Abstract
Keywords
Introduction
Nasal obstruction is a prevalent complaint in primary care and significantly affects the quality of life and work efficiency and can exacerbate lower airway diseases and sleep apnea.1,2 However, the symptoms can stem from various causes, making it crucial to have a comprehensive understanding of the pathophysiology of nasal obstruction to guide treatment and clinical advice.
Previous research has shown that the supine posture can increase nasal resistance compared to the seated posture.3,4 Our previous research reported that both supine and prone postures notably elevated nasal resistance in 30 healthy subjects relative to the seated posture. This was determined using a combination of self-reported assessments and objective measures, with the prone posture exhibiting a more pronounced effect on subjective feedback [subjective evaluation by visual analog scale (VAS), seated vs supine: 0.77 ± 1.17 (mean ± standard deviation) vs 1.80 ± 1.85, P < .001; seated vs prone: 0.77 ± 1.17 vs 2.47 ± 2.47, P < .001; supine vs prone: 1.80 ± 1.85 vs 2.47 ± 2.47, P = .017; objective examination by acoustic rhinometry with minimal cross-sectional area (mCSA), seated vs supine: 1.00 ± 0.23 (mm2) vs 0.89 ± 0.30, P = .002; seated vs prone: 1.00 ± 0.23 vs 0.87 ± 0.32, P = .009]. 5 We extended our investigation to 30 patients with allergic rhinitis (AR) and found similar objective findings compared to the healthy control group, indicating increased nasal resistance with postural changes (objective investigation with acoustic rhinometry with mCSA for patients with AR, seated vs supine: 1.11 ± 0.29 vs 1.01 ± 0.39, P = .014; seated vs prone: 1.11 ± 0.29 vs 0.95 ± 0.29, P < .001; supine vs prone: 1.01 ± 0.39 vs 0.95 ± 0.29, P = .022). 6 Despite these findings, there is still a lack of current research reporting the effect of the recumbent posture on nasal patency in individuals with AR. Hence, we conducted the present study, employing questionnaires, acoustic rhinometry, and video endoscopy to investigate the impact of postural changes on nasal resistance.
Materials and Methods
Participants With and Without AR
In this study, patients with AR from the outpatient department provided informed consent and were subsequently included. We employed the VAS to assess the severity of symptoms over the past week, encompassing nasal obstruction, rhinorrhea, sneezing, and nasal itching. Scores on the VAS ranged from 0 (no discomfort) to 100 (extremely bothersome).7,8 The following criteria determined eligibility for the study: a cumulative VAS score exceeding 120 points for the 4 aforementioned symptoms; serum immunoglobulin (Ig) E levels surpassing 120 kU/L (as measured by ImmunoCAP®; Phadia AB); and a positive response to at least one inhaled allergens, such as grass pollen, house dust mite, animal dander, or mold, as identified through multi-allergen simultaneous testing.5,6 Participants were excluded from the study based on the following criteria: presence of severe nasal septal deviation (NSD); history of sinonasal procedures, encompassing inferior turbinoplasty, NSD correction, endoscopic sinus surgery, nasal tumor excision, or skull base surgeries; prior diagnoses of malignancies in the nasal cavity, sinus, or nasopharynx; endoscopic findings of nasal polyps or sinusitis; current smoking habits; or an upper respiratory tract infection within the preceding month. Those on AR-specific medications, such as steroid nasal sprays or antihistamines, were mandated to discontinue their use for a minimum of 2 weeks prior to study commencement.
In cases where a patient has a previous computed tomography (CT) scan, we evaluate NSD by drawing a line in the coronal view from the insertion of the nasal septum at the crista galli to the insertion of the septum at the anterior nasal spine. In addition, we draw a line from the insertion of the nasal septum at the crista galli to the apex of the NSD. The angle formed by these 2 lines is then assessed. If this angle is less than 10°, NSD is not considered significant for the study. 9 If CT images are not available, a thorough nasal endoscopy is conducted to exclude patients with significant NSD as a subjective determination.
The control group consisted of healthy volunteers devoid of AR. To qualify for the control group, participants needed to have VAS scores for the primary AR symptoms below 30 points and serum IgE levels under 120 kU/L. The exclusion parameters mirrored those set for the AR patient group.
Informed consent was obtained from all participants in this study. The present study was reviewed and approved by the Institutional Review Board of Chang Gung Memorial Hospital and was given the number 201801720B0C501.
Assessment of Body Postures
The aims of the present study include 4 body postures, which were the seated, supine, left recumbent, and right recumbent postures (Figure 1). To minimize bias arising from a nasal adaptation from posture to posture, each subject was asked to draw lots to determine the order in which the 4 postures were to be measured (a random approach to measurement). For example, according to the results of the lottery, the subject first maintains a supine posture, then goes to left recumbent, then seated, and finally right recumbent posture. The subjects were asked to remain in each posture for about 15 minutes before being measured with the subjective and objective evaluations. Participants underwent subjective-objective measurements in 4 postures: seated, supine, left recumbent, and right recumbent, following the sequence they were assigned. All assessments were conducted in a controlled environment with a humidity range of 50% to 60% and a consistent temperature between 25°C and 26°C.
