Abstract
Introduction
Benign central airway stenosis is a scarcely encountered respiratory condition characterized by an obstructive airway, which can cause severe breathing difficulties and pose a significant threat to life. 1 The causes of benign central airway stenosis are diverse, including prolonged tracheal intubation, tracheostomy, benign airway tumors, airway trauma, and idiopathic stenosis. 2 The clinical manifestations of benign central airway stenosis often encompass difficulty in breathing, difficulty in speaking, chronic cough, a sense of foreign body in the throat, wheezing, and respiratory distress. 3 Irrespective of the patient’s medical history, the mucosa of the stenotic area can be observed under the microscope, displaying features of epithelial damage, excessive inflammatory response, and proliferative scar formation, all of which contribute to the narrowing of the airway lumen and result in the observed clinical symptoms. 4 Although abnormal inflammatory responses are often associated with pathogenesis, whether excessive inflammatory responses play a role in the onset and recurrence of the condition has not been fully clarified. To further delve into the etiology of benign central airway stenosis, this study explored its pathogenesis at the genetic level. Benign central airway stenosis poses a great challenge to respiratory and thoracic surgeons. 5 Currently, the most common treatment methods include surgical reconstruction and bronchoscopic interventions. 6 Surgical airway reconstruction is typically used for complex and refractory stenosis cases, while bronchoscopic interventions are frequently utilized for simpler cases. 7 However, the incidence rate of airway restenosis following these treatments is as high as 40%-70%, posing a considerable financial burden on patients and significantly impacting their postoperative quality of life. 8
In recent years, bronchoscopic interventions have gained increasing popularity in clinical practice. 9 The combination of balloon dilation and electrical ablation, cryotherapy, Nd-YAG laser, drug injection, tracheal stent placement, etc. can quickly open up the airway and relieve symptoms.10,11 However, these methods may stimulate the growth of local granulation tissue, leading to restenosis, and also cause postoperative complications such as airway mucosal swelling and difficulty in hemostasis.12,13 Therefore, there is a need to explore new treatment techniques that can be performed under bronchoscopic guidance, reduce surgical trauma, and better address clinical challenges. Coblation, a technique based on high-energy plasma, was initially used in orthopedics and can achieve both tissue ablation and vessel sealing. Its application in otolaryngology and head-neck surgery is also increasing.14-16 Currently, there is no published data on the combination of coblation with bronchoscopy for the treatment of benign central airway stenosis. This study aims to assess the therapeutic effect and treatment prospects of this technique on large airway stenosis and to explore the pathogenesis at the genetic level.
Materials and Methods
Methods
The ethical approval for this study was granted by the Ethics Committee of The Second Hospital of Hebei Medical University. A retrospective study was conducted on patients with benign central airway stenosis who were treated at The Second Hospital of Hebei Medical University from August 1, 2017 to September 30, 2020. All patients provided informed consent and underwent the prescribed treatment procedures. Detailed records were maintained for each patient, including individual characteristics such as age, gender, etiology, treatment procedures, and follow-up results.
Assessment of airway stenosis
In this study, considering the irregular shape of stenosis caused by granulation tissue growth in the airway, the narrow airway luminal area was assessed as the measurement parameter using a computed tomography scanner (Optima ct660, General Electric, Chicago, Illinois, USA, slice thickness: 1.25 mm). All patients with large airway stenosis in this study had stenosis at different sites in the trachea. To accurately describe the degree of tracheal stenosis, the Myer-Cotton grading system was used. The tracheal lumen area at the site of maximum narrowing (determined via 3D reconstruction) was compared with the normal lumen area both proximally and distally to the stenosis to calculate the percentage of stenosis. The calculation method for the degree of stenosis was as follows: degree of stenosis = (initial lumen area − postoperative lumen area)/initial lumen area × 100%. The severity of tracheal stenosis was classified into four grades: grade 1: stenosis ≤ 50%, grade 2: stenosis = 51%-70%, grade 3: stenosis = 71%-99%, and grade 4: complete tracheal obstruction. The frequency and duration of re-treatment following follow-up were recorded, and the re-treatment effect was compared with that of the initial treatment.
Patient data
The study encompassed patients with benign central airway stenosis who sought treatment at The Second Hospital of Hebei Medical University from August 1, 2017 to September 30, 2020. Inclusion criteria were as follows: (1) patients with benign central airway stenosis caused by various primary or secondary factors; (2) those with dyspnea symptoms that could not be relieved by conservative treatment; (3) those with benign central airway stenosis who were intolerant of surgical intervention; (4) those with benign central airway stenosis who refused surgical treatment and chose bronchoscopic intervention. Exclusion criteria included the following: (1) children or adolescents under 18 years of age; (2) patients with active pulmonary or tracheobronchial tuberculosis; and (3) those with severe underlying diseases or coagulation disorders who could not tolerate bronchoscopic diagnosis and treatment.
A total of 32 patients (n = 32) were included, including 8 females and 24 males, with a mean age of (51.22 ± 14.18) years; 1 patient presented with granulation tissue growth after T-tube insertion, 23 patients had a history of tracheotomy, and 8 patients had a history of tracheal intubation. There were varying degrees of scar stenosis and granulation tissue growth at the tracheostomy site and tracheal intubation site.
Surgical technique
Preoperative examination
To improve preoperative examination, routine examination and evaluation were performed on patients before bronchoscopic intervention, including electrocardiography, pulmonary function test, arterial blood gas analysis, complete blood count, coagulation function test, and preoperative screening. The surgical risk and patient tolerance were assessed while discontinuing anticoagulant and antiplatelet medications prior to the procedure. Thin-slice computed tomography scans with contrast enhancement and pathological diagnosis were used to identify the nature, extent, location, and scope of the lesions in the tracheal lumen. Informed consent was obtained for bronchoscopy and interventional therapy after informing both the patient and their family members about the risks as well as common postoperative complications associated with bronchoscopic intervention. Prior to the procedure, a comprehensive examination of the trachea and lobar bronchi was performed to determine the location, extent, and type of stenosis.
Surgical procedure
All patients underwent the procedure under general anesthesia in the operating room. The patient was placed in a supine position and different ventilation methods were used depending on the etiology. For patients with post-tracheotomy stenosis using silicone tracheotomy tubes, a direct connection to the anesthesia machine was feasible. If metal tracheotomy tubes were used, they were replaced with tracheal tubes before connecting to the anesthesia machine. For patients with post-endotracheal intubation stenosis, high-frequency jet ventilation was provided with a rigid bronchoscope. Every patient was given dexamethasone at 0.5 mg/kg.
