Abstract
Introduction
According to the “World Report on Hearing” published by the World Health Organization (WHO) in 2021, hearing impairment affects more than 1.5 billion people globally, and predictions show that around 2.5 billion people will have experienced some degree of hearing loss by 2025. 1 The most common cause of hearing loss in older adults worldwide is presbycusis, or age-related hearing loss (ARHL). 2 The WHO estimates that approximately 164.5 million people aged >65 years worldwide had hearing impairment in 2012. 3 With the current increasing age of the global population and increasing life expectancy, more than 500 million people over the age of 60 are expected to have ARHL by 2025. 4
ARHL usually manifests as an irreversible, progressive disorder, with hearing loss beginning in the high-frequency range and gradually progressing to the low-frequency range. 5 Several recent studies have revealed that ARHL can cause cognitive impairment and that it is an independent high-risk factor for several neuropsychiatric disorders, including dementia, depression, and Alzheimer’s disease. 6 Uncorrected hearing loss not only affects physical and cognitive functions, but can also result in serious withdrawal from social activities. 7
Tests of liver function can be used as important indicators of pathological metabolic changes and are extensively used in clinical diagnostics. They represent the metabolic, immunological, and repairing abilities of the body.8-10 Moreover, there is a higher incidence of liver pathologies, impairing liver function, in older adults. 11 Impaired liver function may be a predictive factor for sudden sensorineural hearing loss. 12 A retrospective cohort study showed that the occurrence and development of ARHL are common, patients with high serum albumin (ALB) levels are less likely to experience hearing deterioration, while patients with high blood bilirubin levels show poor prognoses regarding ARHL. 13 This may be because ALB is an important antioxidant component that has a protective effect on the auditory system. While, bilirubin can directly cause neurotoxic damage to the auditory system after crossing the blood-brain barrier. 13
Although several studies have explored serum markers as indicators of risk for hearing loss, there have been no studies investigating the relationship between liver function and ARHL. Therefore, this retrospective study analyzed several common indicators, including ALB, globulin (GLB), the albumin-to-globulin ratio (AGR), alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total bilirubin (TBIL), direct bilirubin (DBIL), and prealbumin (PAB), to elucidate whether the indicators are potential risk factors for ARHL. Furthermore, we grouped patients with ARHL according to their degree of hearing loss and analyzed whether liver function indicators could predict the severity of hearing loss in ARHL.
Materials and Methods
Study Population
The present study included 107 patients diagnosed with ARHL from May 2022 to September 2023 at our hospital, with pure-tone average (PTA) values exceeding 20 dB Hearing level (HL) being considered as indicative of hearing impairment. This study enrolled patients with ARHL aged ≥60 years with average PTA values >20 dB HL at 0.125, 0.25, 0.5, 1, 2, 4, and 8 kHz. The exclusion criteria were: (1) the presence of otologic disorders affecting a diagnosis of ARHL, including conductive hearing loss; acoustic trauma or chronic otitis media; ear surgery; or Ménière’s disease; (2) severe organ dysfunction including liver or kidney dysfunction or malignancy that significantly affected liver function; (3) patients with incomplete clinical data; and (4) patients taking drugs that interfere with liver function. We recruited 107 sex- and age-matched older adults with healthy hearing from our regular medical checkup population for comparison. The same exclusion criteria were applied to the control group.
Data Collection
A detailed medical history of baseline characteristics such as sex, age, and body mass index (BMI) was collected. Venous blood samples were collected from all participants between 06:00 and 07:00 after overnight fasting. The blood samples were then subjected to liver function tests using an automated biochemical analyzer. The AGR was calculated by dividing the ALB level by the GLB level. After blood samples were collected, hearing examinations performed on the same day measured air-conducted and bone-conducted PTA at 0.125, 0.25, 0.5, 1, 2, 4, and 8 kHz; the tests were performed by an audiologist in a standardized shielded room. The degree of hearing loss was assessed using average hearing thresholds (0.125-8 kHz) for the frequencies damaged in both ears.
Statistical Analysis
SPSS 27.0 (IBM Corp) and GraphPad Prism 9.0 (GraphPad Software) were used for all statistical analyses. Descriptive variables in this study are presented as median (interquartile range), mean ± standard deviation, or percentage. The means of normally distributed variables were compared using unpaired
Results
Clinical Characteristics
A total of 107 patients with ARHL and 107 age- and sex-matched healthy participants were included. There were no significant differences in age, sex, or BMI between the 2 groups (Table 1). The ARHL group showed significantly higher GLB and DBIL levels than the control group (
Clinical Characteristics of the ARHL and Control Groups.
