Abstract
Introduction
Neuroendocrine tumors (NETs) are a rare type of tumor that originates from neuroendocrine cells, which are characterized by their ability to synthesize and secrete neuropeptides and hormones as well as the expression of neuroendocrine markers such as synaptophysin and chromogranin. 1 These tumors can occur at any location but are commonly found in the lungs, pancreas, rectum, appendix, and small intestine. On the other hand, laryngeal neuroendocrine neoplasms (LNEN) are even rarer, accounting for less than 1% of all laryngeal tumors, 2 and primarily manifest in the supraglottic region (57.9%-95.5%). 3 Symptoms in affected individuals vary based on the location and size of the tumor, with common local manifestations including discomfort in the pharynx and larynx, hoarseness, and swallowing difficulties. In addition to complications arising from the space-occupying effects of the tumor, attention should also be directed toward complications resulting from the endocrine functions of NETs, such as paraneoplastic syndromes. While paraneoplastic syndromes are less common in LNEN compared to NETs occurring in other locations, there have been reported cases. 4 Risk factors for LNEN include smoking, human papillomavirus (HPV) infection, among others.3,5,6 As understanding of LNEN deepens, its classification has evolved, leading to occasional challenges for clinicians during certain periods. 7 The evolution of classification is shown in Table 1.The most recent classification standard is the 2022 WHO Classification of Head and Neck Neuroendocrine Neoplasms, which aligns with the classification for gastrointestinal NETs.8,9 From the perspective of the evolution of NETs classification, there is a gradual trend toward unification in the classification of all NETs, including those of the digestive and respiratory systems.
Classifications of Laryngeal Neuroendocrine Neoplasms.

Abbreviations: NET, Neuroendocrine tumor; WHO, World Health Organization.
Methods
This study is a single-center investigation, where all patients underwent surgical treatment, and the authors were directly involved in patient care. The surgeries were collectively performed as a treatment group. On admission, comprehensive laboratory tests, contrast-enhanced computed tomography scan of the neck and chest, neck ultrasound, electronic laryngoscopy with narrow-band imaging, pathological biopsies, and other examinations were conducted to assess the primary tumor, neck lymph nodes involvement, and the presence of distant metastasis. Following a multidisciplinary team discussion (MDT), individualized treatment plans were formulated based on TNM staging and the patient’s specific condition. If the primary tumor could be completely excised orally, transoral laser microsurgery (TLM) was performed. In cases where the tumor was too large for complete oral excision, open partial supraglottic laryngectomy (OPSL) was undertaken based on the tumor characteristics. For cases with cTxN1~3M0, neck lymph node dissection was carried out. If cT1N0M0, observation or ipsilateral neck lymph node dissection was considered. Postoperatively, additional radiotherapy and chemotherapy were administered based on the tumor’s classification and TNM staging at the time, following a treatment regimen similar to that for laryngeal squamous cell carcinoma (LSCC). In November 2023, after a reexamination of pathological slides by head and neck surgical pathologists according to the 2022 WHO Classification of Head and Neck Neuroendocrine Neoplasms, the patients were reclassified. Patient demographics, smoking index, P16 expression, preoperative biopsies, pathological classification at the time of surgery, current pathological classification, Ki67/mitoses, surgical methods, and prognosis were recorded and analyzed using SPSS 26 for Kaplan-Meier survival analysis. The basic characteristics and prognosis of the 11 patients are summarized in Table 2. The pathological classification and prognosis of 11 patients were illustrated using a Swimmer plot, as shown in Figure 1.
Characteristics of Patients, Tumors, and Treatment.