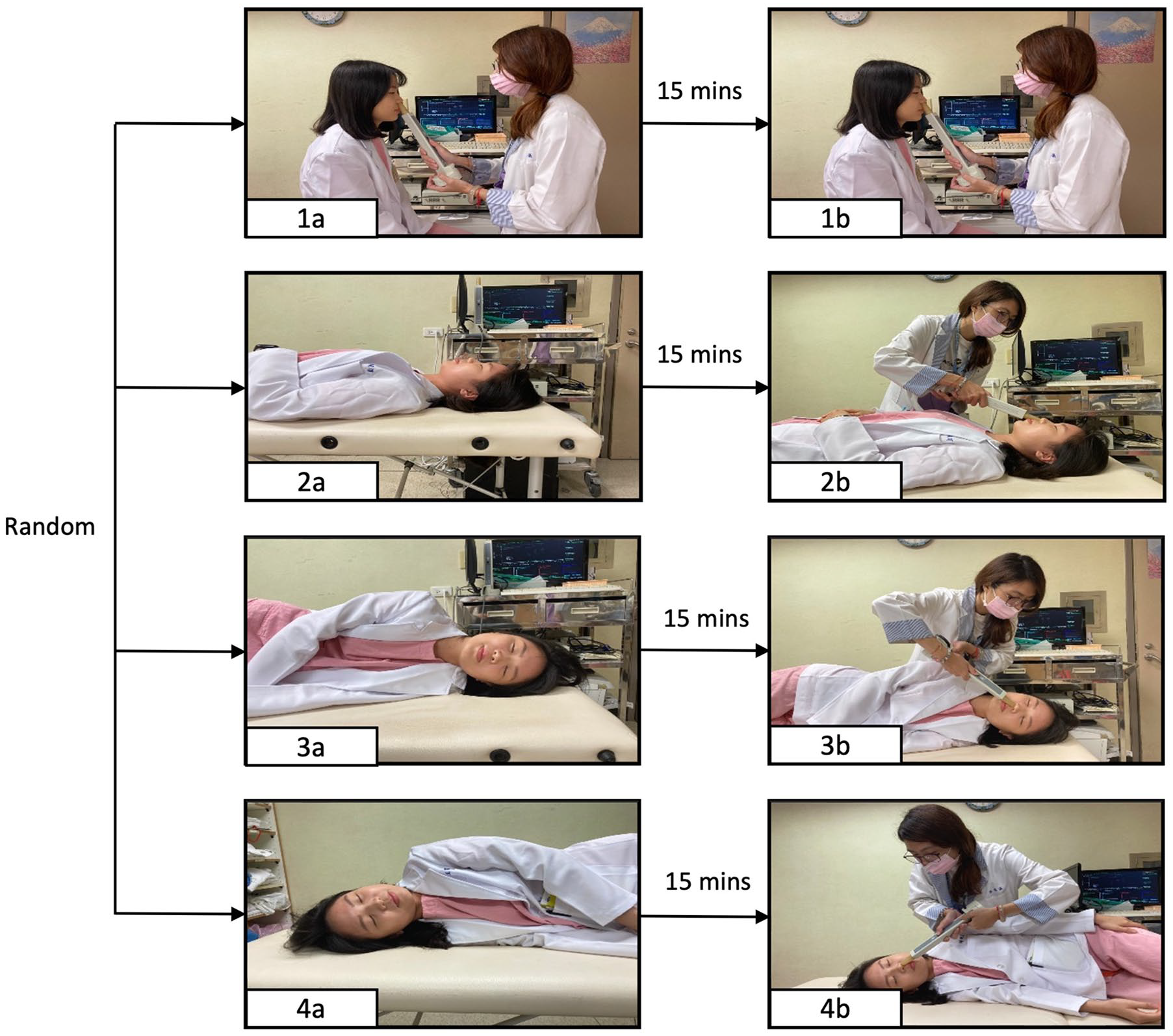
Flowchart of the postural examinations. Questionnaires, acoustic rhinometry, and video endoscopy were performed with subjects in randomly assigned postures, including seated (1a and 1b), supine (2a and 2b), left recumbent (3a and 3b), and right recumbent (4a and 4b) postures. Each time the posture was changed, the subject was required to wait for 15 minutes before being subjectively and objectively assessed.
Subjective and Objective Measurements
All participants responded to VAS questionnaires with a scale of 100 mm in length to assess the sensation of nasal blockage in 4 postures. The left side of the scale (0 mm) corresponded to a “completely unobstructed nose,” while the right extreme (100 mm) indicated a “completely obstructed nose.”
With the device (#31000 “Hood” Eccovision Acoustic Rhinometer), the mCSA of bilateral nasal cavities were measured separately, and the sum was recorded. For evaluating nasal airflow, we utilized the data from the initial MCA indicated by the test results, which correlates to the nasal valve region, to gauge the extent of nasal patency. 10 Proper, noninvasive nasal adaptors were used to prevent air leaks, and one experienced assistant conducted all measurements. Furthermore, we directly observed turbinate mucosal changes with different postures by performing the rigid endoscopic examination.