The employed techniques involved standard bronchoscopy (Olympus, Shinjuku, Tokyo, Japan) and micro laryngoscopy (Storz, Germany) using a self-retaining laryngoscope and a rigid telescope. At the start of the surgical operation, a rigid bronchoscope (Storz, Tuttlingen, Germany) was inserted orally under the guidance of a video bronchoscope (Olympus, Japan), and the rigid bronchoscopic sheath was fixed below the glottis to fully expose the stenotic site. The video bronchoscope was used to clear airway secretions, providing a clear view for the surgical team. Local anesthetics consisting of an appropriate dose of 2% lidocaine were administered. Then, a low-temperature plasma radiofrequency ablation electrode was inserted into the bronchoscopic sheath to ablate the tissue using a laryngeal-specific wand. The ablation was performed using the ARS600 low-temperature plasma surgery system (BONSS CHINA) with an AC403 integrated plasma radiofrequency ablation electrode. The coblate setting was set at 7 watts and the coag setting was set at 3 watts. A flow controller regulated the synchronous dripping of normal saline, with the speed set primarily at 80-100 drops/min. The tip of the coblation wand was faced laterally, and coblation was performed with a medial-to-lateral and superior-to-inferior approach. An extended-length, ultrafine coblation wand (ArthroCare, Austin, Texas, USA) was inserted down to the level of the stenotic site under direct vision. Under direct vision, coblation was performed, and the stenotic segment was ablated as thoroughly as possible while preserving the mucosa over the tracheal cartilages. The distance between the distal end of the video bronchoscope and the ablation electrode was approximately 2 cm, providing a clear view of the ablation effect without damaging the bronchoscope. The synchronous suction function of the video bronchoscope was used to help remove the electrolyte and necrotic tissue. The wand was used to ablate the tissue from the center of the stenotic segment spreading outwards. The ablation direction was kept parallel to the long axis of the trachea to avoid damaging the normal tracheal wall structure. For patients in Myer-Cotton grade 4, biopsy forceps could be utilized to explore the direction of ablation while ablating. For patients who were ventilated through a tracheostomy tube, when the ablation reached above the tube, temporary removal of the tube was necessary to cease ventilation. The ablation electrode could then be inserted through the fistula following tracheostomy and retrograde ablation could be performed to remove the scar and granulation tissue. For patients with severe stenosis, Nd:YAG laser (MY100C, Ligenesis, Beijing, China) could be used to ablate the center of the stenosis before placing a balloon (CRE BALLOON, BOSTON and SCIENTIFIC, AMERICA) for repeated expansion, ensuring smooth contact of the electrode with the stenotic area. Electric coagulation hemostasis was used to control minimal bleeding in the ablation area, whereas adrenaline solution (1:10000) could be injected via the bronchoscope for cases with significant bleeding. Triamcinolone (80 mg/mL) was injected submucosally following Coblation. Upon completion of the procedure, for patients who could not breathe freely without support, a T-tube or silicone stent was typically employed as a substitute for the intubation tube, which could reduce the incidence of granulation.
Postoperative indicators
After surgery, patients were routinely monitored in the post-anesthesia care unit for 1-4 hours before being transferred back to the general ward. A follow-up bronchoscopy was performed 24-48 hours postoperatively to remove liquefied necrotic tissue, blood clots, and secretions, and inhaled budesonide, oral expectorants, or local injection of triamcinolone was given as needed. If necessary, biopsy forceps were used to remove granulation tissue. Patients received follow-up bronchoscopy at 1, 3, 6, 12, and 24 months after surgery to evaluate clinical symptoms and determine whether additional intervention was required. The prognosis was recorded accordingly. The study endpoint was defined as patient death, emergency tracheal intubation, or tracheotomy.
Statistical Analysis
SPSS 26 software package (Armonk, New York, USA) was used for data analysis. Fisher’s exact test was used to compare the degree of stenosis, and a paired t-test was used to analyze the differences in airway stenosis before and after treatment.
Genetic Analysis
Genetic regulation is believed to play a role in the development of benign central airway stenosis. The advent of novel technologies, such as next-generation sequencing, has led to significant advancements in genomic research. Big data bioinformatics analysis provides core data for identifying reliable and functional differentially expressed genes. In this study, the publicly available, annotated, and freely shared genomics datasets submitted by the international research community were analyzed. The GSE189336 dataset related to gene expression was mainly obtained from the GEO database (Bethesda, Maryland, USA). First, the literature search and dataset selection were conducted to compare the overall gene expression in the laryngotracheal mucosa between healthy subjects and patients with benign central airway stenosis. Second, data processing, statistical analysis, and identification of differentially expressed genes were performed. Raw data were downloaded from the selected databases, and differentially expressed genes were identified, with a significance level of P < .05. Relevant differentially expressed genes were screened for pathway analysis. Enrichr and TRRUST datasets (Houston, Texas, USA) were used to perform enrichment analysis on genes and transcription factors with the GSEA software (Cambridge, Massachusetts, USA). Gene functional annotation was performed on upregulated and downregulated genes. The heatmap of differentially expressed genes was plotted, with red representing increased expressions and blue representing decreased expressions.
Results
Therapeutic Effect
A total of 32 patients (n = 32) who underwent a total of 43 treatments were included, including 8 females and 24 males, with a mean age of (51.22 ± 14.18) years; 1 patient had granulation tissue growth after T-tube insertion, 23 patients had a history of tracheotomy, and 8 patients had a history of tracheal intubation. There were varying degrees of scar stenosis and granulation tissue growth at the tracheostomy site and tracheal intubation site. All patients were followed up for a median duration of 18 [1-24] months, and their symptoms improved after treatment. Computed tomography scans were performed to assess the efficacy of airway reconstruction using multi-planar reconstruction. At the final postoperative assessment, all patients showed postoperative improvement in airway stenosis, without any major surgical or postoperative complications (Figures 1-3). All patients were treated with coblation. Different auxiliary treatment methods were used according to the degree and location of stenosis, including simple ablation treatment, ablation treatment + T-tube placement, and ablation treatment + stent placement. In some patients with severe stenosis (grade 4 stenosis), the direction of the trachea cannot be determined due to atresia of the tracheal lumen. In this case, if we solely rely on coblation treatment, there is a risk of inadvertent damage to the lateral wall of the trachea, leading to airway fistula formation. Therefore, our approach involves the initial utilization of NDYAG ablation to meticulously treat the central stenosis area and subsequently gradually expand the wound. The balloon catheter can be maneuvered smoothly through the narrow site with this procedure, resulting in clear exposure of the distal tracheal structure upon pressure expansion, followed by coblation for treatment. The purpose of performing NDYAG ablation and balloon operation is to minimize the occurrence of complications rather than treating, and refraining from being primarily utilized for therapeutic purposes.
