Abstract
Introduction
Advantages of microvascular free flap procedures include the transfer of a rich vascular supply that allows tissues to withstand subsequent radiotherapy and permits preservation of the osteogenic potential of transferred bone. Despite a high overall success rate, various postoperative complications for all types of free flaps have been described including wound infection, hematoma, facial paralysis, necrosis of soft tissue, fistula formation, skin defects, donor site morbidity, and vascular compromise leading to flap loss.1-3 Of these complications, vascular compromise and flap necrosis is especially feared as it can lead to unfavorable functional and cosmetic outcomes often requiring revision surgery. 2
As coronavirus disease 2019 (COVID-19), or severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, rose in prevalence, reports of related coagulation abnormalities increased.4,5 Coagulopathy in COVID-19 presents distinctly from other causes of coagulation abnormalities including bacterial sepsis-induced coagulopathy or disseminated intravascular coagulation and is associated with an increased risk of elevated fibrin degradation product levels, venous thromboembolism, complicating pulmonary embolism (PE), and death in severe cases.5,6 Furthermore, the excessive inflammatory response seen in patients with COVID-19 contributes to the elevation of pro-inflammatory cytokines, which may trigger necrosis and contribute to tissue and organ damage.7,8 These changes may contribute to complications such as wound dehiscence and hematoma formation. Given the reliance of free flap survival on adequate blood supply, it is imperative that coagulation defects and endothelial damage be prevented in patients undergoing this procedure.
Few studies have described free tissue transfers in patients positive for COVID-19. Nassar et al presented a case of lower extremity free tissue transfer failure in a previously healthy, asymptomatic patient who tested positive for COVID-19, 9 while Benmoussa et al reported a case of late postoperative flap failure and necrosis in a COVID-19-positive patient who received a fibula free flap. 10 COVID-19 testing guidelines regarding surgery vary by institution, and many guidelines now recommend against routine preoperative COVID-19 testing in asymptomatic patients. 11
Current data regarding the treatment and prevention of COVID-19 associated coagulopathy are limited, and no consensus exists on whether free flap procedures should be delayed in COVID-19 patients. Furthermore, very few studies exist on the long-term effects of COVID-19 infection, especially in the head and neck cancer population and on free flap outcomes. It is possible that lingering effects such as inflammation and coagulopathy may influence outcomes in this population. The aim of this study was to determine whether patient complications differ by COVID-19 infection history among patients undergoing any kind of free flap reconstruction. To our knowledge, this report outlines the largest analysis, to date, of free flap complications associated with COVID-19 infection status.
Patients and Methods
This retrospective review study utilized the TriNetX Research Network (Cambridge, MA). Initial data were collected on August 27, 2023, during which the TriNetX database contained electronic medical records from over 110 million individuals and 78 health care organizations. The TriNetX database Research Network includes 120,829,986 patients as a whole and the database uses data from 20 years prior when running analyses (August 27, 2003, to March 5, 2024). An additional analysis was conducted on March 5, 2024, comparing outcomes in patients who had a COVID-19 infection within 2 weeks of surgery. The TriNetX Research Network provides users with uniformly processed clinical data, including demographics, diagnoses, imaging, laboratory results, medications, procedures, and other interventions. TriNetX is compliant with the Health Insurance Portability and Accountability Act (HIPAA). Due to TriNetX containing de-identified patient information, the Penn State Institutional Review Board reviewed and approved STUDY00018629 as exempt and informed consent was not required.
The database was queried using diagnosis [International Classification of Diseases-10 (ICD-10)] and procedure (Current Procedural Terminology) codes to identify patients between 18 and 90 years of age with a history of microvascular surgery between 2013 and 2023. History of microvascular surgery was defined by ICD-10 codes listed in Appendix A. A cohort was created including patients who had free flap surgery and a diagnosis of COVID-19 within a year of surgery. The control group included patients who underwent free flap surgery with no history of COVID-19 infection. COVID-19 diagnosis codes used to create the cohorts can be observed in Appendix A. For additional timing analysis, cohorts were created for 1 group that had surgery within 2 months of COVID-19 infection, and a second cohort with COVID-19 infection between 2 months to 1 year prior to surgery. All patients who had free flaps with and without COVID-19 were included. Patients over the age of 90 were excluded, as well as patients who satisfied the inclusion criteria more than 20 years ago.
Using the Compare Outcomes tool within the TriNetX database, the incidence of postoperative complications within 3 months of surgery was determined. Postoperative complications assessed included sepsis, deep vein thrombosis (DVT), stroke, acute respiratory distress syndrome, PE, surgical site infection (SSI), generalized infection, flap failure, gangrene, dehiscence, hematoma, seroma, readmission or subsequent admission to the intensive care unit (ICU), and death. Specific ICD-10 codes for these postoperative complications can be seen in Appendix A. These methods closely mimic the recently published study by Taghioff et al assessing the influence of the COVID-19 vaccination on microsurgical complications. 12
To balance potential confounding variables, additional analyses were conducted after matching the cohorts according to demographics (age, sex, race, and ethnicity) as well as preoperative comorbidities, including tobacco use (Z72.0), hypertension (I10-I16), chronic ischemic heart disease (I25), diabetes (E08-E13), overweight/obesity (E65-E68), and chronic obstructive pulmonary disease (J44).
