Abstract
Introduction
Nasopharyngeal melanoma (NPM) is a rare malignancy with high rates of recurrence, metastasis, and vascular invasion. The estimated incidence of NPM is only about 0.3 per 1,000,000, per year, 1 which typically results in a poor prognosis, with a 5-year overall survival rate of only 10% to 30% due to its late onset, susceptibility to misdiagnosis, and high rates of local recurrence and metastasis.2,3 So far, the pathogenesis of NPM is unknown. Some scholars believe that cigarette smoke and formaldehyde in the air have a certain relationship with their pathogenesis, 4 but this has not been proven. Here, we report a rare case of NPM in a 59-year-old man without metastases and infiltrates, and review the relevant literature.
Case Reports
A 59-year-old male patient with no previous history of melanoma presented to the hospital for treatment of persistent nasal congestion and earplugs for 2 months. On endoscopy, a large, fleshy, smooth, grayish-black, nontender tumor was found in the nasopharynx that blocked the entire posterior nostril. Contrast-enhanced computed tomography (CT) scan shows a soft-tissue mass in the nasopharynx, approximately 2.1 cm × 1.8 cm. Enhanced magnetic resonance imaging showed that T2-weighted images showed quasi-circulatory hyperintensities with uneven pressure of fat in the posterior wall of the upper nasopharynx, while T1-weighted images showed slightly shorter and equally strong shadows. The margins are clear and there is no invasion of