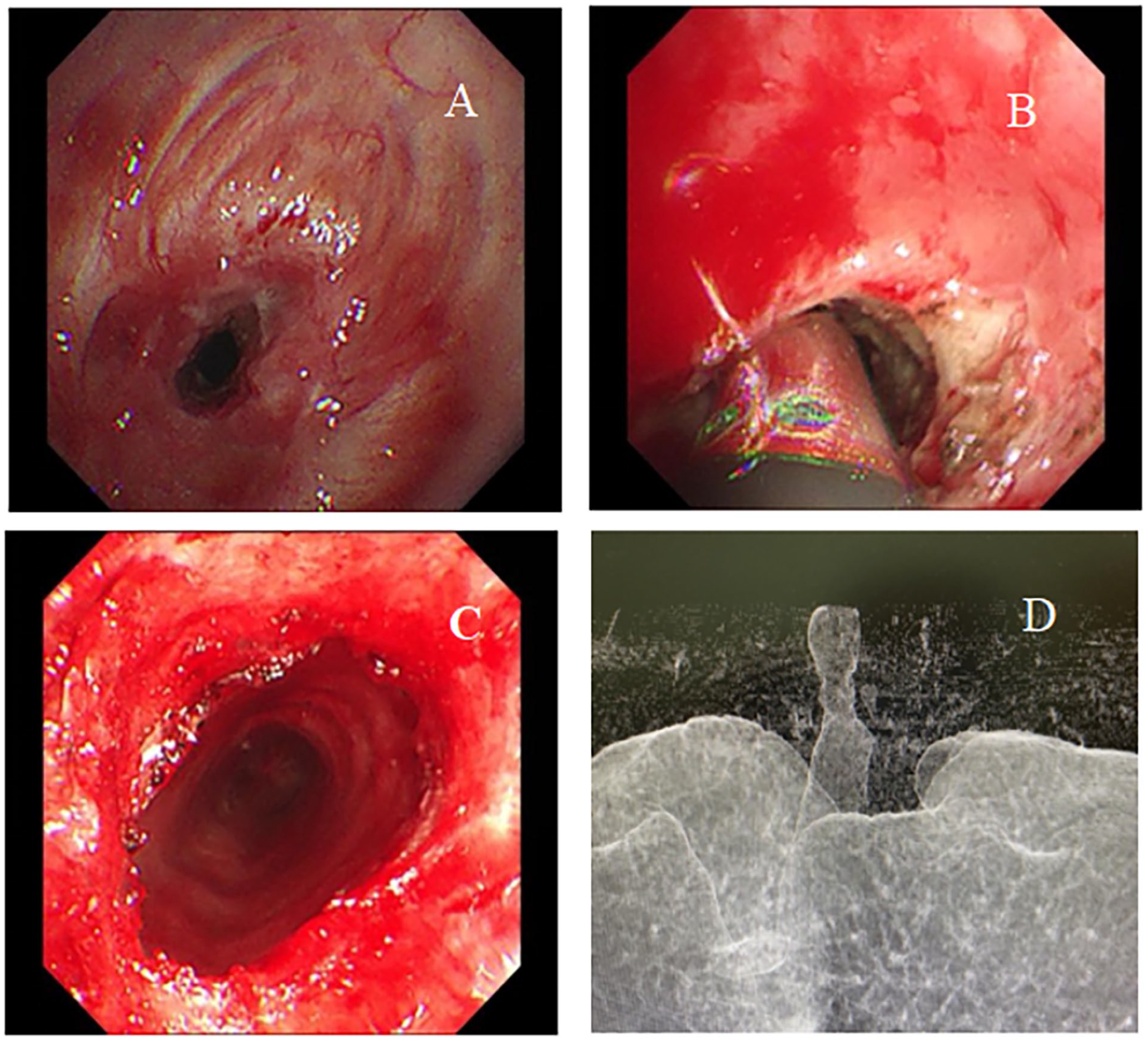
Coblation treatment of a patient with tracheal stenosis. (A) Tracheal stenosis. (B) Performing coblation treatment. (C) The outcome of the procedure. (D) Initial computed tomography scan showing tracheal stenosis.
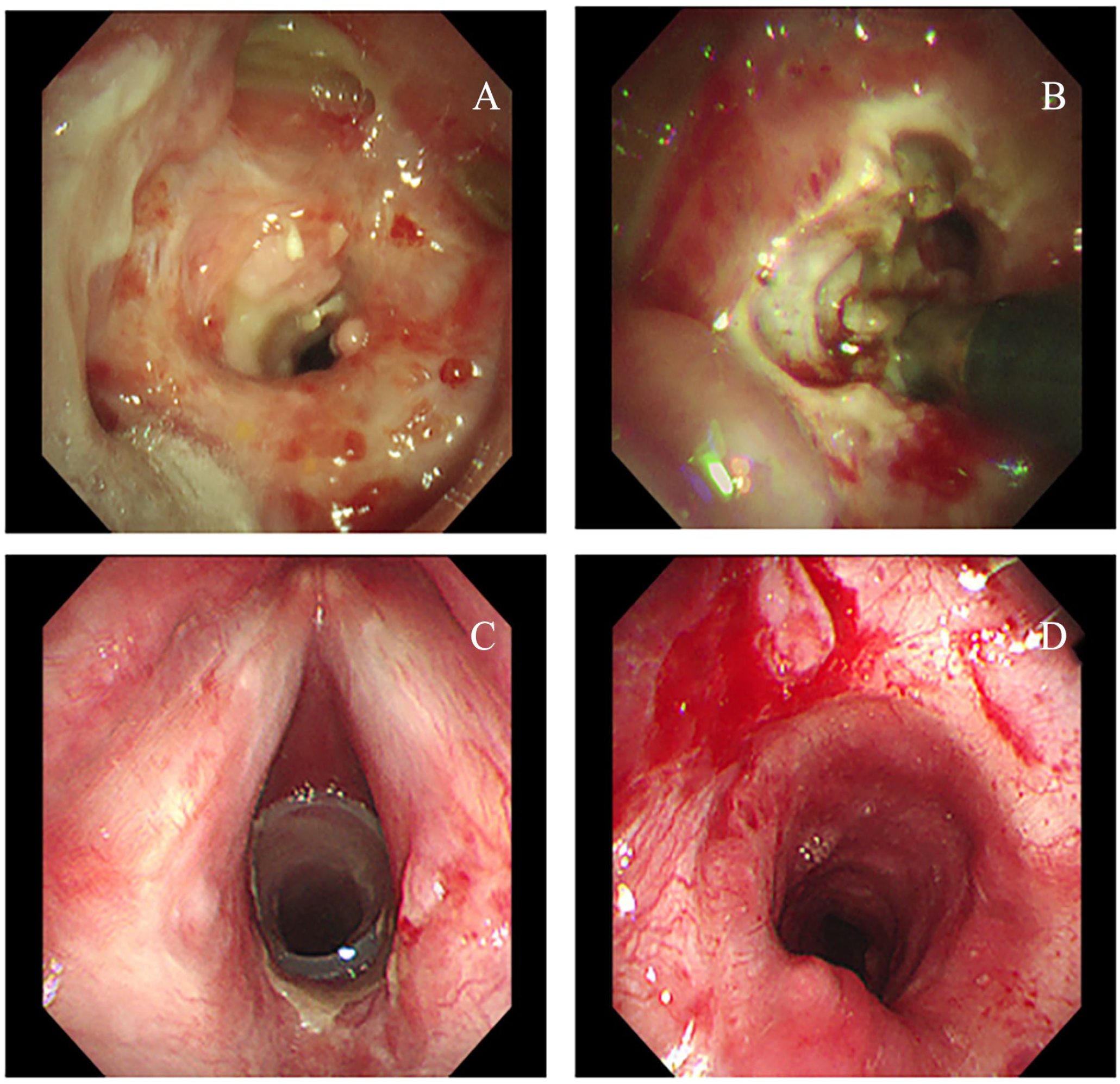
Coblation treatment of a patient with tracheal stenosis. (A) Tracheal stenosis. (B) Performing coblation treatment. (C) Placement of a silicone stent. (D) After the removal of the silicone stent.
