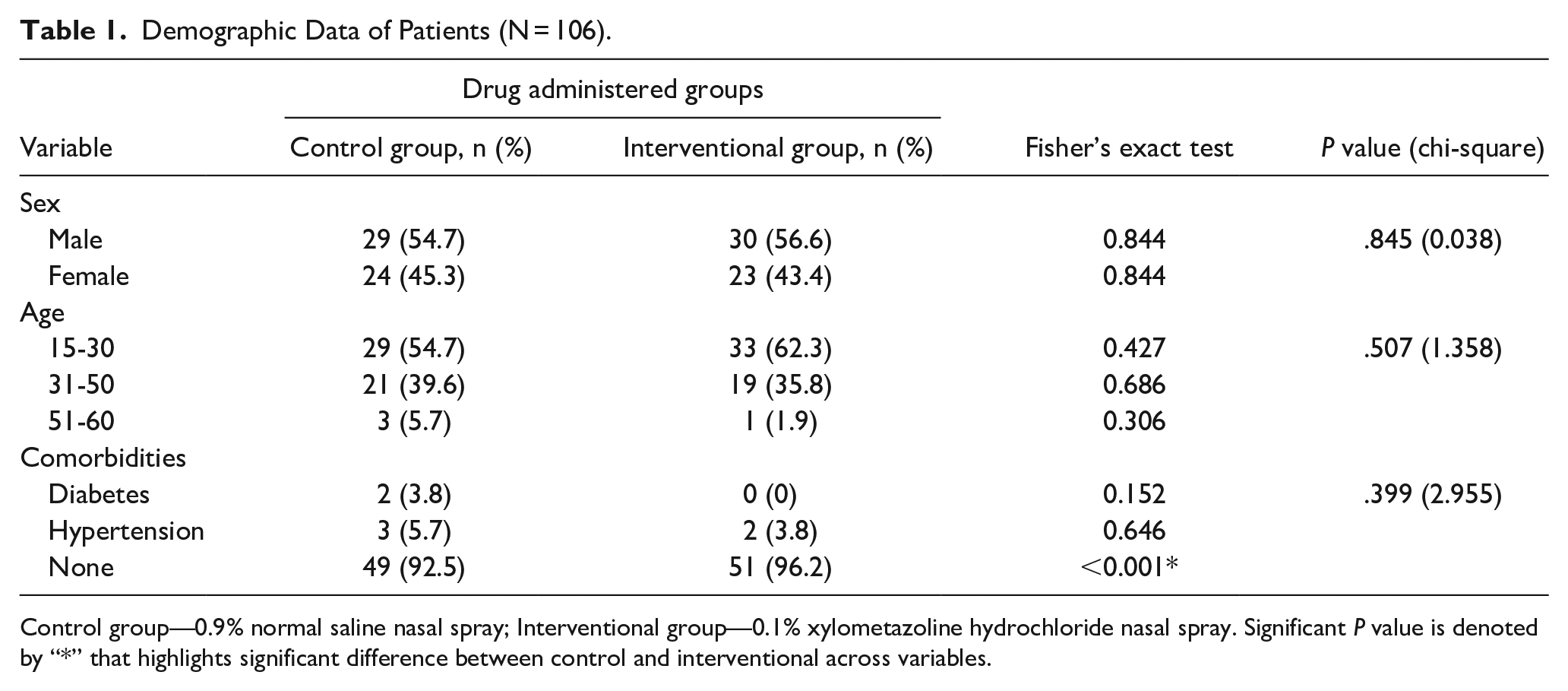
Control group—0.9% normal saline nasal spray; Interventional group—0.1% xylometazoline hydrochloride nasal spray. Significant P value is denoted by “*” that highlights significant difference between control and interventional across variables.
Postoperative Symptom
Patients in control group (0.9% normal saline nasal spray) had significantly higher incidences of bleeding, nasal blockage, headache, and pain by the end of the third day of recovery when compared to patients in intervention group (0.1% xylometazoline hydrochloride nasal spray). Moreover, persistent bleeding and nasal blockage were observed in some control group patients, unlike in intervention group, where symptoms were nearly absent by the end of the seventh day of recovery (Table 2).
Postoperative Self-Reported Symptom in Both Groups (N = 106).