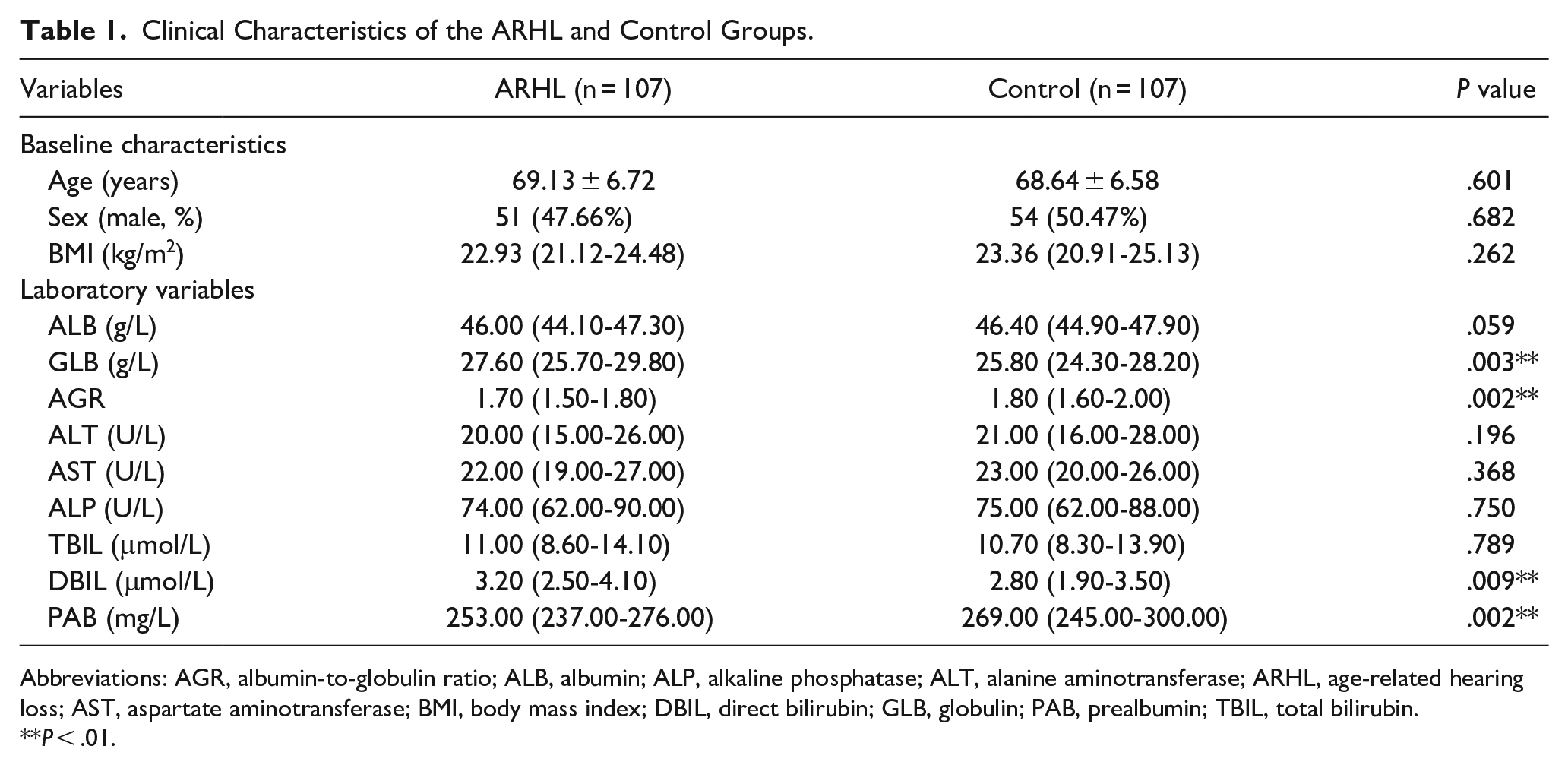
Abbreviations: AGR, albumin-to-globulin ratio; ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; ARHL, age-related hearing loss; AST, aspartate aminotransferase; BMI, body mass index; DBIL, direct bilirubin; GLB, globulin; PAB, prealbumin; TBIL, total bilirubin.