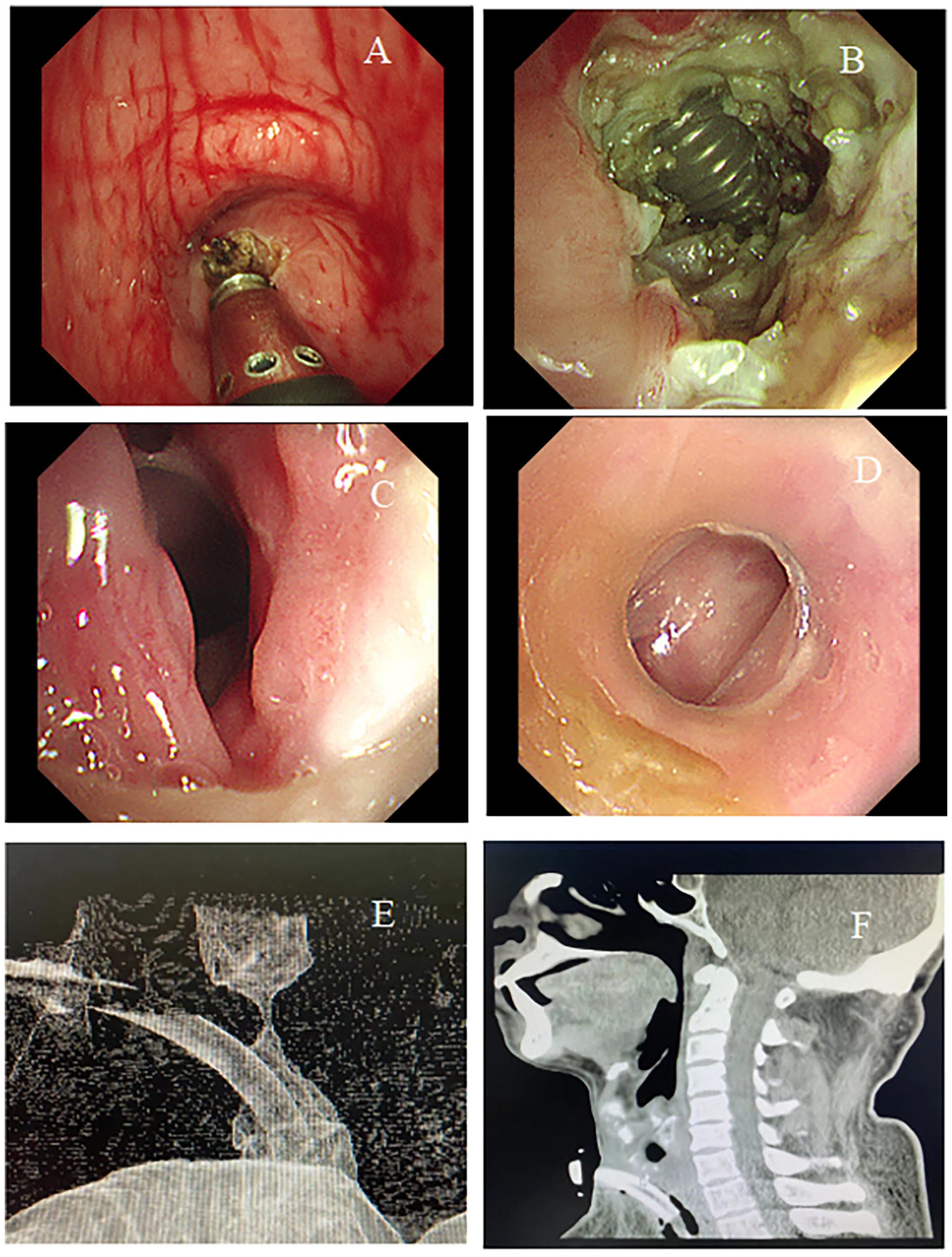
Coblation treatment of a patient with benign central airway stenosis. (A) Performing the coblation treatment. (B) Tracheotomy tube exposed after ablation. (C) Placement of a T-tube. (D) Outcome of the treatment. (E) A computed tomography scan of the air structure showed tracheal stenosis and a metal tracheotomy tube. (F) Minimum intensity projection showing stenosis.
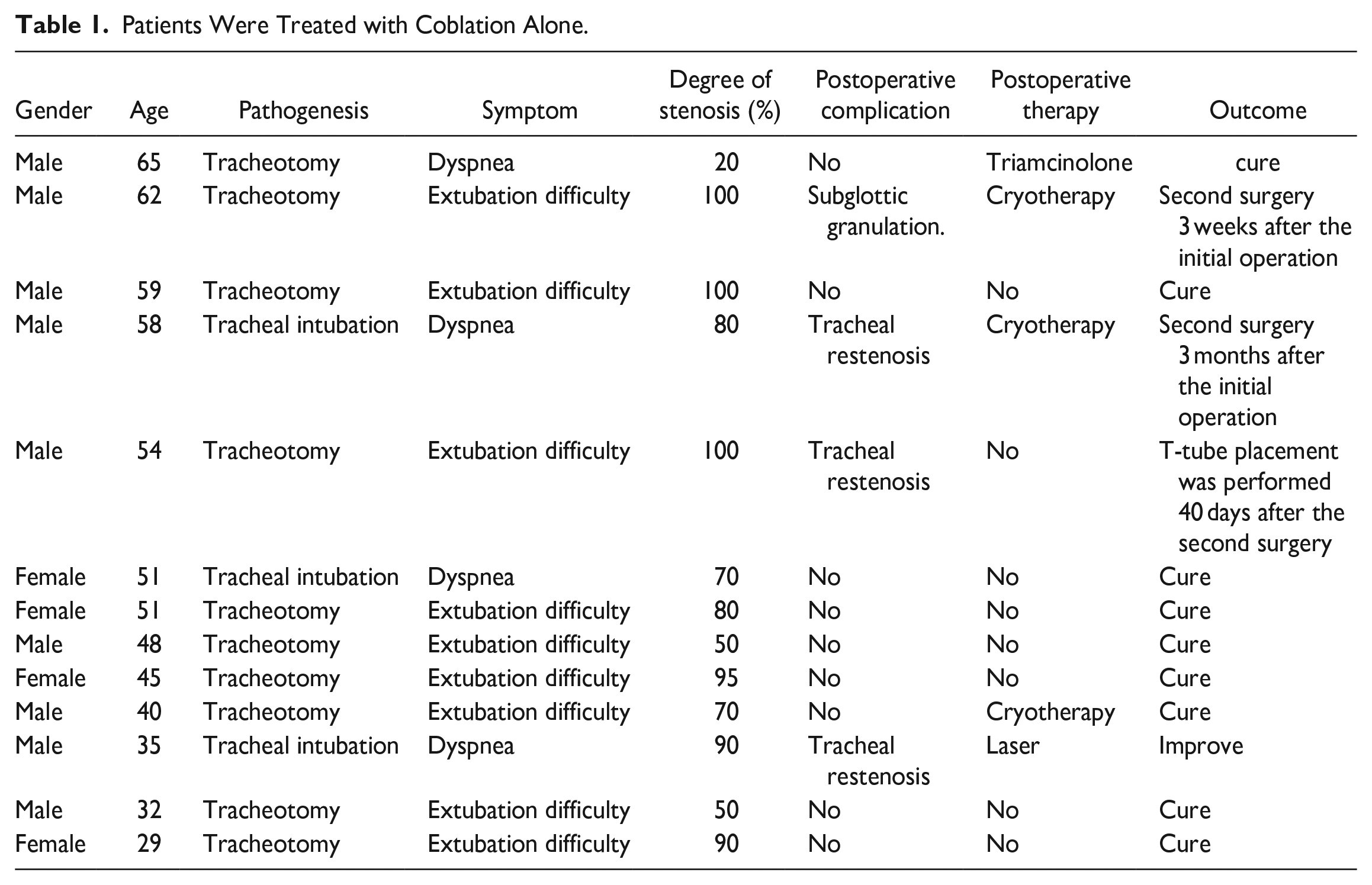
The majority of patients only required one coblation treatment, while only 5 cases required additional surgical treatment (Tables 1-3 and Figure 4).