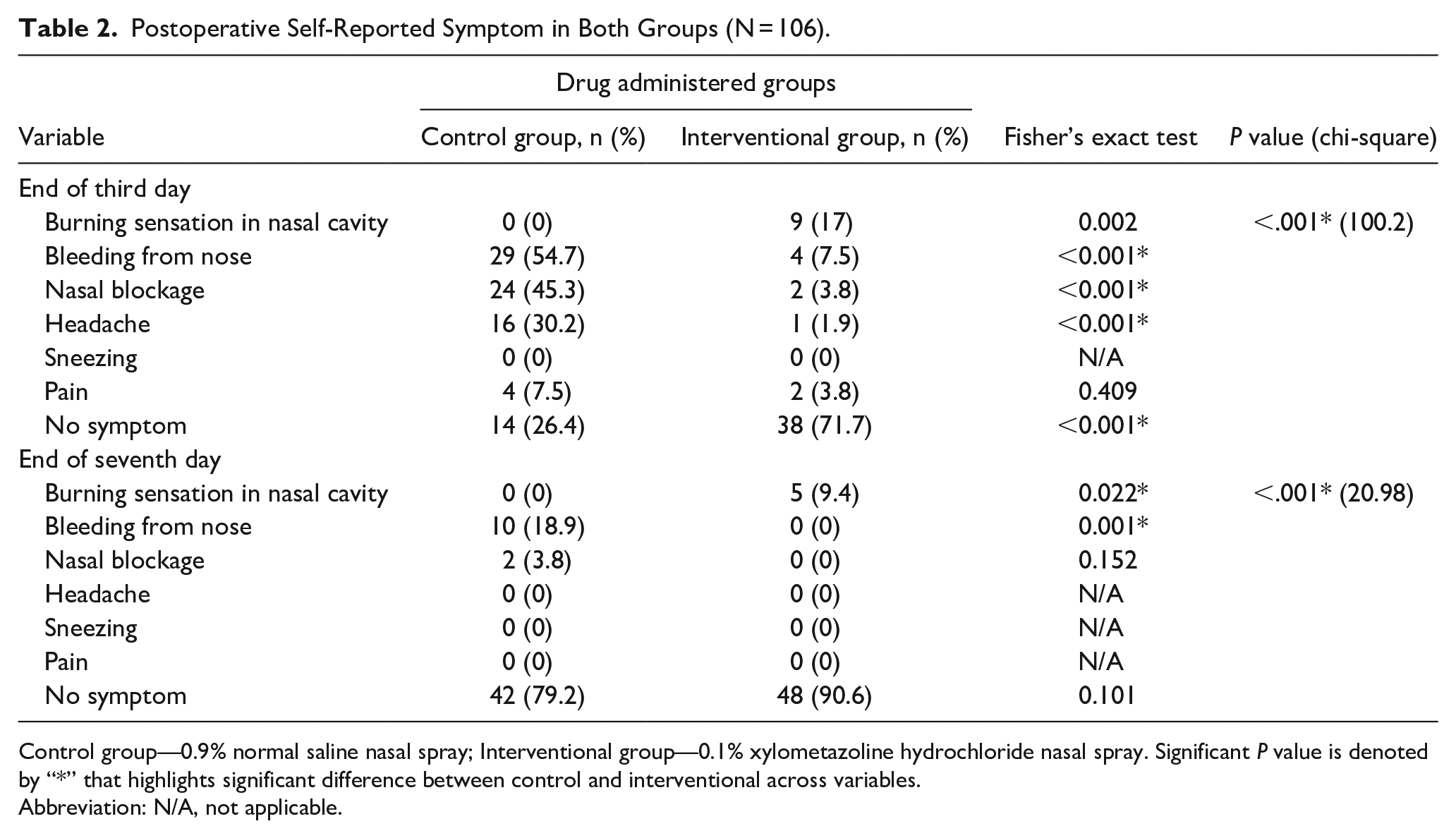
Control group—0.9% normal saline nasal spray; Interventional group—0.1% xylometazoline hydrochloride nasal spray. Significant P value is denoted by “*” that highlights significant difference between control and interventional across variables.
Abbreviation: N/A, not applicable.
