Abstract
Introduction
Sublingual immunotherapy (SLIT) has been utilized for the treatment of atopy for several decades with use dating back to the 1930s. SLIT gained prominence with the first trial in 1986 by Dr Glenis Scadding’s group, followed by several trials focusing on efficacy.1-3 Before the development of SLIT, subcutaneous immunotherapy (SCIT) remained the primary form of allergen immunotherapy (AIT) after its development over a century ago by Leonard Noon. SCIT is highly effective and is regarded as the gold standard for allergic rhinitis.4,5 However, the use of SCIT has been associated with adverse events including local reactions and systemic reactions such as anaphylaxis or death. Fatalities associated with SCIT are estimated at 1 in 2 to 2.5 million doses given and occur mostly during the escalation phase of treatment.6-8 Fatal reactions have been attributed to human error while administering SCIT. In a 2004 survey of physicians, 57% reported at least one occurrence of incorrect SCIT administration to patients. 9 Due to the potential severe systemic reactions associated with SCIT, SLIT has been regarded as a safer alternative and is slowly gaining favor.
Despite the increased use of SLIT across Europe, SLIT has been slower to gain popularity in the United States. Surveys done through the American College of Allergy, Asthma and Immunology (ACAAI) assessed the use of SLIT by US physicians. In 2007, only 5.9% of allergists reported using SLIT, mainly attributed to a lack of Food and Drug Administration (FDA) approval. 10 By 2011, SLIT use remained low at 11.4%, 11 but increased to 73.5% in 2018 following FDA approval for standardized grass, ragweed, and dust mite sublingual tablets.12-14 Still, in 2018, American allergists reported the main reason for lack of SLIT utilization to be a lack of FDA-approved multi-antigen protocols. 15
With growing SLIT utilization, new questions emerge regarding safety and potential side effects. The most common adverse events associated with SLIT are local intraoral reactions which usually occur at the initiation of treatment.16,17 There have been scarce reports of systemic reactions and early anaphylaxis.18,19 This article aims to address the safety profile of SLIT by analyzing prospective controlled trials of SLIT to determine the most common adverse events associated with SLIT, rates of local versus systemic reactions, and rates of anaphylaxis. Additional information on discontinuation of treatment was also addressed.
Methods
A comprehensive literature search was performed. Articles were identified in PubMed, Cochrane Database, and Scopus using search terms “oral immunotherapy” or “sublingual immunotherapy” and “allergic rhinitis” or “environmental allergy” and “drug-related side effect” or “side effect” or “anaphylaxis.” Studies for the FDA-approved SLIT tablets for grass, ragweed, and dust mite were included. Patients were included who were enrolled in a clinical trial during the time of January 1, 2001, to December 31, 2021. Abstracts were independently reviewed by 2 reviewers (T.A.J. and R.O.J.). Case reports, review articles, retrospective studies, and nonhuman studies were excluded. Full texts of included articles were reviewed for all clinical trials. Articles without primary endpoints of side effects or anaphylaxis were excluded. Adverse events from trials were recorded for further analysis as well. Texts without English translation were excluded after appropriate attempts to translate the articles. Data extraction and quality assessment was performed independently by 2 reviewers (T.A.J. and R.O.J.). Differences in data interpretation were resolved by a third reviewer (K.B.H.) The level of evidence for each article was evaluated according to the Oxford Center for Evidence-Based Medicine criteria. The risk of bias (ROB) for randomized studies was assessed via the Cochrane Handbook for Systematic Reviews of Interventions, Version 6.3, 2022. For nonrandomized studies, ROB items included the following: confounding, selection of participants into the study, classification of interventions, missing data, measurement of outcomes, and selection of reported result. For randomized controlled trials, ROB items included the following: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other bias. ROB grading criteria were specified as “low,” “unclear,” or “high.”
Results
Overview of Studies
The initial literature search yielded 186 unique articles. Figure 1 shows the reasons for study exclusion, which resulted in 26 articles for data analysis.20-45 The included studies were published between 2003 and 2021, reporting data on a total of 7827 patients (Table 1). Critical appraisal of studies indicated an acceptably low ROB (Figures 2-4). Potential sources of bias were most pronounced in blinding of participants and personnel and blinding of outcomes assessment. Most nonrandomized studies were considered low risk, with greater potential for bias due to deviations from intended interventions. A funnel plot with Egger’s test (−1.24, 95% confidence interval −4.25 to 1.77, P = .41) demonstrated all studies were within the funnel except for one with no asymmetry, suggesting little publication bias (Figure 2).