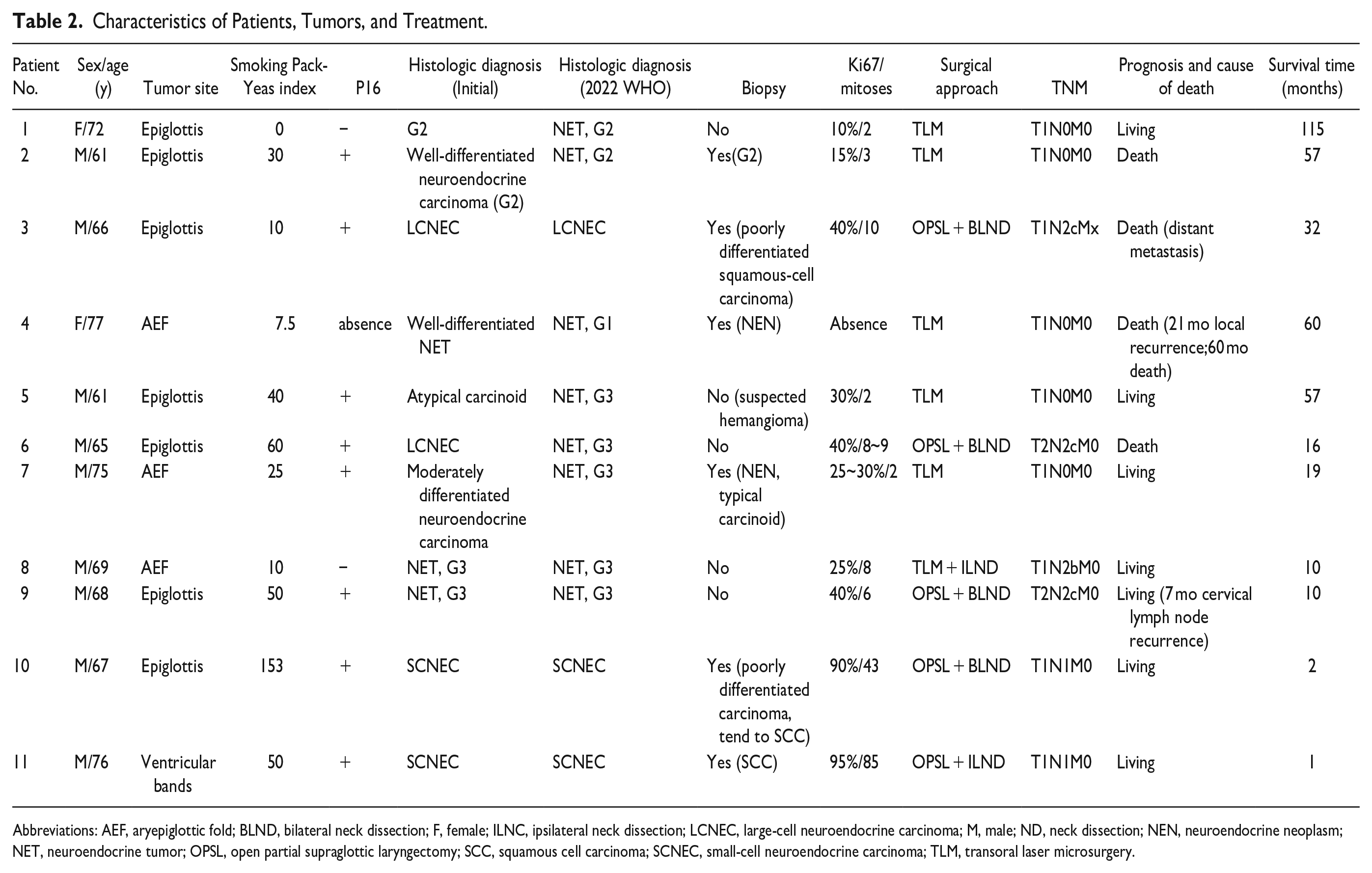
Abbreviations: AEF, aryepiglottic fold; BLND, bilateral neck dissection; F, female; ILNC, ipsilateral neck dissection; LCNEC, large-cell neuroendocrine carcinoma; M, male; ND, neck dissection; NEN, neuroendocrine neoplasm; NET, neuroendocrine tumor; OPSL, open partial supraglottic laryngectomy; SCC, squamous cell carcinoma; SCNEC, small-cell neuroendocrine carcinoma; TLM, transoral laser microsurgery.
