Abstract
Introduction
White lesions of the oral mucosa are commonly encountered in daily practice by dentists, otolaryngologists, and maxillofacial surgeons.1,2 Oral leukoplakia is rarely symptomatic, and the importance of screening and early diagnosis is derived from its frequent association with oral cavity squamous cell carcinoma.3,4 Oral leukoplakia constitutes 85% of all potential malignant disorders occurring in the oral cavity, with a described prevalence of 2.89% to 3.6%, a higher incidence among men.5-7 The etiology of oral leukoplakia is multifactorial.8-12 Consumption of tobacco, whether through smoking or chewing, appears to be the sole direct risk factor implicated in the induction of oral leukoplakia.13-17 The most commonly described locations are represented by mandibular alveolus (25%-40%), buccal mucosa (22%-46%), palate (27%), tongue (26%), and floor of the mouth (19.3%).18-21 Negative prognostic risk factors include being of the female gender, advanced age, having a size greater than 200 mm², and having a Candida albicans infection. HPV, or human papillomavirus, plays a significant role in developing oral leukoplakia. 22 Particularly, high-risk strains like HPV-16 and HPV-18 are associated with an increased likelihood of developing oral leukoplakia and its progression to cancer. 22 Also, different genes involved in DNA damage response and repair pathways have been reported as candidates for cancer susceptibility.23,24 Oral leukoplakia can present carcinoma in situ in 7% to 7.6% of cases,25-27 with nonhomogenous leukoplakia possessing greater malignancy probability (20%-25%).28-31 Visual examination represents the first cost-effective approach, compromised by its inherent subjectivity and the heavy reliance on the clinician’s experience. Toluidine blue staining is employed to supplement visual examination, due to selectively stain areas of dysplasia or malignancy.32,33 However, diagnostic accuracy is sometimes compromised by potential false positives and negatives. As a more sensitive approach, brush biopsy and cytology are often utilized.34,35 While minimally invasive, their sensitivity and specificity can vary, and more severe or deeper dysplastic changes may not be captured. Although tissue biopsy remains the definitive diagnostic method with high diagnostic accuracy, the procedure can present risks and potential discomfort for the patient. In the face of emerging technologies, optical imaging techniques such as Raman spectroscopy (RS) and narrow-band imaging (NBI) are gaining attention.31-34 These innovative, noninvasive techniques can pinpoint subtle structural and biochemical tissue changes in real time. However, their successful implementation requires specialized equipment and expertise for accurate interpretation.
To evaluate and analyze the different diagnostic indications, the advantages, and limitations of each process, we conducted a comprehensive review of the literature on the diagnostic procedures involved in the early detection of oral leukoplakia.
Methods
Study Design
This systematic review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, the Cochrane Handbook for Systematic Reviews of Interventions, and the PICOTS framework (Population, Intervention, Comparison, Outcomes, Timing, and Setting).
Search Strategy
The authors performed a comprehensive literature search in the following electronic databases: PubMed/Medline, Embase, Web of Science, Google Scholar, and the Cochrane Library. The search strategy included the combination of keywords and MeSH terms related to “oral leukoplakia,” “oral precancerous lesions,” “early diagnosis,” “early detection,” “neoplastic lesions,” “oral cavity,” “diagnostic techniques and procedures,” and “biomarkers.” The search was limited to English-language publications with no publication date restrictions.
Inclusion Criteria
Studies were considered eligible for inclusion if they met the following criteria:
Study design: Cross-sectional studies, case-control studies, retrospective cohort studies, prospective cohort studies, primary science articles, and epidemiological studies.
Population: Patients with oral leukoplakia.
Intervention: Procedures or techniques used for the early diagnosis of oral leukoplakia.
Outcomes: Diagnostic accuracy, sensitivity, specificity, positive predictive value, negative predictive value, and other relevant measures.
Exclusion Criteria
Studies were excluded if they:
Were not published in English.
Were case reports, case series, reviews, commentaries, editorials, or letters to the editor.
Focused on diagnosing other oral lesions or conditions without a specific focus on oral leukoplakia.
Did not provide sufficient data to assess the diagnostic accuracy or other relevant outcome measures.
Study Selection and Data Extraction
Two independent reviewers screened the titles and abstracts of the identified articles. Full-text articles were obtained for those that appeared to meet the inclusion criteria or when there was uncertainty. Disagreements between reviewers were resolved through discussion or by involving a third reviewer.
Data extraction was performed using a standardized data collection form. The extracted data included study design, population characteristics, diagnostic procedures, outcomes, and follow-up. In addition, the risk of bias and quality of the included studies was assessed using appropriate tools, such as the Quality Assessment of Diagnostic Accuracy Studies and the Newcastle-Ottawa Scale.
Data Synthesis
A narrative synthesis of the findings was conducted, summarizing the included studies’ main features, diagnostic procedures, treatment modalities, outcomes, and follow-up. The primary objective of this review was to evaluate the diagnostic accuracy of various methods for the early detection of oral leukoplakia. The secondary objective was to compare different diagnostic techniques and identify potential biomarkers that could aid in the early diagnosis of oral leukoplakia. Due to the expected heterogeneity in study designs and diagnostic methods, a formal statistical analysis was not performed. Instead, a qualitative synthesis of the findings was presented, highlighting the strengths and limitations of the included studies.
Results
Study Design and Patient Inclusion
After assessment for eligibility, 16 articles were included for quantitative analysis. The systematic protocol is summarized in Figure 1. The sample sizes varied significantly across the studies, ranging from a minimum of 18 to a maximum of 184 subjects. The studies included patients with various oral conditions, from benign inflammatory lesions to potentially malignant disorders and oral squamous cell carcinoma (OSCC).
