Abstract

Significant Statement
Fetal rhabdomyoma (FRM) is an uncommon benign soft tissue tumor showing skeletal muscle differentiation. It occurs predominantly in the head and neck region of infants and young children. We describe an adult patient presenting with dysphonia and cough for a polypoid glottic FRM in which a SUFU gene deletion was found by molecular analysis. When dealing with a laryngeal polypoid lesion in an adult, FRM should be kept in mind as a very rare diagnosis.
Rhabdomyoma is a benign soft tissue neoplasm showing skeletal muscle differentiation.1,2 Three subtypes (ie, fetal, adult, and genital) are distinguished. The fetal subtype is the rarest, occurs predominantly in the head and neck of infants and young children and in approximately 25% of cases is congenital.1,2 Less than 30 cases of FRM have been reported in patients older than 20 years. 3 Even though fetal rhabdomyoma (FRM) is usually sporadic, it may also occur in association with nevoid basal cell carcinoma syndrome,4-6 a genetic syndrome associated with monoallelic loss-of-function germline mutations of hedgehog signaling pathway (HHSP)-related genes, especially PTCH1 (OMIM #109400) and SUFU (OMIM #620343).7,8 Notably, mutations of HHSP-related genes have been reported also in sporadic FRMs.6,9
Histologically, FRM has been distinguished into 2 variants (classic/myxoid and juvenile/cellular) based on the degree of cellularity, amount of myxoid stroma, and maturity of muscle cells; however, this distinction does not affect the prognosis and the 2 histologic variants frequently coexist blending into each other.1,2
We report here the case of a 31-year-old man presenting at the othorhinolaryngology clinic for a 5 months history of dysphonia and cough. He denied smoking history and previous laryngeal surgery. A flexible transnasal pharyngo-laryngoscopy showed a nonulcerated polypoid lesion involving the glottis (Figure 1A). The polyp disappeared in subglottis with inspiration and appeared in supraglottis with expiration and was interpreted as benign even though a specific diagnosis was not made. It was excised with a carbon dioxide laser. Pathologic examination revealed a soft, unencapsulated, and paucicellular polypoid lesion composed of primitive spindle cells and more differentiated skeletal muscle fibers dispersed in a loose stroma (Figure 1B and C). Mitosis, atypia, necrosis, and a “cambium layer” were absent. Immunohistochemical analysis, performed as described previously,10,11 showed immunoreactivity of both neoplastic cell components for desmin (Figure 1D). The more differentiated skeletal muscle fibers were also immunoreactive for myoglobin. The proliferation index (Ki-67) was less than 5%. Based on these findings, the diagnosis of classic FRM was made. The postoperative course was unremarkable and recurrences were not appreciated for 10 months after surgery. Molecular analysis was also performed on genomic DNA and RNA extracted from paraffin sections. Next-generation sequencing (Ion Torrent, IonGene Studio™ S5 Prime System, Thermo Fisher Scientific) using the Oncomine Comprehensive Assay Plus (OCA+, Thermo Fisher Scientific) detected a deletion in the SUFU gene (chr10: 104263902-104389967) that encodes a key negative regulator of HHSP. 7 Additional alterations were identified in other genes including BRCA2 and TP53 but not in other genes associated with the HHSP including PTCH1, SMO, and GLI1 (Figure 2). The Reactome pathway database was also employed to ascertain the pathways or biological processes associated with the identified altered genes. 12 Notably, processes predominantly linked to DNA repair mechanisms were identified (Figure 3). Based on these results, molecular testing on peripheral blood was recommended to assess for germline alterations.