A rigid endoscopy (4 mm in diameter) was marked 1.7 cm from the front end, which corresponded to the fixed depth from the anterior naris into the nasal cavity we measured while we performed endoscopic examination (Figure 2A and B). After adjustment to each body posture, the endoscope was placed at the nostril, parallel to the Frankfurt horizontal plane, and gently introduced into the nostril until the marked site (1.7 cm from the nostril). The state of the nasal passages in each posture is then captured by a video system. An imaginary horizontal line was delineated across the most prominent section of the inferior turbinate’s head to serve as a reference. The extent of inferior turbinate hypertrophy was determined by dividing the width of the inferior turbinate (distance “b”) by the width of the nasal cavity (distance “a” as shown in Figure 2C). Measurements from both nasal passages were captured for subsequent comparison.
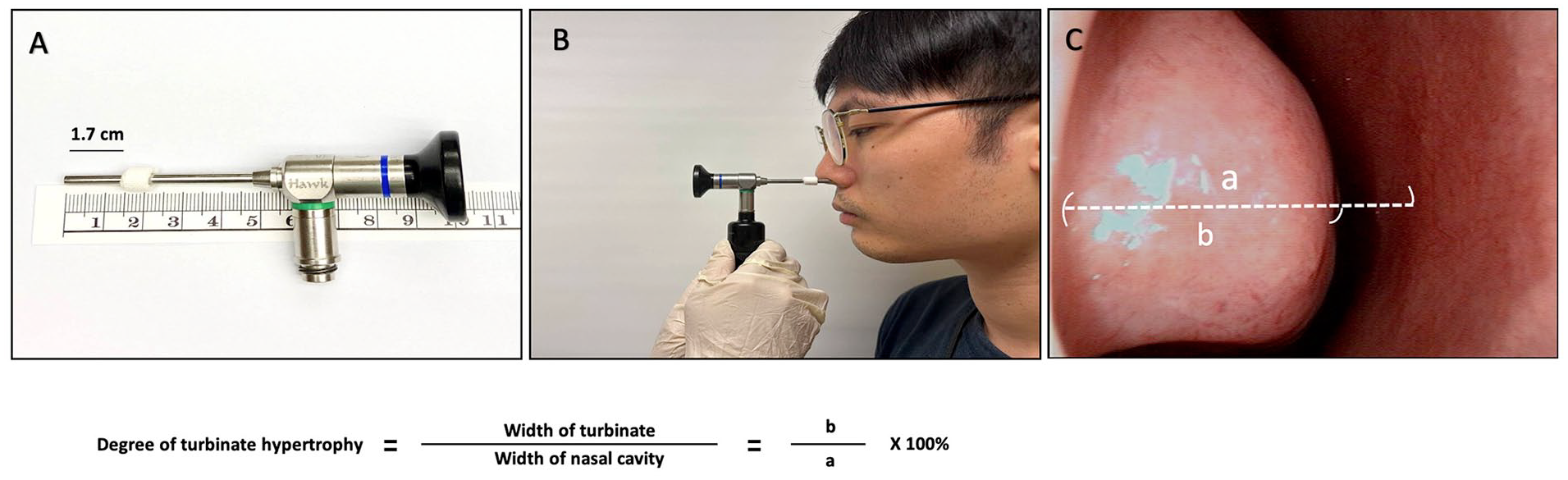
Video-endoscopic measurement of nasal patency. (A) A mark was placed 1.7 cm from the anterior tip of a rigid endoscope. (B) The endoscope was consistently inserted to this depth within the nasal cavity for image acquisition. (C) The most prominent section of the inferior turbinate’s head served as the reference, marked by a horizontal line. The nasal cavity’s width was designated as “a,” while the inferior turbinate’s width was termed “b.” The hypertrophy extent of the inferior turbinate was computed by the formula (“b” divided by “a”) × 100, and the result was expressed as a percentage.
Statistical Analysis
Information on age, serum IgE levels, VAS scores, and mCSA are expressed as mean ± standard deviation. We evaluated data normality using the Kolmogorov-Smirnov test, a robust nonparametric method suitable for our sample size. The unpaired Student’s t test was employed for data comparison. A P value below .05 was deemed statistically significant. For each of the 4 postures, VAS scores, mCSA, and endoscopy outcomes are illustrated using bar and dot plots. Intergroup comparisons were conducted using the paired Student’s t test. Significance levels are denoted as: *P < .05; **P < .01; ***P < .0001.
Results
Study Population
Table 1 presents the fundamental characteristics of the study participants. The cohort included 30 AR patients, evenly split between 15 females and 15 males, with an average age of 37.73 ± 13.40 years and a mean serum IgE level of 247.58 ± 422.41 kU/L. The healthy control group consisted of 19 females and 11 males, who were notably younger than the AR patients (31.1 ± 7.3 vs 37.73 ± 13.40 years, P = .022). On conducting the Kolmogorov-Smirnov test, our data were found to follow a normal distribution, validating the use of the unpaired Student’s t test for analyzing the differences between the 2 independent samples. When seated, AR patients indicated pronounced severity in AR-related symptoms, notably nasal obstruction (VAS score for AR vs non-AR: 30.33 ± 26.58 vs 7.67 ± 11.65, P < .001 by unpaired Student’s t tests) and rhinorrhea (14.33 ± 21.44 vs 4.33 ± 10.06, P = .026 by unpaired Student’s t tests). Although the AR group reported higher VAS scores for sneezing and nasal itching, the differences between the groups were not statistically significant. In addition, the initial mCSA, as gauged by acoustic rhinometry, showed no significant variance between the groups while seated (mCSA for AR vs non-AR: 1.11 ± 0.29 vs 1.00 ± 0.23 cm2, P = .269). Given the significant age difference between the AR group and the non-AR group, we employed analysis of covariance (ANCOVA) to adjust for age as a covariate in all our comparative analyses. This ensured that any detected differences in subjective and objective outcomes were attributable to AR, rather than age disparities.
