Abstract
A neoplasm was found in the left nasal cavity of a 45-year-old woman during electronic laryngoscopy for reflux pharyngitis. She reported experiencing an occasional slight headache in the left parietal region for 1 to 2 years, which she considered a migraine. Electronic laryngoscopy showed a gray, soft, smooth neoplasm in the left nasal meatus, located near the olfactory region blocking the olfactory clef and compressing the left middle turbinate. The neoplasm was resected at endonasal endoscopic surgery. Histological assessments indicated chronic mucus inflammation and cyst formation. This is a rare case because the polyp was large but asymptomatic and originated from nasal septum.
Case Report
A neoplasm was found in the left nasal cavity of a 45-year-old woman during electronic laryngoscopy for reflux pharyngitis. She complained of an abnormal sensation in the throat due to gastroesophageal reflux disease. She reported experiencing occasional slight headaches in the left parietal region for 1 to 2 years, which she considered migraine. She did not have nasal obstruction, nasal bleeding or discharge, or visual or olfactory impairment. Furthermore, there was no history of head trauma or surgery. The physical examination revealed intact skin on the nose, with no rash, redness, or edema on either side. There was no facial asymmetry, weakness, sensation abnormalities, physical activity disorders, or altered consciousness. The electronic laryngoscopy was performed through a nostril and the neoplasm was found accidently. It was reported as a gray, soft, smooth lesion in the left nasal meatus that was located near the olfactory region blocking the olfactory clef and compressing the left middle turbinate. The neoplasm was intact with a clear boundary, solid texture, and did not