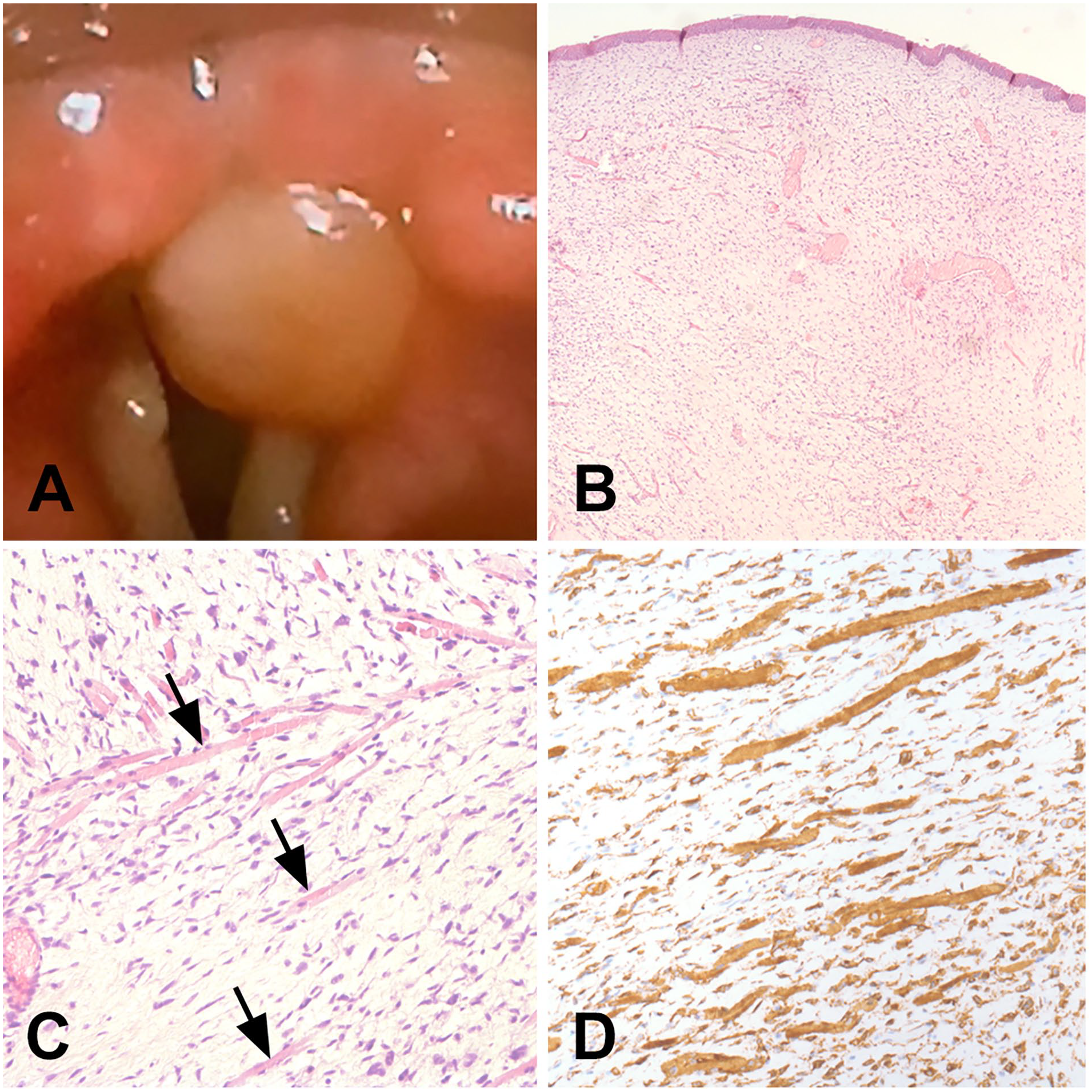
Clinical presentation and low- and high-power magnification of the glottic polypoid lesion are illustrated in A, B (hematoxylin-eosin, 10× magnification), and C (hematoxylin-eosin, 40× magnification), respectively. In C, more differentiated skeletal muscle fibers are identified by arrows. Desmin immunoreactivity of both primitive spindle cells and more differentiated skeletal muscle fibers is illustrated in D (40× magnification).