Characteristics of the Study Population.
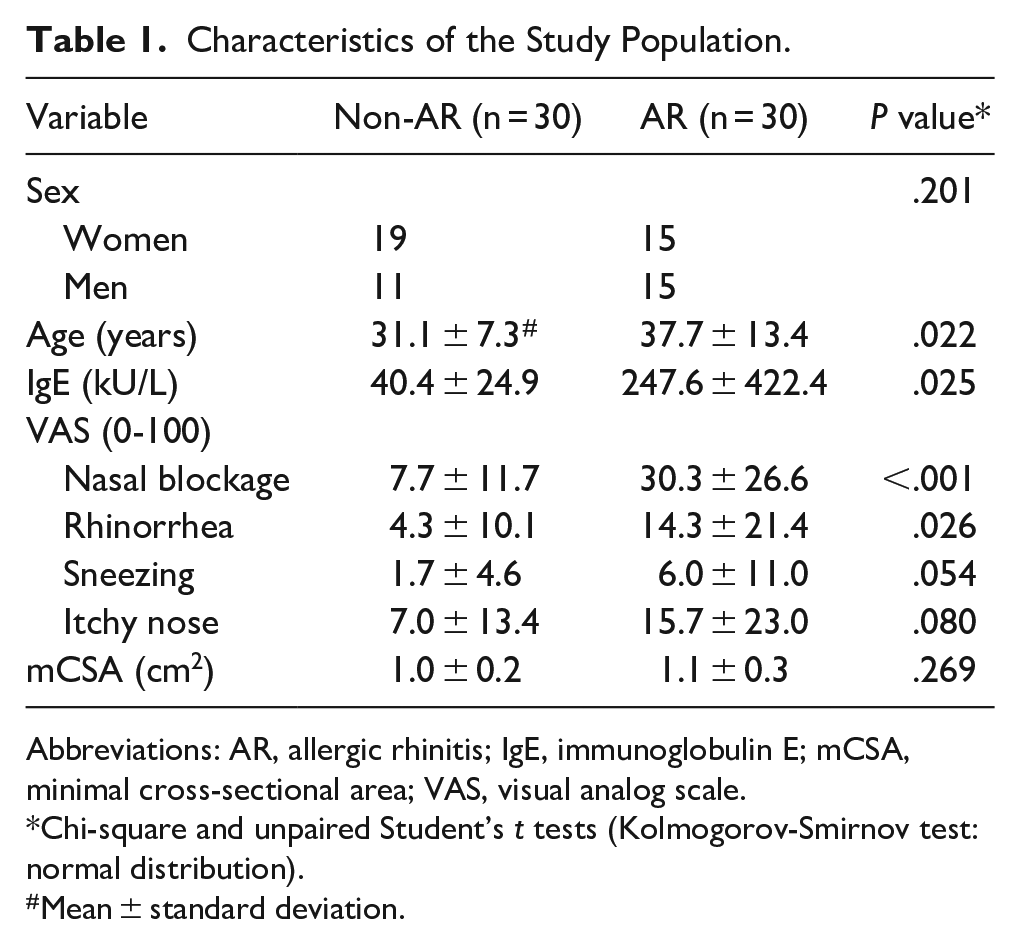
Abbreviations: AR, allergic rhinitis; IgE, immunoglobulin E; mCSA, minimal cross-sectional area; VAS, visual analog scale.
Chi-square and unpaired Student’s t tests (Kolmogorov-Smirnov test: normal distribution).
Mean ± standard deviation.
Subjective Assessment Outcomes
The variance in nasal obstruction of the AR and non-AR groups across different postures was evaluated using subjective VAS scores, with findings illustrated in Figure 3. When compared to the seated posture, healthy individuals reported a marked increase in nasal obstruction in the supine (VAS score in the non-AR group, supine vs seated: 18.00 ± 18.46 vs 7.67 ± 11.65, P < .001; an average increase of 2.35 times), left recumbent (left recumbent vs seated: 16.67 ± 16.26 vs 7.67 ± 11.65, P = .003; an average surge of 2.17 times), and right recumbent postures (right recumbent vs seated: 18.00 ± 18.08 vs 7.67 ± 11.65, P < .001; an average escalation of 2.35 times). Notably, the VAS scores between the left and right recumbent postures in the non-AR group did not differ significantly (left vs right recumbent: 16.67 ± 16.26 vs 18.00 ± 18.08, P = .354), as well as between recumbent and supine postures (left recumbent vs supine: 16.67 ± 16.26 vs 18.00 ± 18.46, P = .508; right recumbent vs supine: 18.00 ± 18.08 vs 18.00 ± 18.46, P = 1.000) (Figure 3A).