Correlation Between ARHL and Liver Function Parameters
As shown in Figure 1, PAB levels were negatively and linearly correlated with ARHL. Next, we constructed logistic regression models on the basis of the liver function parameters (Table 2), with the AGR being precluded due to collinearity between all continuous variables, as assessed by the variance inflation factor. In the univariate logistic regression analysis, with unadjusted ORs of 0.888 (95% CI, 0.796-0.990;
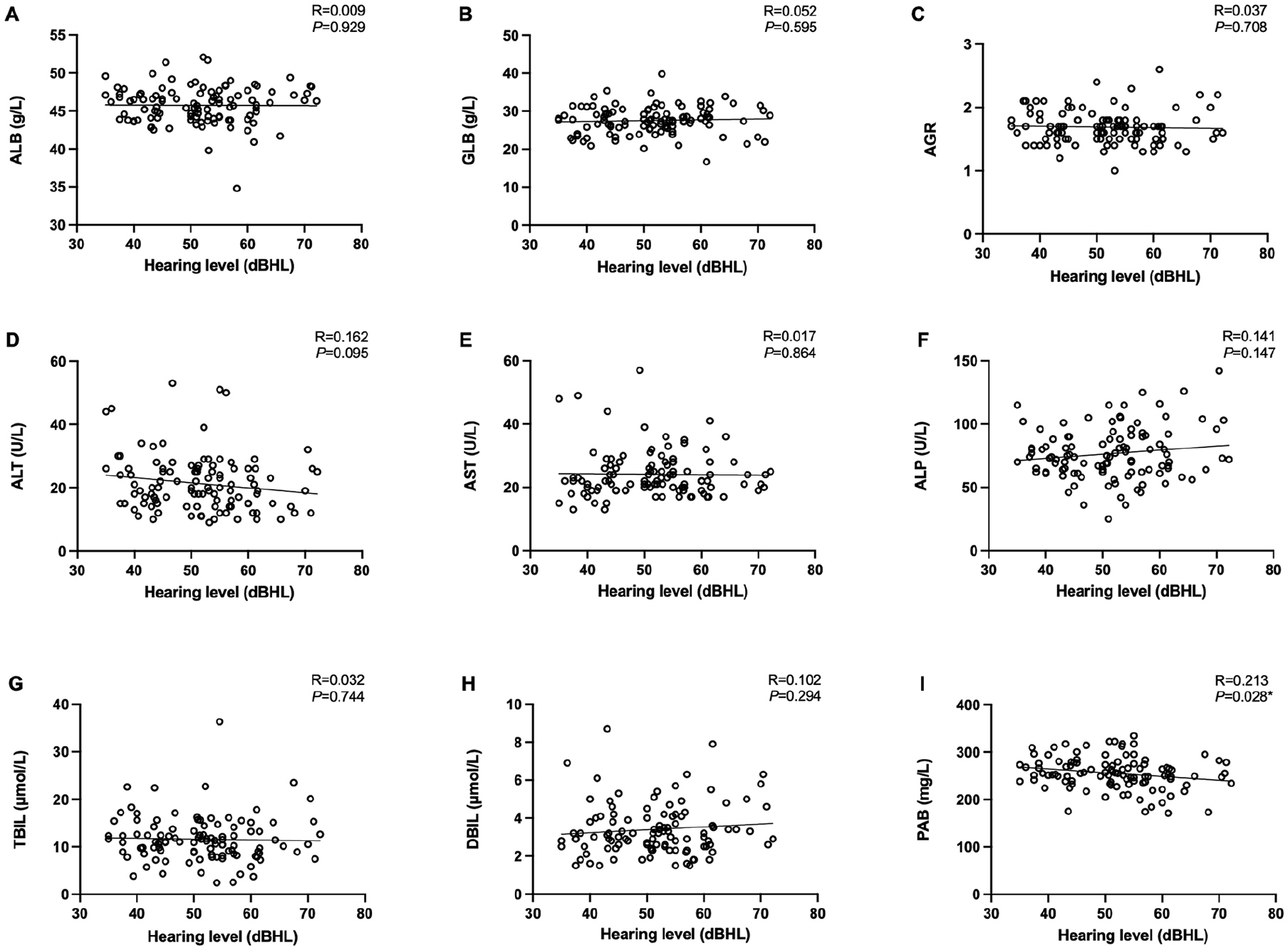
Plots of liver function marker levels versus ARHL severity. (A-I) Represent the relationship between ALB, GLB, AGR, ALT, AST, ALP, TBIL, DBIL, and PAB with ARHL severity, respectively. *
Binary Logistic Regression Analysis of the Relationship Between Liver Function Levels and ARHL.
