Abstract

Significant Statement
Secretory carcinoma (SC) is a salivary gland adenocarcinoma commonly characterized by ETV6::NTRK3 fusion gene. In most cases, it shows an indolent clinical behavior. We report the case a patient with SC of the parotid gland presenting as macrocystic lesion. The diagnosis was made based on cytomorphology and immunohistochemistry, including nuclear positivity for pan-TRK, and confirmed by molecular analysis. Macrocystic SC is an unusual variant of SC and may represent a relevant pitfall for both clinicians and pathologists.
Secretory carcinoma (SC) is a distinct variant of adenocarcinoma of the salivary gland commonly characterized by indolent behavior. 1 It was recognized as a specific entity in 2010 by Skálová and colleagues. 2 Most of the cases harbor the translocation t(12;15)(p13;q25) that results in ETV6::NTRK3 fusion gene.1-3 SC usually presents as a painless slowly growing mass. However, when high-grade transformation occurs, the clinical behavior is more aggressive and the patients are at risk for distant metastasis and show a poor prognosis.1,2,4,5 An unusual variant of SC characterized by macrocystic presentation has been recently reported. 6
In this article, we report the case of a 58-year-old Caucasian smoker male admitted for a 1 year history of a painless swelling in the region of the right parotid gland. It showed an elastic consistency and was mobile on the superficial and deep planes. The covering skin was intact and there was no lateral cervical adenopathy. There was no relevant personal or family history. Ultrasonography performed elsewhere before the admission revealed a macrocystic mass with a peripheral solid component 1.8 cm in maximum size in the superficial lobe of the parotid gland. The mass showed partial peripheral vascularization at power Doppler evaluation. A fine needle aspiration cytology of the lesion was also performed and interpreted elsewhere as consistent with either Warthin tumor or oncocytoma. Based on these findings, superficial parotidectomy was performed.
Histological examination revealed a macrocystic lesion surrounded by a fibrous capsule (Figure 1A). Lining of the cyst consisted of a mono- or multilayered epithelium focally forming luminal papillary projections and showing hobnail features (Figures 1B and 2A). The neoplastic cells showed round to oval nuclei with inconspicuous to small nucleoli and a granular, focally vacuolated, cytoplasm. Atypia, necrosis, invasion of the adjacent salivary gland parenchyma, lymph-vascular, and perineural invasion were not observed. Immunohistochemical analysis, performed as described previously,7,8 showed immunoreactivity of the neoplastic cells for CKAE1AE3, S100 (Figure 2B), and mammaglobin (Figure 2C) but not for p40, p63, DOG1, and androgen receptor. Notably, nuclear immunoreactivity in most of the neoplastic cells was also observed for pan-TRK (Figure 2D). The overall expression of Ki-67 was less than 5%. Based on these findings, the diagnosis of SC was made. As expected by the nuclear immunoreactivity for pan-TRK, whose specificity in both diagnosing SC and in detecting NTRK3 fusion has been reported to be highly specific,9-11 molecular analysis, performed on paraffin sections of the tumor by next-generation sequencing technology, confirmed the diagnosis demonstrating the ETV6::NTRK3 fusion gene. The patient did not receive adjuvant therapy. The postoperative course was unremarkable and recurrences were not observed for 4 months after surgery. Further follow-up is ongoing.