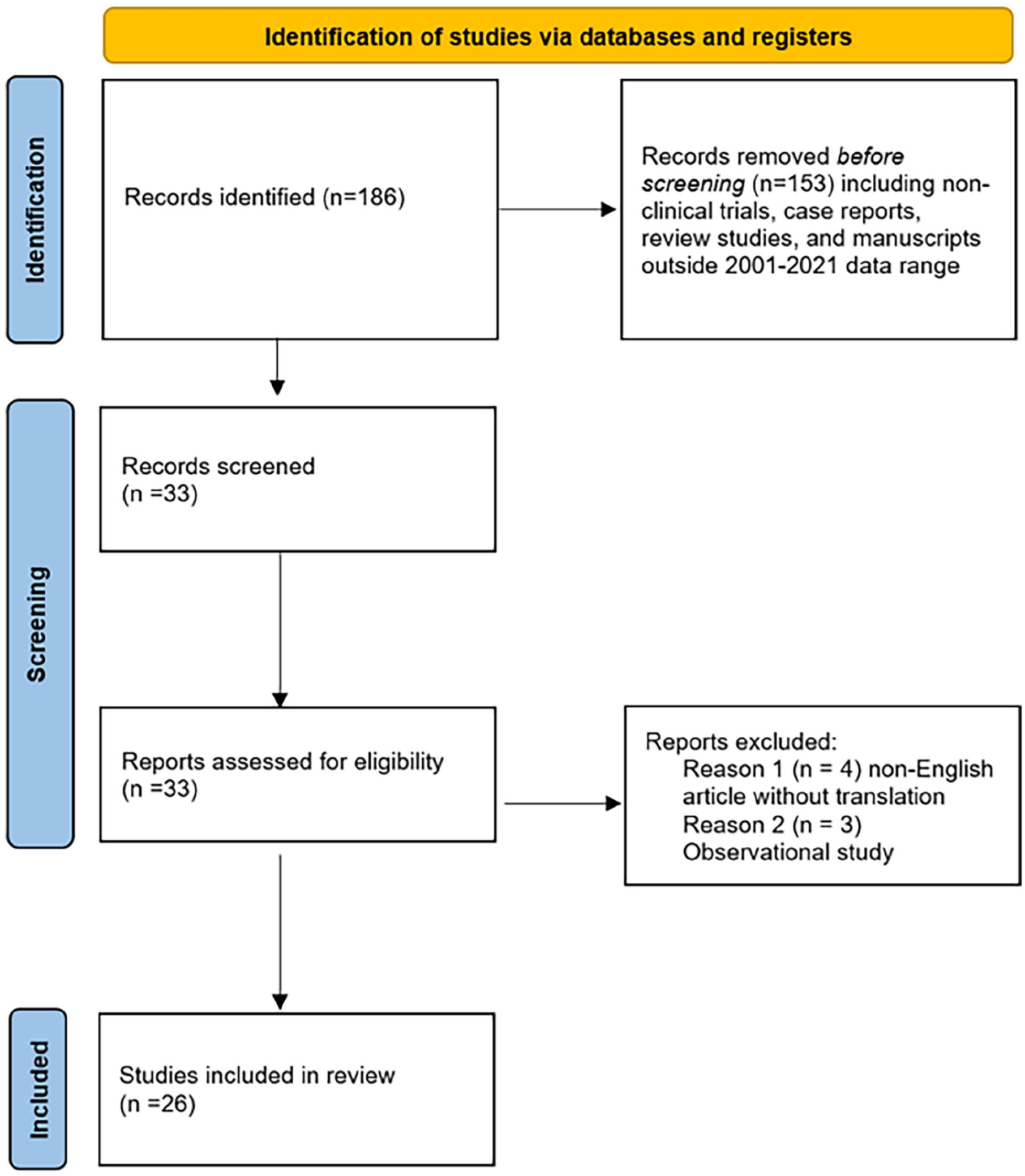
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) diagram for included randomized controlled trials for SLIT. SLIT, sublingual immunotherapy.
Description of Included Studies.