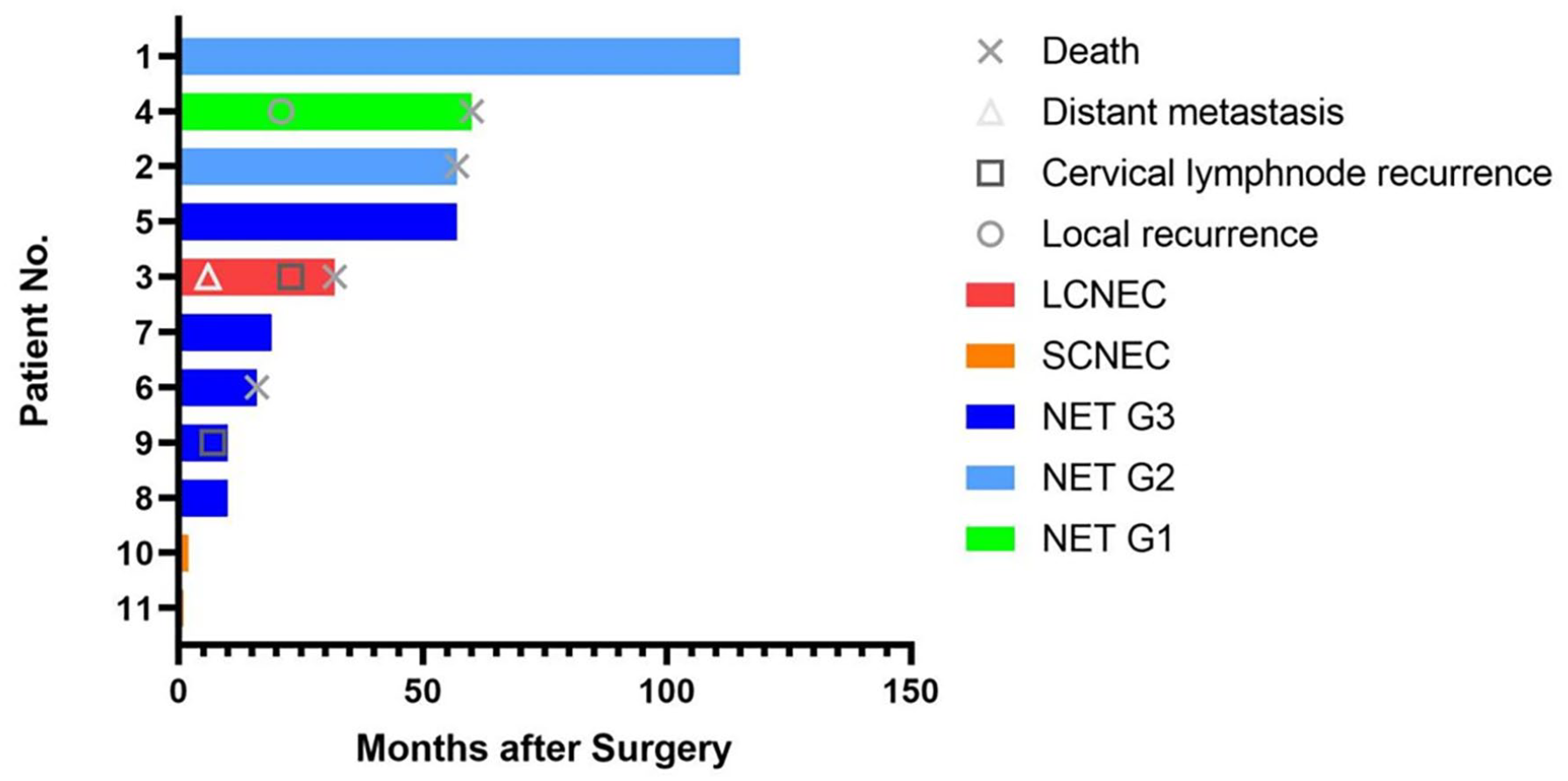
Swimmer plot. Time 0 represents the date of surgery, axis represents survival time (months), Y-axis represents patient No.
