Abstract
Introduction
Medullary thyroid carcinoma (MTC) is a relatively uncommon malignancy arising from parafollicular C-cells that accounts for 0.6% of all thyroid cancers in Korea 1 and 1% to 2% in the United States. 2 While the incidence of MTC is low, its propensity for invasion and metastasis is significant, resulting in a relatively poor prognosis. MTCs manifest locoregional metastasis in 50% to 75% of patients and distant metastasis in 10% to 15% of patients at the time of initial diagnosis. The 5 and 10 year disease-specific survival rates stand at 83% to 89% and 56% to 87%, respectively.3-5 Early detection of risk factors associated with aggressive behaviors hold significant clinical importance in formulating appropriate surgical proposals for surgeons. Aggressive behaviors of MTCs encompass perithyroidal invasion, lymphatic metastases, distant metastases, and recurrence.
Most guidelines recommend that curative therapy for MTCs is primary tumor excision with central lymph node (CLN) dissection.2,6,7 Whether prophylactic lateral cervical lymph node (LCLN) dissection for lymph node negative patient remains controversial.2,8,9 The extent of initial surgery for MTCs is closely related to prognosis of patients, but it is difficult for clinicians to determine whether prophylactic LCLN dissection is required or not. Therefore, detecting preoperative risk factors for LCLN metastasis of MTCs is also very crucial.
Ultrasound examination is a pivotal imaging tool in early detection and staging of MTCs before surgery as well as in follow-up after surgery. However, most studies focused on ultrasonic features of invasive papillary thyroid carcinomas (PTCs). Limited research is available regarding ultrasonic risk factors of aggressive behaviors of MTCs, and results are inconsistent.8,10,11
This study aimed to identify clinical and sonographic predictors for aggressive behaviors in MTCs, including metastasis (both lymph nodular metastasis and distant metastasis), perithyroidal invasion, and adverse outcomes (recurrence/death).
Materials and Methods
Clinical and Pathological Information
This retrospective study was approved by the Institutional Review Board of Affiliated Hospital of Qingdao University (Ethics No. QYFY WZLL 27819), and the need for informed consent was waived. All patients were confirmed as MTCs by operation and histopathology examination according to World Health Organization classification criteria 12 between January 2009 and May 2022 in our hospital. Patients suspected of hereditary MTC were excluded, including those with multiple endocrine neoplasia (MEN) 2, patients with a family history of MTC, and patients who tested positive for RET gene mutation. All patients underwent preoperative ultrasonic examination to find suspicious thyroid nodules and/or cervical lymph nodes. Tumor staging was determined based on the tumor node metastasis (TNM) classification. 13
Clinical data of patient were extracted by one radiologist from our picture archiving and communication system (PACS) systems, including sex, age, date, and details of the operation procedure, distant metastases (present/absent). Postoperative parameters, such as focality of MTCs (unifocal/multifocal), lymphatic invasion (present/absent, and location of the invasive lymph node), and extrathyroidal extension (present/absent) were also recorded. Patient outcomes (including normal, recurrence, and death) were obtained through telephone follow-up and outpatient follow-up. Recurrence or death indicated the end of follow-up. Follow-up time was calculated in days: from the date when nodules were first detected until the date of last follow-up or recurrence/death. In addition, to identify risk factors for predicting LCLN metastasis, we categorized patients with LCLN metastasis at the initial thyroid surgery as the N1b group, and patients with normal calcitonin levels without any lymphatic metastasis or recurrence during follow-up after the initial surgery as the N0-NED group.
Ultrasonic Features of MTCs
Ultrasonic images and video clips of thyroid nodules, cervical lymph nodes, and perithyroidal tissue were extracted and retrospectively reviewed by 2 thyroid radiologists—a junior with 7 years of experience and an expert with 20 years of experience—from our PACS systems. Size (the maximum diameter in transverse or longitudinal view on ultrasound images), composition (cystic/almost complete cystic, spongiform, mixed cystic and solid, solid or almost complete solid), echogenicity (anechoic, hyperechoic or isoechoic, hypoechoic, very hypoechoic), shape (wider-than-tall, taller-than-wide), margin (smooth/ill-defined, lobulated or irregular, extrathyroidal extension), and calcification (none or large comet-tail artifacts, macrocalcifications, peripheral calcifications, microcalcifications) were evaluated according to ACR thyroid imaging reporting and data system (TI-RADS). 14 This system stratifies nodules into TR1 (benign) to TR5 (high suspicion of malignancy) in accordance with the sum of the scores. Besides, tumor location (upper pole, nonupper pole) and association with capsule (subcapsular, nonsubcapsular) was also recorded. Upper pole was identified as the upper third of thyroid lobe along in longitudinal view. 8 A subcapsular location was designated when the nodule was adjacent to thyroid capsule without any intervening parenchyma. 15 In case of multifocal MTCs, only the most prominent or suspicious nodule was analyzed in our study. Results from the expert were included in final statistical analysis. Both radiologists were blinded to clinical and pathological information.
Statistical Analysis
SPSS 18.0 (SPSS, Inc) was used to analyze the data. Continuous variables were presented as mean ± standard diversion and categorical variables were presented as number with percentage. Interobserver agreement was assessed by kappa test. The kappa values were interpreted as follows: 0 to 0.2, slight agreement; 0.21 to 0.40, fair agreement; 0.41 to 0.60, moderate agreement; 0.61 to 0.80, substantial agreement; and 0.81 to 1.0, perfect agreement. Comparisons between groups were performed by student’s t test for parametric variables. Mann-Whitney U test for nonparametric variables, and Chi-square or fisher’s exact tests for frequencies, respectively. Receiver operating characteristic (ROC) curve analysis was performed to determine the optimal cutoff points for tumor size. Binary logistic regression was carried to screen risk factors. Odds ratios (ORs) and 95% confidence intervals were calculated for each variable. Log-rank test was applied to compare the difference in Kaplan-Meier (K-M) curves in postoperative disease-free survival (PDFS). P < .05 was considered as having statistically significant difference. In addition, we adopted forest map drawn by GraphPad Prism version 8.0 (GraphPad Software, Inc) to show results of multivariable analysis.