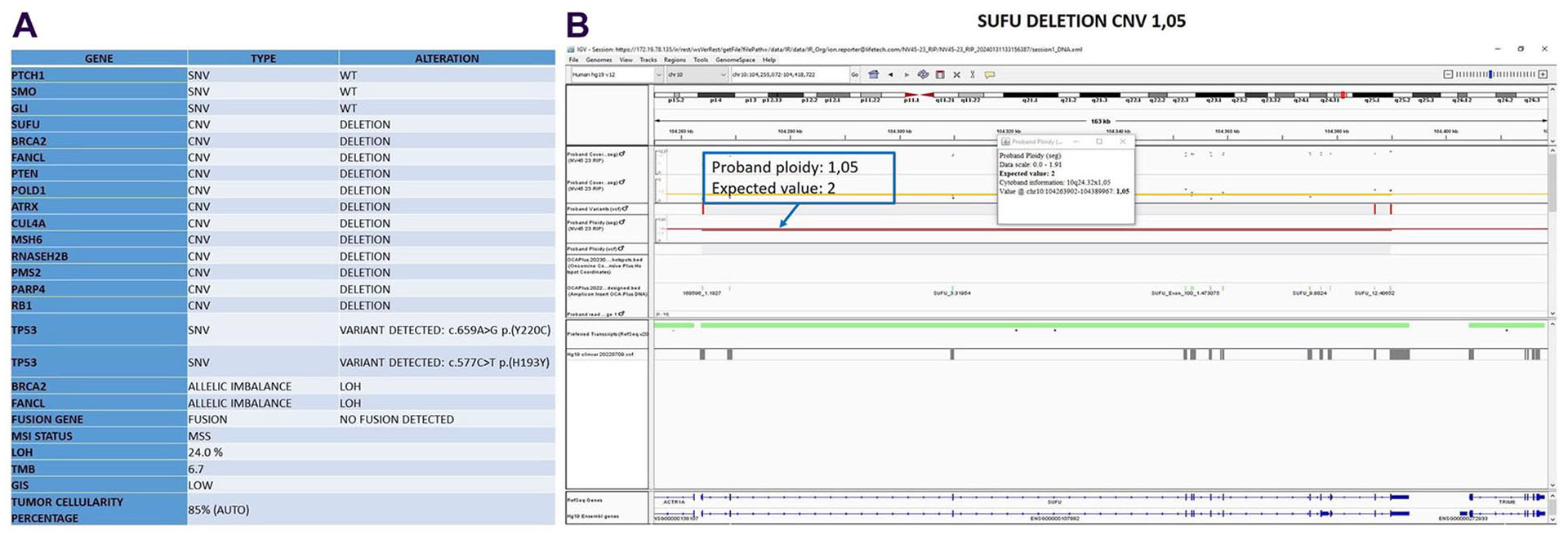
(A) Genetic alterations identified through NGS Oncomine Comprehensive Assay Plus panel. SNV, Single Nucleotidic Variants; CNV, Copy Number Variation; LOH, Loss of Heterozygosity; MSI, Microsatellite Instability; MSS, Microsatellite Stable; TMB, Tumor Mutational Burden; GIS, Genomic Instability Status. (B) Representation of SUFU gene deletion on Integrative Genomics Viewer (IGV, https://www.igv.org/). The panel shows the chromosomal region of the patient (chr10: 104263902-104389967) and its ploidy level compared to the wildtype. This region includes the SUFU gene. Since ploidy refers to the number of copies of each chromosome, we would expect to see 2 copies of each chromosome (diploidy) in a normal sample. In the case of a deletion, a lack or reduction in signal intensity in the affected region will be evident. In our case, the reduction in chromosome copy number in the affected region indicates a lower-than-normal ploidy; and therefore, it appears consistent with loss of signal due to deletion of SUFU gene.
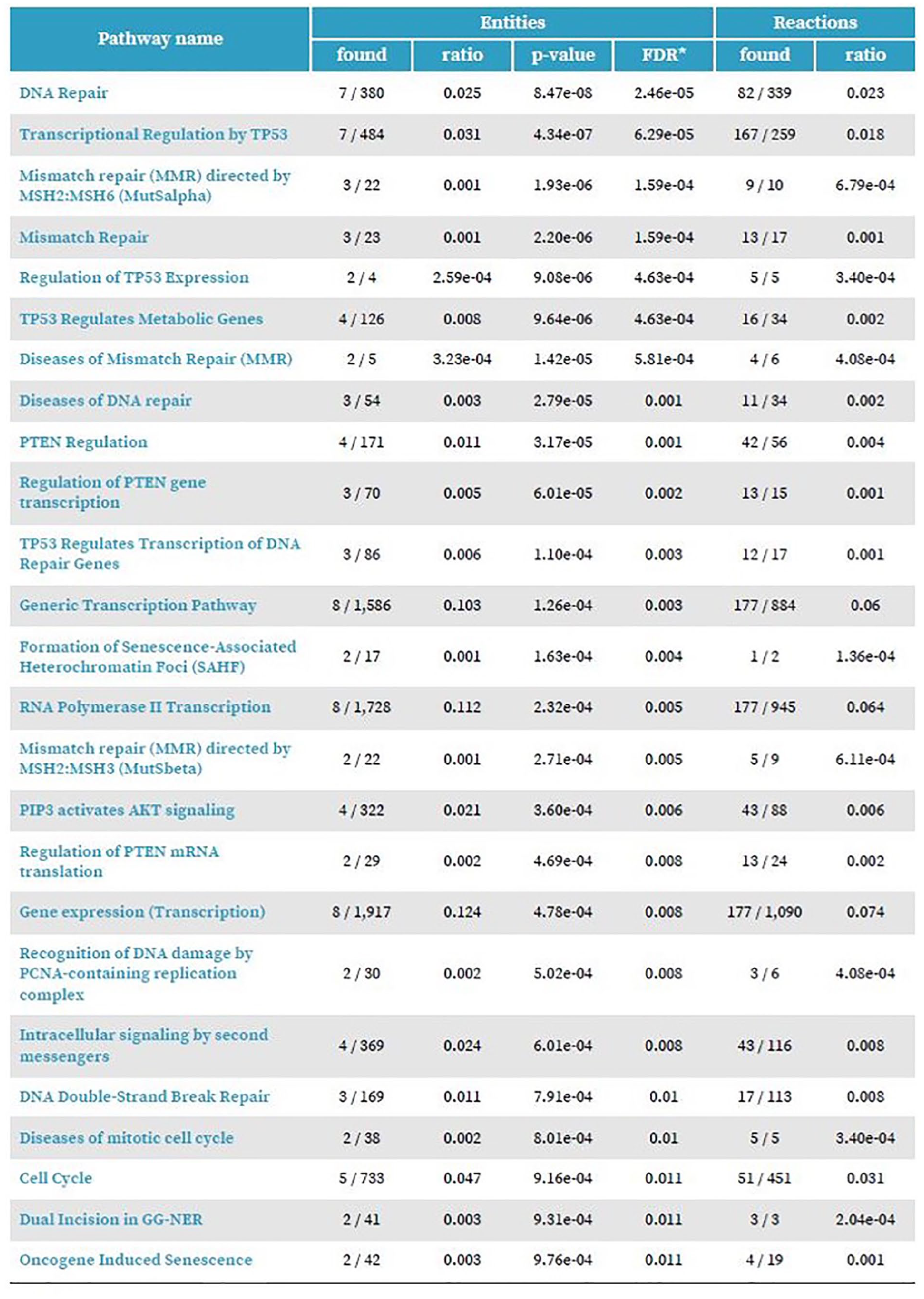
Reactome analysis (https://www.reactome.org/). The panel shows the 25 most relevant pathways sorted by P value. These pathways appear predominantly linked to DNA repair processes.
Otolaryngologists might not be familiar with FRM because development of this tumor in the upper airways is extremely rare.3,12,13 A recent review of the literature revealed that only 8 cases of FRM have been reported in the larynx of adult patients. 13 Clinical presentation is not specific. Hoarseness, airway obstruction, and inspiratory stridor are the most common chief complaints. 