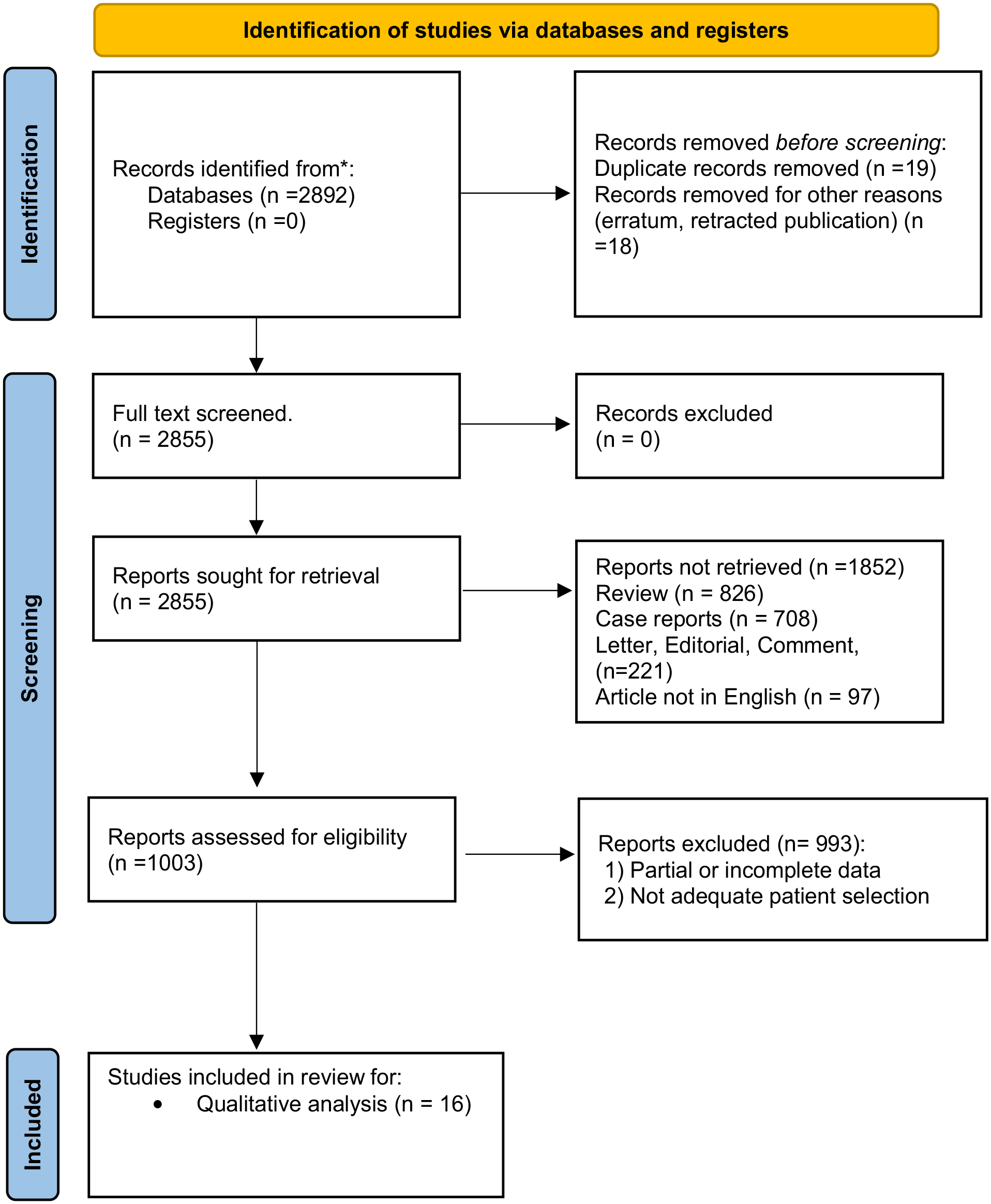
Flow-diagram describing systematic protocol according to PRISMA guidelines. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Methodologies and Outcomes Reported
Several different diagnostic methods were used across the studies. These included salivary microRNA, methylene blue staining, Rose Bengal (RB) staining, blue toluidine staining, Lugol’s iodine staining, RS, elastic scattering spectroscopy (ESS), diffuse reflectance (DR) spectroscopy (DRS), autofluorescence, NBI, high-resolution microendoscopy (HRME), and photodynamic diagnosis.
The sensitivity of the diagnostic methods ranged from 64.3% to nearly 100%. The specificity of the methods ranged from around 60% to 100%. Several studies reported instances of false positives and false negatives. The diagnoses made included normal tissues, dysplasia, potentially malignant disorders (PMDs), and OSCC. Some studies reported on the ability of the diagnostic method to restrict the margins of premalignant lesions or differentiate between different types of oral lesions.
The risk of bias for each included study, according to the Joanna Briggs Institute tool, is summarized in Figure 2.
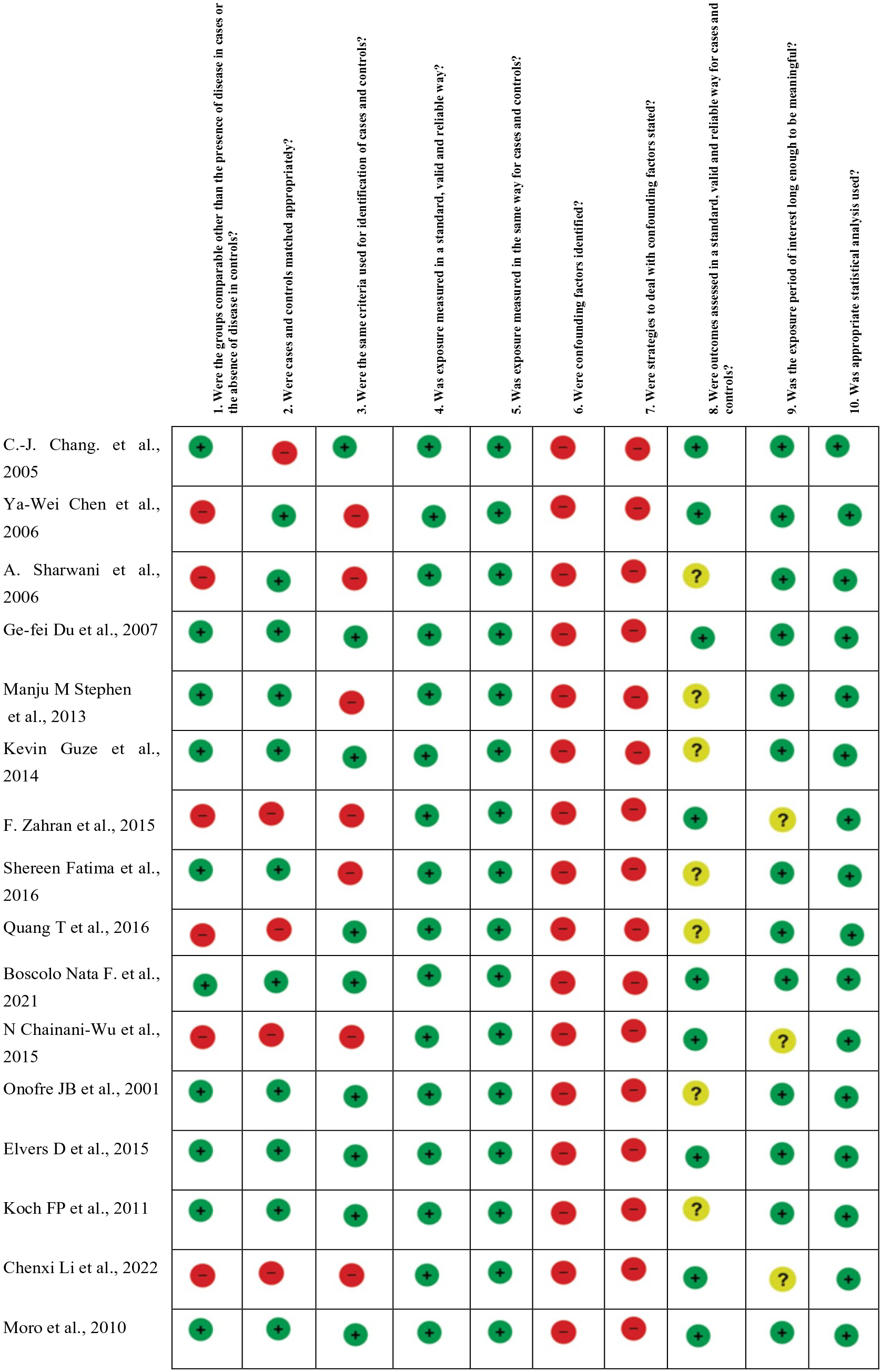
Risk of bias summary author’s judgments for each included study, assessed by the JBI. Critical appraisal checklist for case-control studies. JBI, Joanna Briggs Institute.