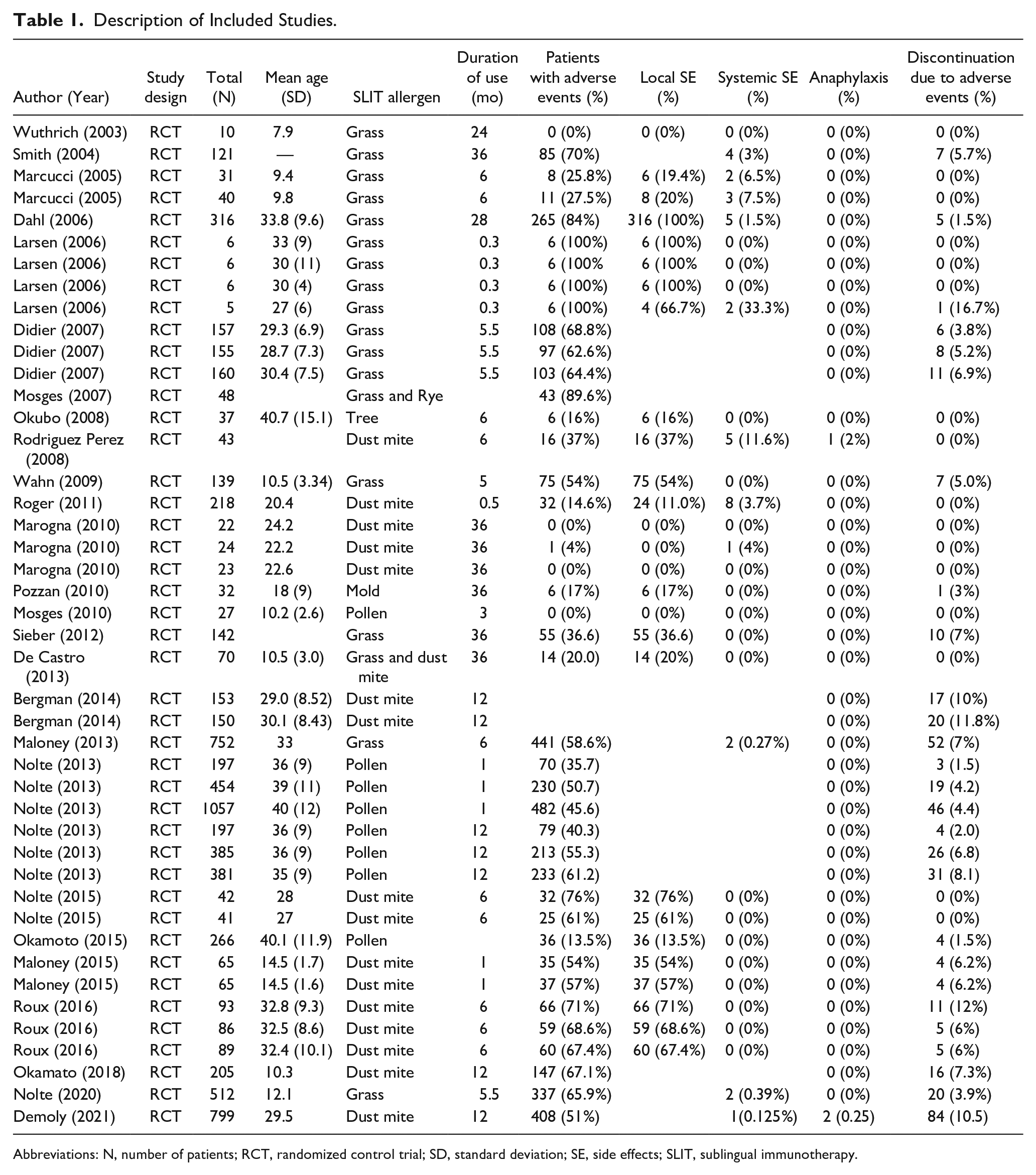
Abbreviations: N, number of patients; RCT, randomized control trial; SD, standard deviation; SE, side effects; SLIT, sublingual immunotherapy.