Patients Were Treated with Coblation Alone.
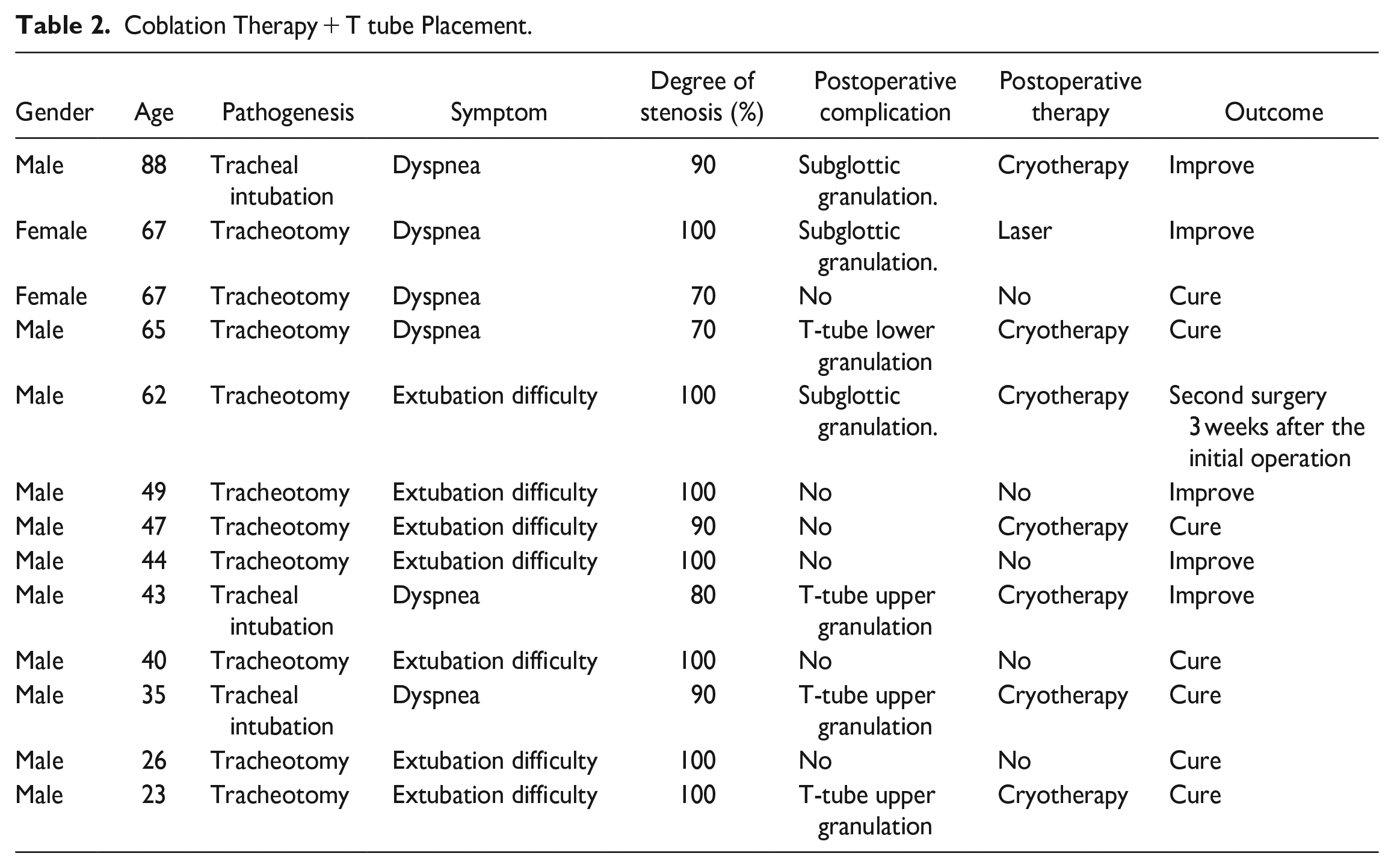
Coblation Therapy + T tube Placement.
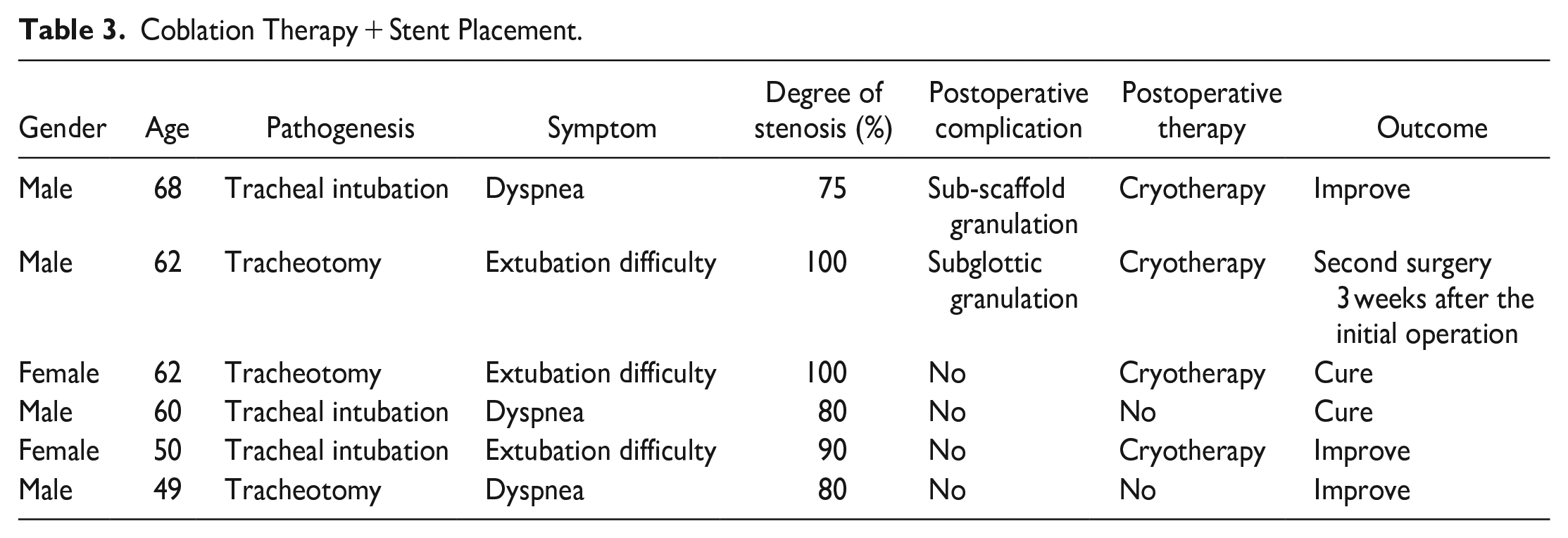
Coblation Therapy + Stent Placement.