Oral Leukoplakia—Diagnosis
Early detection of oral premalignant lesions (ie, leukoplakia) is essential to reduce the high morbidity and mortality rate associated with ensuing oral cancer.36,37 The different noninvasive test approaches available to screen lesions of the oral mucosa are summarized in Figure 3.
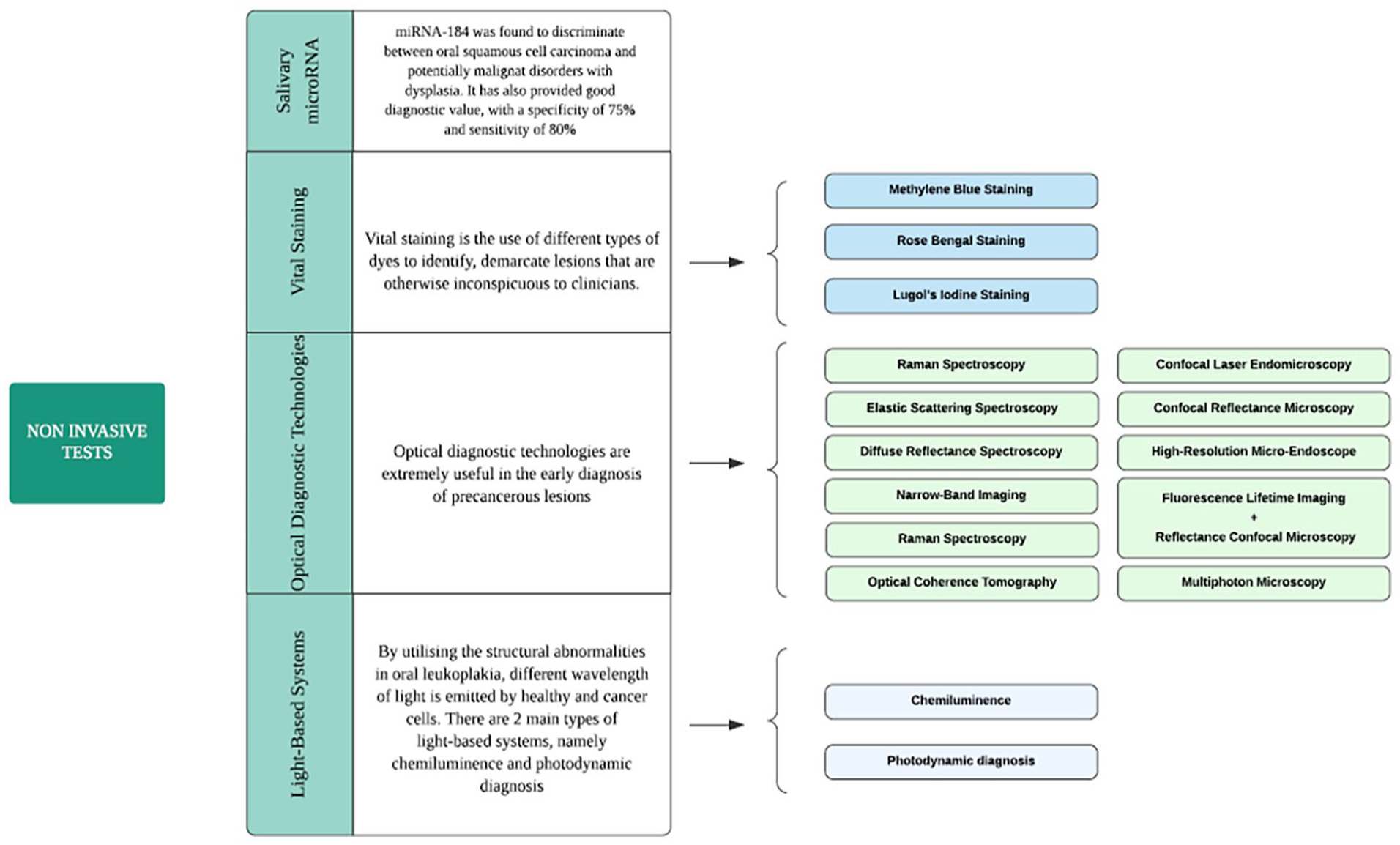
Noninvasive test subclasses and main features.
During oral examination, it is important to identify the following by visual inspection and palpation:
Location
Size
If the lesion is raised
Presence of ulcers
Borders of the lesion—whether it is well-defined or irregular
Patients who are suspected to have premalignant disease will undergo incisional biopsy for histological examination to confirm the diagnosis. However, oral leukoplakia remains a diagnostically challenging lesion that is a potential hurdle for clinicians. 38 It was reported that the 5 year survival rates have not improved despite advancements in treatment. 39 Given the aggressive nature of this condition, the high rates of malignant transformation, and its propensity for early lymphatic spread, early diagnosis is critical in limiting treatment morbidity and maximizing oncologic control.