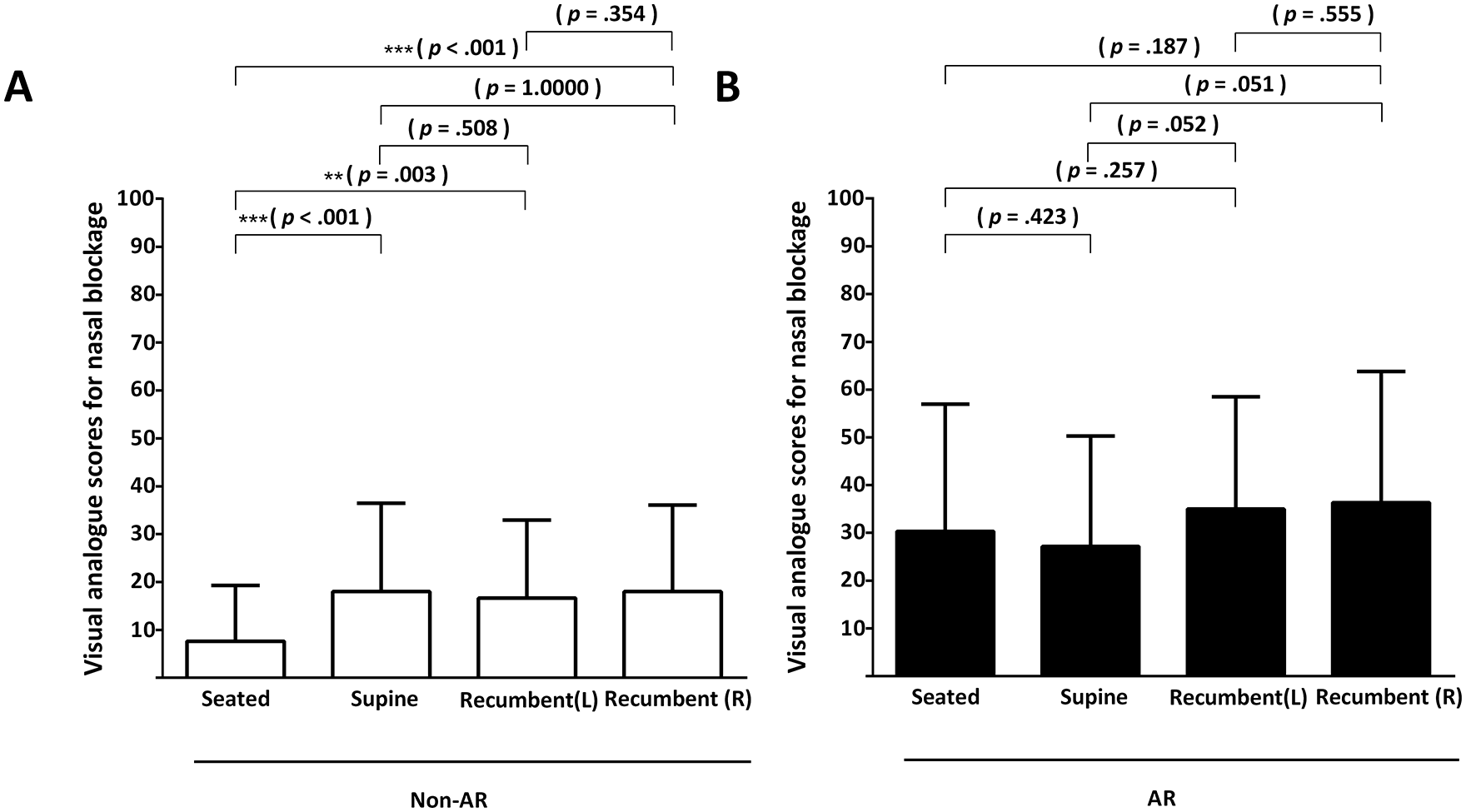
Visual analog scale. In healthy participants (non-AR group) (A) and those diagnosed with AR (B), nasal blockage perception across seated, supine, left, and right recumbent postures were evaluated using the visual analog scale, with scores spanning from 0 to 100 points. AR, allergic rhinitis.
In the AR group, comparisons of VAS scores demonstrated no statistically significant differences between the supine and seated postures (VAS score: 27.17 ± 23.11 in supine vs 30.33 ± 26.59 in seated, P = .423). Similarly, when comparing right and left recumbent postures to the seated posture, the differences in VAS scores were not significant (VAS score: 35.00 ± 23.45 in left recumbent vs 30.33 ± 26.59 in seated, P = .257; 36.33 ± 27.48 in right recumbent vs 30.33 ± 26.59 in seated, P = .187). Furthermore, comparisons between the right and left recumbent postures, as well as between recumbent and supine postures, yielded no significant differences in VAS scores (VAS score: left recumbent vs right recumbent: 35.00 ± 23.45 vs 36.33 ± 27.48, P = .555; left recumbent vs supine: 35.00 ± 23.45 vs 27.17 ± 23.11, P = .052; right recumbent vs supine: 36.33 ± 27.48 vs 27.17 ± 23.11, P = .051) (Figure 3B).