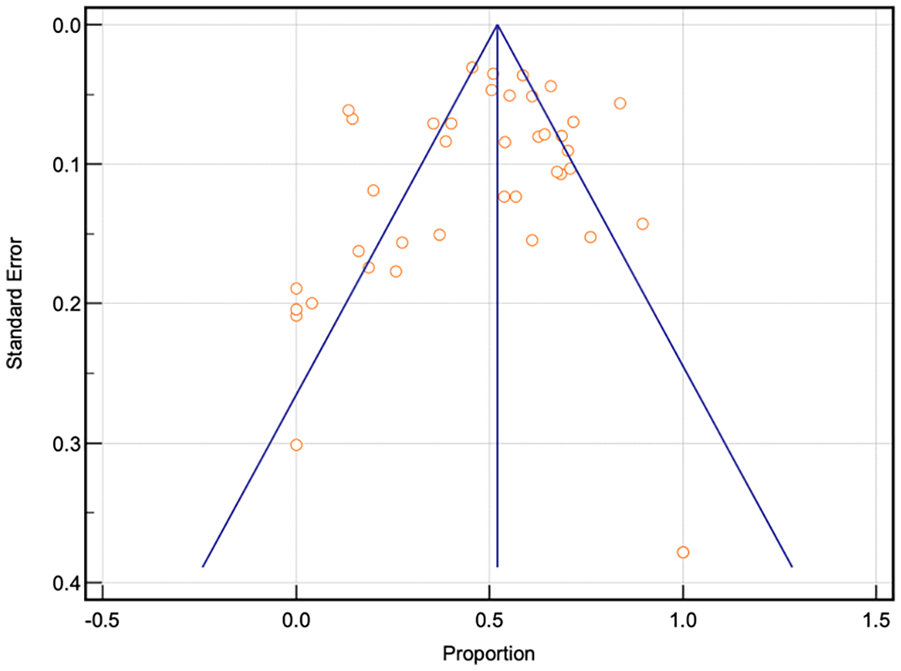
Funnel plot for bias assessment of analyzed articles.
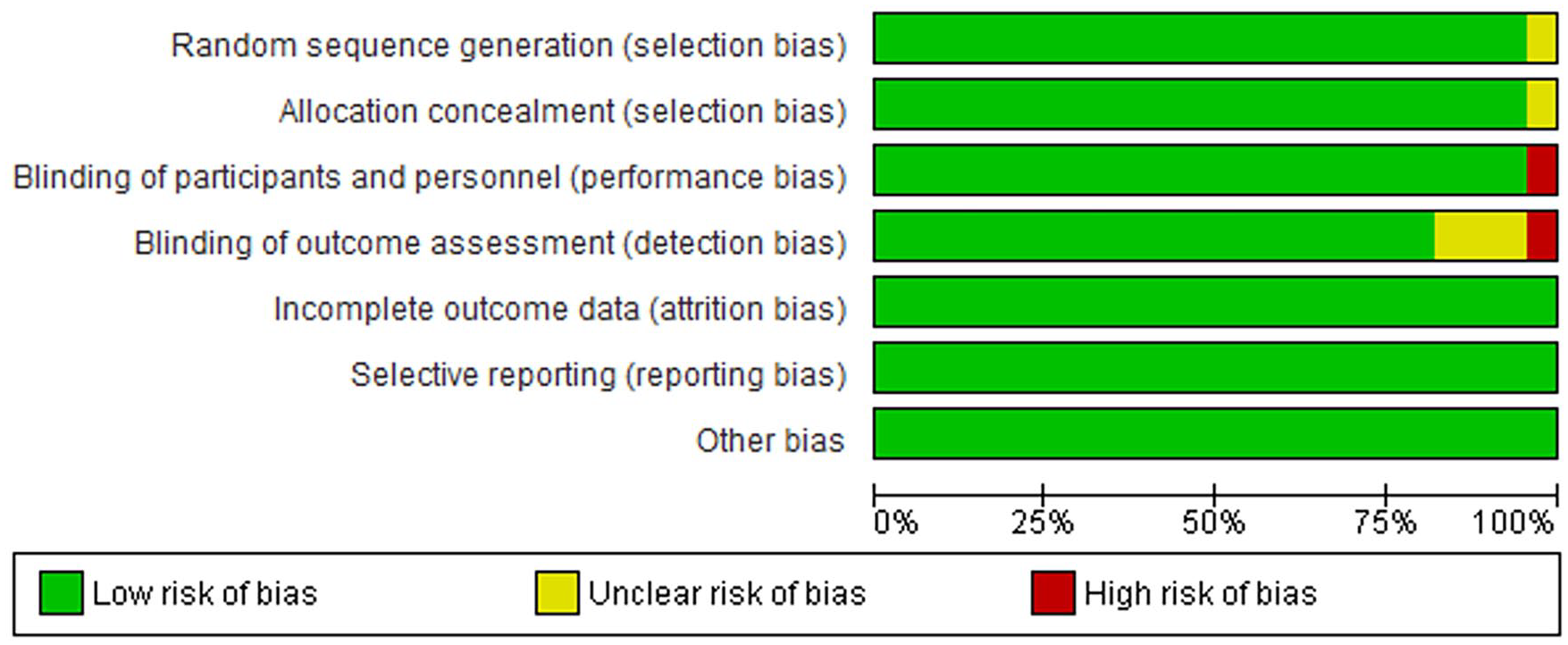
Risk of bias among randomized controlled trials.
