Abstract
Introduction
With the implementation of the concept of multidisciplinary tumor treatment, radiotherapy, chemotherapy, or concurrent chemoradiotherapy are extensively applied in the treatment of head and neck tumors. However, more than 65% of patients with head and neck squamous cell carcinoma may have a risk of recurrence or metastasis. 1 Defect reconstruction in salvage surgery is challenging for patients with recurrence who have received neck radiotherapy.
Postoperative defect reconstruction after head and neck tumor surgery has experienced several stages of development from the era of the local flap to the current period of emphasis on both free and local flaps. The free flap is represented by use of radial forearm and anterolateral thigh flaps. However, using this type of flap has high technical requirements for microvascular anastomosis and is time-consuming. Especially in patients with postoperative radiotherapy, serious problems, including cervical fibrosis and scar formation, may occur, which increases the difficulty of vascular anastomosis. 2 Meanwhile, applying a traditional local flap, such as the pectoralis major myocutaneous flap, may cause an anterior cervical bulky deformity, which may further increase the risk of dysphagia and pharyngeal and esophageal stenoses. 3
In 1979, Lamberty 4 , for the first time, described the anatomical characteristics of the supraclavicular artery island (SAI) flap. In 1997, Pallua et al 5 conducted a detailed anatomical study on the flap and improved the corresponding preparation technology. Subsequently, the SAI flap has been widely used in the reconstruction of head and neck tumors owing to its advantages, such as easy harvest and preparation along with strong plasticity. However, the safety of the SAI flap is uncertain in the clinical application of patients with a history of neck radiotherapy owing to its blood supply and donor site location. Neck radiotherapy was listed as a relative contraindication toward utilizing the SAI flap by Sandu et al, 6 whereas other studies supported the safety of applying this flap after neck radiotherapy.7,8 Due to the small number of studies and low sample size, the safety of using the SAI flap for patients after radiotherapy needs to be further confirmed. This retrospective analysis was performed to accurately evaluate the risk of flap complications and to provide a reference for the application of this technique in patients with a history of radiotherapy.
Materials and Methods
General Data
The data of 61 cases (58 males and 3 females) of head and neck defects reconstructed with the SAI flap between January 2017 and December 2021 were retrospectively analyzed. The median age of the patients was 62 (range, 35-72) years. According to neck radiotherapy history before surgery, patients were divided into the radiotherapy group (16 cases) and the nonradiotherapy group (45 cases). Demographic data from this study have already been partially reported; however, the current objectives, analysis, and findings are novel and have never been published before.
The following data were included in the analysis: preoperative diabetes mellitus history, preoperative albumin level, body mass index (BMI), interval between radiotherapy and surgery, preoperative radiotherapy dose in patients in the radiotherapy group, flap size, postoperative complications, and treatment. Each complication was subjectively graded from mild to severe. Mild complications were defined as those that could be healed by conservative treatment through local dressing change, such as pharyngeal fistula, local infection, flap marginal necrosis, lymphatic fistula, and donor site complications. Severe complications were defined as the loss of flaps requiring secondary surgical repair.9,10 All examinations and treatments in this study were in accordance with the Helsinki Declaration.
Flap Preparation
Preoperative Doppler ultrasound was used to understand the course of the transverse carotid and supraclavicular arteries to ensure flap pedicle vascular integrity. Intraoperatively, based on the location and scope of the defect, the flap was prepared by “point line anterograde dissection.” 11 “Point” was the origin of the supraclavicular artery in the transverse carotid artery. “Line” was the extension line made from the starting point of the supraclavicular vessel for the 1 to 2 cm anterograde dissection along the blood vessel direction. The flap position was designed with the extension line used as the central axis of the flap. The skin, subcutaneous tissue, and deltoid fascia were cut from the distal end of the flap and bluntly separated under the fascia, and the tissue flap was turned up to the proximal end. With the preservation of the external jugular vein, the pedicle was dissected to the starting point of the transverse cervical artery and vein. Simultaneously, with the starting point of the transverse jugular artery as the central axis of rotation, the flap was rotated through the deep surface of the sternocleidomastoid muscle to the defect area of the neck for reconstruction. Following the subcutaneous dissociation of the incision in the donor site of the shoulder, the incision was directly closed and sutured with drainage tube placement. The dressing was changed on the third postoperative day, and the drainage tube was removed on the fifth postoperative day.