TriNetX utilizes matrices of covariates to obtain propensity scores via logistic regression. Matching was conducted via propensity score, nearest neighbor matching, with a caliper of 0.1 times the standard deviation. Statistical significance was defined as P < .05. Statistical analyses were performed within the TriNetX platform. Relative risks and associated 95% confidence intervals (CIs) were used to calculate the risk of postoperative complication in patients with microvascular free flap surgery with and without a history of COVID-19 infection.
Results
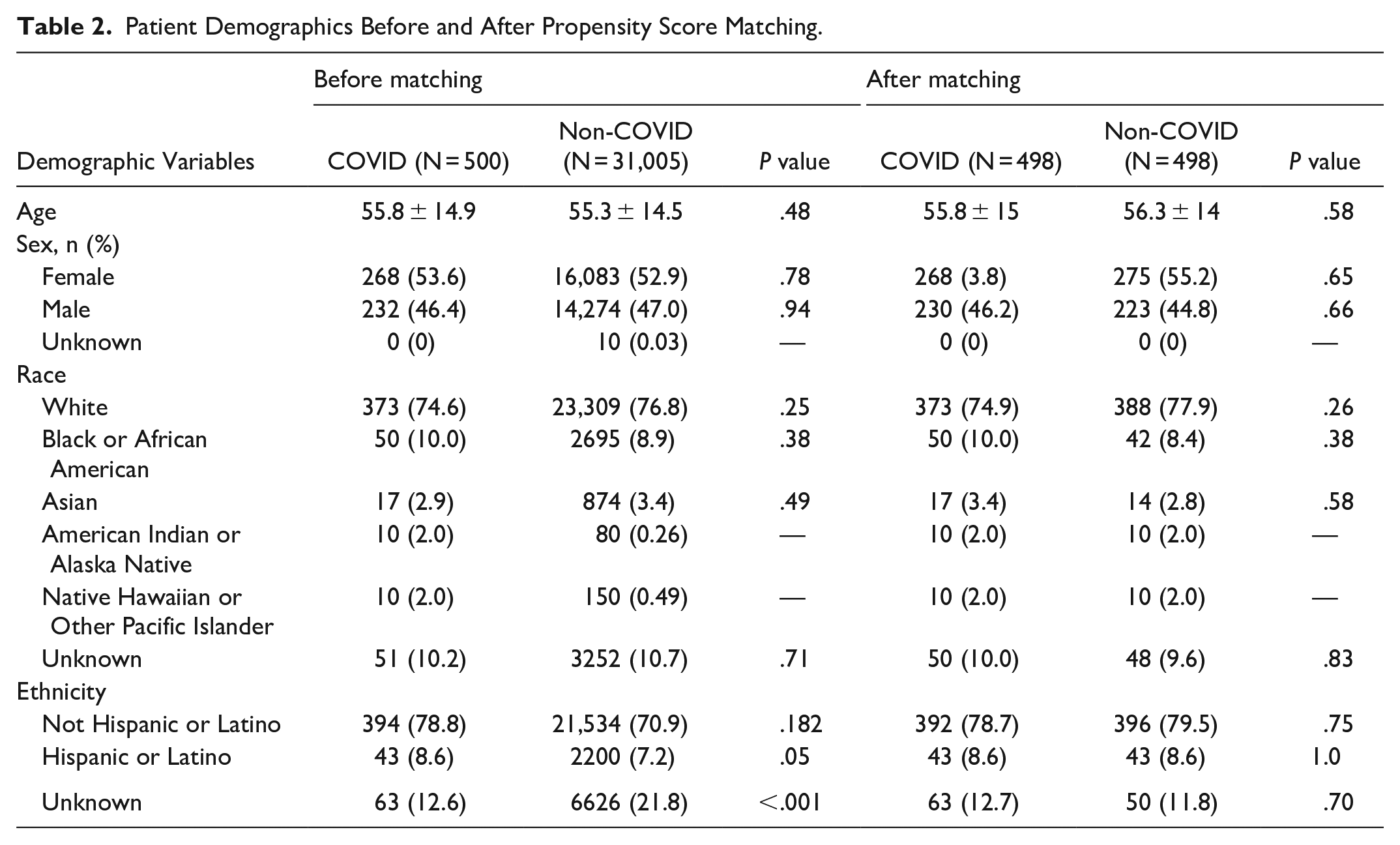
There was a total of 31,505 patients who underwent microvascular free flap surgery from 2013 to 2023. A total of 500 patients had a diagnosis of COVID-19 within 1 year of free flap surgery, 183 patients had a diagnosis of COVID-19 within 2 weeks of free flap surgery (Table 1), and 31,005 patients had no history of a COVID-19 diagnosis before surgery. The majority of patients were female, white, and non-Hispanic or Latino. All demographic differences between cohorts were eliminated after propensity score matching. For outcomes or variables experienced by 1 to 9 patients, the TriNetX database rounds to 10. Therefore, analyses were not completed for all variables with 10 patients. Demographic variables can be observed in Table 2.