bleed on touch. The neoplasm originated from the nasal septum with a thin stalk connecting it to mucosa in that area. Abnormal rhinorrhea was not observed (Figure 1A-C, red arrow). Computed tomography (CT) scan and magnetic resonance imaging (MRI) indicated that the lesion was soft and tissue-like (Figure 2). CT showed a soft tissue-like lesion measuring 5 mm × 22 mm × 21 mm in the olfactory clef (Figure 2A-C, red star). No bone destruction was found. MRI showed an abnormal shadow in the left nasal cavity adjacent to the nasal septum measuring 9 mm × 23 mm × 22 mm (Figure 2D-F, blue star).
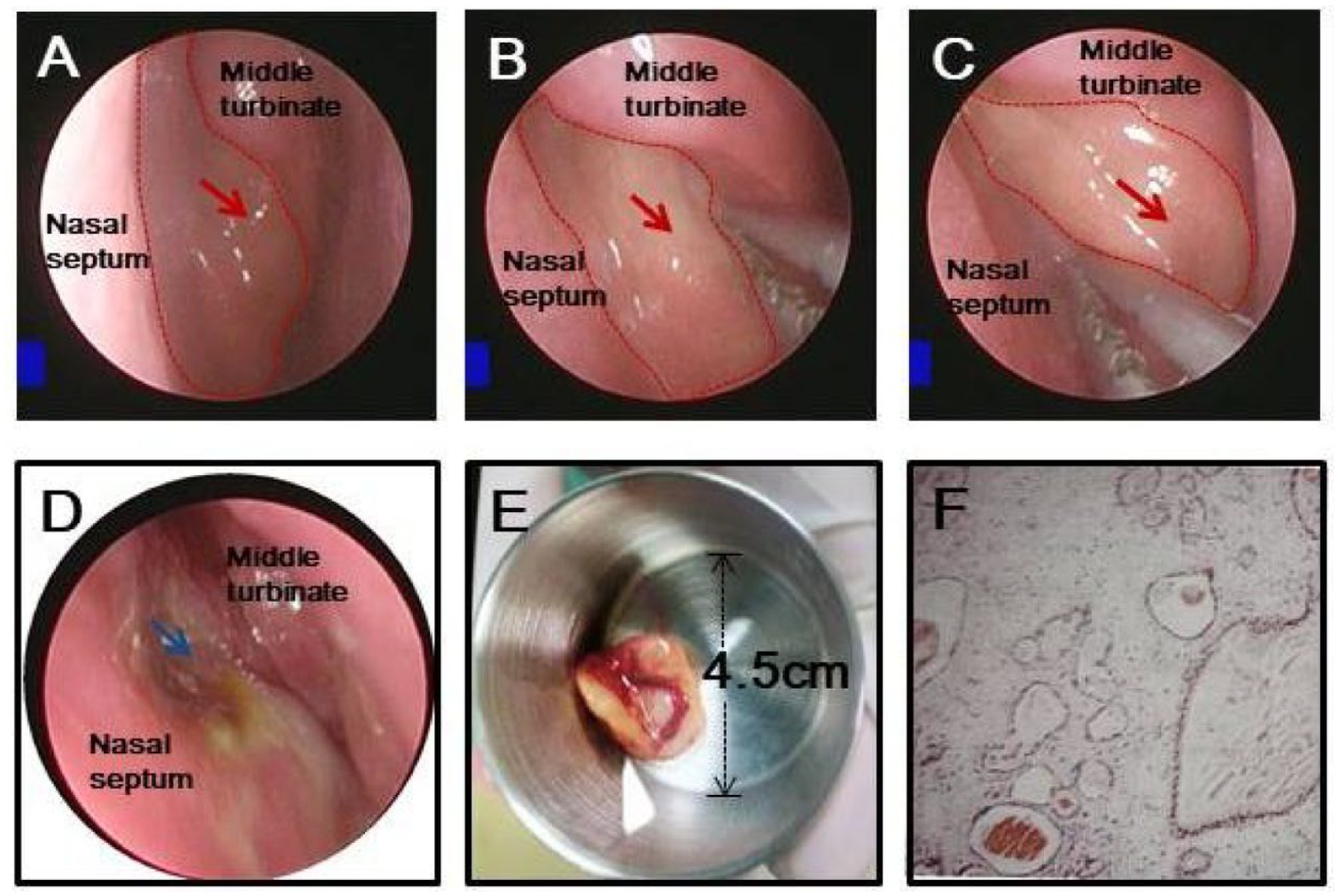
The nasal septum lesion. (A-C) Endonasal endoscopy shows a gray, soft, smooth lesion located near the olfactory region blocking the olfactory clef and compressing the left middle turbinate; (D) the nasal cavity after removing the lesion; (E) the resected lesion; and (F) the pathological images.
Using endonasal endoscopic surgery under general anesthesia, the neoplasm was resected 1 month after detection. The lesion was removed at the stalk, leaving a clear boundary and the blood vessels of the nasal septum were coagulated (Figure 1D and E). The lesion occupied almost the entire olfactory clef. It had a smooth intact capsule, separating it from the middle turbinate, anterior ethmoid sinus, and anterior cranial fossa. Postoperatively, the anterior nostril was packed for 2 days. Histologically, it was reported to be a mucus cyst caused by chronic inflammation, and deemed a polyp (Figure 1F). Ten days postoperatively, endonasal endoscopy showed removal of the lesion and healing of the nasal septum mucosa. The patients’ s left headache disappeared and she felt she could breathe more easily through the left nostril, although she had not realized it was obstructed.