Postoperative Clinical Nasal Endoscopy Findings
Notably, on the third day of recovery, patients in control group (0.9% normal saline nasal spray) displayed more severe incidences of nasal edema, crusting, nonhealed scars, and nasal discharge than patients in intervention group (0.1% xylometazoline hydrochloride nasal spray). In addition, these clinical nasal findings persisted to a greater extent in control group patients compared to intervention group patients on the seventh day of recovery, where the symptoms were virtually absent (Table 3).
Postoperative Nasal Endoscopy Findings in Both Groups (N = 106).
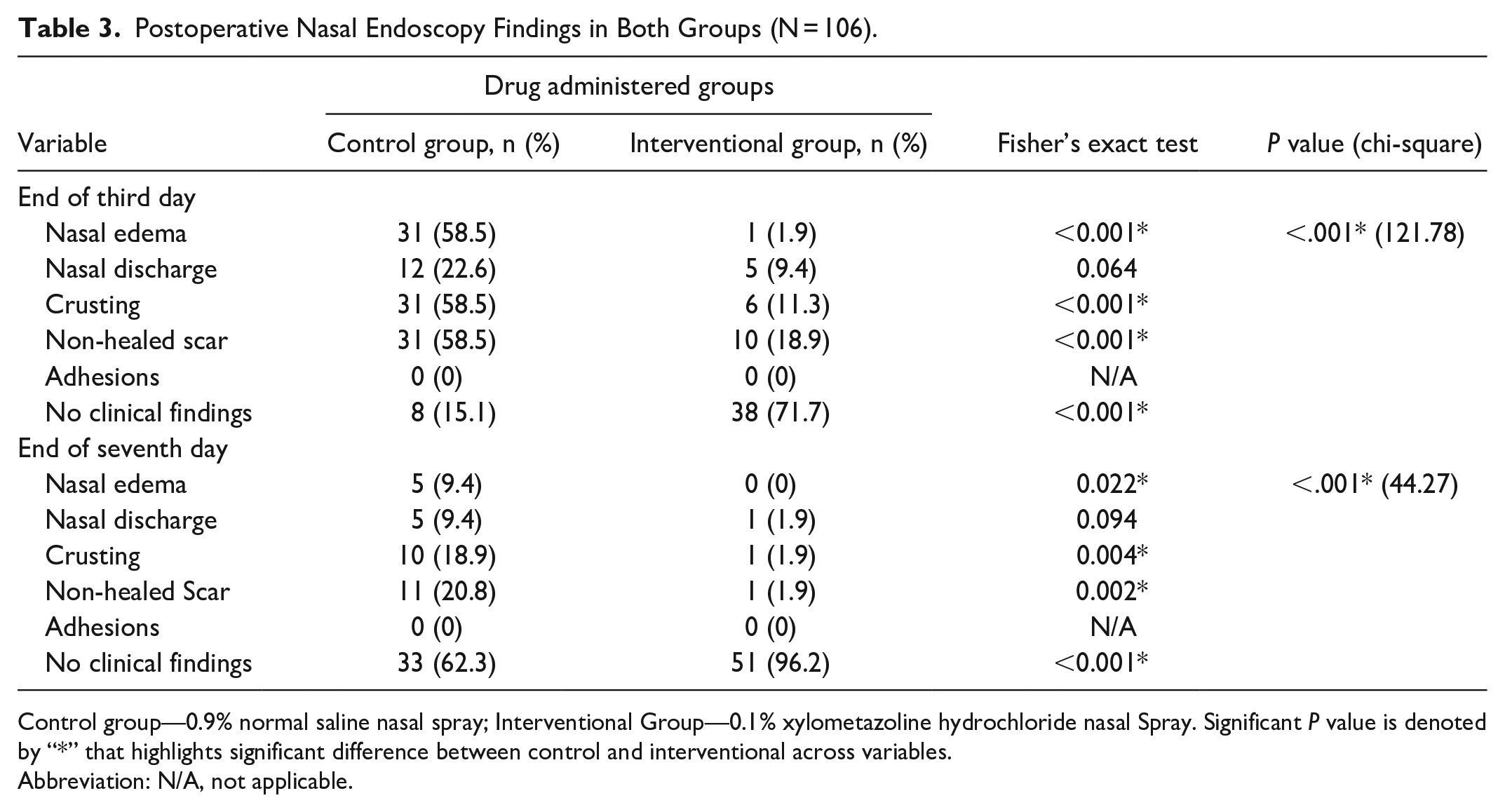
Control group—0.9% normal saline nasal spray; Interventional Group—0.1% xylometazoline hydrochloride nasal Spray. Significant P value is denoted by “*” that highlights significant difference between control and interventional across variables.