Results
Clinicopathologic Features of the Study Subjects
The initial cohort consisted of 141 patients with histologically confirmed MTC. Subsequently, 2 patients with MEN 2 and 10 patients with familial MTC were excluded from the study. Among the remaining 129 patients, voluntary RET gene mutation analysis was conducted in 52 individuals. Nine patients tested positive and were subsequently excluded. Ultimately, a total of 120 patients (59 males and 61 females) were enrolled in the study. The mean age of the cohort was 51.5 ± 12.5 years old (range, 14-76 years old). The general features were summarized in Table 1. Among the patients, 62 had lymphatic metastases, with 48 presenting both CLN and LCLN metastases. Perithyroidal invasion was identified in 27 patients. Before surgery, 4 patients were found to have distant metastasis, including 3 with lung metastases and 1 with metastases to the liver, bone, and adrenal glands. The mean time and interquartile range of follow-up was 1158 and 1588 days respectively, with a follow-up time range 134 to 3865 days in the entire cohort. During postoperative follow-up, another 5 patients were found to develop distant metastases, including 1 with liver metastases, 1 with mediastinal metastases, 1 with mediastinal and thymus metastasis, 1 with mediastinal and lung metastasis, and 1 with bone metastasis.
Clinical and Sonographic Characteristics of the Study Subjects.

Abbreviations: CLN, central lymph node; IQR, inter-quartile range; LCLN, lateral cervical lymph node; ST, subtotal thyroidectomy; TT, total thyroidectomy.
Preoperative Ultrasonic Features of MTCs
Preoperative ultrasonic features were classified according to TI-RADS criteria (Table 2). The kappa value for interobserver agreement of each ultrasonic feature showed high consistency (range from 0.811 to 0.941). Most nodules were classified TR4 and TR5 (25.8% and 72.5%, respectively), and none of the nodules was classified as TR1.
Ultrasonic Features of the Study Subjects.
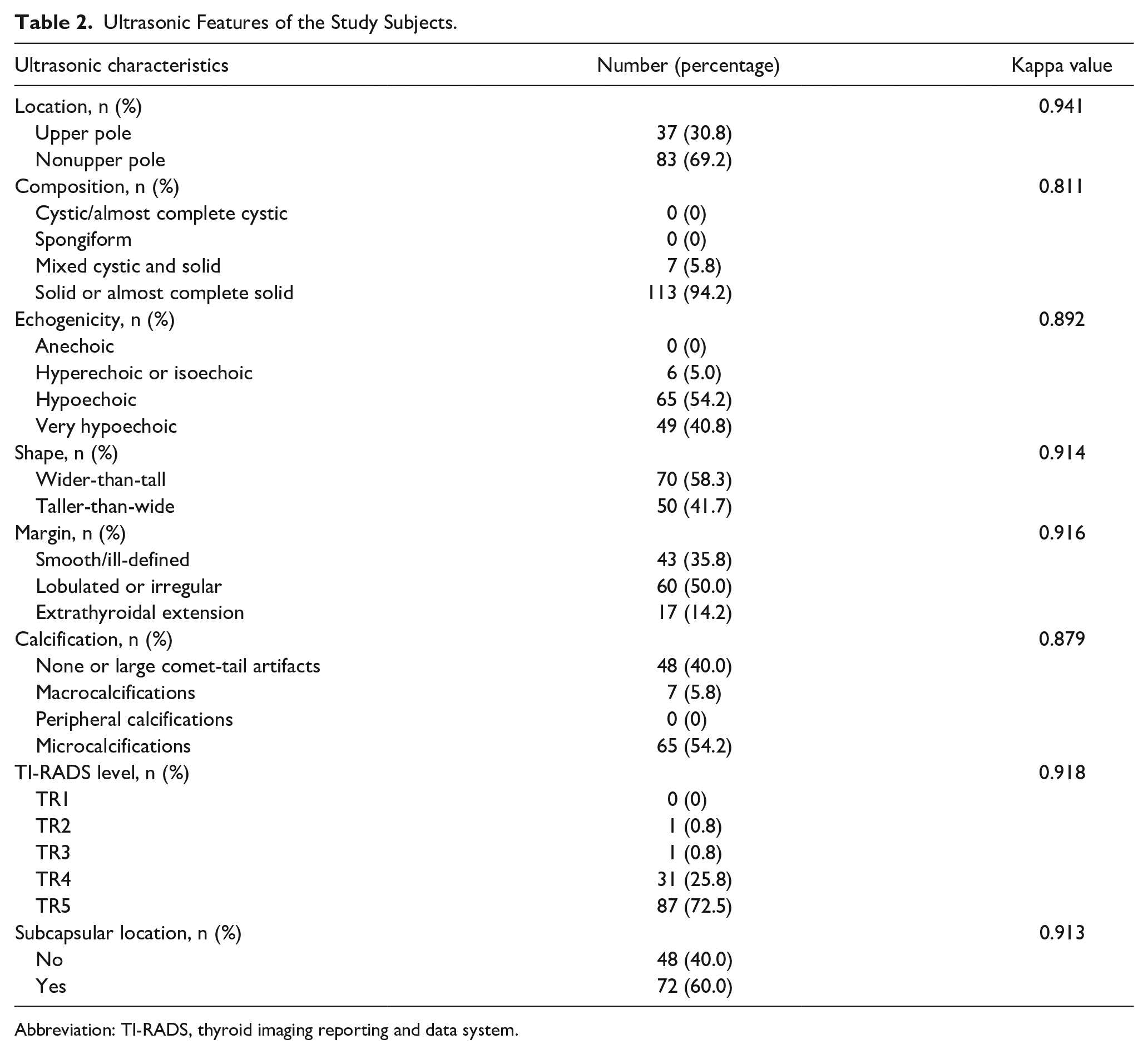
Abbreviation: TI-RADS, thyroid imaging reporting and data system.