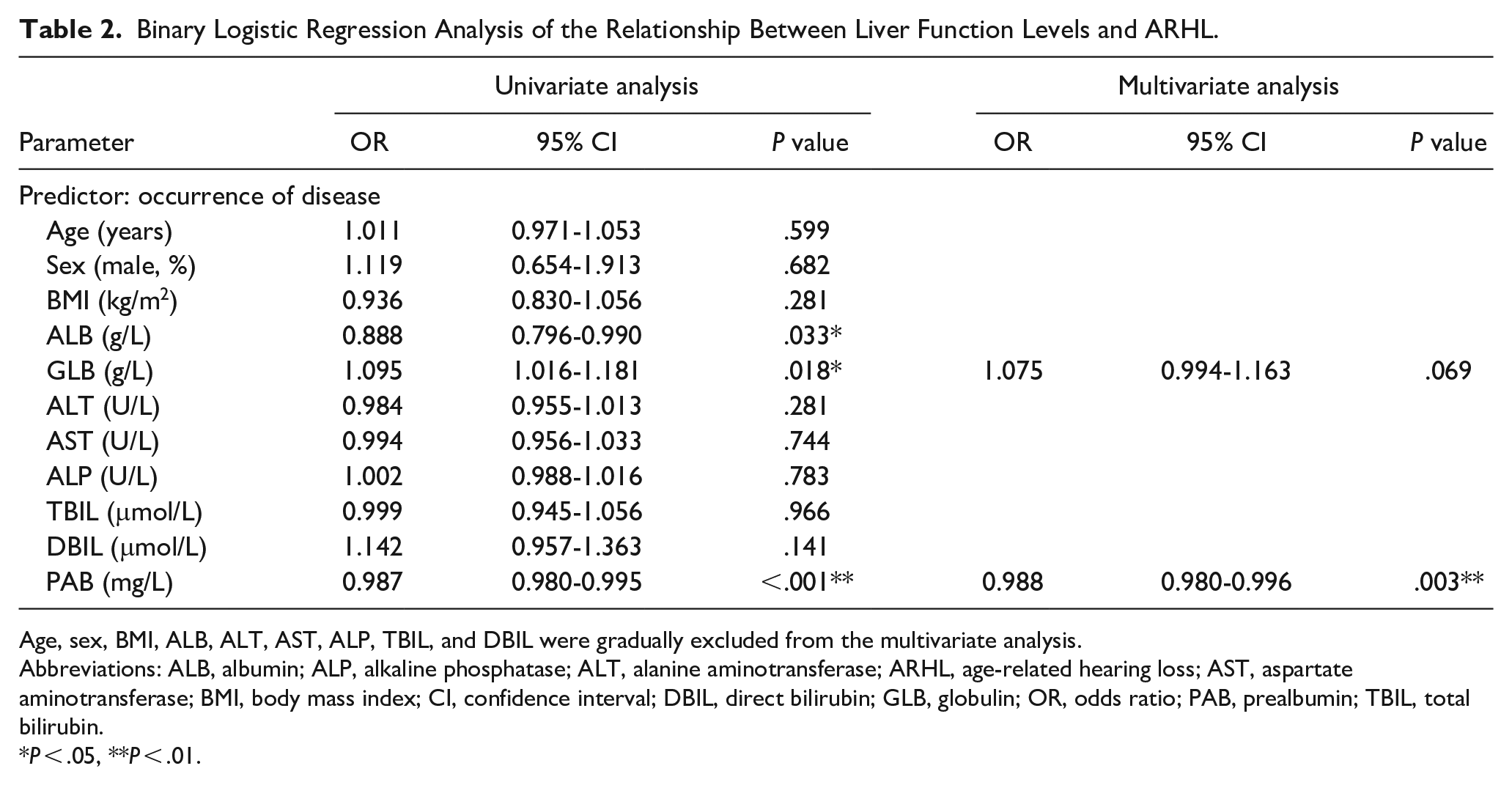
Age, sex, BMI, ALB, ALT, AST, ALP, TBIL, and DBIL were gradually excluded from the multivariate analysis.
Abbreviations: ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; ARHL, age-related hearing loss; AST, aspartate aminotransferase; BMI, body mass index; CI, confidence interval; DBIL, direct bilirubin; GLB, globulin; OR, odds ratio; PAB, prealbumin; TBIL, total bilirubin.
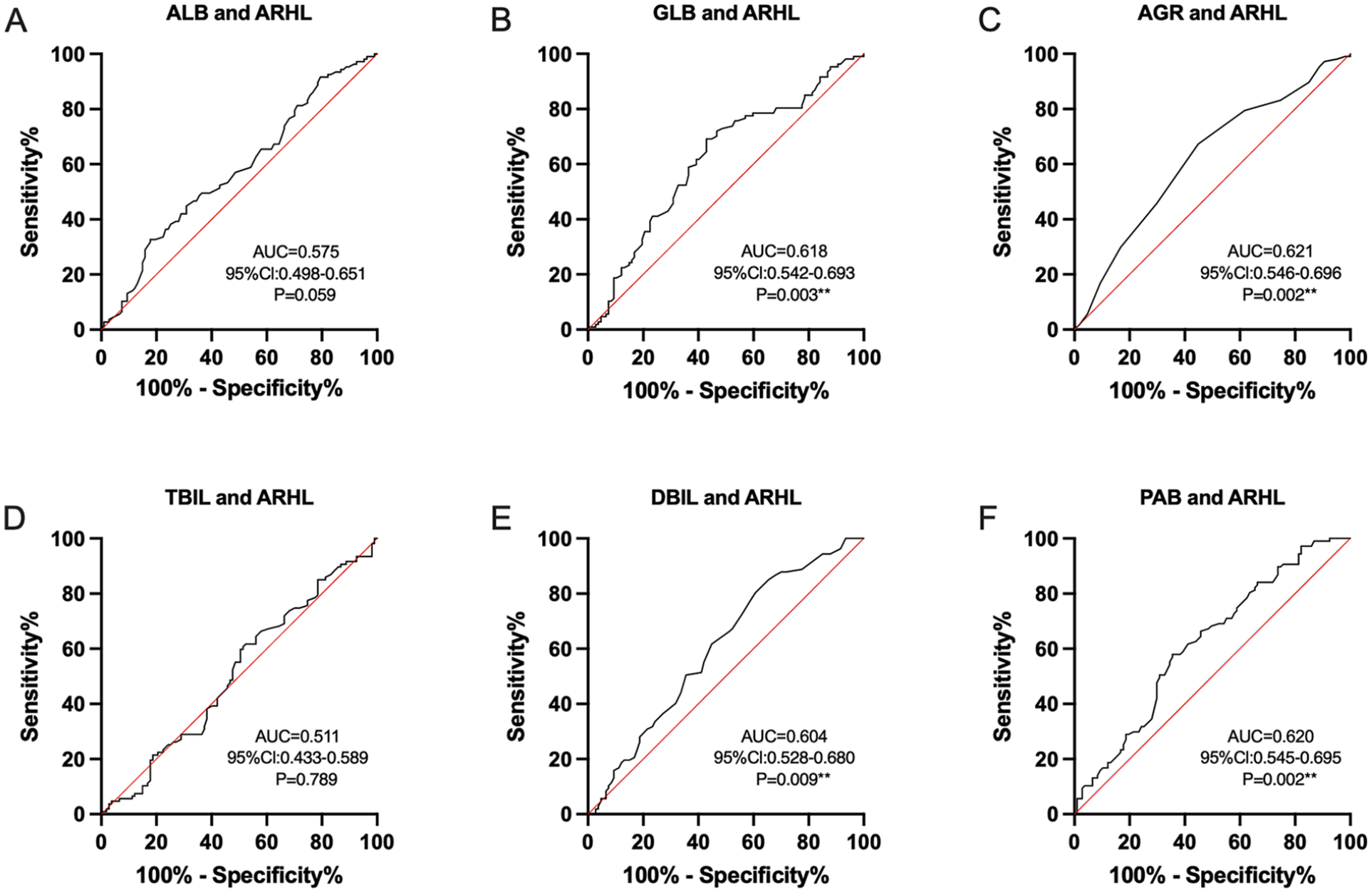
ROC curve analysis of ALB, GLB, AGR, TBIL, DBIL, and PAB levels for the prediction of ARHL. (A-F) represent ROC curves for ALB, GLB, AGR, TBIL, DBIL, and PAB levels, respectively, for the prediction of the occurrence of ARHL. **
Moderately Severe-to-Profound ARHL and Liver Function Parameters
According to the latest WHO report on hearing, hearing loss of over 50 dB qualifies as “moderately severe-to-profound hearing loss,” which means that people with this hearing loss have difficulty hearing during normal conversation.
1
Hence, patients with ARHL were divided into 2 subgroups based on ARHL severity: a moderately severe-to-profound subgroup, with hearing loss >50 dB, and a mild-to-moderate hearing loss subgroup, with hearing loss ≤50 dB. Table 3 shows the baseline characteristics and liver function parameters of both ARHL subgroups. The median ages in the moderately severe-to-profound and mild-to-moderate subgroups were 70.48 and 66.85 years, respectively (
Baseline Characteristics of Participants in the Moderately Severe-to-Profound ARHL and Mild-to-Moderate ARHL Groups.