Statistical Analysis
SPSS 23.0 statistical software was used for data processing. Continuous variables that conformed to the normal distribution were expressed as ‾χ ± s, and those that did not were expressed as medians (lower and upper quartiles). Categorical variables are presented as the number of cases (percentage). In univariate analysis, the independent sample t test or Mann-Whitney U test was used for the intergroup comparison of continuous variables, whereas the intergroup comparison of unordered categorical variables was performed with the chi-square test or Fisher exact test. Univariate analysis was performed on variables between the 2 groups. A P value of <.05 was considered statistically significant.
Results
Comparison of General Data Between the 2 Groups
Overall, the gender, age, BMI, presence of diabetes mellitus, preoperative albumin level, and flap size between the preoperative irradiated and nonirradiated group had no significant difference (Table 1). The most common type of SAI flap for surgical reconstruction in 2 subgroups was hypopharyngeal carcinoma. Specific types of head and neck defects reconstructed in the 2 groups are shown in Table 2.
Comparison of General Data in Patients With Head and Neck Defects Reconstructed With the Supraclavicular Artery Island Flap Between the 2 Groups.
Abbreviation: BMI, body mass index.
Fisher’s exact test.
The flap size was compared using the Mann-Whitney U test, and the statistic was U value. The gender and diabetes mellitus were compared using the Fisher’s exact test. The age, preoperative albumin level, and BMI were compared using the independent sample t test.
Types of Supraclavicular Artery Island Flap for the Head and Neck Defect Reconstruction.
Flap Complications and Treatment
Among the 61 patients, 3 (4.9%) developed severe complications (flap loss) and 8 (13.1%) showed mild complications, including pharyngeal fistula, partial necrosis, lymphatic fistula, and donor site complications in 3 (4.9%), 2 (3.3%), 1 (1.6%), and 2 (3.3%) patients, respectively. No significant difference was observed in the total incidence of complications or in the incidence of mild or severe complications between the radiotherapy and nonradiotherapy groups (Table 3).
Comparison of the Complication Rates Between the Radiotherapy and Nonradiotherapy Groups.

In the radiotherapy group, 1 patient developed complete flap necrosis, one of the severe complications; the patient was a patient with recurrence following concurrent chemoradiotherapy of hypopharyngeal cancer. The patient received radiotherapy at a dose of 73 Gy/39 fx, and the interval between radiotherapy and surgery was 0.6 years. Following flap necrosis, the patient was provided with a pectoralis major myocutaneous flap for reconstruction. One patient with recurrent laryngeal cancer developed a pharyngeal fistula. The patient received radiotherapy at a dose of 70 Gy/35 fx, and the interval between radiotherapy and surgery was 6 years. The pharyngeal fistula had healed after dressing change. Furthermore, another patient with pharyngeal fistula after surgery for hypopharyngeal carcinoma developed marginal flap necrosis, which also healed after dressing change. The patient received radiotherapy at a dose of 60 Gy/33 fx, and the interval between radiotherapy and surgery was 0.66 years.
In the nonradiotherapy group, 2 patients developed complete flap necrosis. Among them, 1 patient with hypopharyngeal cancer (T4aN1M0) received reconstruction with a pectoralis major myocutaneous flap following flap necrosis. Another patient with Merkel cell carcinoma of the auricle received reconstruction of the defect of the parotid gland; however, vascular crisis occurred on the second postoperative day. Through emergency exploration and vascular pedicle decompression, the patient was found to have postoperative skin flap necrosis, and free flap repair was performed again. All patients with mild complications healed after conservative treatment and dressing changes.
Factors Related to Flap Complications
In the univariate analysis, a low preoperative albumin level was the only risk factor for postoperative complications after reconstruction using the SAI flap (P < .05). No significant difference was observed in the effect of radiotherapy on postoperative complications between the 2 groups (P = 1.000; Table 4).
Relationship Between Related Factors and Flap Complications.

Abbreviation: BMI, body mass index.
Fisher’s exact test.
The flap size was compared using the Mann-Whitney U test, and the statistic was U value. The gender, diabetes mellitus, and preoperative radiotherapy were compared using the Fisher’s exact test. The age, preoperative albumin level, and BMI were compared using the independent sample t test.