Noninvasive Tests
The new noninvasive test that this article discusses can be used to aid the identification and diagnosis of oral leukoplakia. Different diagnostic methods for leukoplakia evaluation are summarized in Table I.
Systematic Table for Different Diagnostic Methods for Leukoplakia Assessment.
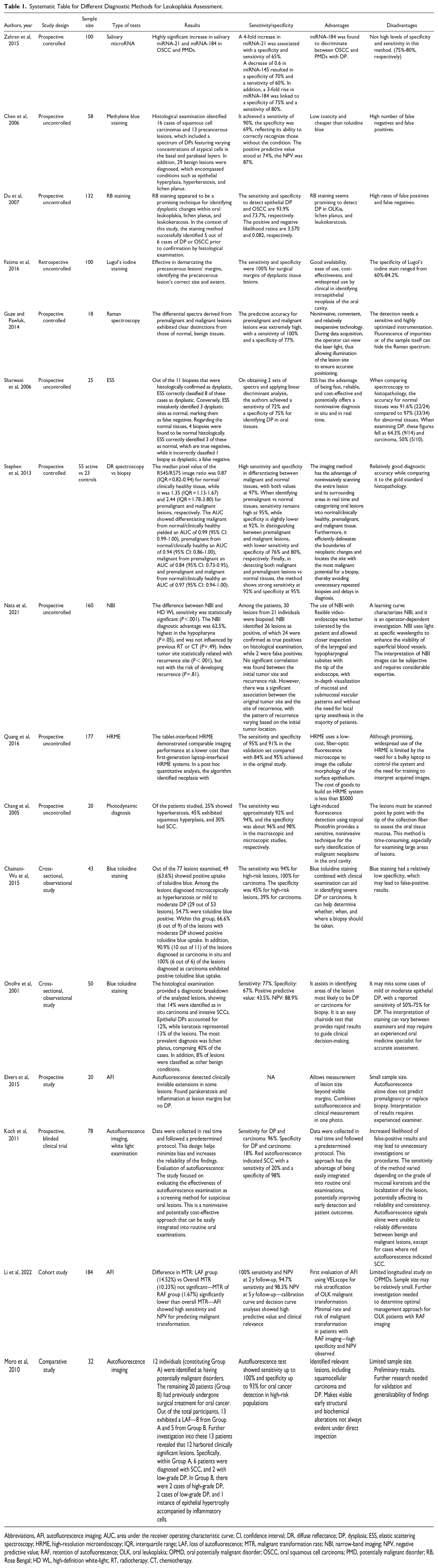
Abbreviations, AFI, autofluorescence imaging; AUC, area under the receiver operating characteristic curve; CI, confidence interval; DR, diffuse reflectance; DP, dysplasia; ESS, elastic scattering spectroscopy; HRME, high-resolution microendoscopy; IQR, interquartile range; LAF, loss of autofluorescence; MTR, malignant transformation rate; NBI, narrow-band imaging; NPV, negative predictive value; RAF, retention of autofluorescence; OLK, oral leukoplakia; OPMD, oral potentially malignant disorder; OSCC, oral squamous cell carcinoma; PMD, potentially malignant disorder; RB, Rose Bengal; HD WL, high-definition white-light; RT, radiotherapy; CT, chemiotherapy.
Salivary biomarkers
The technique involves the collection of saliva samples from patients, which is a noninvasive, easy-to-perform, and stress-free procedure. Once the saliva sample is collected, it is processed to extract the microRNA. This extraction can be done using various methods, including commercial kits. The levels of specific microRNAs associated with oral leukoplakia are measured after the extraction. This is often done using quantitative real-time polymerase chain reaction (qRT-PCR), a technique that precisely measures the amount of a specific RNA. The levels of these microRNAs are then compared to a control group or established thresholds to determine whether they are elevated or reduced. In 2015, there was a study that investigated the use of 3 salivary microRNAs (miRNA-21, miRNA-184, and miRNA-145) as markers for oral cancers. 40 This study isolated RNA from saliva samples using the microRNA Isolation Kit (Qiagen), and miRNA expression analysis was performed using qRT-PCR (Applied Biosystems). This study showed a highly significant increase in salivary miRNA-21 and miRNA-184 in OSCC and PMDs. The miRNA-184 was found to discriminate between OSCC and PMDs with dysplasia. It has also provided good diagnostic value, with a specificity of 75% and sensitivity of 80%. 6 The usage of salivary interleukin-6 (IL-6) has also been investigated. A study involving 40 patients showed that IL-6 levels were elevated in leukoplakia with coexisting periodontitis and periodontitis patients compared to healthy controls. Within the group of patients with leukoplakia, IL-6 levels also correlated with the severity of dysplasia. 41 Several comparative studies have collectively advanced the understanding of other salivary biomarkers in the detection and monitoring of oral diseases with potential malignant transformations.42-47 Agha-Hosseini and Mirzaii-Dizgah 42 identified increased salivary p53 in patients with plaque-like Oral Lichen Planus (OLP), suggesting a higher malignancy risk compared to erosive OLP, while Jacob et al 43 found elevated salivary total sialic acid (TSA) levels in oral precancer and OSCC, indicative of disease progression. Complementing these findings, Varun et al 44 reported that salivary Her2/neu levels were significantly higher in OSCC than in PMDs and controls, underscoring its potential as a localized biomarker for malignancy. The study by Jancsik et al 45 reinforced the concept that saliva testing could be an effective and reliable method for the early detection of OSCC, particularly in high-risk populations such as those with diabetes. A salivary proteomic analysis was conducted to identify potential biomarkers for OSCC, utilizing Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE) and Matrix-Assisted Laser Desorption/Ionization Time-of-Flight/Time-of-Flight (MALDI TOF/TOF) mass spectrometry, the researchers found elevated levels of annexin A8, peroxiredoxin-2, and tyrosine kinase in the saliva of diabetic individuals, proteins previously associated with cancer and OSCC in saliva. In the study led by Punyani and Sathawane, 47 the focus was on evaluating the salivary levels of IL-8 in patients with oral precancer and OSCC to understand its potential as a biomarker. The research revealed that salivary IL-8 concentrations were significantly higher in OSCC patients compared to both the precancer group and healthy controls, indicating its promise as a biomarker for OSCC but not for oral precancerous conditions (OPCs), potentially informing prognostic decisions and treatment strategies. Salivary and serum alkaline phosphatase (ALP) and lactate dehydrogenase (LDH) levels were also assessed among individuals with varying tobacco use and oral health statuses to explore their potential as early diagnostic markers for oral lesions. 48 The study, with a sample size of 500 subjects, demonstrated a significant increase in both serum and salivary ALP and LDH levels in patients with precancerous and cancerous oral lesions, especially those with poorly differentiated oral cancer, potentially aiding in early diagnosis and intervention. The potential of using albumin level measurements as a diagnostic tool, but further details on the statistics, would be necessary to assess the significance of these findings. The study by Metgud and Patel 49 analyzed albumin levels in 45 individuals across 3 groups—healthy controls, patients with oral premalignancy, and patients with oral malignancy. They observed a decrease in serum albumin levels in patients with oral premalignancy and malignancy, and an increase in salivary albumin levels in these same patient groups, although specific numerical values and P values were not provided in the abstract. In the study by Vajaria et al, 50 serum and salivary levels of TSA/total protein (TP) ratios and α-l-fucosidase activity were evaluated in 100 oral cancer patients, 50 patients with OPCs, and 100 controls. The researchers found that both serum and salivary TSA/TP ratios and α-l-fucosidase activity were significantly higher in the patient groups compared to the controls, indicating that these biomarkers could be useful in monitoring the progression of oral cancer. The effect of antioxidant treatment on salivary biomarkers has been assessed in the literature. The study by Rai et al 51 explored the effects of curcumin, a component of turmeric, on patients with precancerous oral lesions including oral leukoplakia, oral submucous fibrosis, and lichen planus, as well as on healthy individuals. The research showed that curcumin intake correlated with increased levels of antioxidants vitamins C and E, and decreased levels of oxidative stress markers malonaldehyde and 8-hydroxydeoxyguanosine in serum and saliva, which was statistically significant post clinical cure of the lesions (P < .05).
