Abstract
Introduction
Stuttering is a multifactorial pronunciation disorder represented by frequent prolongations, repetitions, or blocks of spoken sounds. The most prevalent form of stuttering is developmental stuttering, which first manifests in childhood. 1 Between 80% and 90% of developmental stuttering starts by 6 years of age, and the condition affects approximately 5% of children. 2 Stuttering is one of the most well-known speech disorders, but the underlying neurological arrangements are still being debated. In addition to genetic components, there are also significant nongenetic factors. 3
This study suggests that infection with group A beta-hemolytic Streptococcus (GABHS) has been a significant inherent cause of stuttering until the mid-1900s with the discovery of penicillin in 1943. 4 The primary identity theory is that stuttering relates to an autoimmune response to upper airway infections, targeting explicit molecules, for example, within the basal ganglia. 5 This study also suggests that GABHS infections may have continued to promote stuttering, to some extent, up to the present, though more unusually so. If so, initial diagnosis of such patients would be of significance. Child patients suffering from an immediate attack of stuttering after an upper airway infection may be especially significant to evaluate for possible GABHS infection.
The primary reasons for this theory originate from 3 propositions. First, information contained in medical documents from the 1930s strongly suggests that there was 1 type of medical issue in particular that was associated with the onset of childhood stuttering with unexpected frequency, namely, GABHS upper airway infections. 6 Stuttering shares many similarities with Tourette syndrome—both begin in childhood, have a male-to-female prevalence of 4:1, have waxing and waning trends, are made worse by anxiety, are conjoined with tic motions, suggest brain malformations of the basal ganglia, are worsened by dopamine agonists, and manifestations are ameliorated by dopamine antagonists. 7
The aim of this survey was to examine the association between streptococcal serology and stuttering in children.
Methods
We conducted this study in the Child and Adolescent Psychiatry and Otorhinolaryngology Clinics of 2 referral hospitals, Osmangazi and Eskisehir City Hospitals, in Eskisehir in 2020 to 2021. This study was approved by the Ethical Committee of the University.
The study was conducted according to the principles of the Declaration of Helsinki. All the participants and parents were fully informed about the study and signed informed consent forms.
Initially a questionnaire was completed by an authorized physician for each case.
Study group: This group consisted of 26 children with stuttering, based on the Stuttering Severity Instrument administered by a child and adolescent psychiatrist and speech-language therapist after clinical and entire laboratory or imaging studies.
Control group: This group consisted of 25 children who had been hospitalized for elective pediatric otorhinolaryngology surgery. These subjects were age-matched with cases. They were visited by a child and adolescent psychiatrist and speech-language therapist before surgery. We utilized their extra blood (which was taken for their routine blood tests before their individual surgery) for the serologic tests.
Removal criteria: All subjects with proven etiology for neuropsychiatric expressions (eg, tumor, vasculitis, brain anomalies, metabolic disorders, encephalopathy, encephalitis, etc) were accepted by clinicians before admitted to the study.
Blood specimens (2 ml) were obtained from all subjects and centrifuged. The specimens were transferred and stored frozen at −20°C in our research laboratory. Enzyme-linked immunosorbent assay (ELISA) tests (MyBioSource) were done for specific antistreptococcal antibodies [antistreptolysin O (ASO), anti-deoxyribonuclease B (anti-DNase B), and antistreptokinase). The results were determined by cutoff control as proposed by the manufacturer. The antibody titers (IU/ml) of subjects were compared and statistically analyzed.
Statistical Analysis
All analyses were performed using SPSS 26.0 (IBM Company) software. Chi-square values were computed for all categorical variables, with P < .05 deemed significant. The Mann-Whitney U test and the Student’s t test were also conducted. The sensitivity, specificity, and positive predictive value of tests were computed.
Results
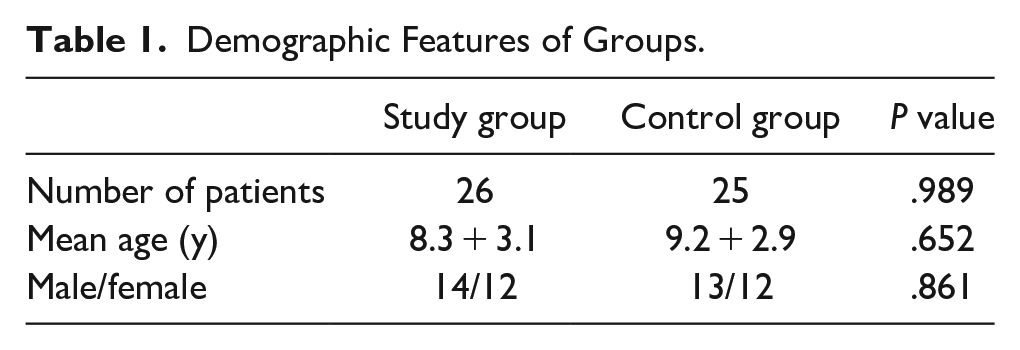
The 26 cases with stuttering with a mean age = 8.3 + 3.1 years were compared with the 25 healthy controls with a mean age = 9.2 + 2.9 years (Table 1).
Demographic Features of Groups.