Abbreviation: N/A, not applicable.
Postoperative Drug-Associated Side Effects and Patient Satisfaction
Both the nasal sprays had no side effects. Ultimately, the patients of intervention group were more satisfied that were given xylometazoline-hydrochloride nasal spray as compared to control group patients that were given normal saline nasal spray (P value <.001*; Figure 2).
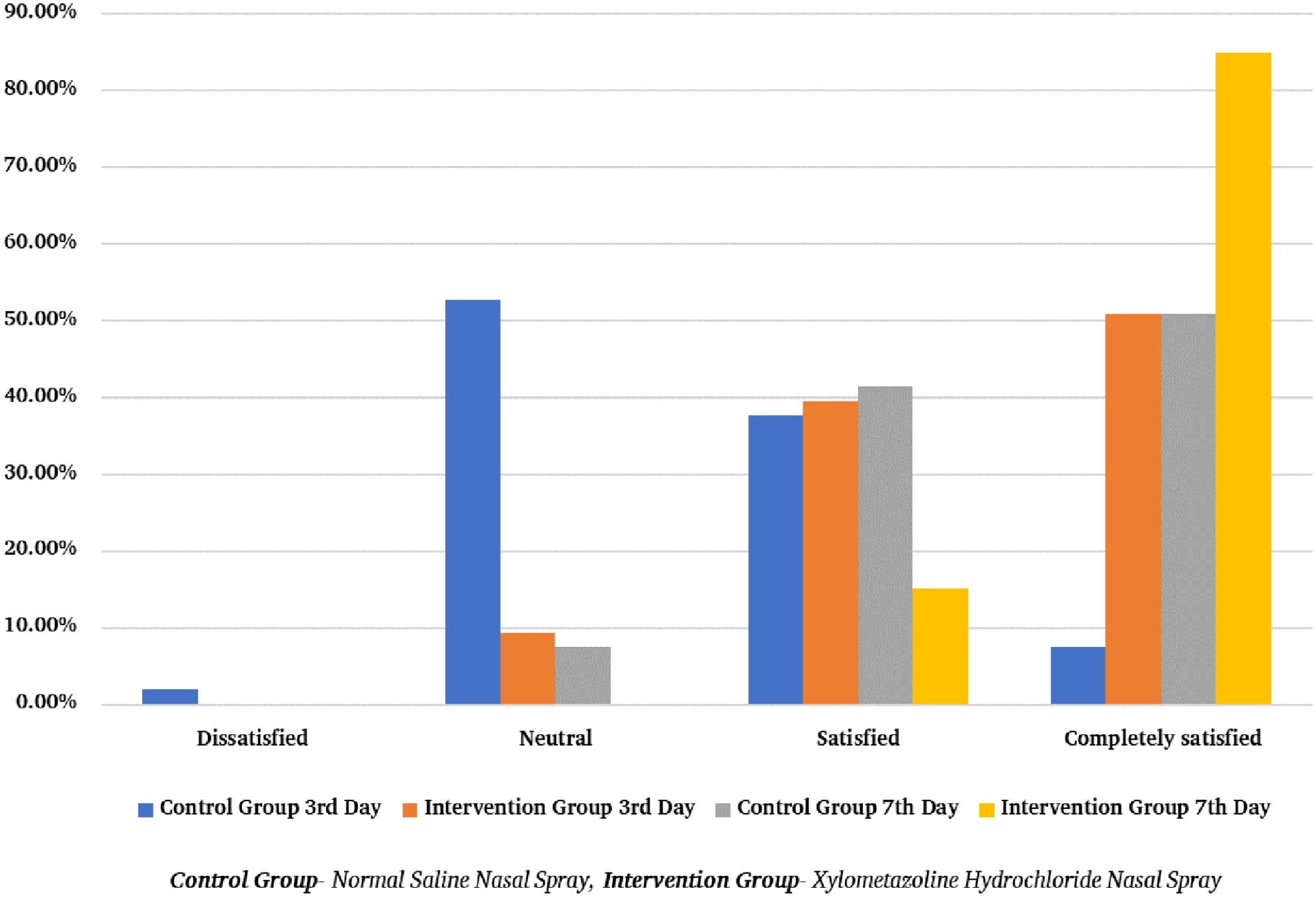
Postoperative patient satisfaction (N = 106).