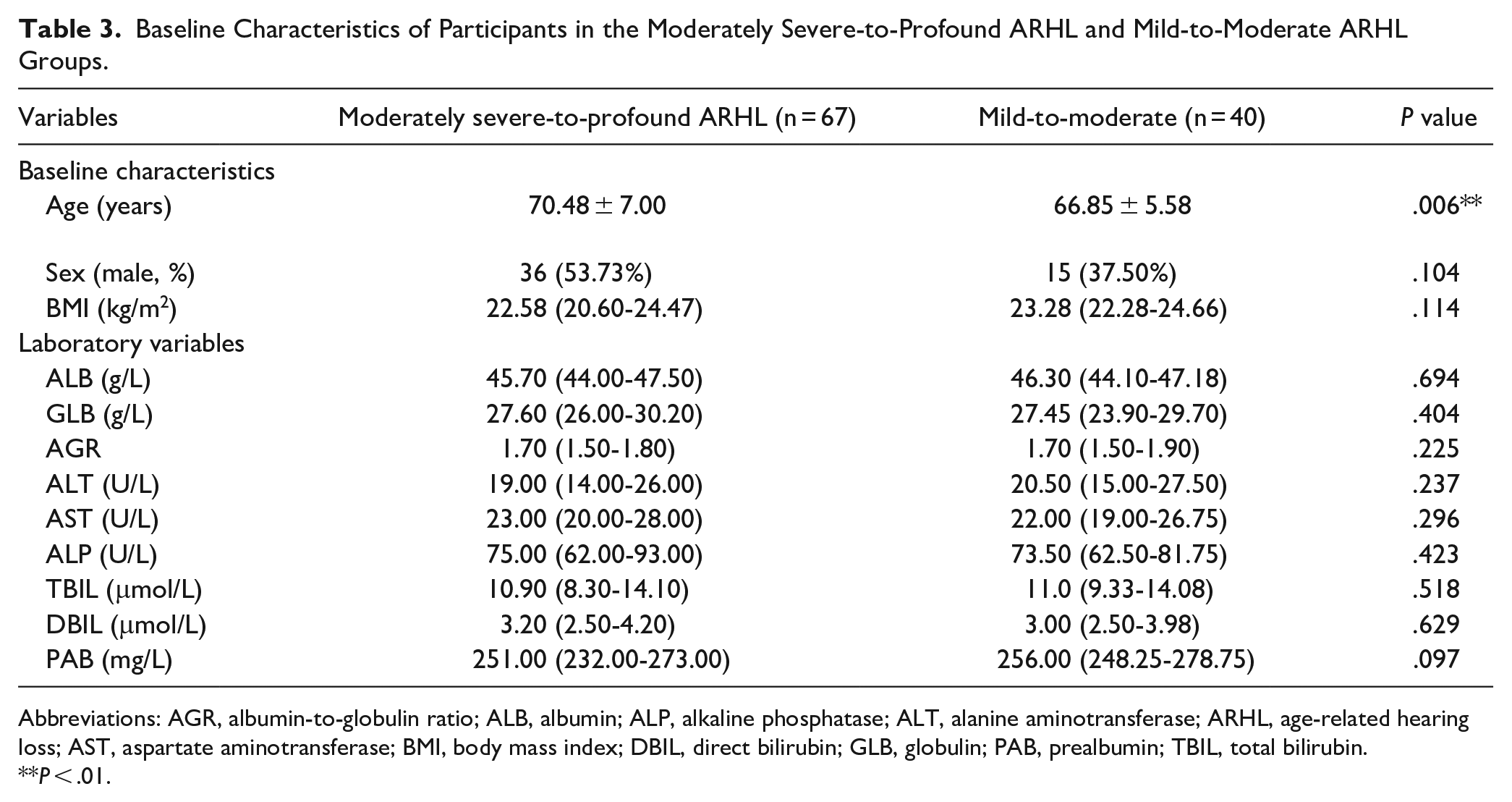
Abbreviations: AGR, albumin-to-globulin ratio; ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; ARHL, age-related hearing loss; AST, aspartate aminotransferase; BMI, body mass index; DBIL, direct bilirubin; GLB, globulin; PAB, prealbumin; TBIL, total bilirubin.
Binary Logistic Regression Analysis of the Relationship Between Liver Function Marker Levels and ARHL Severity.
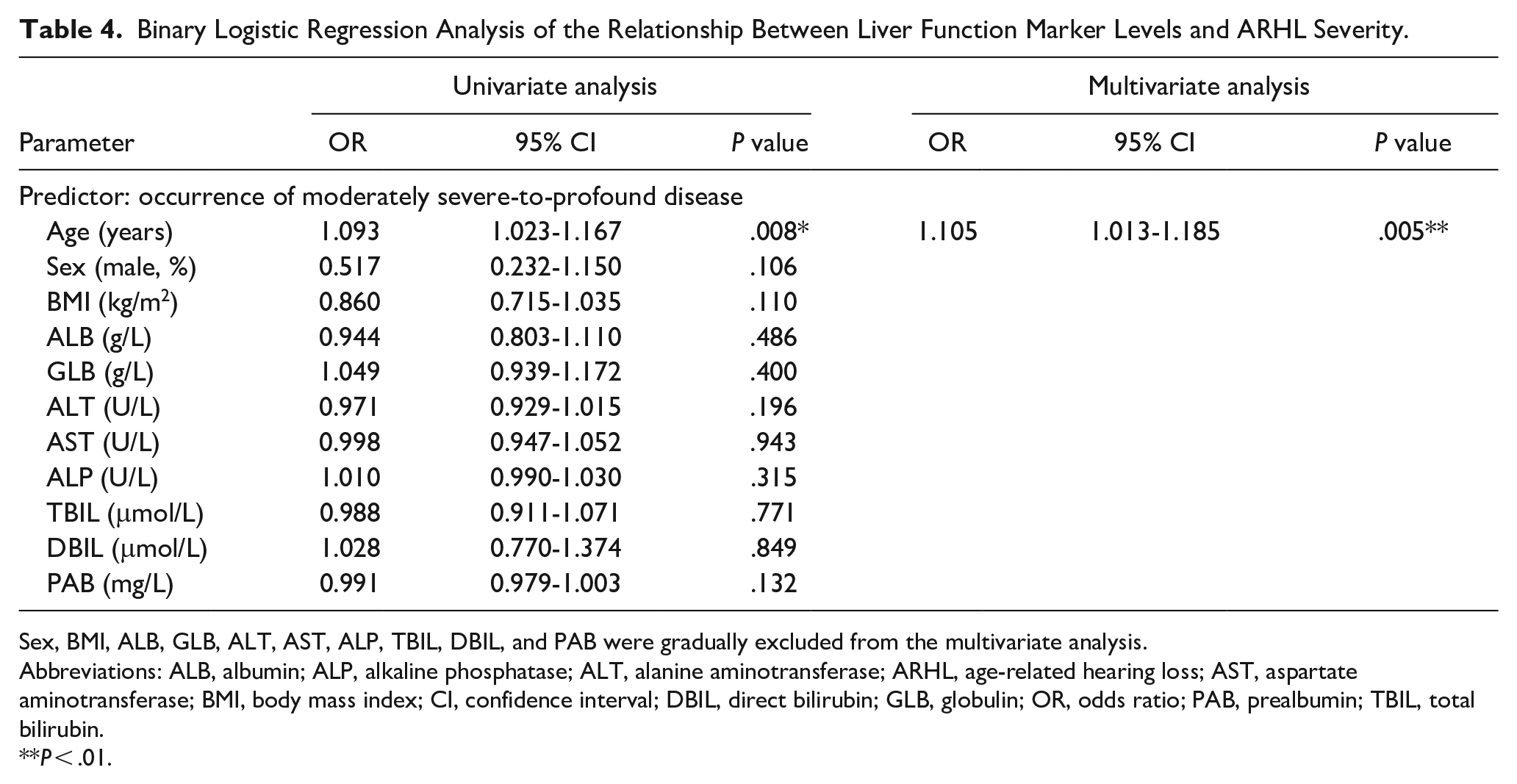
Sex, BMI, ALB, GLB, ALT, AST, ALP, TBIL, DBIL, and PAB were gradually excluded from the multivariate analysis.
Abbreviations: ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; ARHL, age-related hearing loss; AST, aspartate aminotransferase; BMI, body mass index; CI, confidence interval; DBIL, direct bilirubin; GLB, globulin; OR, odds ratio; PAB, prealbumin; TBIL, total bilirubin.
Discussion
ARHL is a progressive and degenerative disease, and most of the causes of its development are unknown. The proportion of people with ARHL is likely to increase as a result of the aging of the global population. As the body ages, the auditory system undergoes various changes. Various components of the inner ear, including hair cells, vascular strips, spiral bands, and auditory nerve fibers, experience irreversible degradation, which affects the transmission of ions and signals. 14 In addition, decreased immune function causes persistent chronic inflammation, and decreased enzymatic activity diminishes the ability of the body to remove toxins, which can ultimately result in hearing loss.15-17
ARHL and other age-related diseases share common etiologies and often involve low-grade chronic inflammation, a relationship referred to as “inflammaging.” Aging can lead to immune system dysfunction, resulting in an accumulation of unresolved inflammation in the body. 18 Several molecular pathways have been identified that are linked to aging and low-grade inflammation, such as the inflammasome, which may be triggered by age-related oxidative stress; clustering of senescent cells; and reduced autophagic activation. 