Typical Case
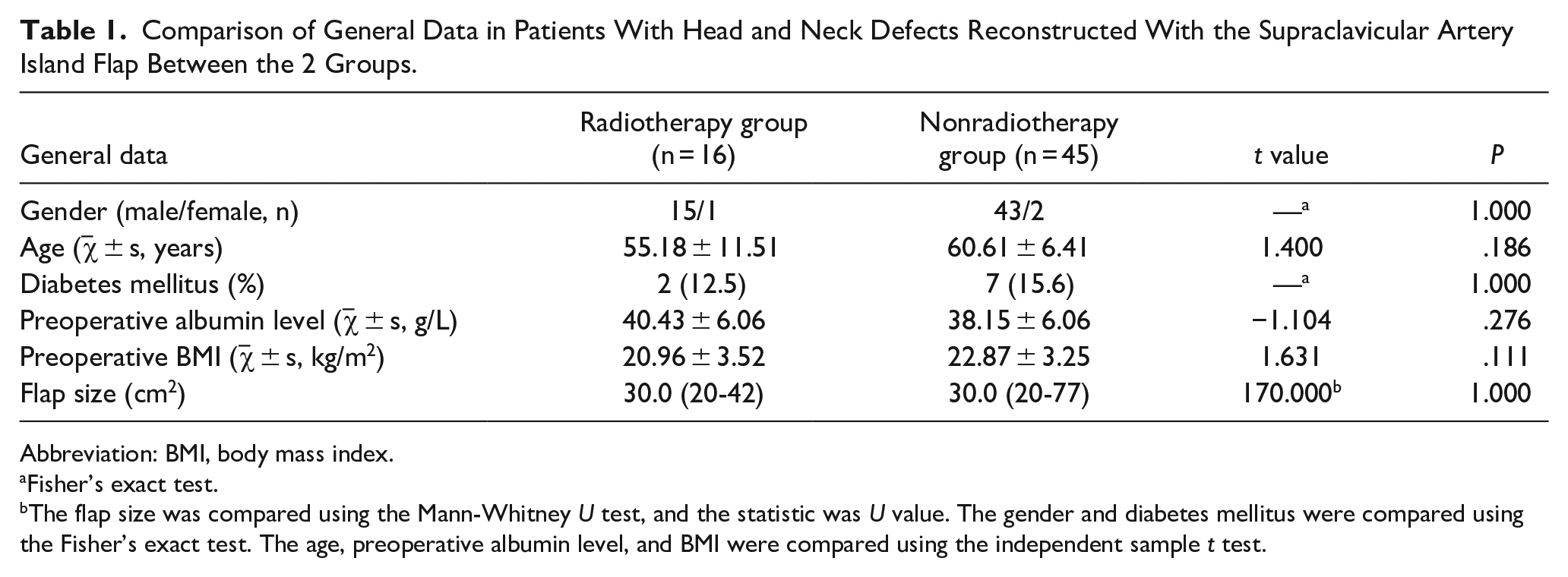
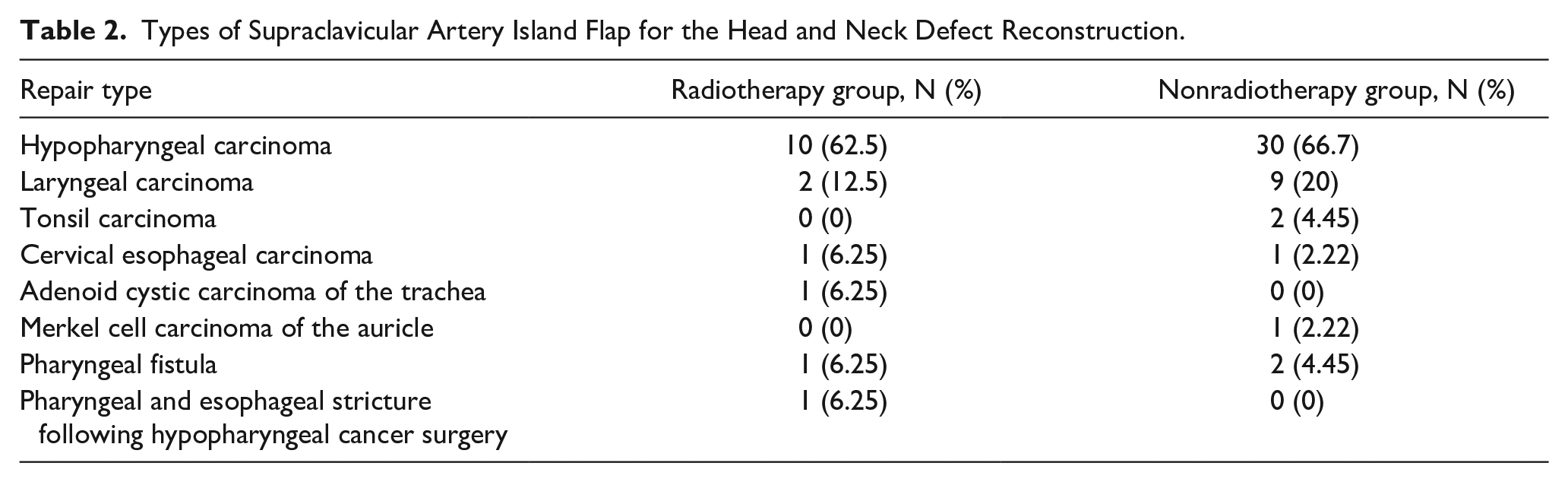
A 64-year-old male patient underwent total laryngectomy with partial pharyngectomy + bilateral selective neck dissection + tracheostomy due to hypopharyngeal carcinoma (T3N1MO) and was prescribed a soft liquid diet after surgery. Thirty-five days postoperatively, the patient received radiotherapy at a dose of 60 Gy/30 fx. Two months after radiotherapy, the patient developed dysphagia with gradual aggravation, which prompted a visit to our hospital. Physical examination showed that the skin of the neck was dark in color and hard in texture given post-tracheostomy status. Gastroscopy showed that the distance to the esophageal inlet was approximately 16 cm from the incisor, with a narrow inlet that did not allow endoscopic passage and with no erosion or mass. Imaging examination showed no obvious mass in the larynx after surgery. Ultrasound vascular labeling showed that the transverse carotid artery could be traced; however, the supraclavicular artery could not be displayed. Subsequently, under general anesthesia, the patient underwent endoscopic exploration of the hypopharyngeal esophagus for collecting pathological tissue + esophageal stenosis reconstruction + local cervicothoracic flap repair. No tumor was found in the local pathological examination during surgery. The length of esophageal stenosis was approximately 2 cm, and the absent esophageal cavity was approximately 4 cm × 3 cm. The “point line anterograde dissection” was used to determine the development and direction of the supraclavicular artery, prepare the SAI flap (6 cm × 7 cm), and reconstruct the tubular structure of the esophagus. Direct suture by pulling compresses the SAI flap. Therefore, in our practice, the perforator flap of the deltoid branch of the thoracoacromial artery was used to repair anterior cervical skin defects (Figure 1). The wound healed well, and the patient returned to a normal diet (Figure 2).