Discussion
A DNS is the most common condition worldwide,1,2 and can lead to nasal obstruction and congestion, which in turn can cause additional symptoms. Surgical intervention in the form of septoplasty can alleviate these symptoms. Common symptoms following nasal surgery due to postoperative inflammation include nasal obstruction, rhinorrhea, pain, and bleeding, among others. 11 These symptoms may last for several days or weeks. The post-septoplasty recovery period is crucial for optimal recovery and symptom relief. Although several regimens5,6,8,9 have been proposed to expedite post-septoplasty recovery, a standardized approach has yet to be established.
We present the results of a multicenter, randomized, triple-blind study conducted at 2 institutes in Karachi, Pakistan, between June 2022 and June 2023. This study elucidates the efficacy of 0.1% xylometazoline hydrochloride nasal decongestant spray in preventing post-septoplasty symptoms, clinical nasal findings, adverse effects, and evaluating patient satisfaction. The placebo used in the study was normal saline, and the intervention consisted of nasal decongestant spray contained 0.1% xylometazoline hydrochloride. To eradicate bias, the study recruited a diverse group of participants aged 15 to 60 years and included only septoplasty cases. The study was meticulously blinded, and patient monitoring spanned a period of 7 days.
In our clinical practice, it is customary to recommend the administration of nasal irrigation with saline solutions,12-14 or topical/oral steroids9,15 subsequent to nasal surgical procedures, with the intention to aid in clearing secretions, detritus, and crusts, reducing the risk of postoperative adhesions, promoting mucosal healing. However, complete amelioration of the symptoms like nasal obstruction due to edema or discharge, bleeding, and side effects remains elusive for a considerable proportion of patients with the use of aforementioned regimens. This predicament is most likely attributed to the occurrence of postoperative vasodilation and swelling of the nasal mucosa. We hypothesize that the incorporation of nasal decongestants into the post-septoplasty care regimen could yield significant advantages for patients’ recovery. This conjecture is founded on the vasoconstrictive properties inherent to nasal decongestants, 16 which hold the potential to mitigate symptoms of nasal blockage and bleeding by counteracting the vasodilatory responses linked to inflammation, which in turns helps in preventing other symptoms occurring from blockage or bleeding.
Despite the well-known properties of decongestant spray, only 2 studies have investigated its effects in the context of postnasal surgery care. The study conducted by Dağli et al, 6 involving the application of a combination of 0.05% saline and oxymetazoline nasal spray in comparison to normal saline in post-septoplasty symptom relief, revealed that the use of oxymetazoline in conjunction with saline as a nasal spray leads to a reduction in symptoms following septoplasty (bleeding, crusting, and pain) over 2 weeks, as opposed to the use of normal saline alone. Another, study conducted by Humphreys et al 17 involving the use of 0.1% xylometazoline hydrochloride nasal spray did not reveal any discernible advantages in alleviating pain when compared to the use of saline nasal spray in the context of nasal surgeries (functional endoscopic sinus surgery and septoplasty) when observed on day 10. However, the results of our investigation indicate that the administration of 0.1% xylometazoline hydrochloride nasal spray does indeed reduce post-septoplasty symptoms and nasal manifestations, with significant improvements on the third day of drug use and complete recovery in the majority of patients by the seventh day, with no adverse effects, in comparison to the saline spray. This discrepancy between our findings and those of previous studies may be attributable to the balanced distribution of sample sizes between both groups, the duration of follow-up, the administration of drugs in our study, and ensuring the correct method of drug delivery. Another contributing factor may be the disparate use of assessment tools; whereas earlier studies relied on Visual Analog Scale scores to determine the severity of bleeding, pain, and nasal crusting only, whereas, our study focused on the absence and presence of multiple post-septoplasty symptoms, nasal manifestations, and adverse effects, which is a strength of our study. The administration of these regimens individually rather than in combination and only in the context of septoplasty cases is a further important distinction. The inclusion of nasal splints and additional medications in the protocols of the aforementioned investigations, which was not the case in our study, may also contribute to disparities. Another distinguishing factor would be the exclusion of patients who had Functional Endoscopic Sinus Surgery (FESS), any trauma or previous surgery.