19 Inflammation has been identified as a pathophysiological mechanism contributing to ARHL in numerous preclinical and population-based medical research studies. 20 Aging cochleae show increased levels of interleukin-1 beta and tumor necrosis factor alpha. 21 In addition, genetic variations in genes related to inflammatory mediators were shown to be associated with an increased risk of hearing loss in a study in older Japanese adults.22,23 Verschuur et al 23 reported that the expression of systemic inflammation-related markers, such as C-reactive protein, interleukin-6, neutrophils, and leukocytes, was upregulated in patients with ARHL and correlated with the degree of hearing impairment. Lassale et al, 24 in the English Longitudinal Study of Aging, also found a correlation between hearing loss and white blood cell counts.
The main finding of the present study is that low serum PAB levels were an independent risk factor for ARHL. PAB, also called transthyretin, plays an important role in the transport of thyroxine and retinol and is synthesized mainly by the liver. 25 Recent clinical studies investigating PAB have found that serum levels of PAB were downregulated in different pathological conditions such as inflammation, protein malnutrition, end-stage liver disease, and malignancy. 26 In addition, serum PAB levels may be predictive of respiratory diseases, burns, systemic sclerosis, and cardiac surgery.27-30 Serum PAB levels are negatively correlated with stroke severity in young individuals and are significantly reduced in patients who experience recurrent ischemic stroke.31,32 Although the biological mechanisms and the role of serum PAB in ARHL are unknown, we hypothesize that these associations are mediated by several pathways.