Univariable and Multivariable Analysis of Aggressive Behavior of MTCs
Patients were divided into metastatic groups (62 patients, 51.7%, including lymphatic metastases and distant metastases), and nonmetastatic groups (58 patients, 48.3%). Table 3 displayed the results of univariable analysis. Metastasis was more frequently observed in male patients with larger and multifocal tumors. Furthermore, the optimal cutoff tumor size between 2 groups was 2.55 cm and area under curve (AUC) was 0.737 (0.649-0.825) according to ROC analysis. Regarding sonographic characteristics, there were significant differences in margin (P < .001), calcification (P = .013), and subcapsular location (P < .001) in metastatic groups versus nonmetastatic groups. No statistical difference in location, composition, echogenicity, or shape of nodule was found between 2 groups. To define independent risk factors for metastasis, multivariate logistic regression analyses were performed for all variables that had a univariable test P value <.05. The results indicated that only male sex (OR: 3.109, P = .019), size (OR: 1.914, P = .002), lobulated or irregular margin (OR: 3.101, P = .029), and multifocality of tumors (OR: 9.159, P < .001) were identified as independent risk factors for metastasis of MTCs (Figure 1).
Results of Univariable Analysis of Aggressive Behavior of MTCs.
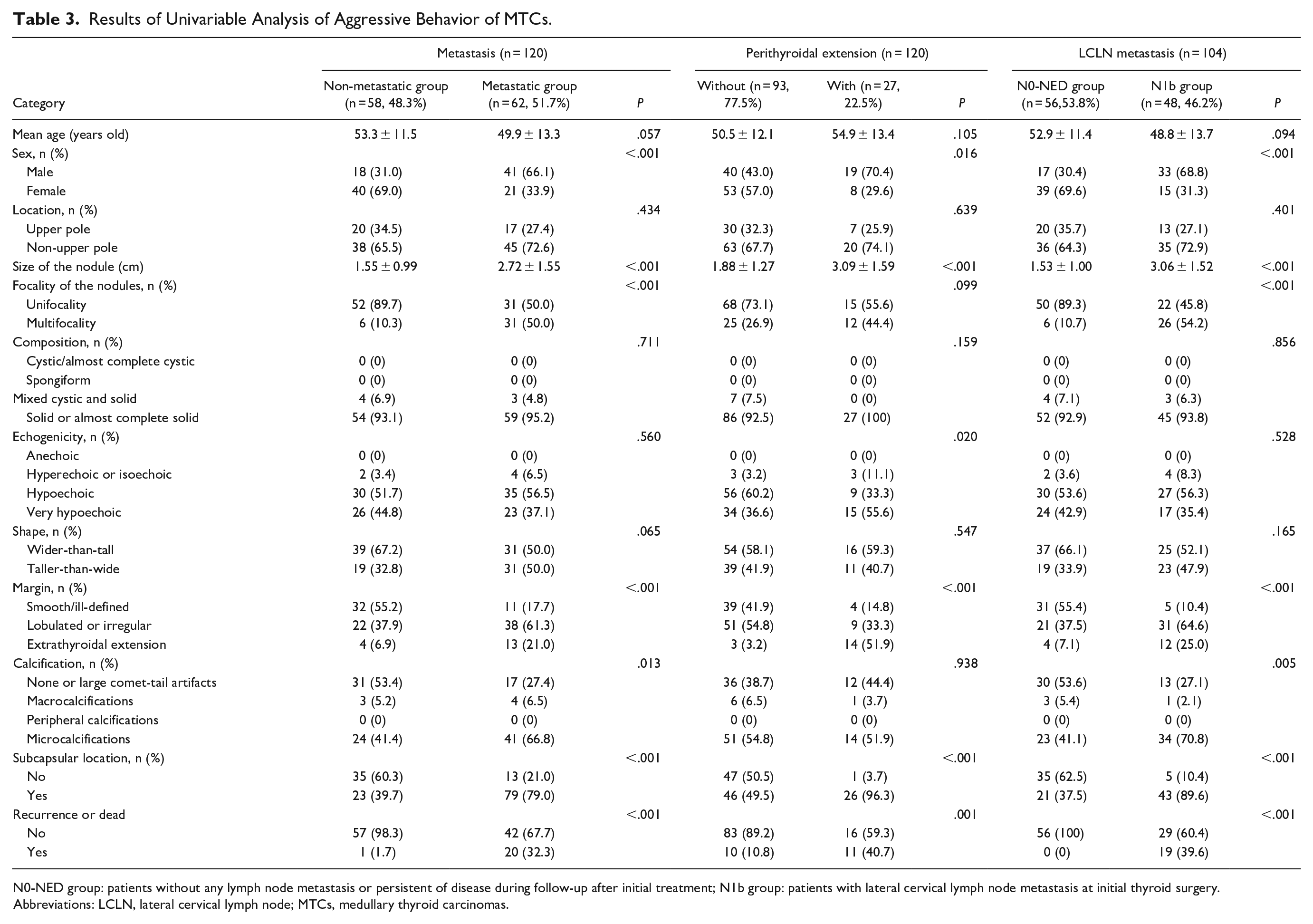
N0-NED group: patients without any lymph node metastasis or persistent of disease during follow-up after initial treatment; N1b group: patients with lateral cervical lymph node metastasis at initial thyroid surgery.
Abbreviations: LCLN, lateral cervical lymph node; MTCs, medullary thyroid carcinomas.
