Abstract
Neonatal suppurative parotitis is a rare condition that is diagnosed primarily through clinical evaluation. The prognosis is generally good. In this report, we present the case of a 21-day-old female infant who was treated in our ENT department for neonatal suppurative parotitis. The patient presented with a feverish, hard, red, and tender swelling of the parotid loge which displaced the right earlobe outward. In addition, purulent exudate was observed from Stensen’s duct. A computed tomography scan of the cervical and cerebral regions revealed a swollen right parotid gland with heterogeneous density and enhancement, but no detectable collection. The patient received intravenous antibiotics for 48 hours, resulting in a favorable outcome without the need for surgical treatment. The aim of this study is to emphasize the diagnostic and therapeutic aspects of this clinical condition. Suppurative parotitis should be suspected by the clinician in newborns who present with an inflammatory preauricular swelling, with or without contributing factors. The outcome is generally favorable, and complications are rare with timely and appropriate medical treatment.
Introduction
Parotitis is an inflammatory or infectious condition of the parotid gland that can be viral, recurrent, or chronic. 1 Although it is common in adults, suppurative acute parotitis is rare in children and even more so in newborns. There are some case reports and small case series that have described neonatal suppurative parotitis (NSP) in newborns, but there is little information on the contributing factors for this condition. 2 This report presents a case of NSP in a neonate who has cared for in our department.
The aim of this work is to highlight the diagnostic and therapeutic aspects of this clinical condition.
Case Presentation
A 21-day-old female infant presented to the ENT emergency department with a febrile context, preauricular swelling, decreased oral intake, and irritability for the past 2 days. The patient was the result of an uneventful pregnancy followed by a spontaneous vaginal delivery at term without any intrapartum trauma or complications. The patient was breastfed at home and had normal stool. Before admission, the mother noticed that the baby became irritable during feeding and spent less time at the breast. This was followed by an inflammatory swelling in front of the right ear that rapidly increased in size, along with local signs of inflammation. The parents reported no history of trauma to the newborn’s head or face, and the mother denied any history of skin infections or breast tenderness, as well as any drug use by herself or the baby. During the physical examination, the newborn presented with a fever of 38.9°C. The patient was alert, nontoxic, and well hydrated. An inflammatory swelling was observed in front of the newborn’s right ear, extending across the angle of the mandible to the area under the earlobe (Figure 1a). The swelling was hard, red, and tender, and displaced the right earlobe outward. Purulent exudate was noted from Stensen’s duct. The remainder of the medical examination was unremarkable. The C-reactive protein level was 250 mg/L. A lumbar puncture was performed, and the results were normal. Samples of purulent material from Stensen’s duct, blood, cerebrospinal fluid, and urine were sent for culture. A cervical and cerebral computed tomography (CT) scan was performed, which showed a heterogenous dense swollen right parotid gland with enhancement and no detectable collection, along with significant infiltration of the right parotid and masticatory spaces. These findings were consistent with acute NSP (Figure 2). The newborn received intravenous antibiotic therapy with ceftriaxone (100 mg/kg/day) and gentamycin (3 mg/kg/day). After 48 hours of admission, the newborn showed clinical improvement with reduced erythematous swelling (Figure 1b). Staphylococcus aureus was identified in the culture of purulent exudate from Stensen’s duct, while other cultures showed no pathogens. The newborn was treated with oral cefpodoxime for 10 days. After 2 weeks of follow-up, the newborn showed a favorable outcome.