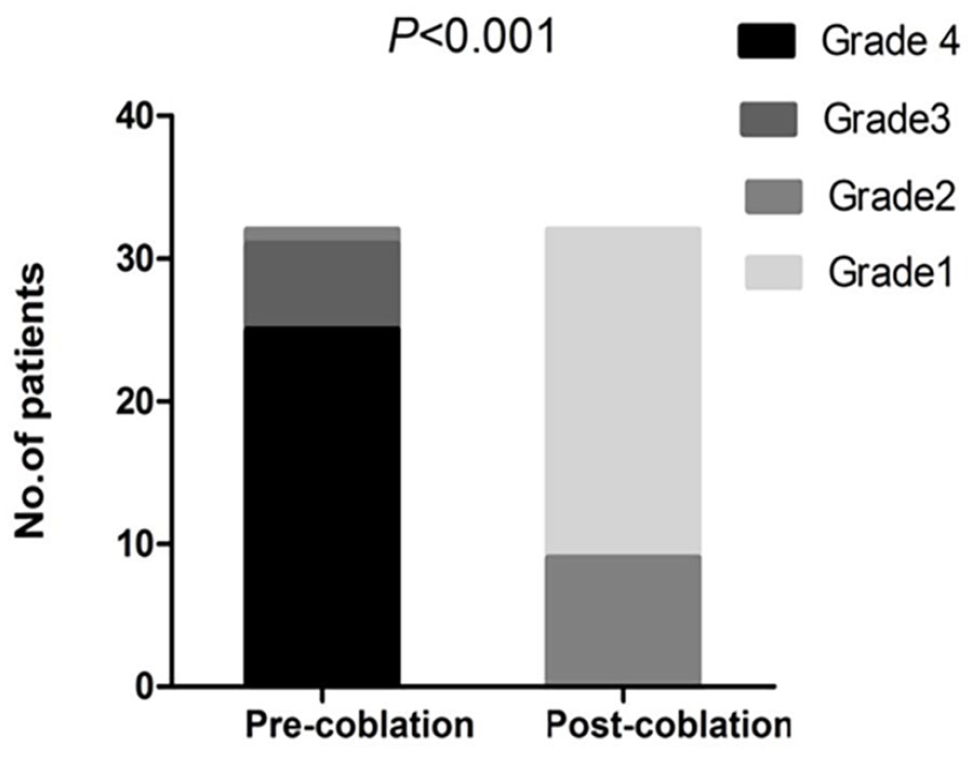
Change in the grade of tracheal stenosis before and after coblation treatment.
Genomic Analysis
Genomic analysis showed significant changes in the gene expression of the airway mucosal layer in patients with benign central airway stenosis. Compared with healthy individuals, patients with benign central airway stenosis exhibited multiple genes with aberrant expressions. A total of 91 differentially expressed genes were identified, including 44 upregulated genes. The upregulated genes exhibited characteristics of promoting inflammatory responses, including regulation of the interleukin, interferon, and NF-κB signaling pathways. Some