Vital staining
Vital staining uses different types of dyes to identify and demarcate lesions that are otherwise inconspicuous to clinicians. It can also enhance the characteristics of lesions. This mode of identifying oral lesions is cheap, easily available, and sensitive. Various types of staining, such as methylene, RB, and Lugol iodine, are described in the literature with high sensitivity and specificity in detecting oral lesions.
Methylene blue has been utilized since 2007 as a tool to detect oral mucosal lesions. The strength of methylene blue lies in its ability to stain cells that contain large amounts of nucleic acid, indicating rapid cell division, a common characteristic of malignant or potentially malignant cells. When applied, methylene blue is absorbed by cells in the oral cavity, resulting in a more intense coloration.
Its reported specificity, which is the test’s ability to identify those without the disease correctly, is around 66% to 69%. On the other hand, the sensitivity of methylene blue, or the ability to correctly identify those with the disease, is higher, ranging from 90% to 91%. However, it is important to note that while methylene blue staining is useful, it is not definitive. Areas that appear stained may still be benign, and not all areas of concern will necessarily take up the stain. Therefore, methylene blue staining is typically used as an initial screening tool, and any areas of concern are usually followed up with more definitive tests, such as a biopsy. 42
RB staining, a dye derived from fluorescein, has been effectively used to identify oral conditions such as leukoplakia, lichen planus, and leukokeratosis. In one study, RB staining accurately identified these conditions in 132 patients. 43 RB staining offers advantages over other stains, such as toluidine blue (TB).44-46 The adjunctive role of TB in detecting and managing oral premalignant and malignant lesions can be useful in identifying high-risk areas of lesions for biopsy and predicting molecular changes and behavior of oral premalignant lesions, 45 especially on lesion margins, expedite the decision-making process for biopsy, and guide treatment strategies. Onofre et al found that TB staining showed a sensitivity of 77% and specificity of 67% in detecting oral epithelial dysplasia, in situ carcinoma, and invasive squamous cell carcinomas. 46 The positive predictive value of the staining method was 43.5%, and the negative predictive value was 88.9%. While TB staining demonstrated reliability in detecting certain lesions, its limitations suggest that it should be used in conjunction with clinical judgment and biopsy for accurate diagnosis. Conversely, RB does not stain inflammatory cells, enhancing its specificity for potentially malignant cells. Moreover, RB has shown superior sensitivity, correctly identifying those with the disease, with a 90% to 100% rate compared to TB’s 38% to 100% sensitivity. 47 However, RB staining has limitations. While it does not stain inflammatory cells, it may yield false positives in certain situations. In addition, like all techniques, the interpretation of RB staining results can be subjective and dependent on the observer’s expertise and experience. The combination of RB staining with autofluorescence spectroscopy has shown promising results in animal studies, achieving a 100% sensitivity and 87.5% specificity in detecting dysplasia in vivo. 48 Yet, these impressive results have not been replicated in human clinical studies, representing an area for future research.
The RB conjugated gold nanorod (GNR) platform represents a novel approach to detecting oral leukoplakia. The technique combines the attributes of RB staining with the unique properties of GNRs. RB is a dye that selectively stains certain cells, while GNRs are tiny rod-shaped gold particles that can be used as efficient probes to detect specific molecular events in cells. The GNRs are conjugated, or linked, with RB in this combined technique. When applied to a tissue sample, the RB targets and binds to certain cells, and the GNRs enhance the visibility of the bound RB, especially under specific types of light. This can lead to an improved uptake of RB into the cells, making potentially malignant cells more visible. Preliminary studies on this technique are promising, with evidence suggesting that it could significantly aid in detecting oral cancer cells. 49 However, the method has limitations. For one, it requires specialized equipment to prepare the RB-conjugated GNRs and to visualize the results, which could limit its use to certain well-equipped laboratories or clinical settings.
Furthermore, interpreting the results requires specific expertise and understanding of both RB staining and nanotechnology, which may not be universally available. As for Lugol’s iodine staining, this is a time-tested method that involves applying an iodine solution to the oral tissue. The iodine reacts with the glycogen stored in cells. Normal cells, which have a regular amount of glycogen, will stain brown. In contrast, potentially malignant cells, which often have enhanced glycogenesis and loss of cell differentiation, will not take up the stain and appear pale in comparison. The sensitivity and specificity of Lugol’s iodine stain have been reported to range from 87.5% to 92.7% and 60% to 84.2%, respectively. 50 While these figures suggest a high accuracy level, the method has limitations. For example, it can sometimes yield false positives and negatives and may be less effective in patients with certain conditions that affect glycogen storage. In addition, interpreting Lugol’s iodine staining results can be subjective and requires experienced clinicians. When Lugol’s iodine is combined with RB staining, the sensitivity and specificity in detecting leukoplakia are similar or slightly reduced. This suggests that while combination techniques can potentially enhance diagnostic accuracy, the interaction between different staining methods must be carefully considered to avoid a reduction in effectiveness. One concern is patient discomfort. Applying Lugol’s iodine can cause a burning sensation ranging from mild to moderate, potentially leading to a less-than-comfortable experience for the patient during the procedure. Another issue is the potential for nonspecific staining. Lugol’s iodine is designed to react with glycogen in cells, but it can also stain other substances like mucous. This nonspecific staining can complicate the interpretation of results, potentially leading to inaccuracies.