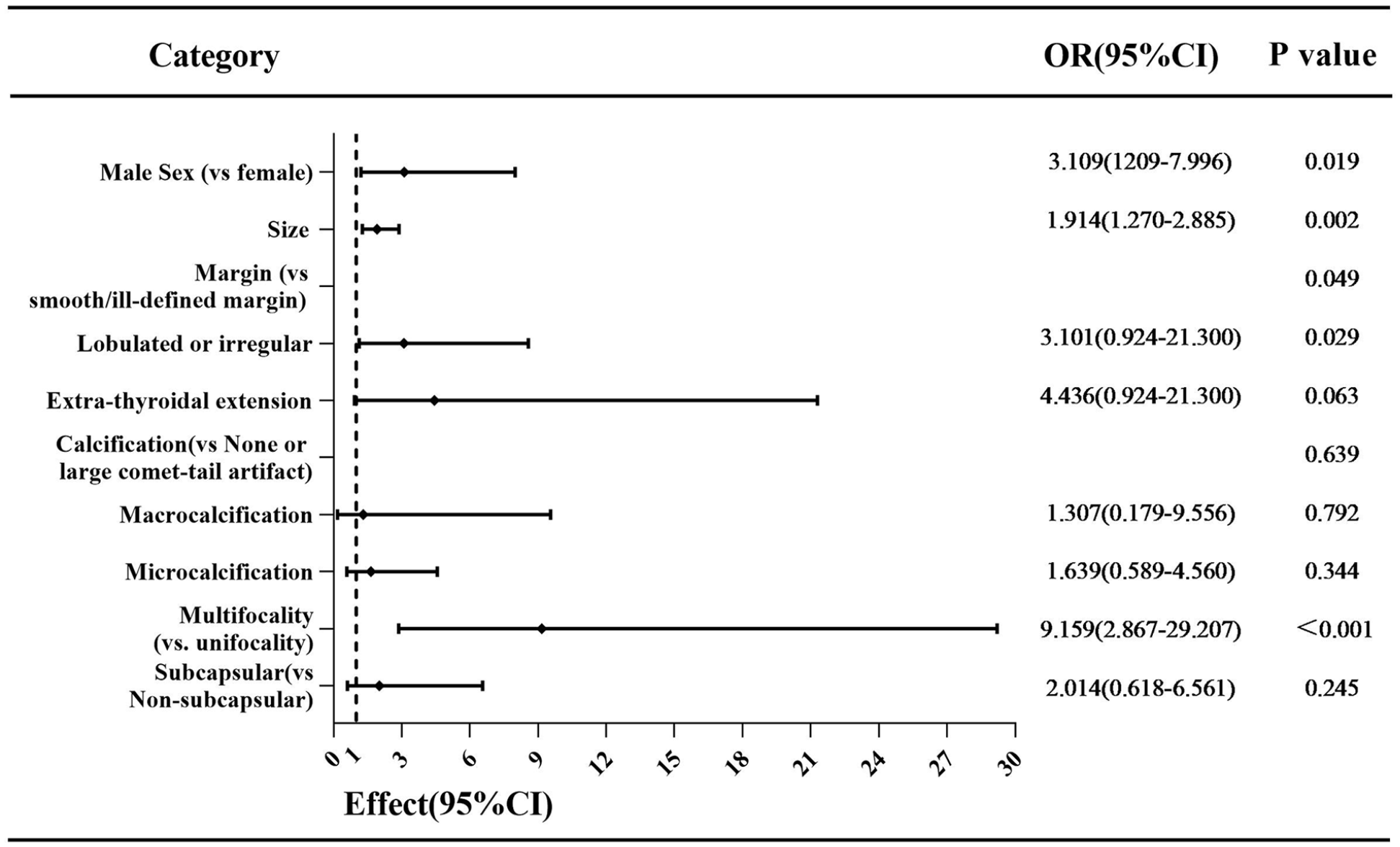
The forest plot of multivariable analysis result for metastasis of MTCs. MTCs, medullary thyroid carcinomas.
Table 3 and Figure 2 showed results of univariable and multivariable analysis between groups with/without perithyroidal invasion respectively. Only male sex (OR: 5.316, P = .018), extrathyroidal extension (OR: 20.360, P = .001), and subcapsular location (OR: 12.735, P = .021) were independent risk factors for perithyroidal invasion of MTCs.
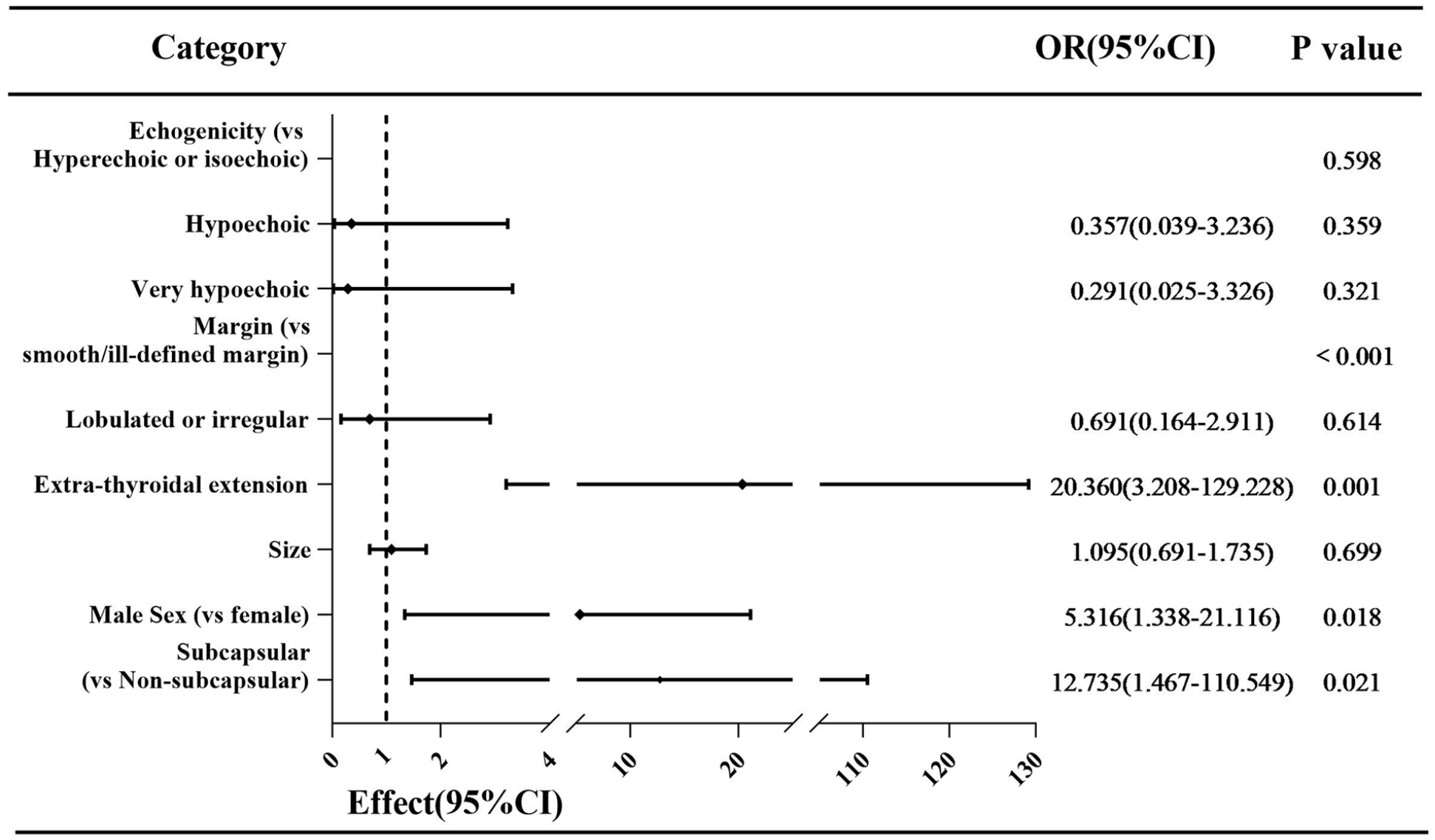
The forest plot of multivariable analysis result for perithyroidal invasion of MTCs. MTCs, medullary thyroid carcinomas.
Disease recurrence (including dead) rates were 32.3% in metastatic group and 40.7% in perithyroidal invasion group, which were significantly statistic different with groups without metastasis (1.7%, P < .001) and groups without perithyroidal invasion (10.8%, P = .001) (Table 3). K-M curves showed PDFS of patients differed significantly in groups with/without metastasis (P < .001), and in groups with/without perithyroidal invasion (P < .001) (Figure 3).
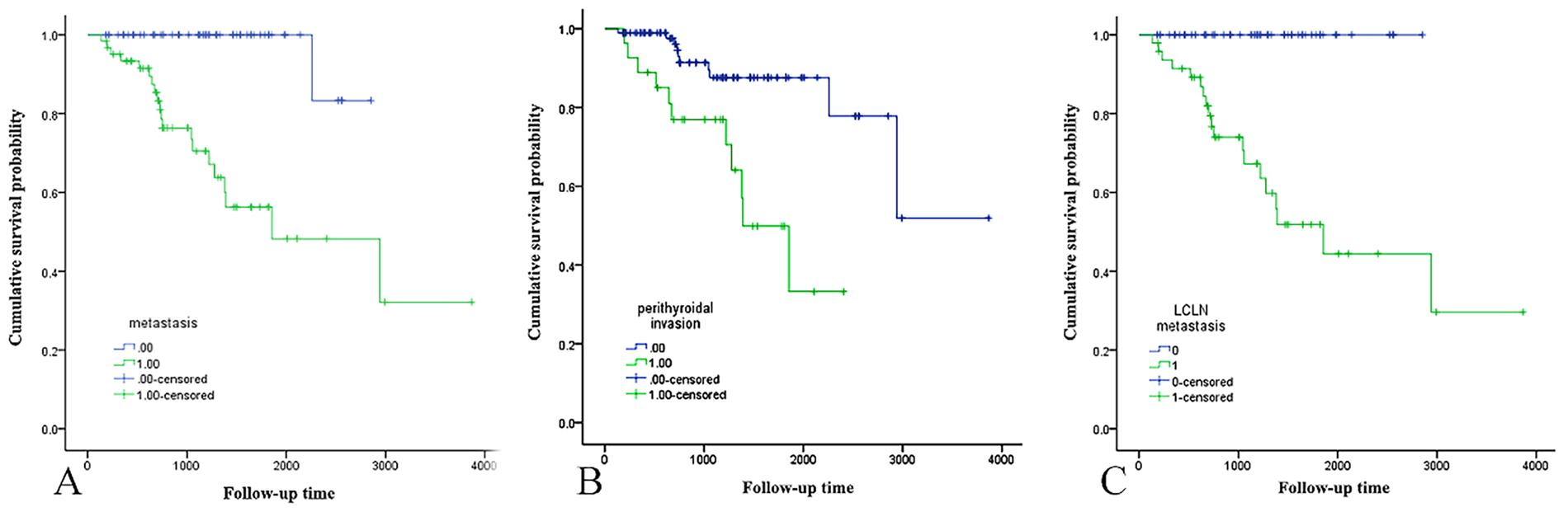
(A-C) Display K-M curves of PDFS between groups with/without metastasis, groups with/without perithyroidal extension, and groups with/without LCLN metastasis, respectively (P < .001). K-M, Kaplan-Meier; LCLN, lateral cervical lymph node; PDFS, postoperative disease-free survival.
Univariable and Multivariable Analysis of LCLN Metastasis of MTCs
This study also estimated suspicious characteristics associated with LCLN metastasis in MTCs. According to the presence or absence of LCLN metastasis, recurrence, and postoperative calcitonin level, the study subjects were reselected and stratified into N1b group (48 patients, 46.2%) and N0-NED group (56 patients, 53.8%). Results of univariable analysis were presented in Table 3. Sex of patient, size, focality, margin, calcification, and subcapsular location of the tumor differed significantly between the groups. On multivariable analysis, male sex (OR: 5.154, P = .012), size (OR: 2.130, P = .012), lobulated or irregular margin (OR: 5.860, P = .015), and multifocality (OR: 17.884, P < .001) of the tumor were significantly associated with LCLN metastasis in MTCs (Figure 4), and similar with results of multivariable analysis between the entire metastatic groups/nonmetastatic groups. ROC analysis showed that the optimal cutoff points for tumor size was 2.15 cm (AUC: 0.732, 0.630-0.835). None of patients in N0-NED group progressed to recurrence or death during follow-up. K-M curve indicated a significant difference in PDFS (Figure 3).