Differences in Complications in Patients With a COVID Infection Within 2 Weeks Before Surgery, After Propensity Score Matching.
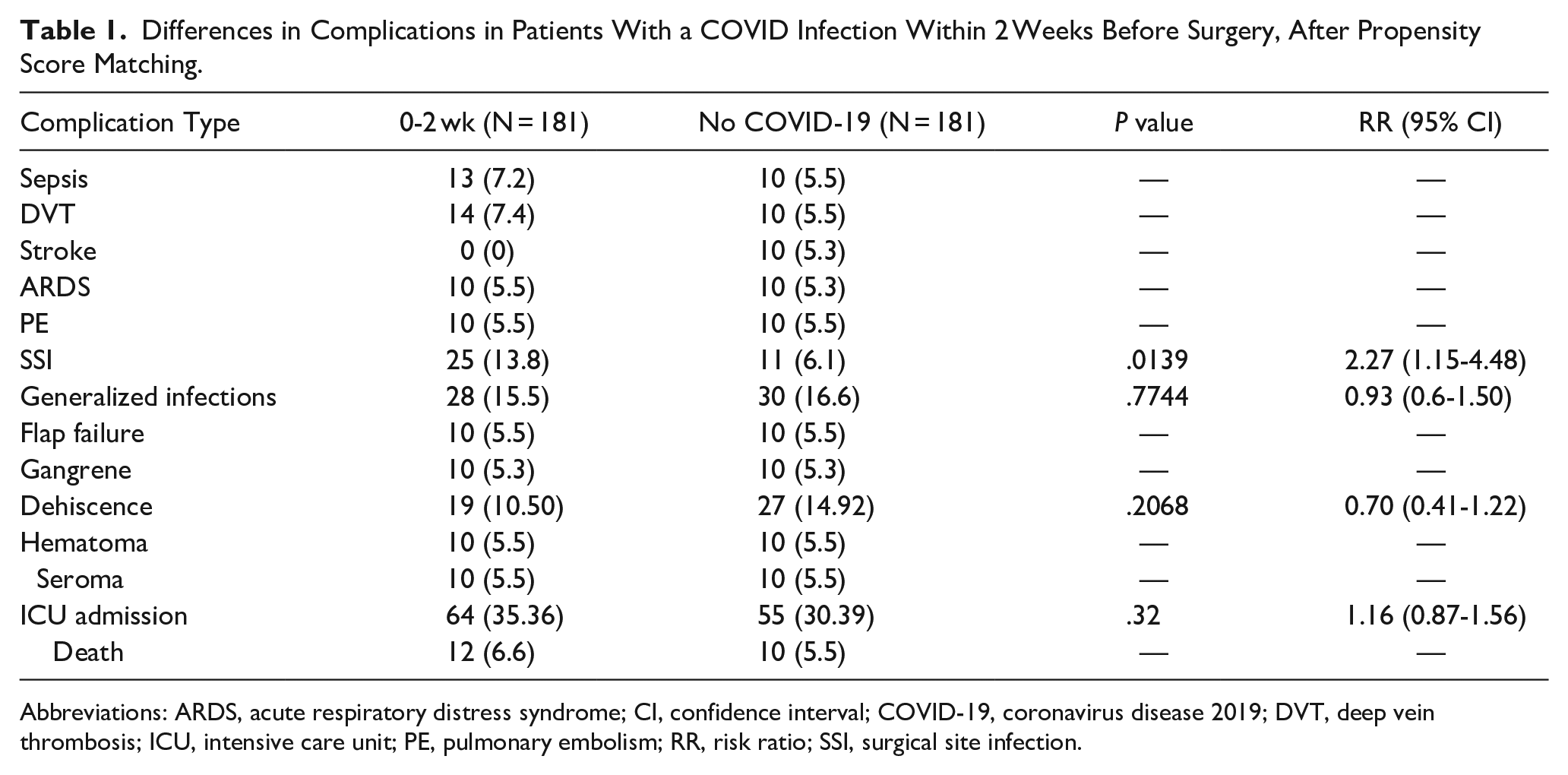
Abbreviations: ARDS, acute respiratory distress syndrome; CI, confidence interval; COVID-19, coronavirus disease 2019; DVT, deep vein thrombosis; ICU, intensive care unit; PE, pulmonary embolism; RR, risk ratio; SSI, surgical site infection.
Patient Demographics Before and After Propensity Score Matching.