In addition, if a biopsy is required following Lugol’s iodine staining, the iodine can interfere with subsequent histological examination. The presence of iodine can mask crucial cellular details, making it more challenging to reach a definitive diagnosis. The procedure also requires some preparation from the patient. To achieve optimal results, patients are often advised to refrain from eating or drinking for several hours before the process, which can be inconvenient.
Optical diagnostic technologies
Highly sensitive and specific optical diagnostic technologies have been developed to accurately pinpoint subtle aberrations before they morph into malignant conditions.51-53 These include autofluorescence imaging (AFI), an approach that captures the natural emission of light by biological molecules in tissues when excited by a light source. 54 Autofluorescence has emerged as a valuable tool in assessing oral leukoplakia, a potentially premalignant condition,55-58 detecting signs of dysplasia, parakeratosis, and mucosal inflammation in the borders of homogeneous oral leukoplakia. The findings demonstrated that autofluorescence can reveal clinically invisible extensions of leukoplakia beyond their visible margins, with areas of autofluorescence loss correlating with parakeratosis. 56 Most important, autofluorescence primarily detects nondysplastic lesions associated with mucosal inflammation and parakeratosis. 57 Li et al reported that time-dependent receiver operating characteristic curve analysis demonstrated that AFI had high sensitivity and negative predictive value for predicting malignant transformation, with excellent predictive and clinical relevance. Overall, autofluorescence holds promise as a diagnostic aid for assessing and characterizing oral leukoplakia lesions. 58 NBI is another technique that enhances the visibility of vascular structures on the mucosal surface by using specific wavelengths of light. Confocal microscopy, on the other hand, offers high-resolution, real-time images of tissue structures at various depths. However, they come with their own sets of challenges. A prominent limitation lies in interobserver variability, which refers to the differences in the interpretation of the same data or images by different observers. This variability can stem from factors like the observers’ unique experiences, their level of training, and individual subjective perspectives. To tackle the issue of interobserver variability, efforts are underway to standardize the interpretation of data and images obtained from optical diagnostic technologies.
RS is a noninvasive diagnostic technique that distinguishes premalignant and malignant lesions from normal mucosa or benign lesions. This cutting-edge optical technology has shown promising results in vivo studies, demonstrating a sensitivity of 93.7% to 100% and a specificity of 76.7% to 77%.58,59 The technique operates based on the Raman effect, named after the Indian physicist C. V. Raman. It involves the scattering of light in a different direction with a change in energy, which provides a specific “fingerprint” for the chemical bonds in a molecule. When a laser light shines on a tissue, most of it scatters in the same energy it originally had, but a small portion of light scatters with slightly shifted energy. This energy shift provides detailed information about the molecular composition of the tissue, enabling the differentiation of normal, benign, and malignant tissues. To enhance the sensitivity of RS, gold nanoparticles with an ultrathin silica or alumina shell are often used. These nanoparticles can amplify the Raman signal, thereby increasing the accuracy of the technique. The process involves spreading a monolayer of these nanoparticles over the surface to be probed. The nanoparticles conform to the different contours of the substrate, providing a detailed map of the tissue surface. The Raman signals from these nanoparticles are then measured, providing insights into the molecular composition of the tissue. 59 Despite the promising results of RS, it is important to note that the technique requires specialized equipment and trained personnel for accurate interpretation. In addition, the use of nanoparticles necessitates careful handling and disposal procedures to avoid potential environmental and health impacts. As such, while RS represents a significant advancement in the early detection of oral leukoplakia, it is one piece of a larger diagnostic puzzle that must be integrated with other techniques and considerations.
ESS leverages a pulsed xenon-arc lamp to examine lesions. Directing light onto the tissue interacts with the cells and structures within, causing the light to scatter in various directions while maintaining its original energy, a phenomenon known as “elastic” scattering. The scattering pattern gives insights into the tissue’s characteristics, such as cell size, shape, and composition. These patterns can change when cells become cancerous or precancerous, providing a potential method for early detection of conditions like oral leukoplakia. Despite its advantages, including noninvasiveness, real-time results, and the ability to examine tissues at a cellular level without dyes, ESS interpretation requires specialized knowledge and can be complicated. It can be challenging to discern between changes caused by malignancy and those resulting from other conditions like inflammation.
In addition, ESS typically examines a relatively small area at a time, potentially missing abnormalities outside the probed area. In a small study involving 25 patients, ESS demonstrated a sensitivity of 72% and a specificity of 75%. 60 While promising, these results suggest there is still room for improvement in the accuracy of ESS in detecting oral lesions.
Diffuse reflectance spectroscopy (DRS) operates on the principle that the morphological and cytological changes occurring in tissues during carcinogenesis result in altered patterns of light reflection and absorption. 61 This diagnostic tool employs an image recording device to capture the DR images of oral lesions. When utilizing the oxygenated hemoglobin spectral ratio (R545/R575) algorithm, the sensitivity and specificity of DRS have been reported to be 95% to 97% and 97% to 100%, respectively.62,63 These figures underscore the potential of DRS as an effective diagnostic tool for identifying oral lesions. It has been suggested that DRS could be as effective as histopathology, the gold standard for diagnosing oral lesions. 64 Despite its promise, the application of DRS requires specialized equipment and trained personnel, and its effectiveness can be influenced by factors such as the patient’s individual characteristics and the specific location of the tissue being examined.