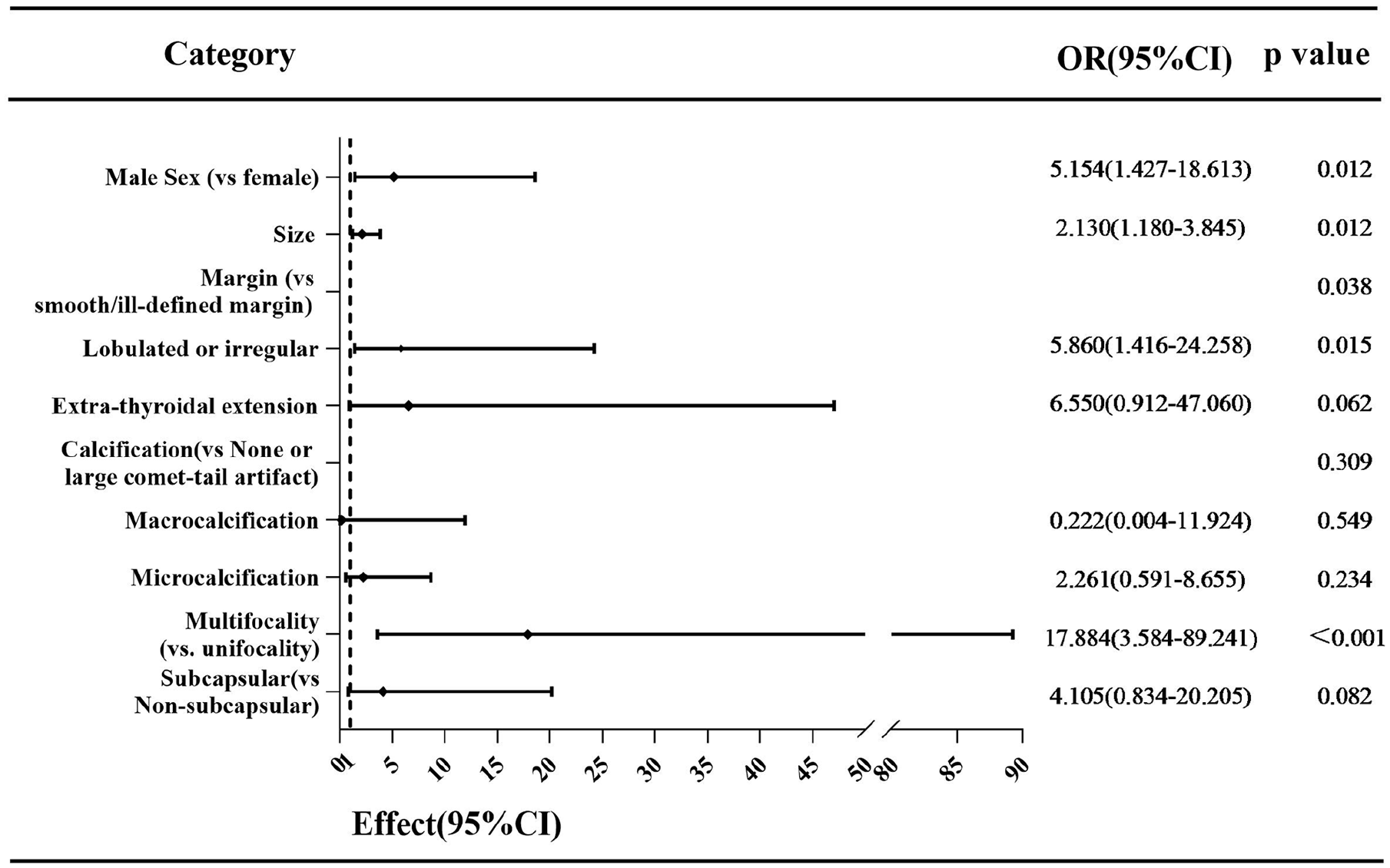
The forest plot of multivariable analysis result for LCLN metastasis of MTCs. LCLN, lateral cervical lymph node; MTCs, medullary thyroid carcinomas.
Discussion
MTCs have more aggressive behaviors than other differentiated thyroid cancers. The rate of metastases and perithyroidal invasion in this study reached up to 51.7% and 22.5%, respectively, consistent with previous reports.3,4 K-M curves indicated a significant difference in PDFS among the 3 groups (P < .001). Similarly, Zhou et al 16 concluded that patients with lateral lymph node metastasis (LLNM) had a higher recurrence rate and cancer-specific survival compared with patients without LLNM (P = .0038). Inconsistent with our results, survival proportion curves were different only between groups with/without perithyroidal extension (P < .001), but not between groups with/without metastasis (P = .473) in Ning and Kim’s study. 17 Given the poorer outcomes associated with MTCs, only a few literatures have reported the related factors influencing prognosis, and results were discordant.8,16-19 Therefore, it is particularly crucial and urgent to further investigate risk factors for aggressive behaviors in MTCs.
The present study was designed to evaluate risk factors of aggressive behaviors in MTCs, including metastasis and perithyroidal invasion in entire cohort study and LCLN metastasis in N1b group/N0-NED group. In terms of clinical features, only male sex was identified as a risk factor for metastasis, perithyroidal invasion, and LCLN metastasis. Regarding ultrasonic characteristics, large size (≥2.55 cm for metastasis, ≥2.15 cm for LCLN metastasis, respectively), multifocality, and lobulated or irregular margin of nodules were independent risk factors for metastasis, as well as for LCLN metastasis. Whereas extrathyroidal extension and subcapsular location were risk factors for perithyroidal invasion.