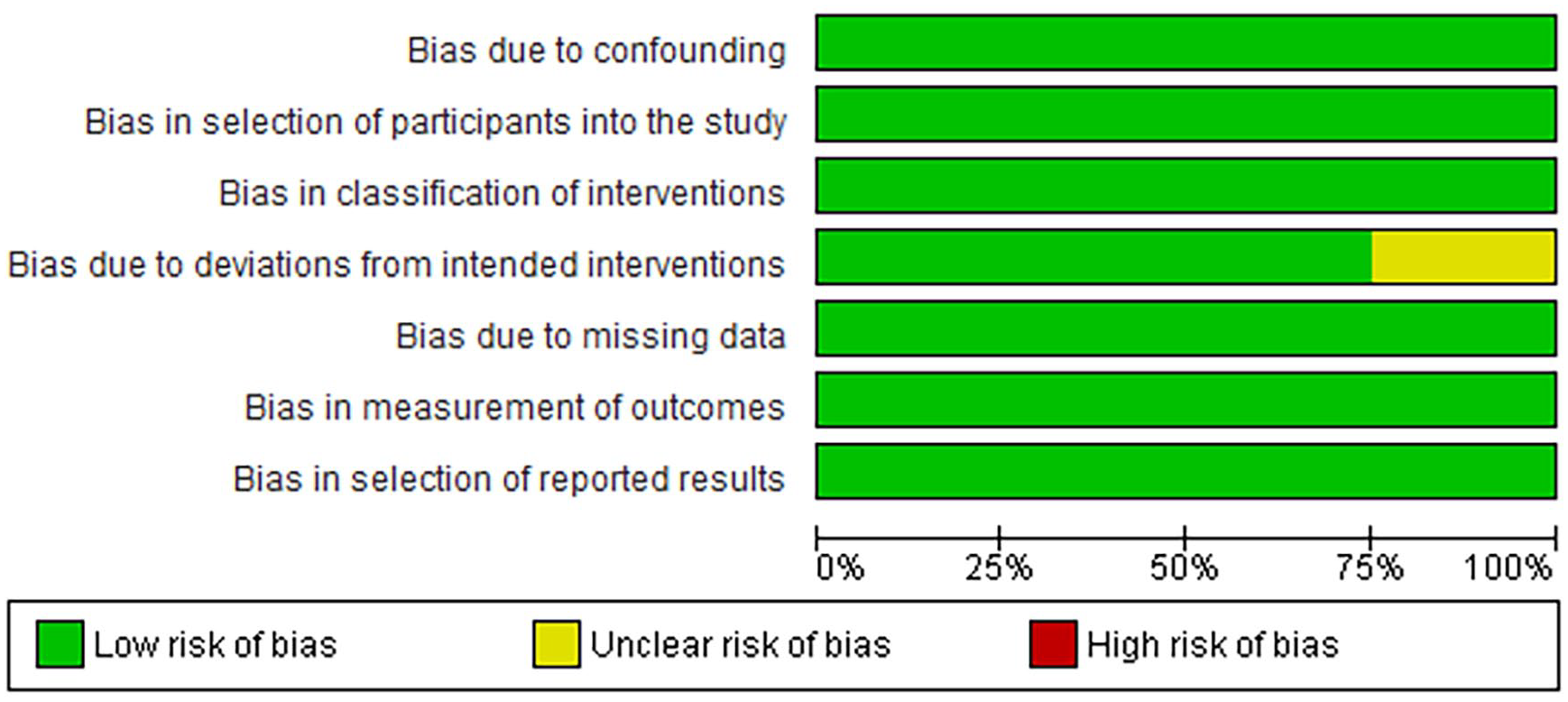
Risk of bias among nonrandomized controlled trials.
Demographics
In total, 7827 patients were evaluated with a mean age of 25.91 years (SD 10.11). The mean duration of treatment was 11.54 months. This represented 2,749,178 daily doses of SLIT. All studies evaluated single-antigen protocols, with most allergens being either grass (40.9% of studies) or dust mite (38.6% of studies). Most antigens were delivered in tablet form (75%).