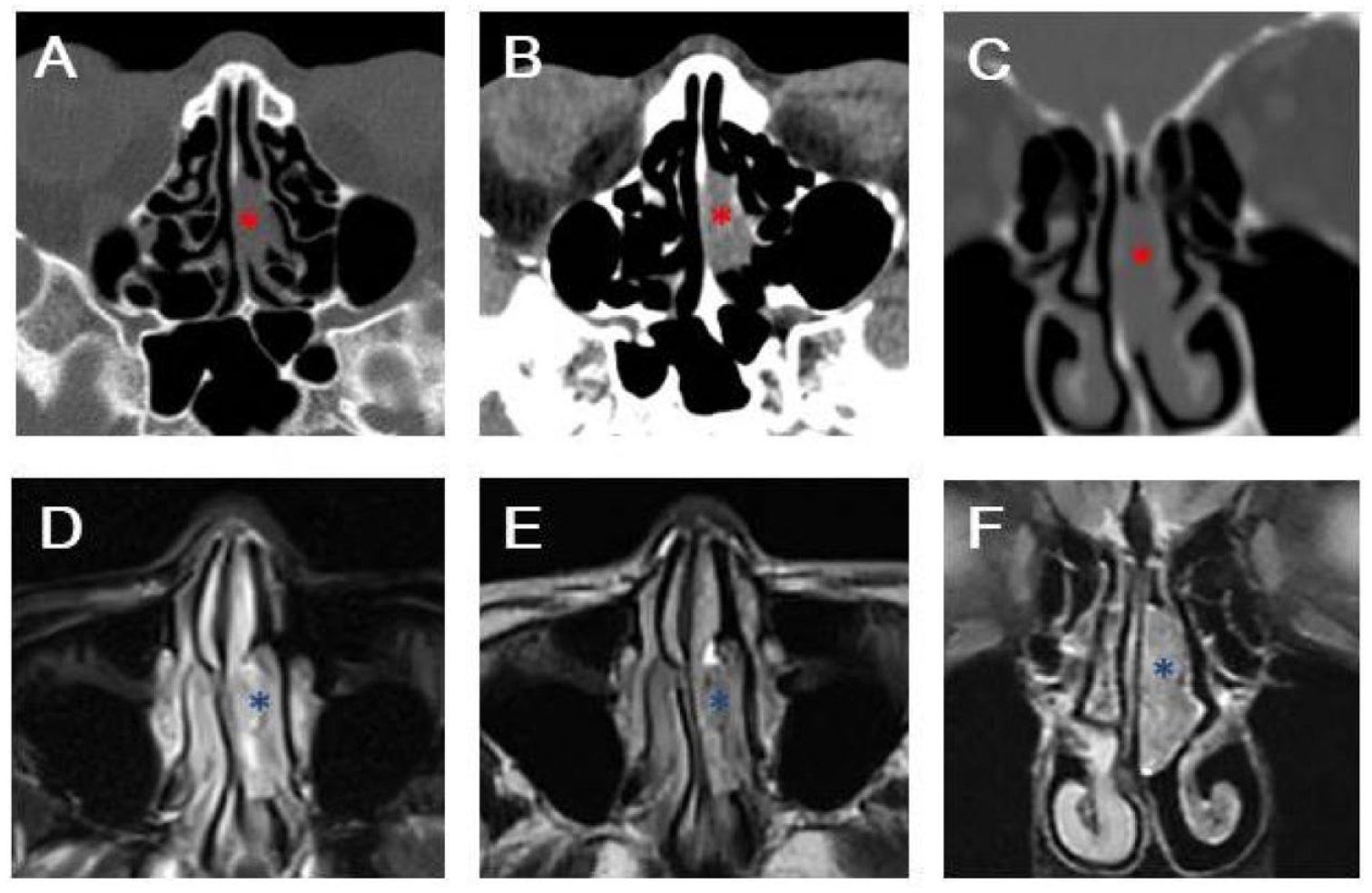
Imaging of the lesion originated from the left nasal septum. (A-C) CT of the nasal cavity and sinus. *The lesion in olfactory clef on the left. Axial images of the (A) bone and (B) soft tissue windows; (C) Coronal images of the lesion; (D-F) MRI of the nasal cavity and sinus. *The lesion. Long, uneven, and slightly shorter T1 and slightly longer T2 signal shadows can be seen in the middle nasal passage of the left nasal cavity. Flair imaging shows high signal intensity and diffusion weighted imaging (DWI) shows no obvious diffusion restriction. The lesion measures 9 mm × 23 mm × 22 mm and is adjacent to the nasal septum. The boundary with the middle turbinate is unclear, while the boundary with the ethmoid sinus and anterior cranial fossa is clear. (D) T1 session, (E) T2 session, (F) coronal T2 images. CT, computed tomography; MRI, magnetic resonance imaging.