Male sex was a risk factor in 3 groups, and the OR value was quite high (OR = 3.109 for metastasis, 5.316 for perithyroidal invasion, 5.154 for LCLN metastasis). The age of patients was not identified as a risk factor in any of the 3 groups, which is consistent with previous studies.16,17 In a large number of MTC patients study conducted by Zhou et al, 16 they systemically retrospectively reviewed clinical characteristics of 714 patients with MTC in the Surveillance, Epidemiology, End Results (SEER) database, 35 patients in their department and concluded that male sex rather than age was a significant independent predictor for LCLN metastasis. However, Ito et al 20 have found that male sex and advanced age are independent risk factors for LCLN metastasis. Two articles published in 1996 21 and 2000, 22 respectively, also concluded that male sex and old age were more apt to progress adverse outcomes. On the contrary, Oh et al 8 discovered that neither age nor sex was risk factor for predicting LCLN metastasis. Therefore, prospective researches are needed to further verify these conclusions.
TI-RADS is likewise applicable for evaluation of MTC nodules, demonstrating high interobserver agreement, as confirmed by previous researches.17,23 Similarly, the kappa value for interobserver agreement of ultrasonic feature was high in our study (range from 0.811 to 0.941). Based on these findings, TI-RADS could potentially be employed to investigate ultrasonic predictors of aggressive behaviors in MTCs, as reported in a few previous studies.17,24
Previous studies indicated that MTC nodules tended to present benign sonographic features,10,25-28 including solid, “wider-than-tall” in shape, and smooth margin. Interest-ingly, MTCs in this study frequently manifested solid hypoechoic/very hypoechoic with lobulated/irregular margin or extrathyroidal extension, and microcalcification (Figure 5). Zhou et al 11 and Yun et al 23 reported that smaller MTC nodules more frequently exhibited spiculated/microlobulated/ill-defined margins, while tumors larger than 1 cm tended to have a smooth margin, and concluded that MTCs of different size might manifest distinct sonographic characteristics. Our research showed the opposite result, large tumors (a diameter of larger than 1 cm) tended to exhibit lobulated or irregular or extrathyroidal extension (70.9%, 61/86) and small tumors (nodules of 10 mm or less in diameter) more frequently displayed smooth/ill-defined margin (18/34, 52.9%). We speculated that observed discrepancy in our findings could be attributed to distinct subsets of MTCs with varying genetic backgrounds and growth patterns. Besides, the resolution of ultrasound equipment and the experience of radiologists might also have contributed to these varied results.
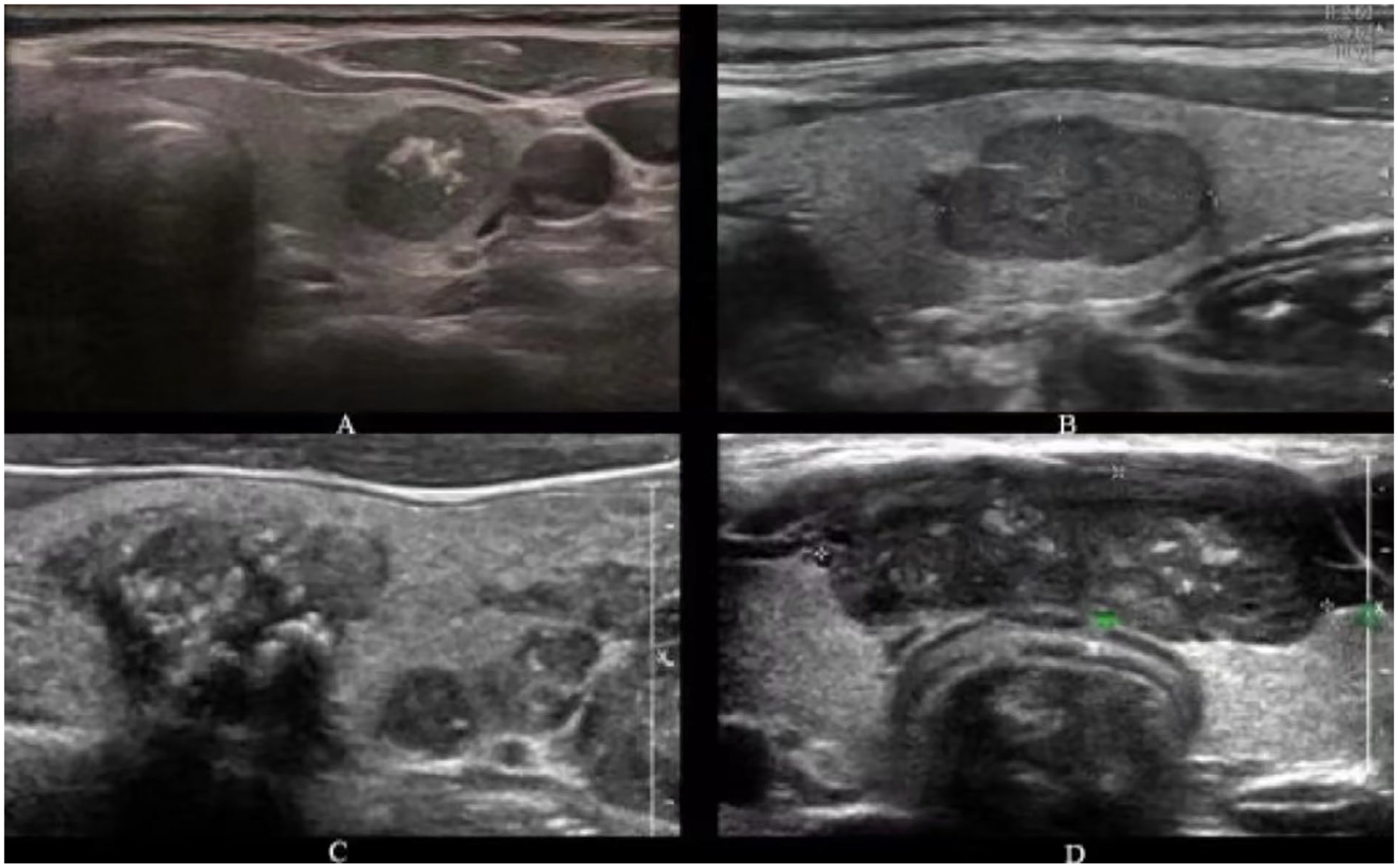
Ultrasonic images of MTCs with different margins: (A) smooth; (B) lobulated; (C) irregular; (D) extrathyroid extension. The first second patients had no lymphatic metastasis at initial surgery and no recurrence/distant metastasis during follow-up. The third patient with multifocality was a 42 year-old male and developed distant metastasis before surgery, including liver, bone, and adrenal metastasis. The fourth patient with extrathyroid extension was a 52-year-old male and progressed mediastinal and thymus metastasis at 1390 days after surgery. MTCs, medullary thyroid carcinomas.