First, the association between PAB levels and ARHL is likely correlated with the inflammatory response. Notably, inflammation plays an important role in neurological disorders such as stroke, Parkinson’s disease, and Alzheimer’s disease, where low levels of PAB have been found, suggesting that serum PAB levels are negatively correlated with the severity of inflammation. 33 Patients with chronic pancreatitis have lower levels of PABs than healthy people, often indicating protein-calorie malnutrition. 34 PAB is believed to function within the immune system as a component of the enhanced cytokine system, exerting suppressive influences on inflammation. 35 Thus, low PAB levels indicate activation of systemic inflammation. 36 Therefore, this could also explain that, in our study, serum PAB levels were higher in the healthy control group than in the ARHL group. The anti-inflammatory effect of PAB may play an important role in the development of ARHL.
Second, the absence of hair cells and spiral ganglion neurons, reductions in the number of synaptic connections between auditory neurons, thinning of the auditory cortex, and reductions in gray matter volume were all found to be related to the development of hearing impairment in ARHL.37-40 This suggests that ARHL is inextricably linked to neural remodeling, and recent studies have shown that PAB improves nerve regeneration.41,42 Thus, low PAB levels may also adversely affect the development of ARHL by interfering with nerve regeneration.
Third, increasing evidence links oxidative stress with ARHL. 21 The development of ARHL is influenced by a combination of aging, comorbidities, lifestyle, and environmental and genetic factors, 43 and these factors increase the generation and accumulation of reactive oxygen species (ROS) in the body. 44 In addition, studies on oxidative senescence have suggested that the decline in function associated with aging is caused by damage to DNA, lipids, and/or proteins due to the accumulation of ROS. 45 In the mouse cochlea, aging was found to be associated with lipid peroxidation, oxidative DNA damage, and reduced expression of glutathione-binding proteins, as well as catalase and other antioxidant enzymes.46,47 Clinical studies have found that the prevalence of high-frequency hearing loss in older adults correlated significantly with reduced serum levels of melatonin, an ROS scavenger. 48 Interestingly, recent articles have identified PAB as a protein with a very high susceptibility to modification and oxidation and as a potential biomarker for monitoring ROS imbalance under different oxidative stress scenarios. 49 Therefore, the pathological mechanisms associated with ROS may be another reason for the usefulness of the PAB level as an indicator for ARHL.
The present results also suggested that higher age is a risk factor for the severity of ARHL. The incidence of hearing loss doubles in each decade of life starting with the second decade. 50 Furthermore, the incidence of hearing impairment with a moderate (or greater) severity is also positively correlated with age worldwide, with numbers surging from 15.4% in individuals aged 60 to 69 years to 58.2% in those aged >90 years. 1 Thus, long-term exposure to multiple risk factors for hearing loss causes the ear to accumulate more and more damage throughout the life cycle, which has a significant effect on the development and progression of ARHL.
Limitations
The present study had some limitations. First, as a preliminary study, the reproducibility and validity of the results need to be confirmed in future studies. Second, despite the use of strict inclusion and exclusion criteria, this was a single-center trial with a limited number of participants, and the findings may have been influenced by selection bias. Third, markers of liver function were measured only during study enrollment, and long-term assessments and follow-ups regarding these values and hearing ability were lacking in this study. A more comprehensive long-term assessment should be performed to accurately reflect the final hearing status of patients with ARHL.
Conclusions
In summary, this study investigated the relationships between ARHL and markers of liver function. PAB levels were negatively correlated with the degree of hearing loss, and lower PAB levels may be a risk factor for ARHL. In addition, age was an independent risk factor predicting the severity of ARHL. Therefore, these findings provide insight into preventive strategies for ARHL. Larger prospective clinical studies and further basic research are needed to validate our findings and explore the exact pathological mechanisms.
Footnotes
Acknowledgements
Thanks to all study participants.
Author Contributions
Di Qian designed and supervised the study. Yanmei Feng and Haibo Shi provided suggestions on data collection and evaluation. Yemeng He, Lin Zhou, and Quanran Lin analyzed the data. Liang Xia and Xiaoyan Chen wrote the manuscript. Tao Wang and Ying Wang participated in revising the manuscript. All authors approved the final version.
Availability of Data and Materials
The raw data supporting the conclusions of this article have been provided by the authors without undue restriction.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the First Grant (2020YFC2005201) of the Chinese National Key Research and Development Programme (2020YFC2005200), the National Natural Science Foundation of China (82171139, 82371152), and the Shanghai Municipal Commission of Science and Technology (18DZ2260200).
Ethical Approval
This study was reviewed and approved by the Sixth People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine (2021-218-[1]). The research process was fully compliant with the Declaration of Helsinki, and all participants provided written informed consent.