Discussion
Nasal polyps are an inflammatory response to the local microenvironment. Their pathophysiology may involve autonomic nervous system dysfunction, abnormalities of carbohydrate metabolism, and vasomotor imbalance. Allergies and inflammation are major postulated pathogenic causes. Other hypothesized mechanisms include the adenoma and fibroma theory, an increase in tubule-alveolar glands, and structural defects such as nasal septum deviation, mucosal edema, mucosal inflammation, and epithelial rupture. The role of inflammatory mediators such as interleukins,1-3 transforming growth factor beta, basic fibroblast growth factor, leptin, and HPV-11 has been evaluated. 4 Bernstein proposed the multivariate theory for the pathogenesis of polyps. 5 Gravity and negative pressure are also thought to play a role in polyp growth. 4
Nasal polyps commonly arise from the lateral walls due to the increased exposure to air turbulence. Polyps originate in the anterior ethmoidal sinus mucosa and at contact points between the uncinate process and middle turbinate. The maxillary sinus and sphenoid sinus can also be involved. Nasal polyps rarely originate from the nasal septum and their pathogenesis remains unclear. 6 Few extending to the choana are ever reported.3,7-10 One inflammatory septal nasal polyp was reported in 2015. 11 Bleeding and fibroepithelial polyps from the nasal septum have also been reported.12-14
Nasal polyps can cause nasal obstruction, snoring, rhinorrhea, headache, anosmia, and nasal bleeding. Antrochoanal polyps are associated with allergic disease, including allergic rhinitis, bronchial asthma, and aspirin sensitivity syndrome. 15 The air stream can trigger inflammation due to mechanical pressure related to nasal septum deviation. 8 The airflow on the concave side becomes more substantial than that on the convex side.
Our patient denied a history of allergic disease or inflammation in the nose. She had no nasal obstruction, snoring, rhinorrhea, headache related to the nose, anosmia, or nasal bleeding. The only possible cause of this polyp was a nasal septum deviation to the right, and the polyp was on the concave side of the deviation. However, the patient did not have a stuffy nose because of the deviation, suggesting there was less increased airflow on the concave side of the deviation. Unlike septochoanal polyps rooted in the posterior septum, the polyp in this case arose from the anterosuperior part of the nasal septum and nearly filled the left olfactory clef. Ours is a truly unusual case of a large asymptomatic polyp originating from the nasal septum.
It remains possible that the polyp grew gradually and slowly, enabling the nose to adapt to the changes. It is unfortunate that we did not measure the airflow volume in this case. The relationship between the side of the septochoanal polyp and the direction of septal deviation is rarely discussed. A few septochoanal polyps that arose on the concave have been documented, and one on the convex side.16,17 The airflow effect is worth considering.
Interestingly, postoperatively the patient never experienced another headache that she had thought to be migraine. We believe that the patient had a rhinogenous headache without realizing it. In imaging, we observed contact points between the lateral nasal wall and the nasal septum polyp. There is evidence that patients have a higher incidence of headache when there are intermittent mucosal contact points between the septum and lateral nasal wall. 18
Footnotes
Data Availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant of National Natural Science Foundation of China (No.82205206), Science and Technology Innovation Project of the China Academy of Chinese Medicine Science (No.CI2021A02706), Project in Eye Hospital China Academy of Chinese Medical Science (No.202015/No.202026), Central High-Level Traditional Chinese Medicine Hospital Project of eye Hospital China Academy of Chinese Medical Science (No.GSP5-36/No.GSP5-80).
Ethics Statement
The studies involving human participants were reviewed and approved by the Committee of Medical Ethics of Eye Hospital China Academy of Chinese Medical Science.