Patients who had a COVID-19 infection within 1 year of surgery had a significantly increased risk of sepsis [risk ratio (RR) 1.93, 95% CI 1.33-2.80, P < .001), SSI (RR 2.61, 95% CI 2.03-3.35, P < .001), generalized infection (RR 1.32, 95% CI 1.06-1.63, P = .0130), gangrene (RR 2.22, 95% CI 1.49-3.30, P < .001), dehiscence (RR 1.40, 95% CI 1.11-1.78, P = .0056), hematoma (RR 1.8, 95% CI 1.0-2.26, P = .0498), seroma (RR 2.13, 95% CI 1.21-3.76, P = .0080), ICU admission (RR 1.51, 95% CI 1.30-1.74, P < .001), and death (RR 1.77, 95% CI 1.12-2.80, P = .0146). After propensity score matching for comorbidities and demographic differences, significant differences remained in SSI (RR 2.59, 95% CI 1.61-4.17, P < .001) and gangrene (RR 2.0, 95% CI 1.01-3.96, P = .0416). Postoperative complications can be seen in Table 3. There were no significant differences in DVT, stroke or flap failure between the 2 cohorts before and after matching. When assessing timing of surgical procedures, there was an increased risk of SSI (RR 2.27, 95% CI 1.15-4.48, P = .0139) in patients who had a COVID-19 infection within 2 weeks of surgery. There were no differences in complications when waiting more than 2 months to perform surgery compared to performing surgery less than 2 months after a documented COVID-19 infection (Table 4).
Postoperative Complications in Patients With and Without COVID Before and After Propensity Score Matching.
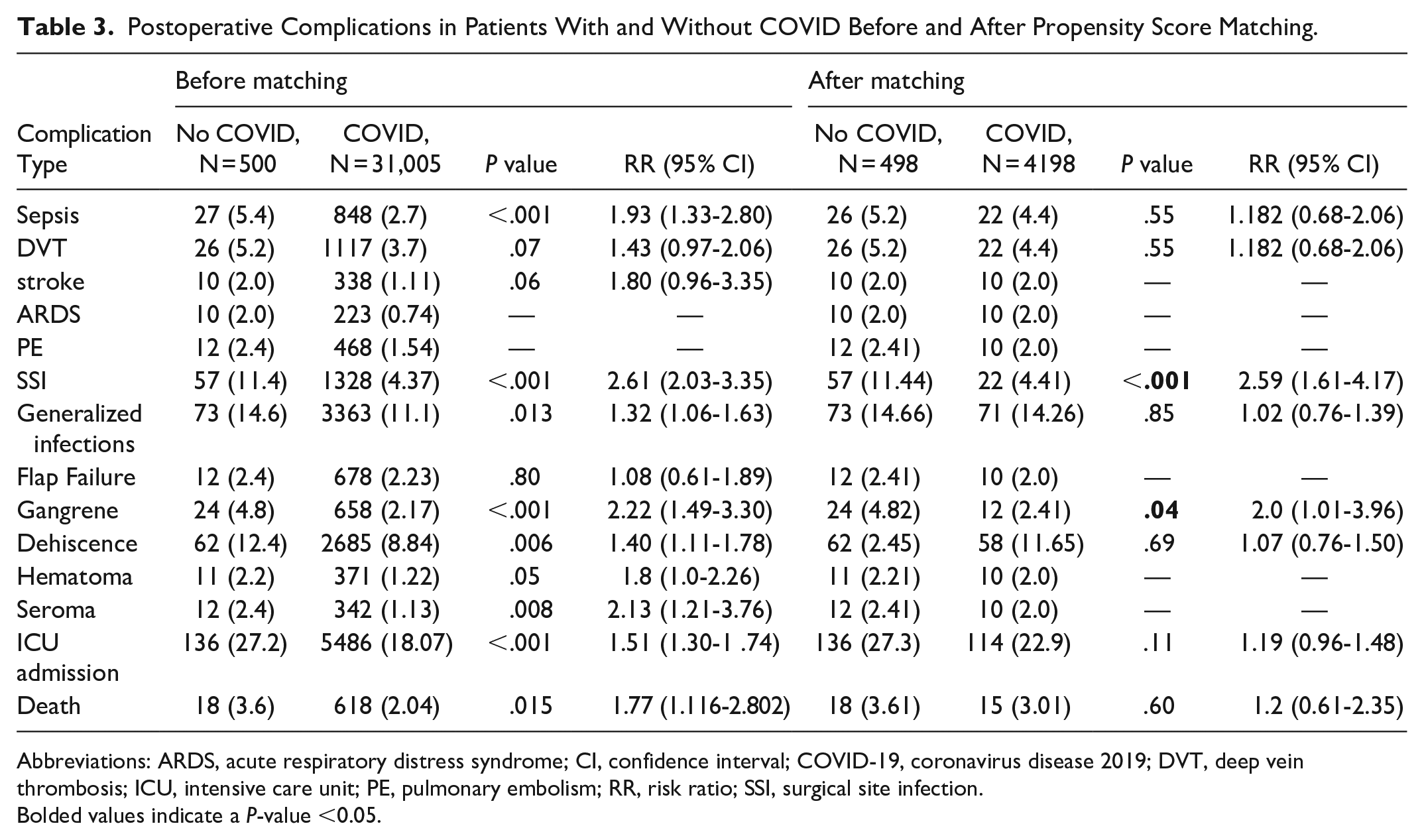
Abbreviations: ARDS, acute respiratory distress syndrome; CI, confidence interval; COVID-19, coronavirus disease 2019; DVT, deep vein thrombosis; ICU, intensive care unit; PE, pulmonary embolism; RR, risk ratio; SSI, surgical site infection.