surrounding tissues (Figure 1). Other tests, such as blood tests, CT of the lungs, CT of the abdomen, and color Doppler ultrasound of the cervical lymph nodes, were unremarkable. After 3 days, the patient underwent surgery and the tumor was completely removed. During surgery, the tumor did not invade deep tissues, and the pharyngeal fossa, eustachian tube, pharyngeal opening, and parapharyngeal space were not affected. Intraoperative cryopathology results showed negative resection margins.
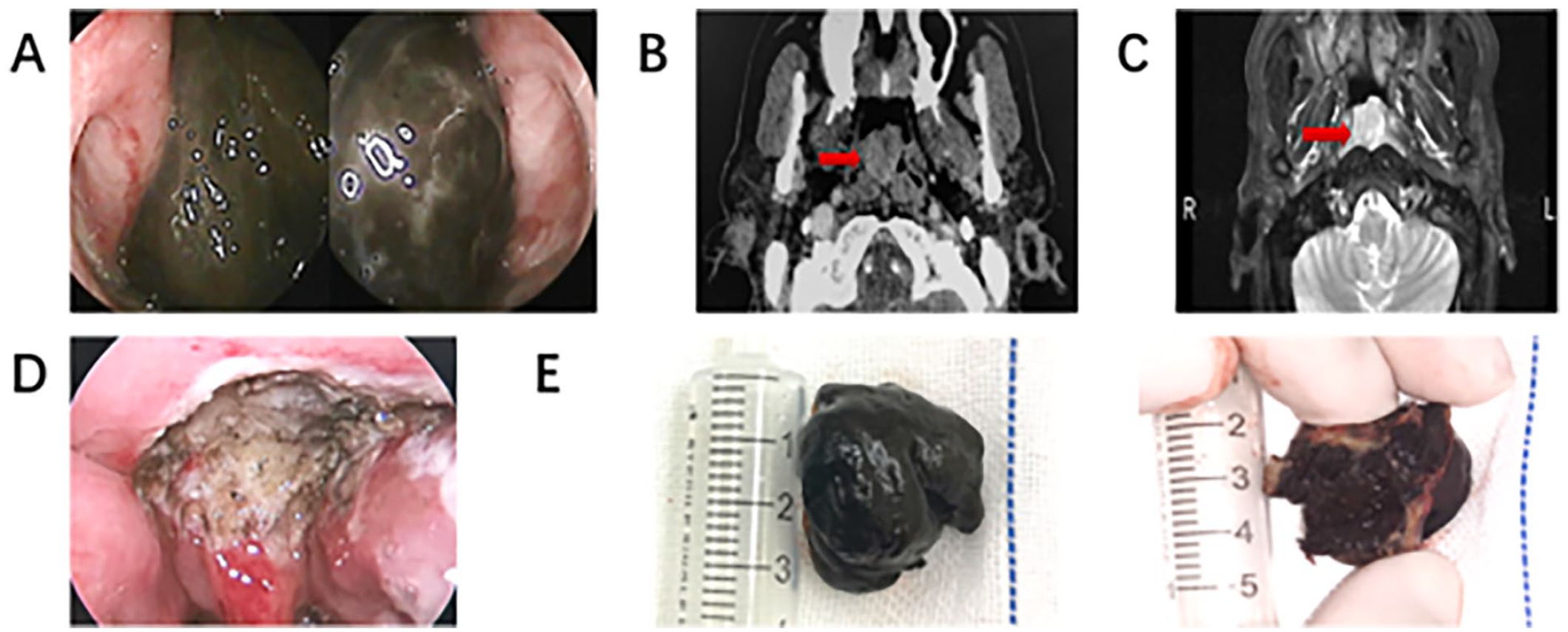
(A) Dark black tumor in the nasopharynx, with a less smooth surface, occluding bilateral pharyngeal recesses; (B) Computed tomography of the nasopharynx showed a soft tissue nodule on the posterior wall of the top of the nasopharynx, with a maximum size of about 2.1 cm × 1.8 cm (arrow); (C) MRI of the nasopharynx showed a quasi-circular posterior wall of the pharyngeal roof with uneven fat pressure hyperintensity, a slightly shorter T1WI and no obvious damage to the skull base (arrow). (4) Postoperative nasopharyngeal section; (E) The tumor is about 3.0 cm × 2.0 cm × 2.0 cm in size, relatively intact, smooth surface, black and red as a whole, solid and irregular, gray-black sections. MRI, magnetic resonance imaging; T2WI, T2-weighted images; T1WI, T1 weighted image.