Objective Assessment Outcomes
Acoustic Rhinometry
Acoustic rhinometry results for both AR and non-AR groups are depicted in Figure 4A and B. We recorded and analyzed the combined mCSA values from both the right and left nasal cavities. For the non-AR group, the data indicated that the supine, left, and right recumbent postures all led to a significant decrease in mCSA when compared to the seated posture (mCSA in non-AR group: 0.89 ± 0.30 in supine vs 1.00 ± 0.23 in seated, P = .002; 0.83 ± 0.26 in left recumbent vs 1.00 ± 0.23 in seated, P = .001; 0.82 ± 0.37 in right recumbent vs 1.00 ± 0.23 in seated, P = .001). A similar trend was observed in the AR patients (mCSA in AR group: 1.01 ± 0.39 in supine vs 1.11 ± 0.29 in seated, P = .015; 0.96 ± 0.37 in left recumbent vs 1.11 ± 0.29 in seated, P = .002; 0.86 ± 0.36 in right recumbent vs 1.11 ± 0.29 in seated, P < .001). When comparing the left and right recumbent postures, the right recumbent posture had a more pronounced effect on reducing mCSA in AR individuals. However, this distinction was not significant in the non-AR group (mCSA in non-AR group: 0.83 ± 0.26 in left recumbent vs 0.82 ± 0.37 in right recumbent, P = .921; mCSA in AR group: 0.96 ± 0.37 in left recumbent vs 0.86 ± 0.36 in right recumbent, P = .023).
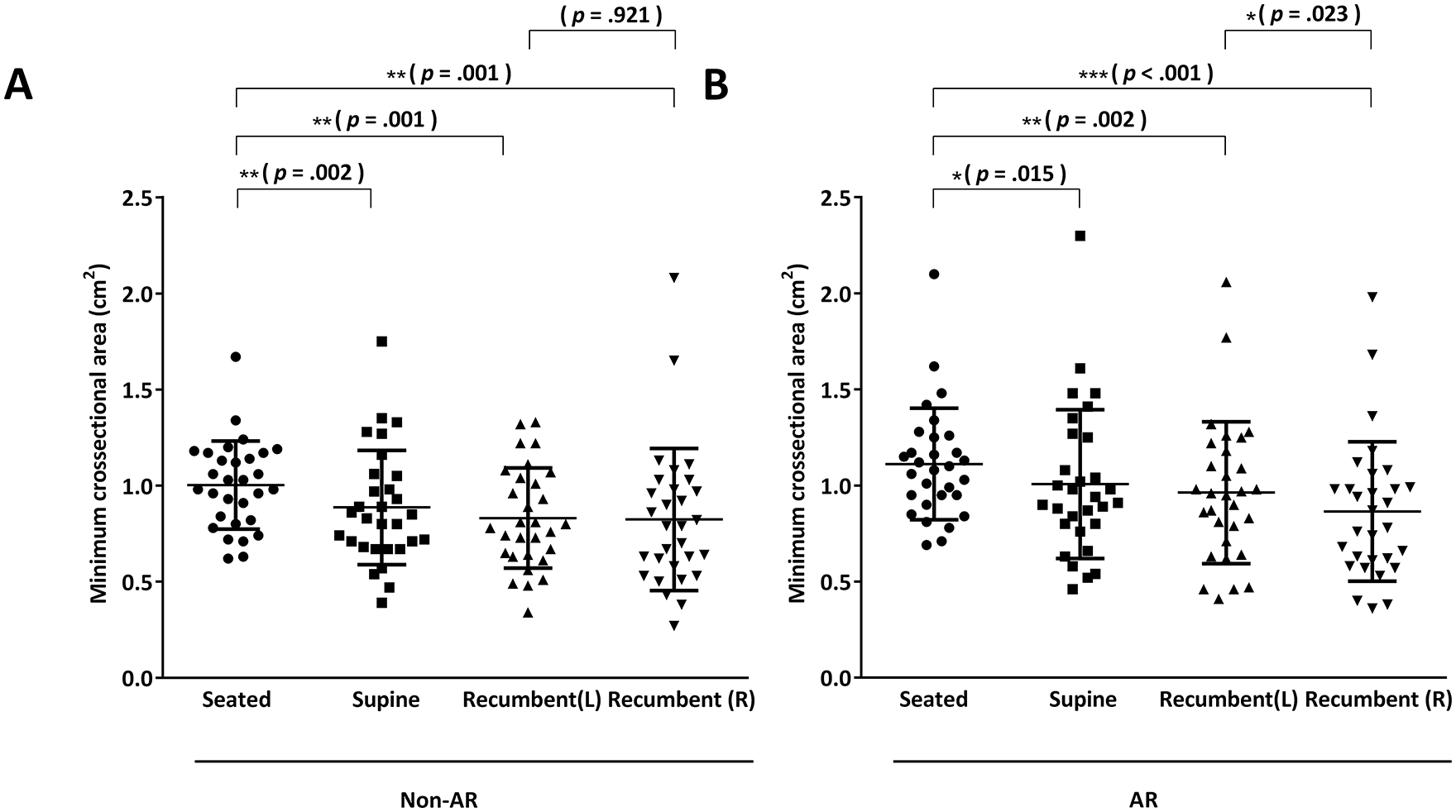
Acoustic rhinometry. Both healthy participants (non-AR group) (A) and those with AR (B) were assessed using acoustic rhinometry to measure nasal cavity patency across seated, supine objectively, left, and right recumbent postures. Outcomes are denoted as the minimal cross-sectional area (cm2), with values from both sides combined for analysis. AR, allergic rhinitis.