Bolded values indicate a P-value <0.05.
Differences in Complications Based on Timing of Surgery After COVID Infection After Propensity Score Matching.
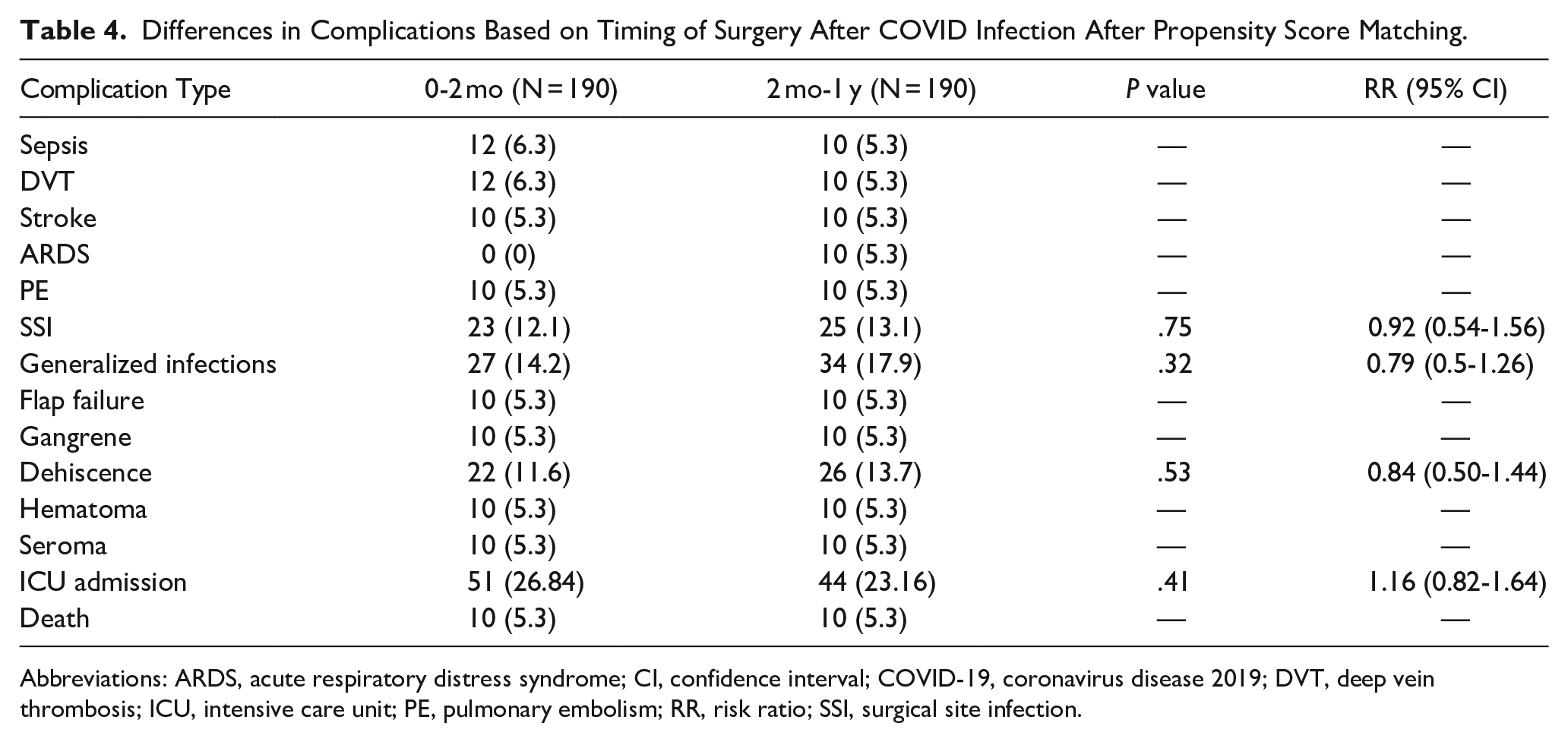
Abbreviations: ARDS, acute respiratory distress syndrome; CI, confidence interval; COVID-19, coronavirus disease 2019; DVT, deep vein thrombosis; ICU, intensive care unit; PE, pulmonary embolism; RR, risk ratio; SSI, surgical site infection.
Discussion
As the COVID-19 pandemic swept over the nation, there were everchanging guidelines for COVID-19 testing prior to surgical procedures. Nowadays, many institutions no longer conduct COVID-19 testing prior to surgery. A previous study showed beneficial effects of the COVID-19 vaccine in decreasing patient complications after free flap surgery. However, to date, there have been no studies that show the impact of a history of COVID-19 infection on free flap complication rates. 10 Therefore, this study aimed to identify the risks of complications in patients with a history of COVID-19 infection who underwent free flap surgery.