The size of the resected tumor is about 3.0 cm × 2.0 cm × 2.0 cm, relatively intact, smooth surface, black and red overall, solid irregular, gray-black sections. Microscopic examination reveals a large number of melanocytes, large vesicular nuclei, prominent nucleoli, and poorly defined cytoplasm. Immunohistochemistry showed that tumor cells were positive for Melan-A, HMB-45, SMA, and S100, and negative for Ki-67 (+20%-30%) and AE1/AE3 (Figure 2). Therefore, the final diagnosis is primary NPM.
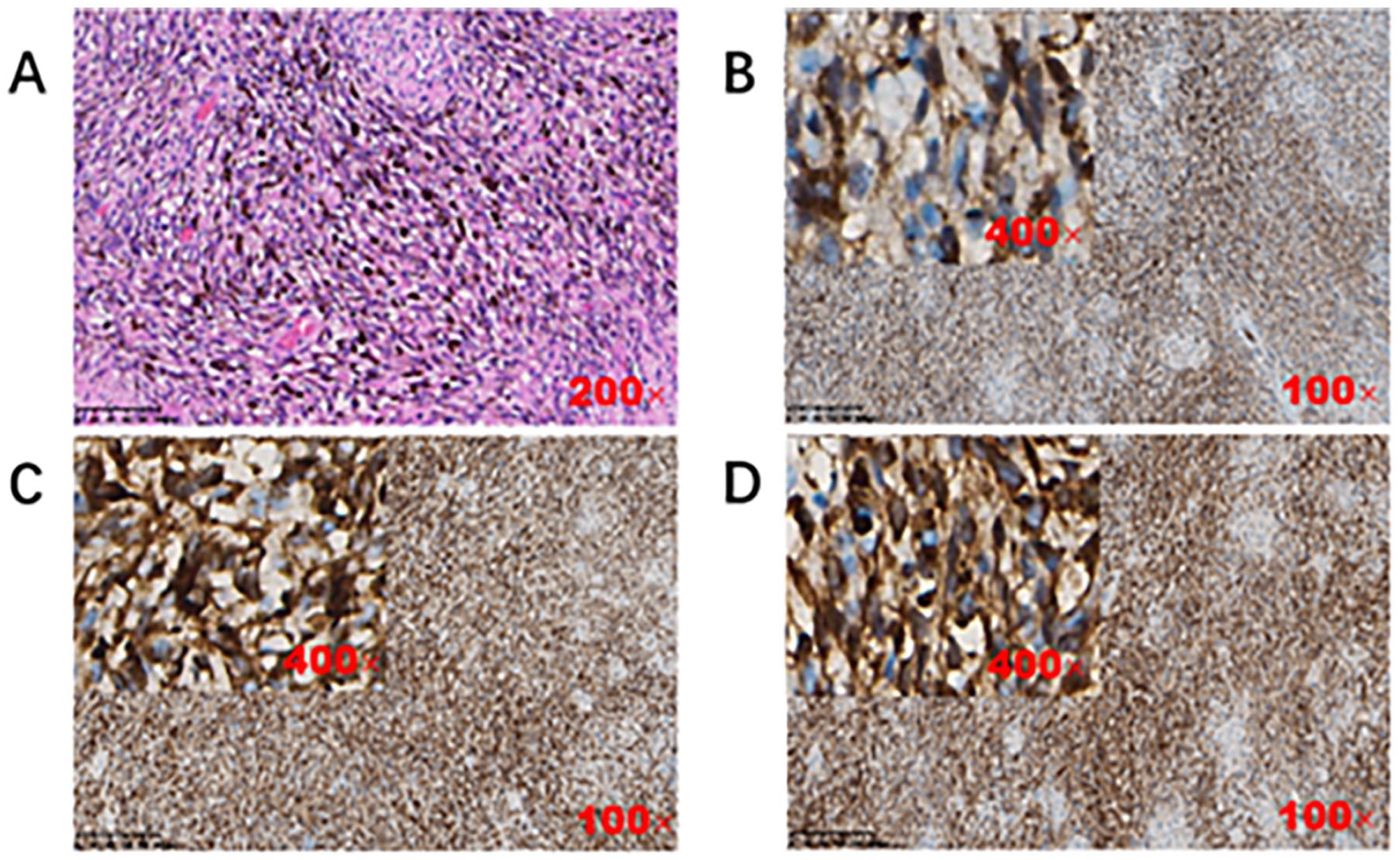
Histopathological findings. (A) HE staining showed a large number of melanocytes, large vesicular nuclei, obvious nucleoli, and unclear cytoplasm; (B) Tumor cells exhibit strong cytoplasmic and nuclear S-100 positivity; (C) Tumor cells exhibit strong cytoplasmic positivity for HMB-45; (D) Tumor cells exhibit strong cytoplasmic positivity for Melan-A. HE, hematoxylin and eosin.
Two months after surgery, the patient received adjuvant radiotherapy (60 gy/30 F) and was in satisfactory condition. Subsequently, we followed the patient for nearly 2 years, and no recurrent or metastatic disease was detected by endoscopy and imaging (Figure 3).
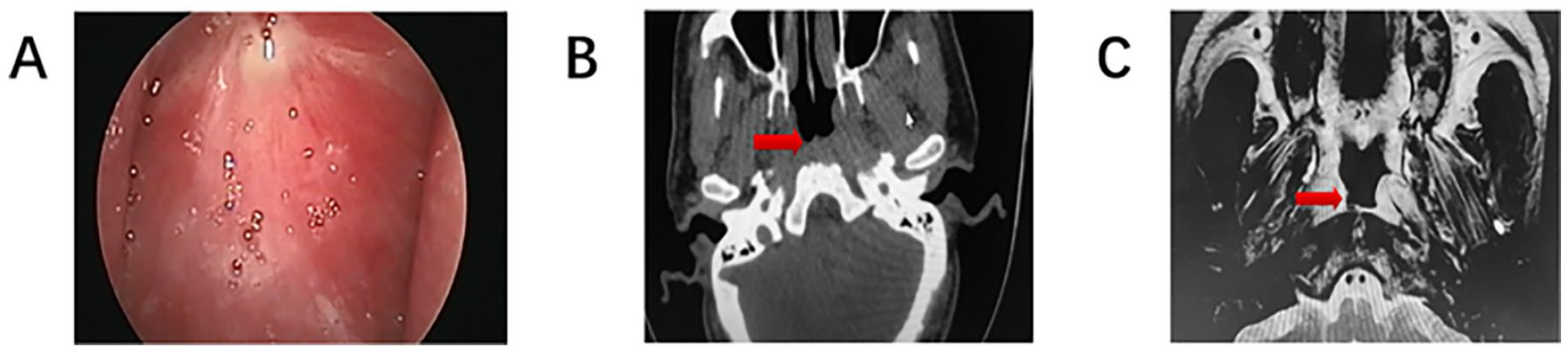
Follow-up 24 months after surgery. (A) Nasopharyngeal mucosa is smooth, local scars can be seen, and there is no mucosal pigment; (B) nasopharynx space-occupying lesions changed after surgery, and slightly thick soft tissue shadow could be seen in the posterior and lateral pharyngeal walls; (C) there was an abnormal signal on the right side of the top of the nasopharynx. Considering the postoperative changes, no definite signs of residual tumor and recurrence were found.