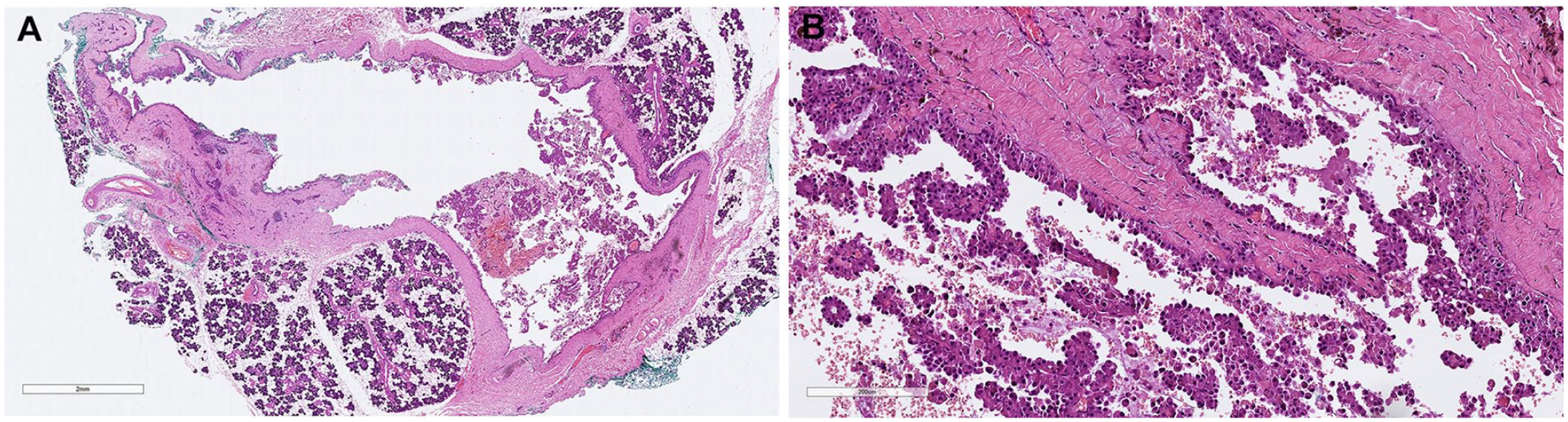
(A) Whole mount section of the tumor. (B) High-power magnification of the epithelial lining of the cyst. It appears either mono- or multilayered and focally forms papillary projections and show hobnail features. (A and B) Hematoxylin-eosin.
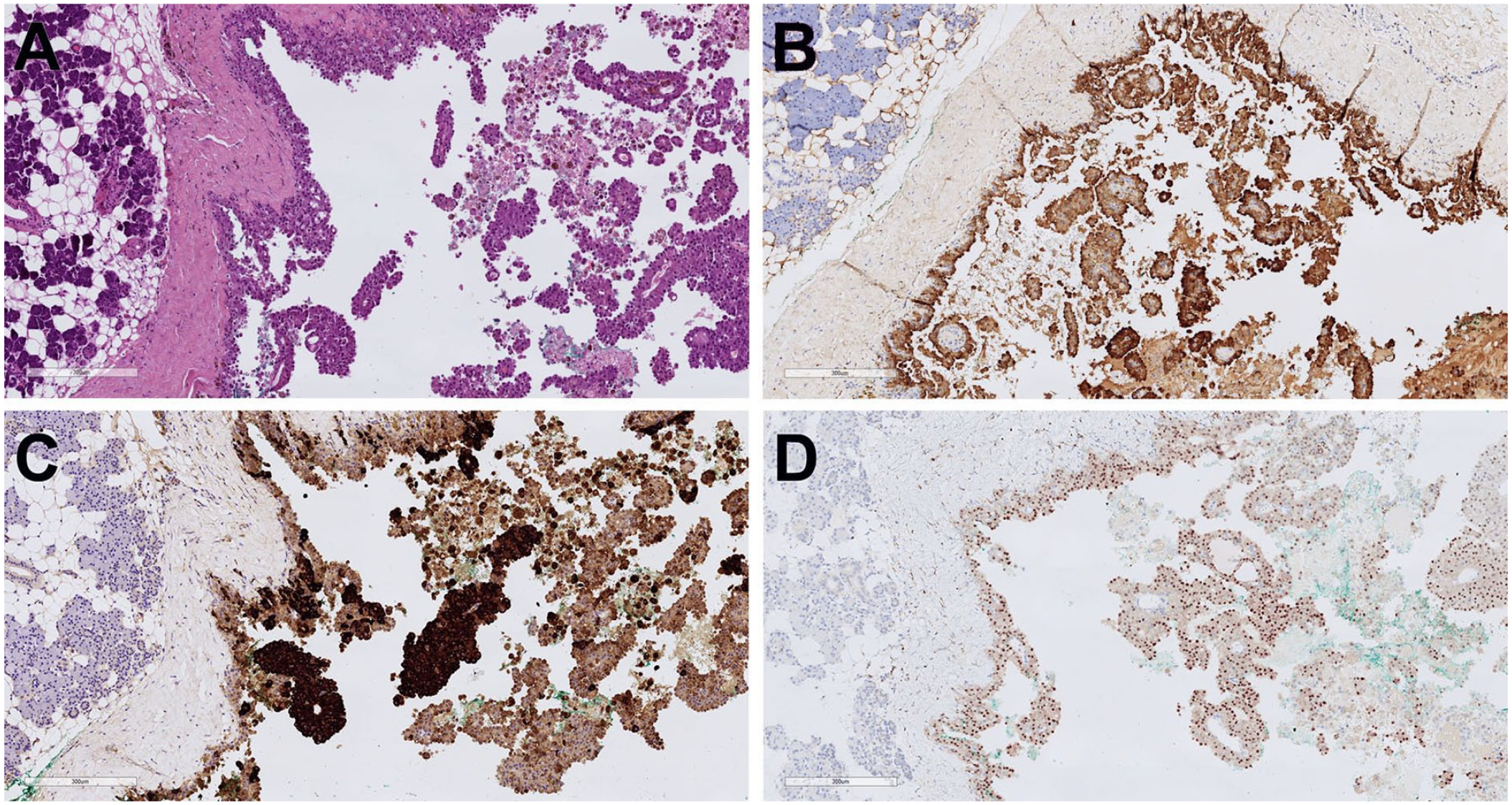
Serial sections of the tumor stained with (A) hematoxylin-eosin and immunostained for (B) S100, (C) mammaglobin, and (D) pan-TRK. The immunoreactivity for pan-TRK is restricted to the nucleus of the neoplastic cells.
The most commonly reported architectural pattern occurring in SC of the salivary gland include microcystic, cribriform, solid, and tubular/glandular.1-3,5 Although these patterns have been reported also to occur in association with macrocysts, a macrocystic variant of SC has been described only recently. 6 This variant may represent a diagnostic challenge for both clinicians and pathologists. Indeed, macrocystic SC may mimic different salivary gland cystic tumor including acinic cell carcinoma, low-grade mucoepidermoid carcinoma, cystadenoma, and intraductal carcinoma. 6 Evaluation of the cytomorphologic features and immunophenotype of the neoplastic cells and the molecular demonstration of ETV6 gene rearrangement can discriminate SC from the other entities. 