of these upregulated genes, such as CXCL8, CD74, IL1B, IL1A, CXCR6, CCL17, NFKB1, NFKB2, NFKBIA, CD55, and HIF1A, exhibited features of acute and widespread inflammatory responses. NF-κB, for instance, regulated inflammation, and its pathways were involved in mediating the overexpression of several inflammatory cytokines (Tables 4 and 5; Figure 5).
Upregulated Genes.
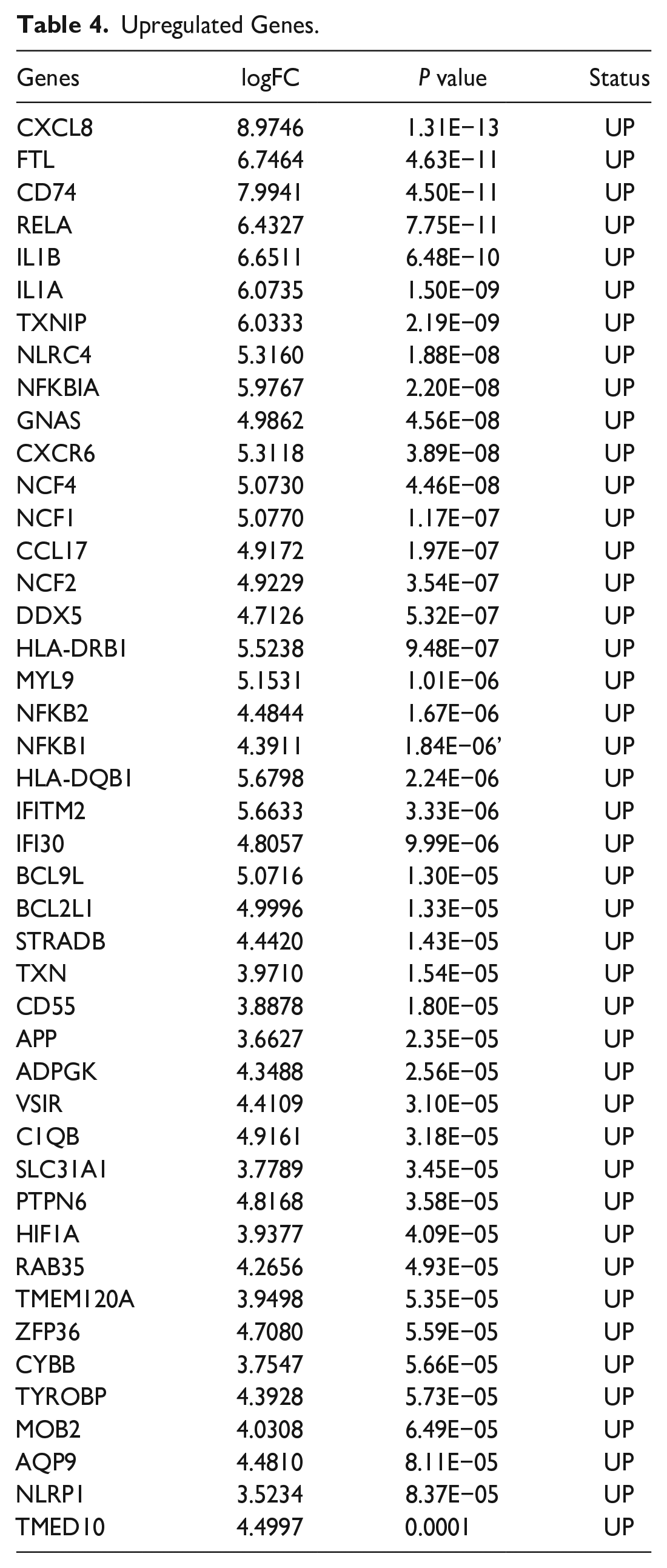
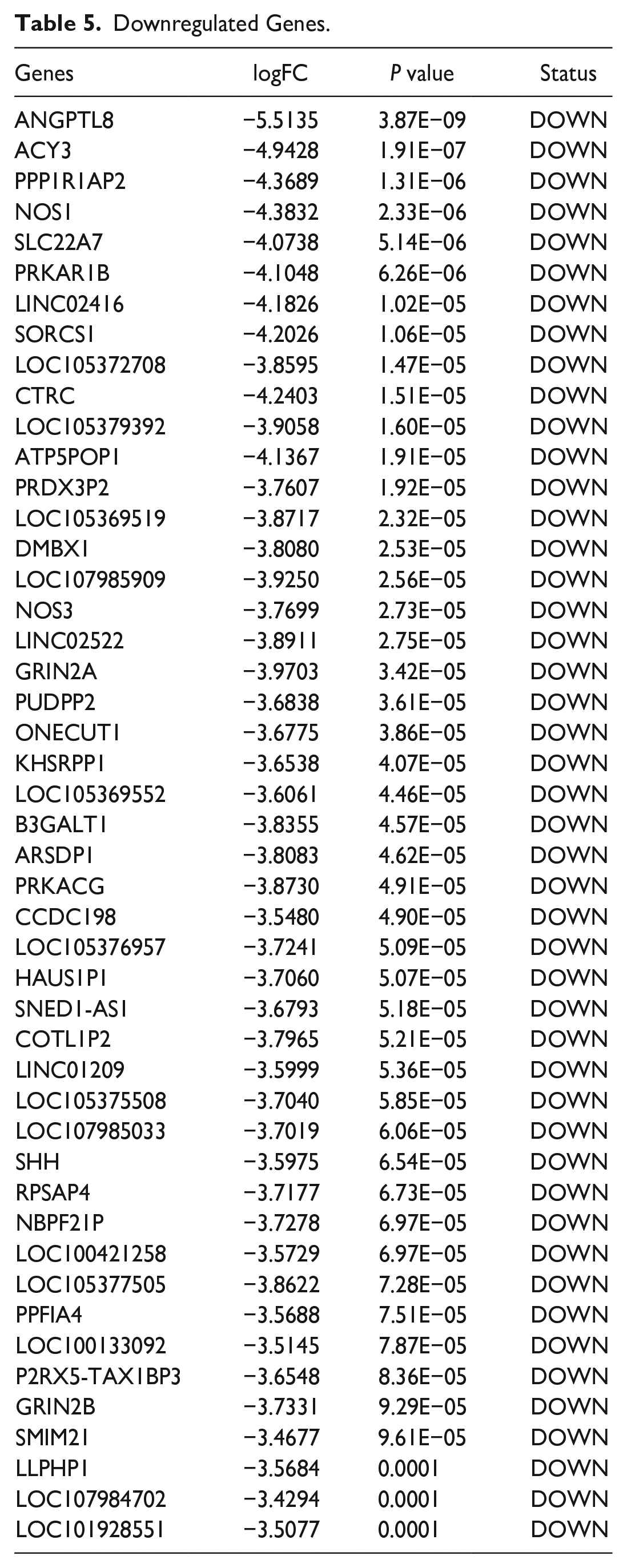
Downregulated Genes.
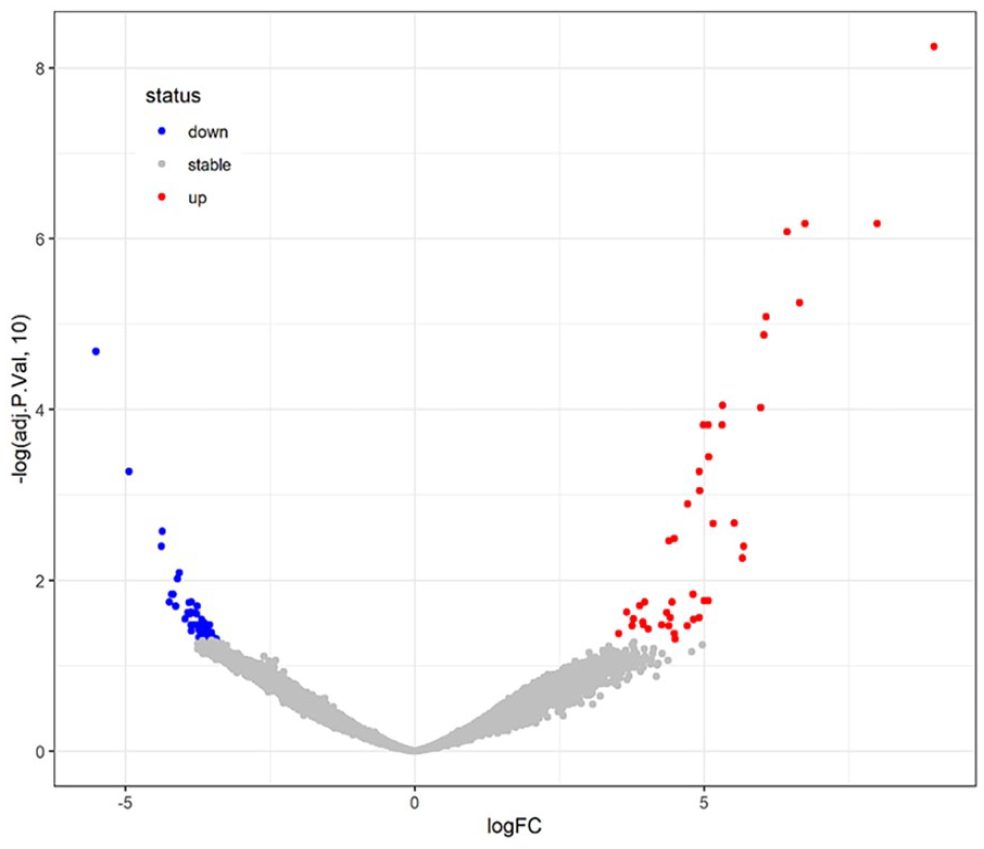
A scatter plot of RNA-seq data shows differentially expressed genes, with red indicating upregulated expression and blue indicating downregulated expression.