Furthermore, our study found that the cohort receiving 0.1% xylometazoline hydrochloride nasal spray did not require any further medications or procedures to reduce symptoms, clinical nasal findings, or adverse effects, resulting in a shorter recovery period. In contrast, the 0.9% normal saline nasal spray group required a variety of interventions at the end of day 7, including the administration of tranexamic acid 500 mg capsules every 8 hours for bleeding management, nasal packing lasting 24 hours, nasal douching to address postoperative nasal endoscopic findings such as crusting, blood clots, and obstruction, and the use of low-dose prednisolone tablets for severe cases of nasal mucosal edema, which is similar to the study by Dağli et al 6 but opposed the study by Humphreys et al 17 because nasal irrigation with normal saline only aids in clearing secretions, detritus, and crusts, which may contribute to recovery to some extent but not completely.
In addition, Tugrul et al 8 investigated using nasal steroids in conjunction with saline irrigation to speed up recovery after septoplasty. Over a 30 day period, their findings demonstrated significant improvements in post-septoplasty symptoms. In contrast, the current study revealed a more pronounced reduction in symptoms within a week. This significant improvement was achieved using a 0.1% xylometazoline hydrochloride nasal decongestant spray, which notably lacked the adverse effects of steroid administration. In addition, steroid use frequently necessitates a progressive tapering-off procedure, which is a disadvantage.
In addition, NSAIDs, paracetamol, and oral enzymes (serrapeptase, bromelain, and papain) are administered postoperatively in tablet form,5,9 varies on geographical location and individual physician practices. Despite these treatments being beneficial for post-septoplasty recovery due to their anti-inflammatory properties, they are not optimal due to their lack of therapeutic properties, first-pass effect, and prolonged duration of use, which cause significant gastrointestinal adverse effects.5,9 In contrast, 0.1% xylometazoline hydrochloride nasal spray circumvents the deficiencies of the aforementioned treatments and promotes faster recovery in a brief period of time with no adverse side effects, as depicted by our study.
In the immediate aftermath of early-stage septoplasty procedures, nasal decongestants can be prescribed to patients. This recommendation is based on their efficacy, favorable patient tolerability, cost-effectiveness, and greater patient satisfaction. Nevertheless, it is essential to recognize that nasal decongestants may cause discomfort during the initial use, which diminishes as the recovery progresses. It is prudent to administer nasal decongestants with caution, as excessive or protracted use can precipitate rhinitis medicamentosa, 18 which was mitigated in our study by using the drug for a brief time. The patient should be made aware of the aforementioned side effect.
The present analysis highlights some limitations that merit careful attention. The study was conducted solely at 2 healthcare institutes in Karachi, Pakistan. Therefore, it is essential to exercise caution when extrapolating the results to various geographical areas or populations with unique demographic characteristics, healthcare practices, and environmental elements. Furthermore, the duration of the study’s follow-up was relatively short, including a time frame of merely 7 days. Although the immediate recovery phase analysis provided valuable insights, the limited temporal scope prevented an assessment of the potential long-term repercussions of using 0.1% xylometazoline hydrochloride nasal spray beyond the specified duration. Future academic research should focus on addressing these temporal gaps by examining the long-term implications, such as the potential occurrence of rebound congestion after discontinuing the medication. Last, the sole limitation associated with the use of 0.1% xylometazoline hydrochloride nasal decongestant spray is a burning sensation experienced by minimal number of patients. This sensation is considered a normal part of the healing process. However, it is important to inform patients of this potential effect prior to initiating treatment.