Positive ASO titers (>200 IU/ml) were observed in 20/26 cases and in 4/25 controls (P < .0001).
Positive antistreptokinase titers (>332 IU/ml) were found in 9/26 cases and in 2/25 controls (P < .0001).
Positive anti-DNase B titers (>140 IU/ml) were observed in 21/26 cases and in 3/25 controls (P < .0001; Table 2).
Number of Cases With Positive Laboratory Titers.
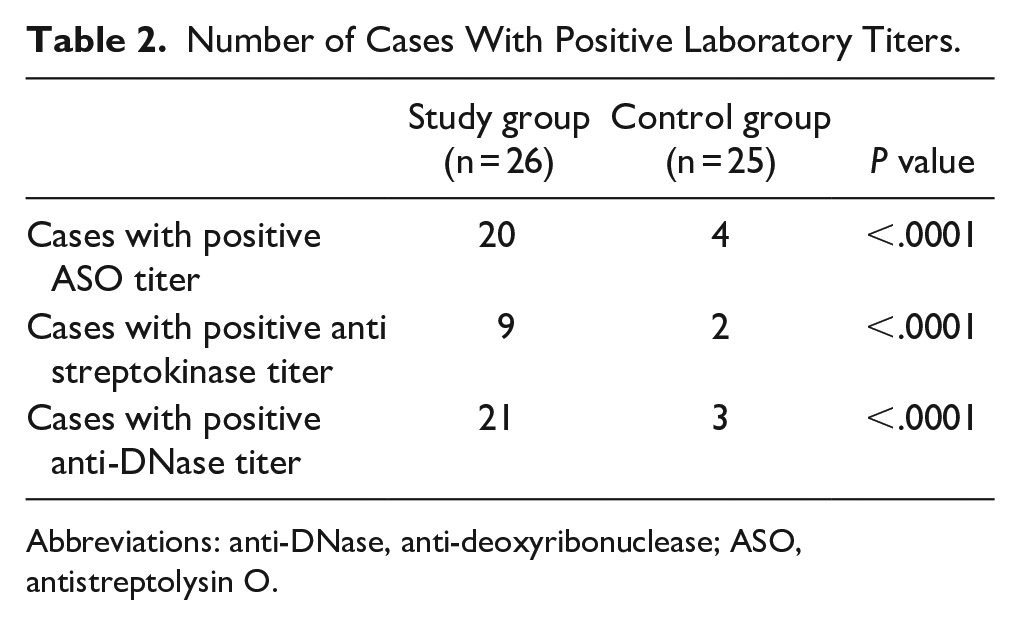
Abbreviations: anti-DNase, anti-deoxyribonuclease; ASO, antistreptolysin O.
Discussion
As anticipated, raised antistreptococcal antibodies were detected in stuttering subjects. All 3 types of antibodies (ASOT, anti-DNase B, and antistreptokinase) were meaningfully higher in stuttering subjects compared to the control group. All of the aforementioned results support the hypothesis that a postinfectious immune mechanism to GABHS may play a role in the pathogenesis of stuttering.
Our outcomes are similar to those of other published studies. In Muller’s study, Tourette syndrome patients presented with higher antistreptococcal levels than age-matched comparison groups of both children and adolescents. 8
In the literature, some authors have distinguished a subgroup of children with early and abrupt onset of obsessive-compulsive disorder (OCD) and/or tic disorders after streptococcal infections [pediatric autoimmune neuropsychiatric disorders associated with streptococcal infection (PANDAS)]. 9
Some authors have revealed that both intravenous immunoglobulin and plasma exchange were useful in reducing neuropsychiatric symptom severity in the PANDAS subgroup. A placebo-controlled investigation revealed that both intravenous immunoglobulin (IVIG) and plasma exchange were useful in reducing neuropsychiatric symptom severity in the group of children with OCD and/or tic disorders. 10
It has also been reported that antibiotic prophylaxis has been efficient in prohibiting persistent streptococcal infections in PANDAS. In addition to the use of anti-tic and anti-obsessional drugs, the use of penicillin during the acute phase and for prophylaxis of tonsillectomy, immune-modulatory treatments such as plasma exchange, IVIG, and so on, have all been described as ameliorating the manifestations.9,11,12
In spite of widespread resistance to antibiotics, penicillin is still the first choice for therapy and controlling of GABHS. Erythromycin is the best option of medication for patients responsive to penicillin.13,14
Our study supports the hypothesis that a postinfectious immune mechanism to GABHS such as PANDAS may play a role in the pathogenesis of stuttering. A combination of throat culture, rapid antigen detection test, and ASO and anti-DNase B, antistreptokinase together with the presence of stuttering is needed to achieve the maximum sensitivity and specificity for detecting GABHS. We advise the use of antibiotic prophylaxis in PANDAS stuttering cases to anticipate repeated streptococcal infections. New studies are required to determine the optimum symptomatic, control, and medicinal strategy, such as preventive tonsillectomy or aggressive treatment (intravenous immunoglobulin, plasmapheresis, etc).
Footnotes
Acknowledgements
We would like to thank Handan Koyuncu, MD, for her assistance in patient follow-up during the study.
Data Availability
It is possible to access data related to the study if needed.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written consent form was obtained from all participants.