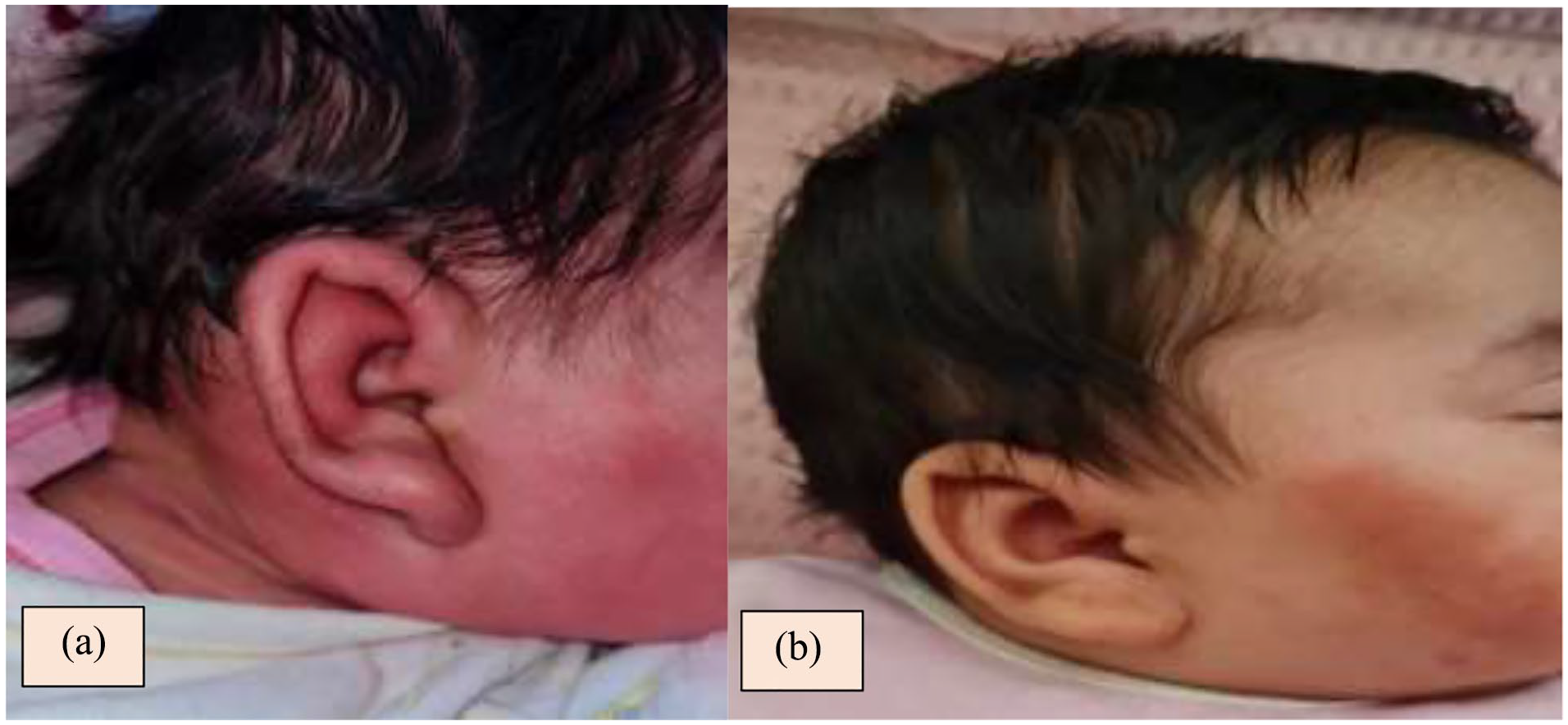
(a) Inflammatory swelling of the parotid region with extension toward the auricle of the right ear. (b) Marked improvement in inflammatory swelling in the parotid region and total regression of inflammation in the right ear after 48 hours of intravenous antibiotic therapy.