Metastasis, especially LCLN metastasis, is prevalent in MTCs and closely linked to prognosis. Sixty-two of 120 patients (62/120, 51.7%) in this entire cohort had lymphatic metastases, 48 patients of them (48/62, 77.4%) had N1b lymphatic metastases. Recently, Zhao et al 24 applied modified TI-RADS classification to stratify MTCs in 2 groups, “malignant” (m-MTC, including as TI-RADS 4b, 4c, 5, 6) or “US-low-suspicious” (l-MTC, including as TI-RADS 2, 3, 4a) and concluded that l-MTC had more indolent character than m-MTC. Different from their study, we analyzed each ultrasonic feature individually according to ACR TI-RADS classification in this study. Here, we used the same TI-RADS classification with Ning and Kim 17 and our results partially aligned with Ning’s findings. Ning demonstrated that margin and focality were independent risk factors for metastasis. In our study, besides margin and focality, size of nodules was also emerged as a predictor for metastasis and LCLN metastasis, consistent with published literatures.8,16 Yun et al 23 reported that microcalcification and irregular shape of nodules were significantly associated with lymphatic metastases in MTCs. However, neither calcification nor shape was a risk factor for aggressive behavior in our study. In a prior study 8 which evaluated the association between preoperative features and LCLN metastasis in MTCs, large size (>1.5 cm), irregular shape, spiculated margin, and subcapsular location were found to be risk factors for LCLN metastasis and MTCs with ≥2 preoperative predictors were at high risk for LCLN metastasis. Nevertheless, we found that subcapsular location of nodules was not a risk factor for metastasis and LCLN metastasis, discordant with the aforementioned study. 8 Simultaneously, we explored preoperative features for predicting perithyroidal invasion and discovered that male sex (OR: 5.316, P = .018), margin (P < .001) and subcapsular location (OR: 12.735, P = .021) were independent risk factors for perithyroidal invasion. In contrast, previous literature identified only male sex and margin as risk factors for perithyroidal invasion. 17 A large sample study involving 568 patients conducted by Lee et al 29 indicated that a subcapsular tumor location could assist clinicians in predicting pathologic extrathyroidal extension of PTCs. Building on Lee’s conclusion and our results, we infer that a subcapsular location in preoperative sonograms could be associated with perithyroidal invasion in MTCs. Perithyroidal invasion is prone to being missed or misdiagnosed during preoperative ultrasonic examinations, as evidenced by 7 cases missed or misdiagnosed in this study (25.9%, 7/27). Therefore, when a nodule is located subcapsularly, it becomes crucial to meticulously observe whether perithyroidal tissues are invaded. As concluded by Ning and Kim, 17 high-frequency ultrasound plays a vital role in predicting perithyroidal invasion of MTCs. We completely agree with this viewpoint.
The strengths of this article lie in the large sample size of MTCs and the comprehensive analysis of ultrasonic features according to ACR TI-RADS. However, our study has some limitations. First, being a single-center cohort study, selection bias was unavoidable. Second, it is possible that a small number of hereditary MTC were included in this study, as 77 of the enrolled patients were not tested for RET, even though they had no family history or clinically confirmed MEN 2. Third, tumor markers such as preoperative serum levels of calcitonin and carcinoembryonic antigen were not evaluated in this study due to incomplete data.
Conclusions
In conclusion, clinical information and ultrasonic characteristics can serve as predictors for aggressive behaviors in MTCs. Patient sex, tumor size, margin, and focality were identified as independent risk factors for metastasis and LCLN metastasis. Consequently, our findings suggest that MTCs in male patients with large size (≥2.15 cm), irregular margin, and multifocality may benefit from treatment with prophylactic LCLN dissection. Male sex, or extrathyroidal extension and a subcapsular location of tumor are predictors for perithyroidal invasion. To better tailor initial treatment method or surgical extent, we should focus more on male patients exhibiting large size, multifocality, subcapsular location, and irregular margin or extrathyroidal extension in clinical practice. In addition, special attention should be given to patients with metastasis/perithyroidal extension/LCLN metastasis, as these individuals have a worse PDFS compared to patients without such complications.
Footnotes
Acknowledgements
The authors thank all participants of this study.
Author Contributions
C.N., C.Z., and S.F. made substantial contributions to conception and design; Q.W. and B.Z. collected data; Y.Z. and S.F. analyzed data; Y.Z. and M.D. wrote the paper; C.N. and C.Z. revised this paper. All authors read and approved the final manuscript.
Availability of Data and Materials
Due to patient privacy protection, materials and data are not publicly available, but are available from the corresponding author for reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was approved by the Ethics Committee of the affiliated Hospital of Qingdao University (Ethics No. QYFY WZLL 27819). The requirements for consent to participate and written informed consent were waived by Ethics Committee of the affiliated Hospital of Qingdao University, for it is a retrospective study. All methods are carried out in accordance with relevant guidelines and regulations.
Consent for Publication
Not applicable.