Discussion
Mucosal melanoma (MM) is a highly aggressive malignant melanoma, accounting for about 1.4% of all melanomas in Caucasians, 5 which occurs in the head and neck region accounting for about 55% of MM. 6 In China, MM is the second largest subtype of melanoma (20%-25%). 7 Tumors can occur in the oral cavity, genital tract, esophagus, nasal cavity/sinuses/nasopharynx, urinary tract, and leptomeninges. 8 Due to the special location, NPM often does not have obvious clinical symptoms in the prelesion stage. The first symptoms of patients are also usually nasal congestion and epistaxis. A retrospective study over 25 years reported that the main symptoms of NPM are epistaxis, nasal congestion, ear tightness, pain, and runny nose. 2 Pain, facial deformity, exophthalmos, diplopia, and other symptoms will also occur after the tumor invades the surrounding tissues.9,10 Under the endoscopy, NPM can appear as a brittle bleeding mass with a smooth or unsmooth surface, and may also appear as blue-black, gray-white, or light yellow lesion. 10 When there is a lack of melanin particles, the lump may also appear as a translucent polypoid mass, which is often incorrectly diagnosed as the common nasal polyp, leading to misdiagnosis and delayed follow-up treatment. 11
The gold standard for diagnosing NPM is histopathological examination. Although the abundant melanin granules in the cytoplasm of this tumor make it easier to diagnose this disease, about 13% to 25% of these tumors lack melanin granules, so they need to be differentiated from other malignant tumors, such as undifferentiated small blue cell tumor of the nasal cavity and paranasal sinus, Ewing’s sarcoma, peripheral neuroectodermal tumor, squamous cell carcinoma, and non-Hodgkin’s lymphoma.12,13 Immunohistochemistry plays an important role in a definitive diagnosis. NPM expresses S-100 protein and melanocyte markers such as Melan-A, HMB-45, tyrosinase, and microphthalmia-associated transcription factor in a variety of ways. 14 The sensitivity of the S-100 protein can be as high as 90%, but because it is not specific, it cannot be used as a marker for the definite diagnosis of MM. 15 The positive rates of HMB-45 and tyrosinase in MM were similar, as are 75%, while Melan-A and MITF were slightly lower, 65% and 57%, respectively. 2 PNL-2 was confirmed to be a high-sensitivity marker for MM in some newer marker research trials, which may be superior to Melan-A and MITF, comparable to HMB-45, and its specificity is superior to S-100. Therefore, PNL-2 can be used as an auxiliary marker for the combined diagnosis of MM with S-100 and HMB-45. 16 Some studies have also confirmed that Sox10 is a highly sensitive and specific marker of melanocytes. If added to the test of conventional melanocyte markers, Sox10 may have a higher diagnostic value than S-100 protein. 17
Surgical resection of the tumor is the first option for the treatment of NPM. Surgeons generally include an external nasal lateral incision approach and nasal endoscopic surgery, which are determined mainly based on the size of the tumor, the extent of invasion, and the surgical level of the operator. In fact, for these 2 different surgical methods, the survival difference is not statistically significant. 18 The intraoperative negative margin is an important factor affecting the prognosis. Currently, according to the range of tumor invasion, a safe negative margin of about 1.5 to 2.0 cm should be provided after resection. 19 However, due to the particularity of the anatomical location of the nasopharynx, the safe margin range should be carefully measured during surgery.
In addition to surgery, radiation therapy and chemotherapy have also been widely used in the adjuvant treatment of NPM. Although no study has confirmed that these 2 methods can improve the survival rate of patients, they can improve the local disease control rate.20,21 In recent years, many new treatment options have emerged, especially targeted therapy (ie, C-kit, NRAS/MEK, BRAF inhibitors) and immunotherapy (anti-CTLA-4, anti-PD-1/PD-L1 antibodies, and anti-LAG-3 antibodies), which have effectively changed the course of MM. For some patients with metastatic or nonresectable NPM, it can become an effective treatment. Currently, there is no standard treatment procedure for NPM, and surgical resection remains the main treatment strategy.
Conclusions
NPM is a rare malignant tumor with a poor prognosis, and few cases have been reported in the literature. Surgical resection is the main treatment. Postoperative adjuvant therapy can improve the local control rate of the disease. Immunotherapy and targeted therapy are expected to become important options for the treatment of NPM in the future. In addition, early diagnosis and comprehensive examination are extremely important for the prognosis of patients. In conclusion, malignant MM originating in the nasopharynx is extremely rare and a detailed differential diagnosis of nasopharynx tumors should be considered.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The patient provided oral informed consent for the information and images of the case in the article to be published.