6 The nuclear immunoreactivity for pan-TRK represents a valid surrogate for diagnosis and for predicting a response to TRK inhibitors in those cases, as that reported here, in which the classic ETV6 exon 5 to NTRK3 exon 15 fusion is present.9-11 However, its negativity does not exclude the diagnosis because fusion partners gene different from NTRK3 (eg, RET, MET)12-15 and other translocations (eg, VIM-RET, CTNNA1-ALK)15,16 have been associated to SC. Even though in our patient follow-up is limited to 4 months after surgery, we expect a prognosis comparable to that previously reported in a series of patients with macrocystic SC 6 who, after a median follow-up of 15 months, were all alive and with no evidence of disease. Indeed, the application of the recently proposed 3-tiered grading and scoring system for SC based on architecture and fibrous septae/fibrosis, nuclear pleomorphism, perineural invasion, lympho-vascular invasion and tumor necrosis, and mitotic activity/Ki-67 labeling index, 17 a macrocystic SC as that reported here, should be expected to fit well within the grade-1 histology that the authors demonstrated to correlate with a favorable prognosis both in terms of overall survival and disease-free survival at 5 and 10 years.
Footnotes
Data Availability Statement
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All the clinic-pathologic investigations detailed in the article have been conducted in accordance with the Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed Consent
Written informed consent for publication of data and images was obtained from the patient.