Results
The follow-up period ranged from 1 to 115 months. Among the cases, there were 8 NETs, with 5 surviving and 3 deceased. One patient died due to local recurrence (discovered 21 months postoperatively, and succumbed 60 months after surgery), another due to the underlying condition, and 1 due to distant metastasis. There were also 3 NEC cases, including 1 large-cell neuroendocrine carcinoma (LCNEC; deceased due to distant metastasis) and 2 small-cell neuroendocrine carcinomas (SCNECs). The Kaplan-Meier survival curve is depicted in Figure 1. All 11 tumors originated in the supraglottic region, with 7 originating in the epiglottis, 3 in the aryepiglottic fold (AEF), and 1 in the ventricular band region. Surgical approaches included TLM in 6 cases and OPSL in 5 cases. Preoperatively, 6 cases underwent pathological biopsy, with 3 accurate reports and 3 diagnosed as squamous cell carcinoma (SCC). Among the 11 patients, 10 were smokers, with 7 being heavy smokers. P16 was tested in 10 patients, with 8 testing positive (80% positivity rate).
In terms of pathological classification and prognosis:
Kaplan-Meier survival analysis for the 11 cases is presented in Figure 3, with an overall 5 year survival rate (OS) of 25.7%.
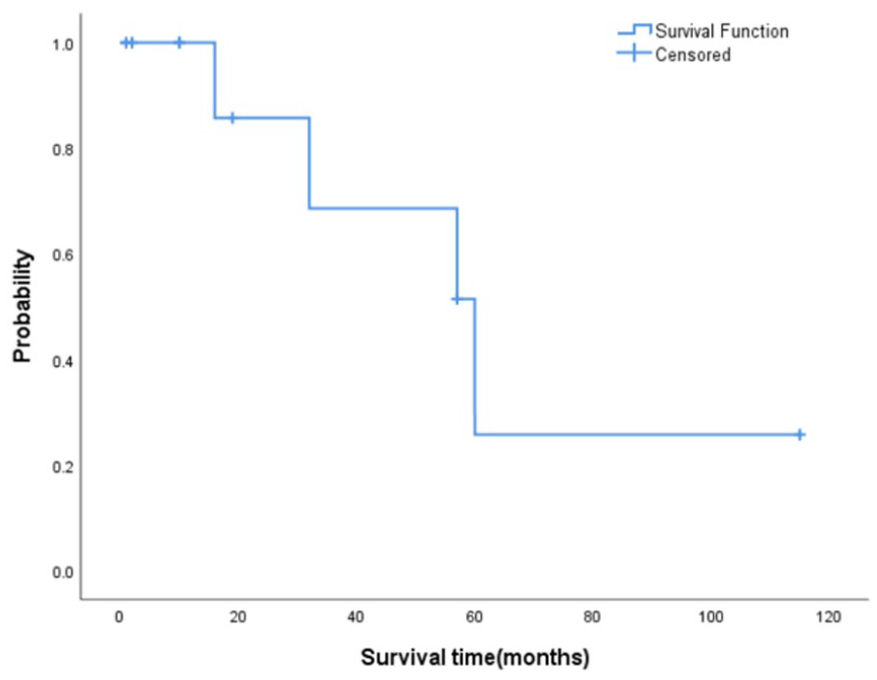
Survival curve of 11 patients with LNEN. LNEN, laryngeal neuroendocrine neoplasms.