NBI utilizes different wavelengths of light to penetrate and identify superficial capillaries or prominent vessels in the submucosal layer. The reflected light is captured by an endoscope, with the image displayed on a screen. In the early stages of carcinogenesis, malignant lesions have distinct angiogenesis characteristics (neoangiogenesis). NBI aids in illuminating these abnormal superficial vasculatures, highlighting the possible presence of oral lesions. 65 Recent meta-analysis has shown that the sensitivity and specificity of NBI are 88.5% and 95.6%. 66 This supports previous studies demonstrating that NBI is useful for identifying and managing oral leukoplakia. In addition, there is an added benefit of identifying the presence of intrapapillary capillary loops, which is an indicator of disease severity. 65
Optical coherence tomography (OCT) creates cross-sectional images of the tissue, using similar principles as an ultrasound machine. It can provide high-resolution images in real time, 67 providing immediate and diagnostic information. Due to the shallow penetration, OCT is useful in identifying oral mucosal lesions. The sensitivity and specificity of OCT have been shown in vivo to be 95% and 97%, respectively. 68 Some studies have combined the use of contrast agents with OCT to improve the sensitivity and specificity of the diagnosis of oral lesions. 69 Limitations of OCT are due to the subjective nature of the interpretation of results.
Confocal laser endomicroscopy (CLE) is another noninvasive technique allowing microscopic imaging under 1000-fold magnification. It is based on tissue laser illumination and subsequent fluorescence light detection. 70 CLE has shown good sensitivity and specificity of 95.3% to 97.3% and 88.1% to 88.9%, respectively, with good interobserver variability. 71 Studies have tried combining the fluorescein probe with CLE to improve specificity and sensitivity. However, the sensitivity of CLE has decreased to 80% while specificity increased to 100%, indicating an improvement in excluding nonmalignant lesions. 71
Confocal reflectance microscopy utilizes laser light at a near-infrared wavelength (830 nm), penetrating the tissue of interest and illuminating a single point. A study that compared confocal reflectance microscopy and histopathology with hematoxylin and eosin staining showed that this is a promising method of diagnosis, with a sensitivity of 96.3%, specificity of 92.3%, positive predictive value of 93%, and negative predictive value of 96%. 72
The HRME is a diagnostic tool that leverages a coherent fiber bundle to capture high-resolution fluorescence images of the tissue in contact with the device’s distal tip. In this setup, a camera plays the crucial role of seizing high-quality digital images, which are then transferred to a computer for further analysis. 73 The HRME provides a noninvasive method to visualize tissue architecture and changes in cellular behavior, potentially aiding in the early detection and diagnosis of conditions like oral leukoplakia. However, this technique requires specialized equipment and trained personnel, and the effectiveness of HRME can be influenced by factors such as the specific location of the tissue being examined and the patient’s individual characteristics.
Fluorescence lifetime imaging (FLIM) is a promising diagnostic technique that analyzes a tissue area of about 1.6 cm × 1.6 cm. It operates by measuring both tissue autofluorescence and the decay of fluorescence over time. This unique approach allows for the potential estimation of the contributions of specific fluorophores like, Nicotinamide Adenine Dinucleotide (NADH), Flavin Adenine Dinucleotide (FAD), and collagen, which are molecules that emit fluorescence when excited by light. The differential presence of these fluorophores can provide valuable information about tissue health and potential abnormalities. Particularly useful in differentiating dysplastic lesions from benign inflammatory lesions, 74 FLIM offers a noninvasive method to identify precancerous or cancerous changes. However, the technique requires specialized equipment and expertise in data interpretation, and its effectiveness can be affected by factors such as the patient’s individual characteristics and the specific location of the tissue being examined.
Multiphoton microscopy represents a fluorescence imaging technique that provides cross-sectional images of tissues at different depths, reaching up to 1 mm. 75 This approach allows for the assessment of cellular invasion beyond the basement membrane, a key characteristic of invasive diseases. By offering a detailed look at tissue architecture and cellular behavior in their native environment, multiphoton microscopy can contribute to the early detection and diagnosis of conditions like oral leukoplakia. In a study conducted by Matsui et al, this technique demonstrated a high sensitivity of 96% and a specificity of 84%, 76 indicating its potential as a precise diagnostic tool. However, like all diagnostic techniques, multiphoton microscopy comes with its own set of requirements, including the need for specialized equipment and trained personnel for accurate operation and interpretation of results.
Light-based systems
Light-based detection systems such as chemiluminescence and photodynamic diagnosis have been developed to aid in diagnosing oral leukoplakia at an early stage. By utilizing the structural abnormalities in oral leukoplakia, healthy and cancer cells emit different wavelengths of light. There are 2 main types of light-based systems, namely chemiluminescence and photodynamic diagnosis.
There are multiple chemiluminescent devices that are available on the market. These devices use a light-based detection system to detect the different wavelengths reflected due to changes in cancer cell morphology. The main devices on the market are ViziLite (Zila Pharmaceuticals, Phoenix, AZ, United States), ViziLite Plus (Zila Pharmaceuticals, Phoenix, AZ, United States), and Microlux/DL (Microlux DL - AdDent, Inc., Danbury, CT, United States).
The oral cavity is first rinsed with acetic acid before being examined under chemiluminescent illumination. This allows the user to differentiate between normal and hyperkeratinized epithelium. Dysplastic or hyperplastic tissue has increased nuclear content that reflects light and hence appears white when viewed at low-energy wavelengths. Conversely, the normal epithelium appears dark. 77
Studies have shown that ViziLite has a high sensitivity of 77.3% to 100% but a low specificity of 0% to 55.56% in detecting oral carcinoma.78-80 However, it has been shown to detect leukoplakia more than other forms of oral lesions. 75 A newer version of ViziLite called ViziLite Plus combines both ViziLite and TB and has been shown to improve the specificity of 75.5% to 78% but a decreased sensitivity rate. The principles of Microlux/DL are similar to ViziLite. It uses a battery-powered LED light that emits blue light. According to Ibrahim et al, the specificity and sensitivity of identifying oral lesions were 100% and 32%, respectively, meaning that it could locate possible lesions but could not differentiate the types of lesions. 80
Photodynamic diagnosis involves treating cells with a photoactivated compound that accumulates more in cells with malignant potential when exposed to photoirradiation. A common compound used is 5-aminolevulinic acid (ALA), which induces the fluorescence of protoporphyrin IX in cancerous and precancerous cells. The procedure involves rinsing the oral cavity with a 0.4% ALA solution followed by exposure to a specific light wavelength of 405 nm. This technique boasts a high sensitivity, as studies have reported a range between 80% and 99%. 81 However, its specificity can be compromised in patients with a history of radiotherapy, as indicated by some studies. 82 Despite this limitation, using ALA and photodynamic diagnosis can provide valuable information for the early detection and diagnosis of conditions like oral leukoplakia. It is worth noting, however, that the effectiveness of this technique can be influenced by factors such as the patient’s individual characteristics and the specific location of the tissue being examined.