Conclusions
In conclusion, this study demonstrates the efficacy of a 0.1% xylometazoline hydrochloride nasal decongestant spray in mitigating symptoms and nasal manifestations following septoplasty. This intervention facilitates a quicker recovery and enhances patient satisfaction. The lack of adverse effects further reinforces the justification for its utilization as a therapeutic adjunct in the recovery process following septoplasty. This approach demonstrates superiority compared to administering the standard saline solution, as well as previous literature encompassing various medication types such as oral enzymes, NSAIDs, steroids, and even oxymetazoline spray. Further research is warranted to validate these results and investigate the most effective time frame for using nasal decongestants, especially 0.1% xylometazoline hydrochloride nasal decongestant spray, in managing patients of different ethnicity and economy status following septoplasty and other nasal surgeries.
Supplemental Material
sj-doc-3-ear-10.1177_01455613241266473 – Supplemental material for Efficacy of 0.1% Xylometazoline-Hydrochloride Nasal Decongestant Spray in Postoperative Sign/Symptom Relief Following Septoplasty: A Randomized Control Trial
Supplemental material, sj-doc-3-ear-10.1177_01455613241266473 for Efficacy of 0.1% Xylometazoline-Hydrochloride Nasal Decongestant Spray in Postoperative Sign/Symptom Relief Following Septoplasty: A Randomized Control Trial by Muhammad Hamza Dawood, Shanila Feroz, Sheza Sohail, Mavra Roshan, Haseefa Perveen and Tasmiha Nizam in Ear, Nose & Throat Journal
Supplemental Material
sj-docx-2-ear-10.1177_01455613241266473 – Supplemental material for Efficacy of 0.1% Xylometazoline-Hydrochloride Nasal Decongestant Spray in Postoperative Sign/Symptom Relief Following Septoplasty: A Randomized Control Trial
Supplemental material, sj-docx-2-ear-10.1177_01455613241266473 for Efficacy of 0.1% Xylometazoline-Hydrochloride Nasal Decongestant Spray in Postoperative Sign/Symptom Relief Following Septoplasty: A Randomized Control Trial by Muhammad Hamza Dawood, Shanila Feroz, Sheza Sohail, Mavra Roshan, Haseefa Perveen and Tasmiha Nizam in Ear, Nose & Throat Journal
Supplemental Material
sj-pdf-1-ear-10.1177_01455613241266473 – Supplemental material for Efficacy of 0.1% Xylometazoline-Hydrochloride Nasal Decongestant Spray in Postoperative Sign/Symptom Relief Following Septoplasty: A Randomized Control Trial
Supplemental material, sj-pdf-1-ear-10.1177_01455613241266473 for Efficacy of 0.1% Xylometazoline-Hydrochloride Nasal Decongestant Spray in Postoperative Sign/Symptom Relief Following Septoplasty: A Randomized Control Trial by Muhammad Hamza Dawood, Shanila Feroz, Sheza Sohail, Mavra Roshan, Haseefa Perveen and Tasmiha Nizam in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
The authors would also like to express their gratitude to UMDC for providing ethical approval for our study and encouraging us to conduct research within the institute and affiliated hospitals. The authors would like to thank Mrs Hina Gul for their contribution in statistics of this study as well 2 doctors namely Dr Osama Taj and Dr Shamim Ahmed for their contribution in data collection. The authors would also like to acknowledge the Research Council of Pakistan for their encouragement and assistance in teaching us how to conduct research.
Author’s Note
S.F. and M.H.D. are the first authors of the study and have contributed equally.
Authors Contributions
S.F. and M.H.D. are the guarantors of the study. Both S.F. and M.H.D. are the first authors of the study and have contributed equally. The contributions are as follows: concept and design, ethical approval, administration of the study, acquisition, interpretation of data, drafting of the manuscript, critical revision of the manuscript for important intellectual content and validation of statistics and procedures. S.S., M.R., H.P., and T.N. are involved in acquisition, interpretation of data, drafting of the manuscript, critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request, as de-identifiable data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The study proposal was approved by the Institutional Review Board of the United Medical and Dental College in Karachi (CGH/Ethics/Retro/2022/20/06/328). All methods adhered to the most recent/appropriate iteration of the Helsinki Declaration on using human subjects in scientific research. Participants gave written informed consent before participating in the study.
Consent
Written Informed Consent was taken from all the patients for their participation during the collection process.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