Endoscopy
Utilizing rigid endoscopy, we assessed the hypertrophy alterations of the inferior turbinate in various body postures, as detailed earlier (inferior turbinate diameter relative to nasal cavity diameter, see Figure 2A-C). Measurements from both nasal passages were documented and depicted separately to represent the extent of inferior turbinate hypertrophy (Figure 5A-D). Irrespective of AR presence, the right inferior turbinate’s head in all participants exhibited pronounced hypertrophy when transitioning from a seated to a right recumbent posture (right inferior turbinate diameter relative to nasal cavity diameter in non-AR group: 73.42% ± 9.50% in right recumbent vs 67.98% ± 10.48% in seated, P = .004; in AR group: 73.11% ± 14.34% in right recumbent vs 61.12% ± 10.18% in seated, P = .015). In the non-AR group, the left nasal passage showed significant turbinate hypertrophy increases in both supine and left recumbent postures compared to seated (left inferior turbinate diameter relative to nasal cavity diameter: 71.68% ± 7.05% in supine vs 66.15% ± 6.09% in seated, P = .018; 70.17% ± 9.61% in left recumbent vs 66.15% ± 6.09% in seated, P = .005). In the AR group, while both supine and right recumbent postures led to turbinate hypertrophy increases, only the left recumbent posture significantly amplified the hypertrophy of the left inferior turbinate compared to the seated posture (67.41% ± 14.48% in left recumbent vs 55.20% ± 8.21% in seated, P = .003).
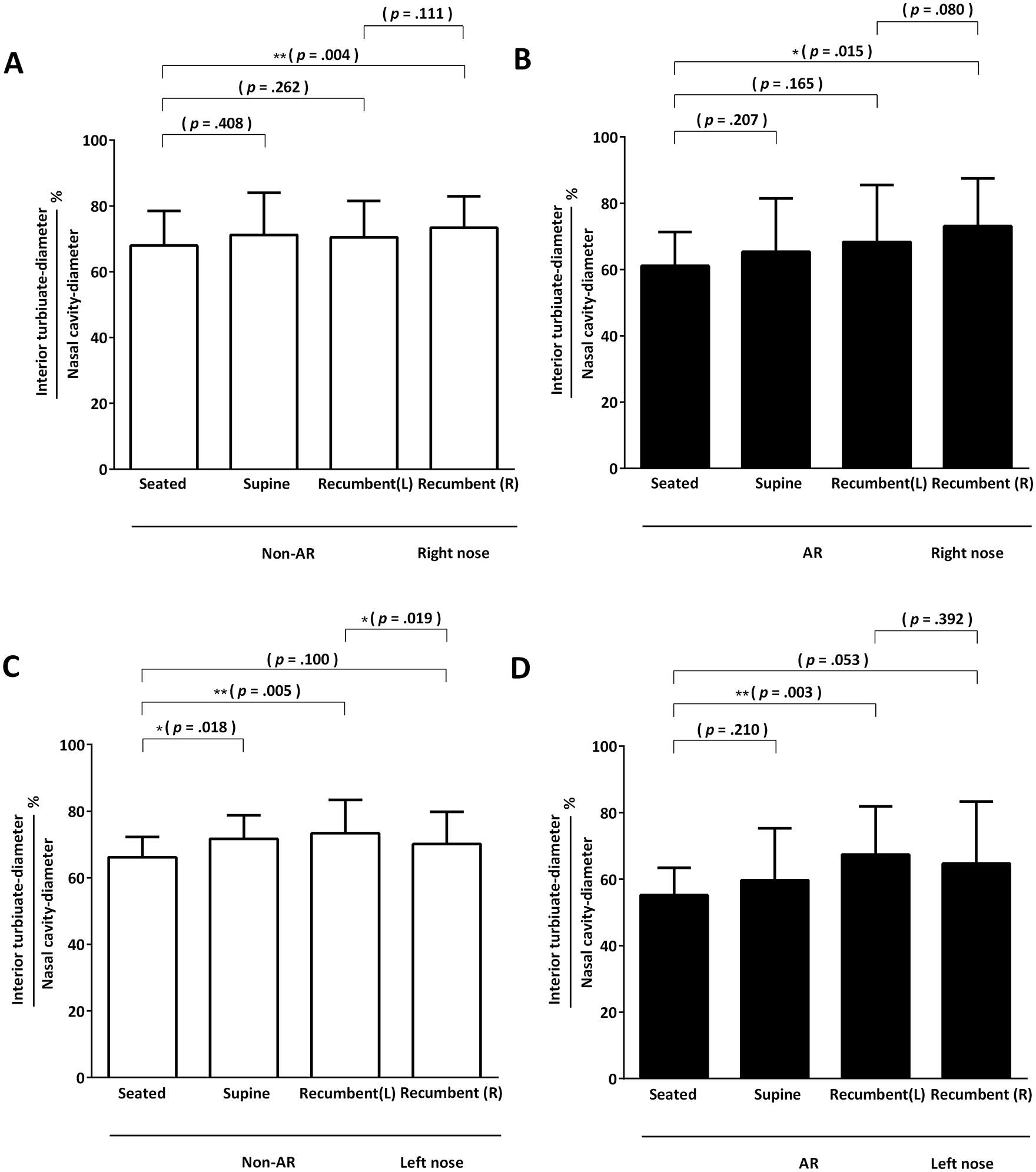
Endoscopic assessment. Both healthy participants (non-AR group) (A, C) and individuals with AR (B, D) underwent video endoscopy for each side of the nasal cavity across seated, supine, left, and right recumbent postures. The outcomes are represented as a percentage derived from dividing the inferior turbinate’s width by the nasal cavity’s width. Results for each nasal side are detailed separately. AR, allergic rhinitis.