Discussion
Although several diagnostic methods for the early detection of the oral cavity neoplastic lesions have been demonstrated, their use currently remains controversial and debated. 20 We aimed to critically describe the noninvasive detection techniques for premalignant oral cavity lesions concerning each method’s sensitivity and specificity. Dentists, ENTs, or maxillofacial surgeons are the specialists who are the first to deal with premalignant lesions of the oral cavity. Identification of such lesions should occur as early as possible. 25 Early diagnosis of premalignant lesions of the oral cavity is essential. It is based on oral screening. The latter could avoid delayed referrals, thus reducing mortality in the SCC. 19 It has been reported that SCC can develop from oral potentially malignant disorders, and its diagnosis is an important preventive step with a major impact on patient survival and future quality of life. 21 However, visual inspection has several limitations, such as the inability to distinguish high-risk benign lesions from other diseases and morbid conditions of the oral mucosa.
Tissue biopsy is an invasive, time-consuming, painful, operator-dependent method frequently not readily accepted by patients. 24 Despite this, oral biopsy remains the gold standard method today. Typically, nonearly detection of a premalignant lesion leads to an advanced stage at diagnosis.82-84 However, several follow-ups have shown that the risk of malignant transformation can persist for over 10 years. For this reason, long-term follow-up with regular checkups by the oral surgeon, maxillofacial, or ENT specialist is required.
The early diagnosis of premalignant lesions of the oral cavity can make use of noninvasive, easy-to-use, and effective methods. 21 Salivary diagnostics is a method that has spread in recent years. 39 Saliva has a very complex composition, including enzymes, antibodies, hormones, antimicrobial elements, and cytokines. 40 The saliva collection is easy, safe, noninvasive, and inexpensive. In recent years, interest has grown in miRNAs (found in various biological fluids, including saliva) being the latter considered as potential markers for diagnosing, prognosis, and evaluating the effectiveness of treating multiple diseases. 6
It is likely that miRNA expression profiling not only allows the identification of neoplastic tissue and its histological origin but also discriminates between different subtypes of malignant lesions. 40 Regarding inflammation, understanding the role of miRNAs in its regulation could be important in helping understand the pathogenesis of a large group of diseases.
Among the diagnostic methods based on vital staining, RB staining and Lugol’s iodine staining must be cited. RB staining was used to delineate the extent of corneal and conjunctival neoplasms. Therefore, such characteristics of RB have enlightened us to perform surveying research on premalignant and malignant lesions of the oral cavity. Lesions more stained by RB had a higher likelihood of being OSCC or epithelial dysplasia than those less stained. 45 Thus, RB staining might have the potency to be used as a diagnostic aid to detect oral premalignant or malignant lesions for clinicians. With regard to the methods based on diagnostics with optical systems, particular interest is given to methods based on fluorescence/autofluorescence and NBI. 64 In soft tissues, potentially malignant lesions and tumor lesions can be detected. Thus, the optical fluorescence system allows for simple, noninvasive, real-time diagnosis and identification of structures and alterations in the oral cavity, revealing lesions that are not easily detectable with lighting. 73
NBI is a new optical technology already widely applied in diagnosing gastrointestinal lesions. Unlike the epidermal tissue, the mucous membrane of the oral cavity has few keratinized layers and lacks appendages deriving from the outlet of the minor salivary glands. 64 Hence the capillaries in the papillae of the connective tissue under the epithelium are hardly observed from the external mucosal surface. While in the healthy oral mucosa, the pegs of the epithelial network and the connective tissue papillae are regularly connected, in cancer, this connection becomes irregular. 76 Therefore, visually, the capillaries will assume an irregular and dense distribution. By observing a tumor lesion under magnification, the proliferation of capillaries can be recognized as a characteristic spotting of the tissue. Another method used for the early diagnosis of oral premalignant lesions is chemiluminescence. 77 Many systems use this method; the 2 most used are the ones based on luminol and based on peroxyoxalate.
Regardless of the system, blue-white light is absorbed by healthy cells and reflected by cells with abnormal nuclei, including dysplastic and neoplastic cells. The acetic acid rinse putatively removes debris and disrupts the glycoprotein barrier on the epithelium’s surface, allowing light penetration. 34 Variable dye uptake was observed between exophytic and ulcerated SCC. The dye showed excellent retention and staining in ulcerates compared to exophytic lesions due to the increased intercellular spaces that allow for better dye penetration.85,86 In conclusion, chemiluminescent light is useful as an additional diagnostic tool for oral cancer care and follow-up Potentially Malignant Epithelial Lesions (PMELs) of subjects treated for the same.
A significant limitation in diagnosing oral potentially malignant lesions, such as oral leukoplakia, is the need for more awareness and knowledge among dental and medical professionals. Despite the availability of various techniques for oral examination, the challenge persists due to a limited understanding of oral leukoplakia and its diagnostic process.87-92 Bridging these gaps by enhancing awareness and knowledge is crucial to facilitate early detection and prevent the progression of OSCC and other potentially malignant lesions in the oral cavity.
Subjectivity in diagnosis due to visual interpretation and the variability in lesion appearance further complicate accurate identification.93,96 In addition, sampling bias during biopsy procedures, the absence of reliable predictive biomarkers, limited accessibility to specialized care, and patient compliance with follow-up appointments all contribute to the challenges in achieving early and precise diagnoses. Overcoming these limitations requires standardized diagnostic criteria, diagnostic techniques, biomarker research advancements, improved accessibility to specialized care, and enhanced patient education and engagement.
Conclusion
The early diagnosis of oral premalignant conditions is crucial for minimizing invasive surgical intervention, providing a better prognosis, and improving the quality of life for patients. Currently, several diagnostic tools for screening are available, enhancing the characterization of suspicious lesions. Today, surgical biopsy and histology remain the primary therapeutic choices, but the advent of salivary biomarkers presents promising new techniques. Scientific progress is continually modernizing diagnostic procedures to facilitate early detection of oral cancer and reduce diagnostic delay. Although any light-based diagnostic device could aid in diagnosing oral mucosal lesions, chemiluminescence examination can delineate oral lesions more effectively, as the edges of the lesions exhibit improved brightness and clarity. Other emerging techniques include OCT and molecular imaging, which offer high-resolution imaging capabilities. Moreover, when combined with quantitative autofluorescence analysis, the autofluorescence-based system could differentiate between tumors and benign oral dysplasia.
Footnotes
Acknowledgements
None.
Data Availability
No new data were created.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki.
Grant Number
No grant was associated.
Informed Consent
Informed consent was waived for the review study.
Trial Registration
The trial registration was waived for the review study.