Side Effect Rates and Anaphylaxis
Regarding side effects, 48.0% of patients included in the study were noted to have side effects while on SLIT. Local side effects were present in 46.19% of patients overall. There were 898 local reactions out of 1944 patients equating to 1 reaction per 3061 doses. Systemic side effects were encountered in 0.921% of SLIT patients. Forty systemic reactions were noted out of 4128 patients equating to 1 reaction per 36,248 doses. Anaphylaxis was reported in 0.38% of patients with 3 events out of 7287 patients equating to 1 reaction per 916,393 doses. Discontinuation due to side effects was noted to occur in 4.32% of patients.
Discussion
With the growing burden of atopic disease including allergic rhinoconjunctivitis and asthma, 46 it is not surprising that SLIT is gaining popularity as a viable form of AIT. Previous literature has shown SLIT to be beneficial for improvement in symptom management. Radulovic et al found significant improvement in overall symptom scores for patients on SLIT compared to placebo. 47 In addition, Durham et al stated these improvements have been estimated to last at least 2 to3 years after discontinuation of therapy. 48 Furthermore, Zielen et al noted an 18.8% reduction in allergic rhinitis medication use for patients taking SLIT for grass pollen. 49 As noted in our meta-analysis, most SLIT studies focus on single allergens, whereas the majority of SCIT protocols in North America are based on multi-antigen treatment as most patients are polysensitizied. 50
Several key findings were noted from our study. Minor local side effects remain common in patients undergoing SLIT. Our study found that over 40% of patients undergoing SLIT within the clinical trials studied experienced local side effects, but this did not deter patients from discontinuing treatment (discontinuation rate: 4%). Higher local side effects have been reported during SLIT, commonly during start of treatment since there is either an accelerated or absent escalation phase unlike traditional SCIT.6,51
While local side effects are common early in treatment, systemic side effects from SLIT are rare. Our study found systemic side effects in 1% of patients. Masuyama et al similarly found systemic side effects to be rare for SLIT patients with a reported frequency of 0.08%.52,53 Physiologically, systemic side effects are less likely due to antigen processing by dendritic cells within the sublingual mucosa. These dendritic cells have a tolerogenic phenotype which may reduce the pro-inflammatory cascade. 18 Comparatively, SCIT may lead to systemic absorption and thus trigger inflammatory cascades through absorption into local vasculature. 54 This study further confirmed the rarity of anaphylaxis for patients undergoing SLIT with a reported rate of less than 0.4% or 1 in 916,393 SLIT doses with none being severe, near-fatal, or fatal events. Comparatively, previous studies have also noted similar low rates of anaphylaxis for SCIT with reported ranges of 0.1% to 0.2% of injections which include near-fatal and fatal reactions. 51
This study’s low SLIT discontinuation rate (4%), despite over 40% local side effects, highlights the high compliance among SLIT patients. SLIT offers patients the ease of at-home self-administration as well as the comfort of avoiding injection administration. 55 Furthermore, given the at-home usage, SLIT has been noted to be more cost-effective and convenient as compared to SCIT. 56
Although comprehensive in nature, this meta-analysis has several limitations. First, this study analyzed only clinical trials and thus retrospective studies were not evaluated. The authors chose to analyze clinical trials given the controlled natures of the studies. Allergy testing and AIT protocols are very heterogenous, and thus it was important to limit some variability. Second, the average total time of treatment at 11.54 months was not ideal; delayed reactions, although less likely, may not have been included or evaluated. Furthermore, treatment attrition may be higher when evaluated over a longer time period. Third, comorbidities were not analyzed due to limited data availability. Finally, we only evaluated patients receiving single-antigen SLIT. Further studies are needed to evaluate long-term efficacy, side effects, and perhaps directly compare rates of reactions between SLIT and SCIT.
Conclusion
In this systematic review and meta-analysis, SLIT patients tolerated treatment well with a low attrition rate. Most of the side effects experienced during treatment were minor and local; systemic and anaphylaxis side effects were rare. SLIT is a safe and useful tool in improving the burden of atopic disease, with studies showing improved allergic rhinoconjunctivitis symptoms, decreasing medication use, as well as asthma amelioration. 57 Further long-term follow-up is necessary to evaluate the overall efficacy profile of SLIT, optimum dosage and length of treatment, single versus multiple antigen therapies, as well as prospective comparisons of SLIT and SCIT. For the treatment of environmental allergies, SLIT remains an important treatment modality with a safe and effective side effect profile which allows for straightforward use for patients.
Footnotes
Data Availability
Data for this systemic review were collected from the cited articles as included in the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the University of Texas Medical Branch Institutional Review Board on October 1, 2021.