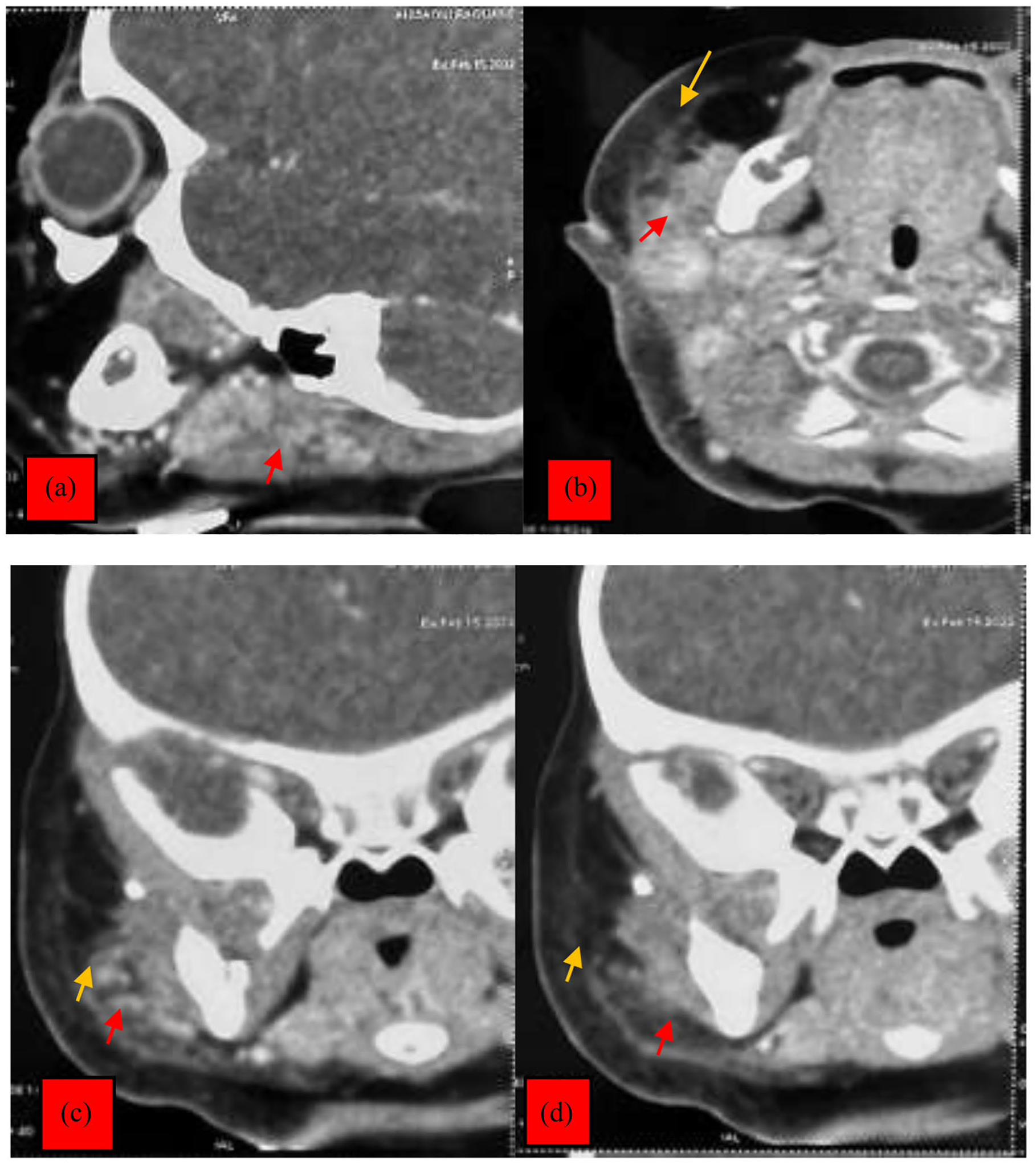
CT scan of the facial bones in (a) sagittal, (b) axial, and (c, d) coronal sections: swollen right parotid gland with heterogeneous density and enhancement without detectable collection (red arrow) significant edematous infiltration of the right parotid and masticatory spaces (yellow arrow). CT, computed tomography.
Discussion
Salivary gland infections are uncommon in newborns. However, the parotid gland is the more susceptible site of infection due to its production of only serous secretions, lacking the bacteriostatic property of the mucoid constituent found in the submandibular gland. The submandibular gland produces a mixture of serous and mucous secretions containing lysozyme and immunoglobulin A.2-4 NSP has a prevalence of 3.6 to 14/10,000 admissions.4,5 A review of the literature identified only 32 cases of neonatal bacterial parotitis over a 35 year period. 6 The condition is more common in male infants, with a sex ratio of 3:1. 6 Risk factors for NSP include prematurity, dehydration, low birth weight, malnutrition, orogastric tube feeding, and ductal stasis.3,5 Prematurity is a major risk factor due to prolonged hospitalization and medical procedures such as the use of an orogastric tube or catheter, nasopharyngeal, and intraoral aspiration. These procedures can cause trauma to the oral mucosa and Stensen’s duct. 7 In addition, premature infants are at a higher risk of dehydration, which reduces salivary secretion and clearance.8-10
Several mechanisms may be involved in NSP. NSP can occur retrogradely in the gland due to decreased saliva production and stasis, congenital modifications in ductal structure, and dilatation of salivary ducts caused by obstruction from a mass or stone. 2 It can also occur via the hematogenous route 10 or through transmission of pathogens from contaminated formula or during breastfeeding.4,9 Todoroki et al 11 suggests that NSP can be caused by congenital Cytomegalovirus infection and maternal methyldopa therapy.