Discussion
Our prior study indicated that in healthy individuals, transitioning from a seated to a supine or prone posture led to a notable decline in nasal cavity patency, as evidenced by both subjective and objective assessments. 5 Despite the prevalence of AR in outpatient settings, limited research has delved into the influence of body posture on this patient group. In this study, we found that among healthy controls, the supine, left, and right recumbent postures markedly exacerbated the subjective sensation of nasal obstruction compared to the baseline seated posture. Objective endoscopic evaluations further highlighted increased hypertrophy of the inferior turbinate’s head on posture change, suggesting a significant reduction in nasal patency across all 3 postures compared to the seated posture.
We further investigated the effects of postural changes on patients with AR in the present study. Interestingly, despite significant decreases in nasal patency observed in both acoustic rhinometry and endoscope measurements, postural changes showed no significant effects on VAS scores for nasal blockage in the AR group. Noticeably, the initial VAS scores were significantly higher in the AR group than in the non-AR group. A possible explanation is that patients with AR suffered from prolonged nasal blockage, and the symptoms might be compensated, which caused persistent nasal blockage perception regardless of body posture.
When the subject changes from a seated to a supine posture, gravity causes an increase in the venous hydrostatic pressure as well as congestion of capacitance of the inferior turbinate, which leads to an increase in nasal resistance. 4 Our results aligned with previous studies and showed that lateral recumbency caused a greater increase in nasal resistance over the dependent side of the nasal cavity compared with the opposite side, whether the presence of AR or not. Although the physiology was not comprehensively understood, a hypothesis is related to increases in internal jugular venous pressure from 4.5 mmHg in the standing posture to 11 mmHg in the recumbent posture. 11 Gravity might cause a disproportional increase in the dependent side of venous pressure, which potentially results in ipsilateral nasal vessel congestion. However, there is still a paucity of definite explanations for the changes in nasal resistance in recumbency, and it warrants more comprehensive research.
AR is often related to respiratory and sleep disorders, such as obstructive sleep apnea (OSA), and potentially deteriorated respiratory conditions. Increased nasal resistance contributed to negative pressure in the oropharynx and oral breathing in OSA patients. 12 Therefore, successful control of AR could alleviate the symptoms of OSA and facilitate further treatment, such as positive airway therapy. 13 In addition, the recumbent posture was proved to cause a larger pharyngeal area, which refers to a less collapsible upper airway, than the supine posture in snoring or OSA patients. 14 According to our study, we can find that, with or without AR, the recumbent posture did not increase nasal congestion compared to the supine posture, based on subjective or objective assessment. In addition, a better understanding of whether nasal surgery, such as septomeatoplasty or inferior turbinoplasty, can reduce the increase in nasal resistance caused by supine and recumbent postures would be an important and helpful research topic to improve the outcome of OSA treatment.
Our study boasts several strengths: We employed random postural changes to assess the impact of these changes on nasal obstruction, mitigating bias from nasal adaptation. Furthermore, we utilized endoscopy to objectively document the inferior turbinate hypertrophy across different postures, a previously unexplored method in related studies. Nonetheless, our study has limitations. We excluded patients with prior nasal surgeries and significant NSD based on rhinoscopy without employing CT to gauge the degree of deviation beyond the curvature angle. Future studies should consider CT to minimize NSD-induced bias. While acoustic rhinometry was used to measure nasal resistance objectively, potential errors might arise from nostril compression during posture changes, especially in the recumbent position. Refining the detection technique and equipment is essential to address this bias.
We used endoscopy to objectively assess the inferior turbinate hypertrophy but recognized its limitations in accuracy. The differences in depth of endoscopy placed into the nasal cavity, the angle created between the endoscope and the horizontal plane, and the differences in the length of the nasal passages between individuals can significantly affect the results of measurements. Future studies may involve precise volumetric CT measurements of the inferior turbinates across different postures to better understand postural impacts on turbinate size.
Conclusion
Our study demonstrated that supine and recumbent postures decreased nasal patency in subjective and objective measurements in healthy individuals compared with seated postures. Compared with the supine posture, the recumbent posture did not further increase nasal resistance, whereas, in the recumbent posture, the nasal resistance at the dependent site was relatively higher than that at the opposite site. In patients with AR, supine and recumbent postures showed similar effects in objective measurements on nasal patency; however, the postural change did not aggravate the subjective perception of nasal blockage in those AR individuals.
Footnotes
Data Availability Statement
The 3 people in Figure 1 (showing the study procedure) and ![]() (showing the endoscopic examination method) of this study are the research assistants who performed this study. They all have permitted the photographs to be used for publication in the article.
(showing the endoscopic examination method) of this study are the research assistants who performed this study. They all have permitted the photographs to be used for publication in the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by the Chiayi branch of Chang Gung Memorial Hospital in Taiwan with the grant number: CMRPG6J0202.
Ethics Statement
The study was performed in accordance with the principles of the Declaration of Helsinki and was approved by the Institutional Review Board of Chang Gung Medical Foundation in Taiwan (No. 201801720B0C501). All participators in this study fully understood the research content and signed the informed consents for participating in the study.