Discussion
Patients with benign central airway stenosis frequently encounter high recurrence rates after treatment, necessitating repeated bronchoscopic interventions or surgery to eliminate growing scars and granulation tissues and maintain airway patency.17,18 The most commonly used bronchoscopic interventions include electrical ablation, balloon dilation, laser ablation, cryotherapy, tracheal stent placement, and tracheostomy. Airway restenosis after treatment occurs in 40%-70% of cases. 8 These treatment methods can easily lead to local granulation tissue growth and difficulties in hemostasis. Coblation, a novel minimally invasive technique, can be utilized in conjunction with other thermal ablation and hemostasis techniques. In recent years, it has been widely used in the treatment of early-stage laryngeal cancer in pediatric surgery and otolaryngology, demonstrating promising outcomes.19-21 The main principle of coblation is to form a plasma layer under a 100 kHz plasma radiofrequency electric field. These charged particles generate enough kinetic energy to break the molecular bonds of target tissue cells at low temperatures (40-70°C), resulting in rapid decomposition of the target tissue into carbohydrates and oxides, and ultimately causing coagulation and necrosis of tissue proteins and blood vessels.22,23 As a result, tissues are reduced, ablated, and removed. 23 In addition, coblation can also close small blood vessels that are cut during the procedure, resulting in hemostasis. Its working temperature is significantly lower than that of electrosurgery and laser therapy, which can significantly reduce the risk of deep and surrounding tissue damage. 24 Moreover, the radiofrequency electrode used in coblation can be bent into different angles according to the actual needs of the surgery, making it adaptable to narrow areas with various shapes and angles and increasing the treatment effect. In this study, the safety and effectiveness of coblation treatment were assessed. This technique successfully eliminated narrow tissues without additional damage to the airway while reducing the stimulation of scar and granulation tissue growth. As a result, the risk of airway stenosis recurrence was significantly reduced, and the time interval for retreatment was substantially extended. In addition, all patients showed improved clinical symptoms without any serious complications. Moreover, the treatment effects on different degrees of stenosis were compared. It was found that coblation was successful for grade 1, 2, 3, and 4 stenosis. To prevent tracheal collapse and excessive granulation tissue proliferation during treatment, a T-tube or silicone stent was placed simultaneously during coblation, depending on the patient’s clinical condition. T-tubes and silicone stents have several advantages over other stents. For example, T-tubes have a good fixation effect and are not prone to displacement; the small diameter of the T-tube reduces direct contact with the tracheal wall, reducing excessive granulation tissue growth caused by stimulation; caregivers can directly remove secretions from the proximal end of the T-tube through the open end. 25 Silicone stents have better tissue compatibility and cause minimal damage to the airway mucosa, thereby reducing stimulation against granulation tissue growth. After follow-up observation for 1-2 years and once the airway structure was recovered, the T-tube or silicone stent was removed.
According to the whole-genome analysis, benign central airway stenosis is mainly characterized by excessive inflammatory response. Studies have shown that the airway changes in benign central airway stenosis resemble those in chronic granulomatous disease, which is a specific immune state with long-term high expressions of pro-inflammatory cytokines, sustained infiltration of neutrophils, and granuloma formation. 26 The results of this study showed that there were significant changes in the gene expression in the airway mucosal layer of patients with benign central airway stenosis. Compared with healthy individuals, patients with benign central airway stenosis had multiple genes with abnormal expressions. There were 91 differentially expressed genes, of which 44 were upregulated. These upregulated genes exhibited characteristics of promoting inflammatory responses, promoting granulation tissue growth, and upregulating genes associated with cancer. First, the upregulated genes facilitated inflammatory response, including regulating interleukin, interferon, and NF-κB signaling pathways. CXCL8, CD74, IL-1B, IL-1A, CXCR6, CCL17, NFKB1, NFKB2, NFKBIA, CD55, HIF1A, etc. displayed characteristics of acute inflammatory reactions. 27 Among them, NF-κB is involved in regulating the inflammatory response, and the NF-κB pathway participates in mediating the overexpression of various inflammatory factors. Hypoxia-Inducible Factor (HIF) is an important transcription factor that initiates stress response and is regulated by NFKB. 28 Second, the upregulated genes promoted granulation tissue growth. FTL, DDX5, and RAB35 are involved in the granulation tissue growth.29,30 TXNIP is overexpressed, which can promote oxidative stress response and lead to further programmed cell death. 31 In addition, NLRC4 is involved in the inflammatory response, mediating the production of IL-1β and IL-18, inducing cell pyroptosis and significantly contributing to inflammatory responses. 32 Third, some cancer-related genes were found to be upregulated. MYL9 can enhance tumor cell proliferation, invasion, and migration, which is closely related to non-small-cell lung cancer. 33 Overexpression of SLC31A1 is involved in cell apoptosis. HIF1A enhances the expression of gamma globulin genes, activates the production of erythropoietin, and regulates the expression of fetal hemoglobin, enabling the body to acquire more oxygen from the air under hypoxic conditions. 34 Overexpression of NLRP1 may lead to hyperactive inflammatory responses and increased susceptibility to cancer. 35 In this paper, it was confirmed that excessive inflammatory response was the major feature of benign central airway stenosis and that the airway mucosa of patients was more consistent with the manifestations of acute inflammation. Prolonged and sustained high expression of pro-inflammatory cytokines and excessive inflammatory responses, accompanied by persistent neutrophil infiltration and secondary granulation, were the main characteristics. A deeper understanding of the pathophysiology of benign central airway stenosis can provide more treatment options in the future, such as local steroid hormone injection or local interferon therapy, which requires further study. We hope that this study will provide direction for new diagnoses and treatments in the future.
This retrospective study had a limited dataset size, along with the presence of multiple pathological etiologies, a short follow-up period, and a potential uncontrolled false discovery rate. In future studies, we will increase the sample size and extend the follow-up time to stratify and analyze the optimal surgical treatment for benign central airway stenosis with different etiologies.
Conclusion
Coblation has demonstrated its efficacy as a reliable treatment option for benign central airway stenosis in adults, exhibiting broad prospects for clinical application. This study was a retrospective analysis with a limited sample size and a short follow-up period. Future investigations will encompass larger sample sizes and extended follow-up periods to stratify and analyze the optimal surgical treatment for benign central airway stenosis with different etiologies.
Footnotes
Availability of data and materials
Data will be available on request.
Credit Authorship Contribution Statement
Haitao Li: Methodology, Conceptualization, Project administration. Xuezhu Ren: Software, Validation, Formal analysis. Haizhong Zhang: Data curation. Shuai Li: Formal analysis. Yuzheng He: Formal analysis. Tianjie Qi: Writing—original draft. Zhigang Cai: Funding acquisition, Supervision, Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project is supported by the Health Commission of Hebei Province (20190509), Hebei Provincial Natural Science Foundation of China (H2021206067), and Hebei Provincial Natural Science Foundation of China (H2020206294).