13 The spectrum of clinical differential diagnosis is huge including cysts and benign (eg, granular cell tumor) and malignant (eg, rhabdomyosarcoma) tumors. Only histological and immunohistochemical analysis let the diagnosis definitely. However, FRM may cause diagnostic challenge due to its morphologic resemblance to embryonal rhabdomyosarcoma from which it can be distinguished, as in the case presented here, for the superficial location and the lack of invasion of adjacent tissue, significant nuclear atypia, infiltrative growth, necrosis, and atypical mitosis.1,2 Its radical surgical excision is commonly curative. Indeed, recurrences have been reported rarely.8,14 A follow-up of at least 1 year after surgery has been suggested 15 even though FRM may recur years after the initial diagnosis. 14 In our case, the demonstration of a SUFU gene deletion confirms that deregulation of HHSP plays a role in the pathogenesis of at least a subset of sporadic FRM.6,9 To date, alterations of genes related to the HHSP have never been reported in FRM involving the larynx. Nevertheless, when dealing with a laryngeal polypoid lesion in an adult, FRM should be kept in mind by otolaryngologists as a very rare diagnosis.
Footnotes
Author Contributions
MF and FZ were involved in clinical care of the patient, performed the excision of the lesion, collected and summarized clinical data, and obtained written consent to publication. RC, MR, and AC performed the morphological analysis of the lesion. BC performed the molecular analysis. BC, EG, and VL evaluated the results of the molecular analysis. MF and AC conceived the idea and drafted the manuscript. All the authors approved the submitted version of the manuscript.
Data Availability
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All the clinic-pathologic investigations detailed in the article have been conducted in accordance with the Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed Consent
Written informed consent for publication of data and images was obtained from the patient.