Diagnosis of NSP is primarily clinical, based on the presence of fever, local swelling, erythema, local warmth, and painful tenderness of the parotid gland, typically occurring between 7 and 14 days after birth. Although purulent discharge from Stensen’s duct is pathognomonic,1,2,5 it is not always present. NSP is usually unilateral, but bilateral cases are extremely rare.6,12
The other possible causes of the condition are trauma, maxillary infection lymphangiomas, hemangiomas, adenomas, and lipomas.2,9
The bacteria responsible for infectious parotitis are those found in the oral cavity, as well as those responsible for invasive infections. 5 S. aureus is the most commonly implicated pathogen. Gram-positive cocci, such as Streptococcus pyogenes, Peptostreptococcus spp., viridian streptococci, and coagulase-negative Staphylococcus spp. are commonly implicated in hematogenous and nosocomial infections, while gram-negative bacilli, including Klebsiella pneumoniae, Escherichia coli, and Moraxella catarrhalis are less frequently involved. Anaerobic bacteria are rarely the cause of such infections.3,13
Recent observations have identified multidrug-resistant strains. 10 Although S. agalactiae (group B Streptococcus) is responsible for 25% to 40% of infections of the newborn. 5 In our case, we isolated multisensitive S. aureus.
Parotid ultrasound can be used to detect a sialolith or parotid abscess. If the patient’s condition does not improve after 2 days, further imaging examinations should be performed to monitor the infection in the retropharyngeal areas or to rule out abscess formation. 2 The gland appears markedly hypoechogenic with heterogeneous areas. Parotid ultrasound is the preferred examination, particularly in children, due to its speed and noninvasive nature. Excluding a differential diagnosis, such as a cyst or other tumor-like mass, 5 is also possible. CT and magnetic resonance imaging have better results in adults. In our case, CT was performed due to the redness in the pavilion and the associated retroauricular area, raising suspicion of associated mastoiditis.
Complications such as facial palsy, fistula formation, mediastinitis, and extension into the external auditory canal are rare since the use of antibiotics. 2 No complications have been observed in this case.
The therapeutic management is mainly medical. A first-generation cephalosporin or a penicillinase-resistant penicillin can be used to cover S. aureus, along with clindamycin or an equivalent antibiotic to cover possible anaerobic pathogens. These are good initial options until better guidance can be acquired from the study of cultures of expressed secretion from Stensen’s duct.4,5 The strain of methicillin-resistant S. aureus may require the use of vancomycin. Cefoxitin, clindamycin, imipenem, or a penicillin with a beta-lactamase inhibitor or the combination of a macrolide and metronidazole provide appropriate coverage of aerobic pathogens. 4
Surgical drainage should only be considered for complicated abscesses or if there is no clinical improvement after 24 to 48 hours of medical treatment.6,9,11 In this case, the patient received intravenous antibiotic therapy without surgical drainage.
The duration of antibiotic therapy is not well defined, but treatment for 7 to 10 days is essential until local signs of inflammation disappear. In premature infants, if there is organ failure or an anaerobic germ has been isolated, the duration of treatment will be prolonged. 10 The patient’s condition improved within 48 hours of receiving intravenous antibiotic treatment, and surgical intervention was not required.
Conclusions
Clinician should consider the possibility of suppurative parotitis in infants who present with an erythematous preauricular swelling, with or without predisposing factors.
Footnotes
Acknowledgements
None.
Data Availability
Data will be made available on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