Initially, the current study determined there was an increased risk of sepsis, SSI, generalized infection, gangrene, dehiscence, hematoma, seroma, ICU admission, and death in patients who underwent free flap surgery within a year of COVID-19 infection. However, many differences between groups were eliminated after controlling for demographic and presurgical comorbidities, with significant risks only remaining for SSI and gangrene. These results demonstrate that COVID-19 infection potentially plays a role in increasing future perioperative infections and perfusion. Notably, these findings may be due to patients having COVID-19 at the time of surgery or potential secondary infections. 13 A systematic review done recently found that as many as 19% of patients had a coinfection and 24% had a superinfection with other pathogens in addition to COVID-19 at the time of diagnosis. While patients in this study were diagnosed with COVID-19 weeks to months prior to surgery, the existence of a persistent superinfection or coinfection may have contributed to postoperative complications. Furthermore, the study reported poor outcomes such as mortality in patients with either coinfection or superinfection. 14 COVID-19-related coagulopathy has also been reported,5,9,10,15,16 with unique features such as increased inflammation and endothelial damage compared to other etiologies of coagulopathy. The resulting increase in inflammatory mediators, endothelial damage, and disruption of normal immune system functioning may help explain the increased rates of SSI and gangrene found in these patients.
Controversies exist in the ideal timing of when to perform surgery after COVID-19 infection. The current study found no differences between performing surgery within 2 months of a COVID-19 infection compared to waiting more than 2 months after infection. Furthermore, we found that patients who had a COVID-19 infection within 2 weeks of surgery were more likely to develop an SSI. A previous study demonstrated a recent COVID-19 infection, regardless of timing within the previous 60 days, was not associated with risk of adverse postoperative outcomes. 17 However, a different study demonstrated an increased 90 day mortality in patients who underwent surgery within 8 weeks after a positive COVID-19 test result. 18 In addition, another study found postoperative mortality was approximately 6 times higher for patients who had a COVID-19 infection within 2 weeks before surgery. 19 It is important to note that these prior studies included all types of surgeries and did not specifically investigate microvascular surgeries. Our results suggest that rates of SSI may be higher in patients who recently had COVID-19 and that this population should be monitored closely.
Many flap surgeries cannot be delayed due to the etiology of the defect, such as for oncologic resection or traumatic injury. This study may help counsel patients regarding the possible complications after surgery and provide a heightened awareness in the surgical team of a possible increase in infectious complications in this population. Several strategies have been reported for preventing and treating SSI, including empiric antibiotic administration and antiseptic skin preparation preoperatively, wound irrigation intraoperatively, and early antibiotic treatment postoperatively.20-24 However, there is a lack of consensus on objective measures and treatment guidelines regarding this issue. 25 Surgeons may consider employing one or all of these measures to prevent SSI in patients with a history of prior or concurrent COVID-19 infection receiving free flap surgery, especially if the COVID-19 infection was detected within 2 weeks of surgery. Several studies have examined the role of anticoagulants in COVID-19,26,27 and surgeons may consider using these medications to prevent or treat coagulopathy in these patients.
During the pandemic, there was a significant decrease in microvascular free flap surgeries performed overall. Interestingly, there was a shift from breast reconstruction to head and neck reconstruction becoming the most common microvascular free flap surgery performed. 28 Some studies also reported a shift to locoregional flaps rather than free flaps due to their relatively lower costs, shorter operative times, and potentially shorter ICU stays compared to free flaps.29,30 Prior case reports demonstrate flap failure in patients who tested positive at the time of microvascular free flap surgery.9,10 Researchers hypothesize that this may be due to COVID-19-related coagulopathy or related conditions such as superinfection or coinfection with other pathogens. 10 To our knowledge, we report the first multicenter retrospective review of microsurgical free flap outcomes in patients with a history of COVID-19 infection. Therefore, this study assists in understanding the longer-term impact of COVID-19 on surgical infections.
In conjunction with prior studies demonstrating decreased risks of free flap complications in patients with COVID-19 vaccination, 12 this study further emphasizes that preventing infection of COVID-19 could have benefits for optimal postsurgical outcomes. However, this study was limited in that results are dependent on clinician diagnosis and accurate documentation due to the retrospective nature. This study would also have benefitted from data on superinfection or concurrent infections, as well as vaccination status, but we were limited by the variables collected by the TriNetX database. Another limitation is that our analysis included all patients with a positive viral infection, but we do not know if this is an asymptomatic positive test due to exposure or if the patient was symptomatic. Because the information is de-identified, an increased investigation of patient charts was not available. In addition, the database rounds to 10 for outcomes or variables experienced by 1 to 9 patients. Therefore, we could not make any conclusions for outcomes that resulted in 10 patients. However, we believe that utilizing this large database provides several advantages due to the size and ability to balance cohorts.
Future studies should investigate optimal timing after COVID-19 infection to perform free flap surgery and determine ways to mitigate potential increased risk of infectious complications such as providing empiric antibiotics to prevent coinfection or superinfection, leveraging vaccination status, or using anticoagulants such as heparin to prevent coagulopathy. A more thorough comparison of locoregional flaps to free flaps in this population may also be beneficial.