Patients with “esophageal stenosis” following the comprehensive treatment of hypopharyngeal cancer. (A) Changes after neck radiotherapy; (B) esophageal defect exploration; (C) supraclavicular artery dissection; (D) SAI flap; (E) perforator flap of the deltoid branch of the thoracoacromial artery. SAI, supraclavicular artery island.
Comparison of the degree of esophageal stricture before and after surgery. (A) Preoperative gastroscopy; (B) preoperative imaging examination; (C) postoperative barium esophagography.
Discussion
Radiotherapy is the primary treatment for head and neck squamous cell carcinoma. However, radiotherapy may induce cervical fibrosis and scar formation, damage vascular endothelial cells, and cause blood supply microcirculation dysfunction, leading to increased difficulty of surgical defect repair.12,13 Meanwhile, radiotherapy may also lead to an impact on the release of growth factors to restrict postoperative healing and increase the incidence of pharyngeal fistula and poor healing. 14 There are multiple choices for reconstruction during salvage surgery for patients with tumor recurrence following radiotherapy, and they depend on the degree of local involvement and extension of the tumor and the practical experience of surgeons.
The SAI flap is a fasciocutaneous flap with a vascular pedicle that is extensively used in head and neck defect reconstructions, including the mouth floor, tongue, oropharynx, hypopharynx, cervical esophagus, face and neck skin. 15 Moreover, we believe that the SAI flap is a suitable choice for head and neck defect reconstruction following radiotherapy. First, the SAI flap has a stable blood supply and simple preparation, with no requirement for microvascular anastomosis, such that the surgery can be completed in the same surgical field. Second, the SAI flap is adjacent to the surgical area and can be used to prepare the contralateral/bilateral SAI flap or to combine with other flaps in the reconstruction of a composite defect.