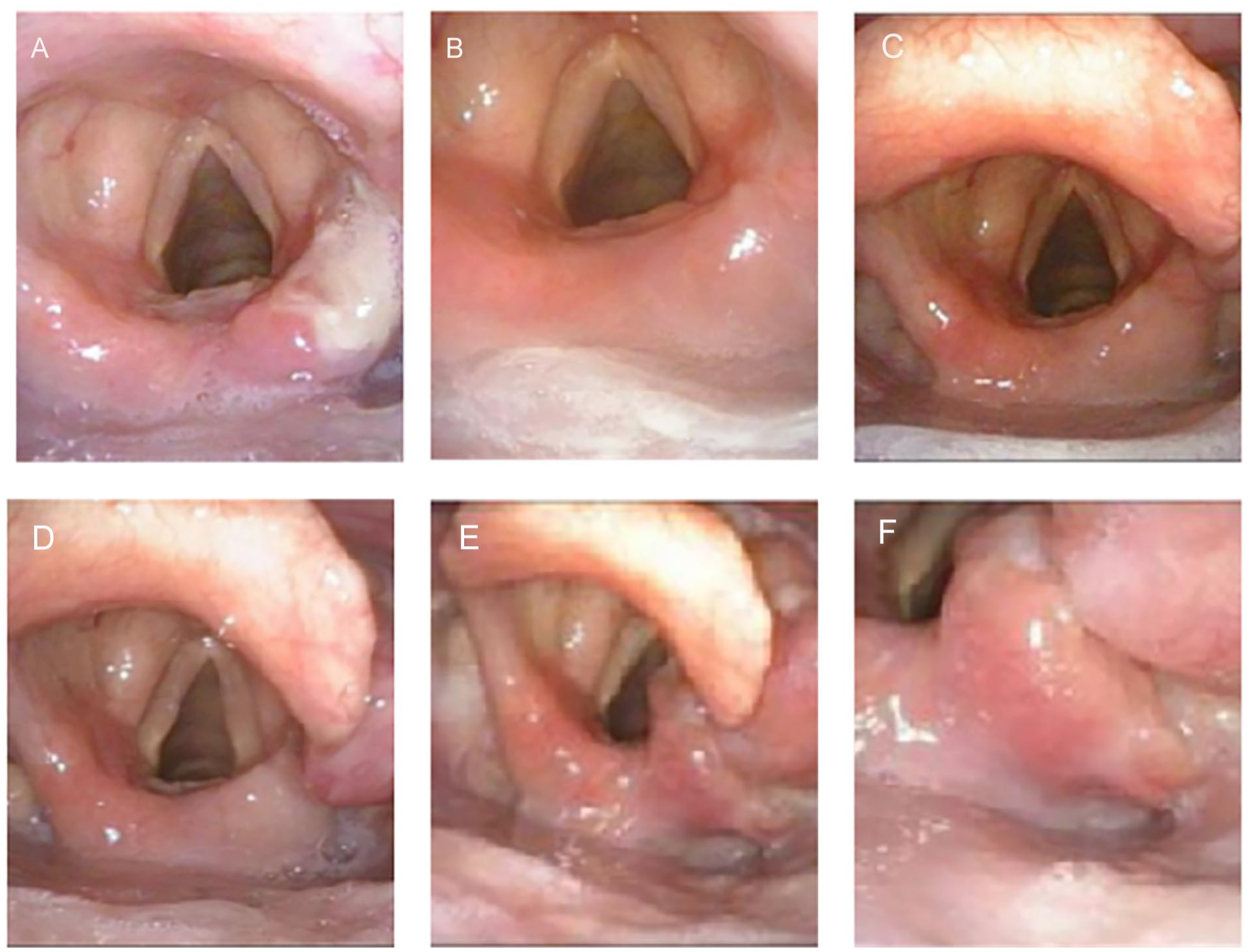
The laryngoscope of NET G1. (A) presurgery, originating from the right aryepiglottic fold; (B) 2 months after surgery; (C) 11 months after surgery; (D) 21 months after surgery, local recurrence; (E and F) 47 months after surgery, local recurrence is more pronounced. NET, neuroendocrine tumor.