Conclusions
This study examines the impact of having a COVID-19 infection within 1 year of microsurgery on postoperative outcomes and complications. COVID-19 infection put patients at an increased risk of SSIs and generalized infections, even after propensity score matching. Of note, specific flap-related complications such as flap failure, gangrene, and dehiscence did not differ in incidence based on a history of COVID-19. Awareness of these data may prepare surgeons and staff for the postoperative patient course.
Footnotes
Appendix
Codes Used Within the Database to Identify Patients, Diagnoses, and Procedures.
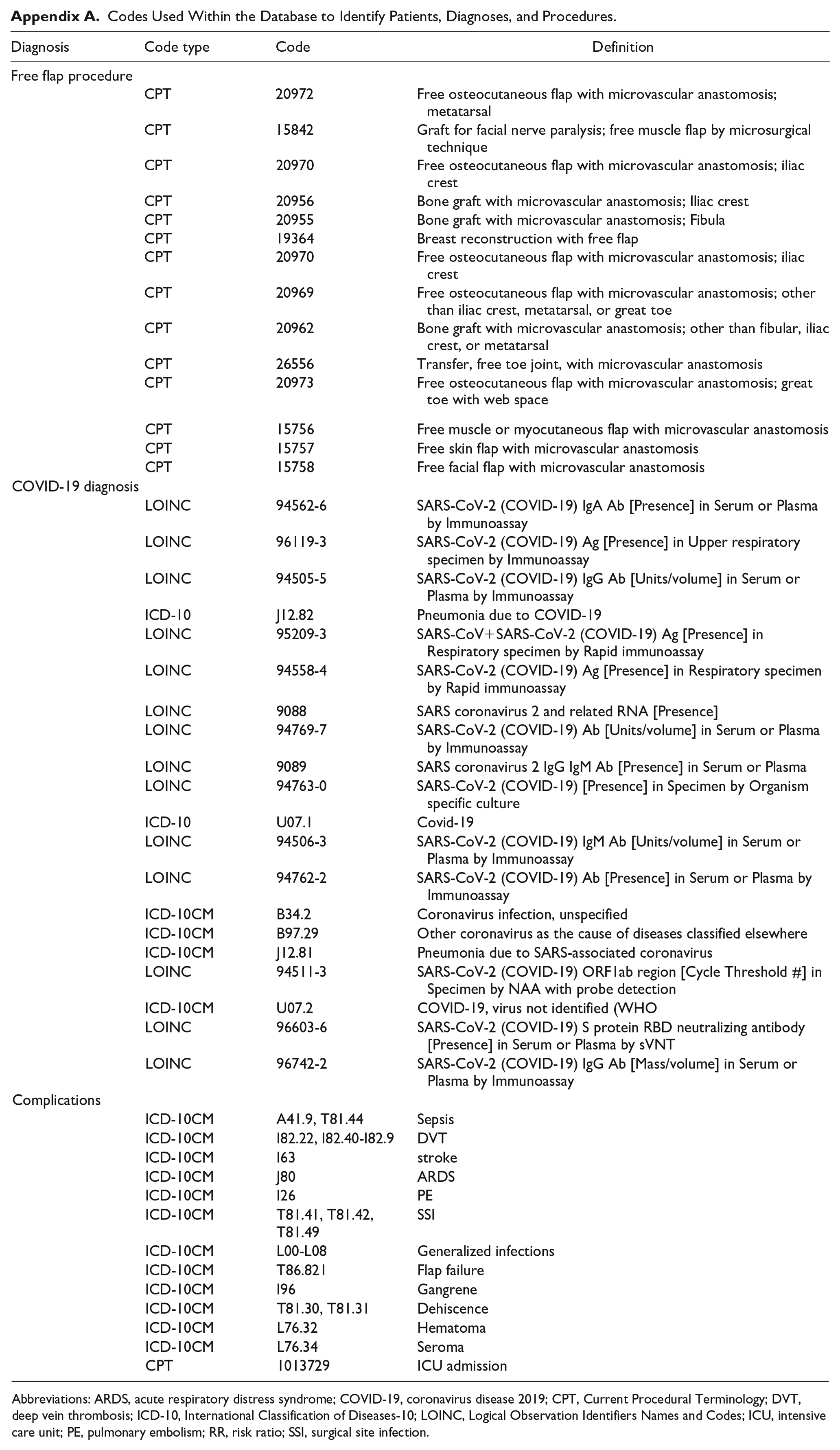
| Diagnosis | Code type | Code | Definition |
|---|---|---|---|
| Free flap procedure | |||
| CPT | 20972 | Free osteocutaneous flap with microvascular anastomosis; metatarsal | |
| CPT | 15842 | Graft for facial nerve paralysis; free muscle flap by microsurgical technique | |
| CPT | 20970 | Free osteocutaneous flap with microvascular anastomosis; iliac crest | |
| CPT | 20956 | Bone graft with microvascular anastomosis; Iliac crest | |
| CPT | 20955 | Bone graft with microvascular anastomosis; Fibula | |
| CPT | 19364 | Breast reconstruction with free flap | |
| CPT | 20970 | Free osteocutaneous flap with microvascular anastomosis; iliac crest | |
| CPT | 20969 | Free osteocutaneous flap with microvascular anastomosis; other than iliac crest, metatarsal, or great toe | |
| CPT | 20962 | Bone graft with microvascular anastomosis; other than fibular, iliac crest, or metatarsal | |
| CPT | 26556 | Transfer, free toe joint, with microvascular anastomosis | |
| CPT | 20973 | Free osteocutaneous flap with microvascular anastomosis; great toe with web space | |
| CPT | 15756 | Free muscle or myocutaneous flap with microvascular anastomosis | |
| CPT | 15757 | Free skin flap with microvascular anastomosis | |
| CPT | 15758 | Free facial flap with microvascular anastomosis | |
| COVID-19 diagnosis | |||
| LOINC | 94562-6 | SARS-CoV-2 (COVID-19) IgA Ab [Presence] in Serum or Plasma by Immunoassay | |
| LOINC | 96119-3 | SARS-CoV-2 (COVID-19) Ag [Presence] in Upper respiratory specimen by Immunoassay | |
| LOINC | 94505-5 | SARS-CoV-2 (COVID-19) IgG Ab [Units/volume] in Serum or Plasma by Immunoassay | |
| ICD-10 | J12.82 | Pneumonia due to COVID-19 | |
| LOINC | 95209-3 | SARS-CoV+SARS-CoV-2 (COVID-19) Ag [Presence] in Respiratory specimen by Rapid immunoassay | |
| LOINC | 94558-4 | SARS-CoV-2 (COVID-19) Ag [Presence] in Respiratory specimen by Rapid immunoassay | |