Furthermore, even for patients with a radiotherapy history, the SAI flap is safe and reliable for head and neck defect reconstruction. The major clinical complications of SAI flap application include total/partial flap necrosis, infection, wound dehiscence, anastomotic and pharyngeal fistulas, and esophageal stenosis. 16 In this study, the rate of complete SAI flap necrosis was 4.9%, whereas that of mild complications was 13.1%, which was close to the rate observed for previously reported patients without radiotherapy (complete necrosis rate, 0%-5.6%; partial necrosis rate, 4.2%-21.8%).17-19 Su et al 20 reported a complication incidence of 38% when applying an SAI flap in patients with previous radiotherapy and neck surgery. 20 Razdan et al 7 reported that complications occurred in 5 (23%) of the 22 cases when the SAI flap was used for patients undergoing surgery after neck radiotherapy. 7 In our study, no difference was noted in the incidence of complications between the radiotherapy and nonradiotherapy groups. It is suggested that radiotherapy history may not be a contraindication for SAI flap application.
To decrease the incidence of postoperative SAI flap complications in patients with a history of head and neck radiotherapy, combined with our experience in 61 cases of SAI, special attention should be given to the following aspects. First, preoperative Doppler ultrasound should be used to understand the course of the transverse carotid and supraclavicular arteries. Digital subtraction angiography is indicated for patients with a previous surgical history and who are dissatisfied with ultrasonic imaging. Second, the transverse carotid artery and supraclavicular artery should be redetermined during the surgery. In flap preparation using the “point line anterograde dissection,” the dissection is initially performed along the transverse carotid artery to determine the existence of the supraclavicular artery, and the island flap is subsequently designed along the direction of the blood vessel (the central axis). This technique can not only screen out supraclavicular artery hypoplasty cases but also avoid damage to the vascular pedicle. 11 Third, for patients without lymph node metastasis in the preoperative evaluation, the posterior edge of the sternocleidomastoid muscle should not be exceeded during the denudation of the vascular pedicle; meanwhile, the pedicle should be simultaneously dissected to the starting point of the transverse jugular artery and vein with the starting point as the central axis of rotation. In this manner, the suture tension in the surgical area is reduced. Finally, when repairing penetrating defects (eg, pharyngeal fistula), reconstruction with double flaps (mucosal and skin sides) can reduce the risk of recurrence and poor healing of the pharyngeal fistula.
Generally, patients with malignant tumors (head and neck tumors in particular) usually have nonspecific systemic symptoms, such as loss of appetite, eating disorders, weight loss, and cachexia. This may further lead to a decrease in the serum albumin level and a decline in physical condition. Previous studies reported an increased postoperative surgical site infection rate in patients with significant preoperative weight loss and lower serum albumin levels than in those with normal levels.13,14 Similarly, in our study, a low preoperative albumin level was identified as an independent risk factor for postoperative surgical site complications. Therefore, early nutritional support and correction of low albumin levels can alleviate delayed wound healing and reduce complications. Accordingly, it is recommended that clinicians improve patients’ protein intake by prescribing consumption of protein-rich food or whey protein powder according to the patients’ condition.
Surgical treatment is the preferred treatment for patients with recurrent head and neck tumors after radiotherapy. However, poor tissue healing is noted following radiotherapy, making surgical treatment prone to complications, such as development of anastomotic and pharyngeal fistulas. In this regard, tissue reconstruction with higher safety and better blood supply should be selected to obtain the optimal therapeutic effect with the least trauma. Significantly, the SAI flap has the advantages of simplicity of surgical procedure, reliable blood supply, large and thin donor site, and absence of influence on the function of the donor site. It is especially suitable for patients with head and neck skin and mucosal defects who cannot receive microsurgery and is worthy of clinical application. The main disadvantage of limiting its impact in this investigation is the small size of the subject population (n = 16), which may result in insufficient statistical efficiency. However, the ratio of 2 groups was approximately 1:3, to avoid their effects on postoperative complications. This generated an impetus to perform prognosis studies on larger sample size populations.
Conclusion
Our data suggest that preoperative radiotherapy does not increase the risk of SAI flap postoperative complications compared with surgical reconstruction alone. In these individuals, adequate preoperative assessment and dietary support may lower the occurrence of postoperative complications.
Footnotes
Author Contributions
This study has been authorized for publishing by all authors. Jing Zhou: mainly drafted and revised the original manuscript. Yiming Ding: was responsible for data acquisition, statistical analysis, and data visualization. Erdong Zuo: assisted in the completion of original manuscript. Jun Wu: gave substantial suggestions on statistics analysis. Yonggang Jin: was responsible for project administration. Xiaohong Chen: contributed to the study conceptualization, design, and supervision.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors report no conflicts of interest. The article has been read and approved by all the authors, and that each author believes that the articlet represents honest work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional guidelines on human research and with the Helsinki Declaration of 1975, as revised in 2008.