Discussion
Within the head and neck region, the larynx emerges as the predominant site for neuroendocrine carcinoma (NEC). Despite constituting less than 1% of all laryngeal tumors, laryngeal NEC (LNEC) stands out as the most prevalent nonsquamous subtype. 16 The supraglottis, particularly the AEF, demonstrates a strong predilection, accounting for 82% of cases Bal et al’s study. 17 In our research, 64% (7/11) originated from the epiglottis, while 27% (3/11) originated from the AEF. The survival rate of NEC occurring in the larynx is higher compared to that arising in other locations, albeit lower than the survival rate of LSCC. 2 Risk factors for LNEC include smoking and HPV infection, with the majority of patients having a history of tobacco use (73.3%-94.2%). 3 The respiratory systems of the larynx and lungs, both exposed to similar tobacco and environmental stimuli, see a higher prevalence of small-cell lung cancer among pulmonary NETs. Rekhtman reported an association between small-cell lung cancer and smoking, and with declining smoking rates, a decrease in small-cell lung cancer incidence was observed, while non-small-cell lung cancer rates increased. 8 In our study, patients with SCNEC were heavy smokers and exhibited P16 positivity. However, laryngeal NETs predominantly manifest as atypical carcinoids, necessitating further clarification of associated risk factors. During electronic laryngoscopy, some NETs may resemble vascular lesions, requiring careful differentiation. Among our cases with preoperative pathological biopsy results, there were 2 misdiagnoses, yielding a misdiagnosis rate of 40%. This underscores the importance of preoperative biopsies in diagnosing LNEC, emphasizing the need for experienced practitioners to obtain samples and skilled pathologists for accurate pathological diagnosis.
NETs originate from neuroendocrine cells (peptidergic neurons), and in the larynx, these cells are scattered within the mucosal epithelium. Laryngeal NETs are classified into 2 major categories based on their tissue origin: epithelial and neurogenic. 18 Prior to the 2022 WHO Classification of Head and Neck Neuroendocrine Neoplasms, epithelial laryngeal NETs were mainly classified as carcinoid tumors, atypical carcinoids, etc. 19 However, various diagnostic terms have been applied to what is now recognized as neuroendocrine tumor/NET (a well-differentiated epithelial NEN) and NEC (a poorly differentiated epithelial NEN; Table 1). The classification of these NETs of the larynx has been a subject of considerable debate over the years, primarily revolving around terminology and classification systems. Various systems have been proposed, with some advocating for consistency with classification systems for neuroendocrine neoplasms in other organ systems (lung and gastrointestinal), while others propose unique systems for the head and neck. 20 The lack of uniform standards, coupled with confusion in classification and a limited number of cases, has impeded in-depth research on laryngeal NEC. As a result, diagnostic standards and treatment approaches often borrow from those established for NETs in the lungs and gastrointestinal tract.
More recently, a unified IARC/WHO terminology framework for all neuroendocrine neoplasms, irrespective of their site of origin, has been proposed.21,22 The 2022 WHO Classification of Head and Neck Neuroendocrine Neoplasms (see Table 1) categorizes tumors based on differentiation into NET and NEC. NETs are further stratified into G1, G2, and G3 based on the presence of cellular necrosis, mitotic figures, and Ki-67 index. NECs are classified into SCNEC and LCNEC. The criteria for mitotic figures are set at 50 per high-power field, with Ki-67 expression assessed in the strongest staining area, counting 500 to 2000 tumor cells, and recalculating. In cases where mitotic figures and Ki-67 indices do not align, a higher grade is recommended. 23 Immunohistochemical stains for several antibodies (INSM1, synaptophysin, chromogranin A) can aid in confirming neuroendocrine differentiation based on appropriate morphology. 24
In our center’s study involving NETs with accompanying neck lymph node metastasis, we employed a method that separately counts the primary tumor and lymph nodes. This classification method provides clear counting criteria, transforming empirical practices into measurable objective data. It has standardized diagnostic criteria, reduced human errors, and facilitated the collection, exchange, and statistical analysis of multicenter cases. However, this classification lacks targeted research on laryngeal NEC’s ability to differentiate between different tumor biological behaviors and prognoses, necessitating large-scale case studies and time validation.