| LOINC | 9088 | SARS coronavirus 2 and related RNA [Presence] | |
| LOINC | 94769-7 | SARS-CoV-2 (COVID-19) Ab [Units/volume] in Serum or Plasma by Immunoassay | |
| LOINC | 9089 | SARS coronavirus 2 IgG IgM Ab [Presence] in Serum or Plasma | |
| LOINC | 94763-0 | SARS-CoV-2 (COVID-19) [Presence] in Specimen by Organism specific culture | |
| ICD-10 | U07.1 | Covid-19 | |
| LOINC | 94506-3 | SARS-CoV-2 (COVID-19) IgM Ab [Units/volume] in Serum or Plasma by Immunoassay | |
| LOINC | 94762-2 | SARS-CoV-2 (COVID-19) Ab [Presence] in Serum or Plasma by Immunoassay | |
| ICD-10CM | B34.2 | Coronavirus infection, unspecified | |
| ICD-10CM | B97.29 | Other coronavirus as the cause of diseases classified elsewhere | |
| ICD-10CM | J12.81 | Pneumonia due to SARS-associated coronavirus | |
| LOINC | 94511-3 | SARS-CoV-2 (COVID-19) ORF1ab region [Cycle Threshold #] in Specimen by NAA with probe detection | |
| ICD-10CM | U07.2 | COVID-19, virus not identified (WHO | |
| LOINC | 96603-6 | SARS-CoV-2 (COVID-19) S protein RBD neutralizing antibody [Presence] in Serum or Plasma by sVNT | |
| LOINC | 96742-2 | SARS-CoV-2 (COVID-19) IgG Ab [Mass/volume] in Serum or Plasma by Immunoassay | |
| Complications | |||
| ICD-10CM | A41.9, T81.44 | Sepsis | |
| ICD-10CM | I82.22, I82.40-I82.9 | DVT | |
| ICD-10CM | I63 | stroke | |
| ICD-10CM | J80 | ARDS | |
| ICD-10CM | I26 | PE | |
| ICD-10CM | T81.41, T81.42, T81.49 | SSI | |
| ICD-10CM | L00-L08 | Generalized infections | |
| ICD-10CM | T86.821 | Flap failure | |
| ICD-10CM | I96 | Gangrene | |
| ICD-10CM | T81.30, T81.31 | Dehiscence | |
| ICD-10CM | L76.32 | Hematoma | |
| ICD-10CM | L76.34 | Seroma | |
| CPT | 1013729 | ICU admission | |
Abbreviations: ARDS, acute respiratory distress syndrome; COVID-19, coronavirus disease 2019; CPT, Current Procedural Terminology; DVT, deep vein thrombosis; ICD-10, International Classification of Diseases-10; LOINC, Logical Observation Identifiers Names and Codes; ICU, intensive care unit; PE, pulmonary embolism; RR, risk ratio; SSI, surgical site infection.
Ethics
This study received ethical approval from The Pennsylvania State University IRB (Approval #00018629) on September 10, 2021. This is an IRB-approved retrospective study, all patient information was deidentified and patient consent was not required.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project described was supported by the National Center for Advancing Translational Sciences, National Institutes of Health, through Grant UL1 TR002014. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