The treatment outcomes are strongly influenced by the histological subtype of laryngeal NEC, with a 5 year disease-specific survival of 100% for typical carcinoid tumors, 52.8% for atypical carcinoid tumors, 19.3% for SCNEC, and 15.3% for LCNEC. 3 In our study, the overall 5 year survival rate was 25.7%. However, the limitations of a small sample size and a relatively short follow-up duration introduce potential errors into the results.
The therapeutic options and prognosis vary significantly based on the pathological types and TNM staging of laryngeal NEC. Combining our 11 cases with existing literature, NET G1 exhibits a relatively benign biological behavior, characterized by slow tumor growth and a low tendency for lymph node metastasis. The preferred treatment approach involves surgical resection, ideally employing transoral laser surgery, which often yields favorable outcomes.2,25 For NET G2, concurrent neck lymph node dissection is recommended during surgical resection, even in the absence of radiological evidence of lymph node metastasis. This precaution is due to early-stage lymph node metastasis that may go undetected by imaging studies. NET G3, when treated with partial laryngeal resection, has shown effective control of the primary tumor. However, it is often associated with lymph node metastasis, especially when double-neck lymph node involvement is present, leading to a poorer prognosis. The postoperative debate revolves around whether to combine surgery with radiotherapy or chemotherapy.
SCNEC and LCNEC exhibit lower survival rates, but they can benefit from chemotherapy.3,26 The treatment approach should be personalized, considering the TNM staging and involving MDTs. 27 Immunotherapy is still in the early stages of clinical exploration. Horn et al’s report suggests that immune checkpoint inhibitors have demonstrated efficacy in treating extensive-stage small cell lung cancer when combined with etoposide and platinum, replacing chemotherapy alone as the standard first-line treatment. 28 In summary, a comprehensive treatment strategy, including surgery, radiotherapy, chemotherapy, and immunotherapy, should be tailored based on the tumor subtype and TNM staging.
The 2022 WHO classification has provided a clear classification standard for head and neck NET, categorizing them into NET G1, G2, G3, SCNEC, and LCNEC. The prognosis of laryngeal NETs is closely associated with pathological subtyping and TNM staging. However, given the rarity of LNEN, further analysis with larger datasets, following a standardized pathological classification, is essential to establish a robust foundation for treatment planning.
Conclusions
LNEN exhibit a poorer prognosis. Beyond traditional approaches involving surgery, radiotherapy, and chemotherapy, novel therapeutic modalities are imperative to enhance survival rates. The prognosis of laryngeal NETs is closely associated with the latest pathological classification and TNM staging. For a more detailed and specific clinical staging, further research involving multicenter large-scale data is needed.
Supplemental Material
sj-png-1-ear-10.1177_01455613241249270 – Supplemental material for Laryngeal Neuroendocrine Neoplasms: Analysis of 11 Cases in a Single-Center Study
Supplemental material, sj-png-1-ear-10.1177_01455613241249270 for Laryngeal Neuroendocrine Neoplasms: Analysis of 11 Cases in a Single-Center Study by Jianlin Yue, Qianqian Zhao, Suyuan Sun, Xinliang Pan and Dayu Liu in Ear, Nose & Throat Journal
Supplemental Material
sj-png-2-ear-10.1177_01455613241249270 – Supplemental material for Laryngeal Neuroendocrine Neoplasms: Analysis of 11 Cases in a Single-Center Study
Supplemental material, sj-png-2-ear-10.1177_01455613241249270 for Laryngeal Neuroendocrine Neoplasms: Analysis of 11 Cases in a Single-Center Study by Jianlin Yue, Qianqian Zhao, Suyuan Sun, Xinliang Pan and Dayu Liu in Ear, Nose & Throat Journal
Footnotes
Author Contributions
All authors were involved in the conception. JY wrote the first draft, which was reviewed and revised by XP and DL. QZ provided the pathological content in this article. SS is responsible for patient follow-up. All authors have read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
