Abstract
Introduction
Sudden sensorineural hearing loss (SSNHL) is one of the common emergencies in otolaryngology, which is sudden and unknown with at least 2 adjacent frequencies hearing loss ≥20 dB within 72 hours, 1 accompanied by varying degrees of nausea and vomiting, tinnitus, vertigo, and other symptoms. SSNHL results from a variety of etiologies of either primary or secondary origin, such as viral infection, vascular insufficiency, and autoimmune disorder, disruptions of inner ear membranes, cerebellopontine angle tumors, or a combination of processes. 2 The incidence of SSNHL in the United States is reported to be about 5 to 27 cases per 100,000 people per year, 3 while the incidence in China was estimated to be 19 cases per 100,000 persons. 4 Analyzing the related factors influencing the prognosis of SSNHL and the establishment of a predictive model for SSNHL prognosis using statistical models is an extremely important issue for individualized treatment of sudden deafness in clinical practice. The alignment diagram, also known as the nomogram, 5 is based on the multivariate regression analysis. It integrates multiple predictors, and then uses a line segment with a scale to draw on the same plane according to a certain proportion, so as to express the relationship between the variables in the prediction model. It is widely used clinically as a visual model for disease diagnosis and prognosis. And it is also one of the current clinical research hotspots in clinical settings. At present, there is limited research on the use of alignment diagrams to establish predictive models for SSNHL prognosis. Therefore, based on the results of multivariate logistic regression analysis, this study constructed a nomogram to establish a predictive model for the prognosis of SSNHL, with the hope of contributing to the exploration of this field.
Methods
Patient Selection and Data Availability
Clinical information was retrospectively collected from patients who were admitted to the Department of Otolaryngology at the Second Affiliated Hospital of Wenzhou Medical University between January 2020 and December 2022, with a primary diagnosis of SSNHL based on the 2015 SSHL diagnostic and treatment guidelines (China). 1 This project was approved by the Ethics Committee of the Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University. Routine blood analysis was performed on the first day of hospitalization for all patients, with venous blood samples collected. Pure-tone audiometry and tympanometry were performed for all patients on admission and at the completion of treatment. Patient history, audiological reports, and laboratory parameters were evaluated by professional otolaryngologists and audiologists. Inclusion criteria for retrospective case analysis were as follows: (1) meeting the diagnostic criteria outlined in the SSNHL diagnosis and treatment guidelines (2015); (2) absence of middle ear pathology, Ménière’s disease, acoustic neuroma, history of head trauma, history of ototoxic drug use, history of noise exposure, and other relevant factors. Transferring to another hospital during treatment, acoustic neuroma, cerebral thrombosis, duration from onset to treatment more than 30 days, and cases with incomplete data were excluded (Figure 1).
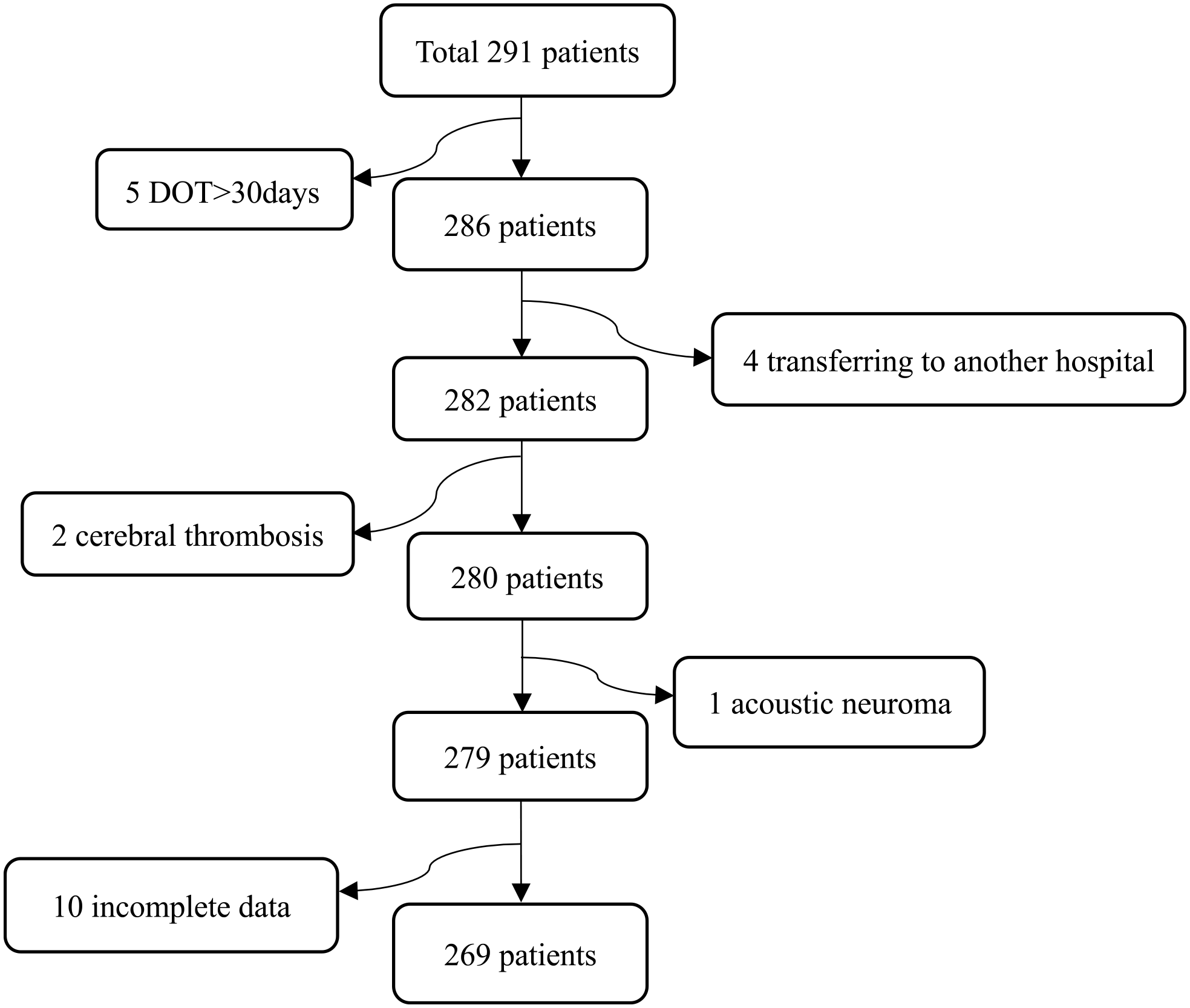
Schematic view of the study workflow. This study included 269 patients affected by SSNHL. DOT, duration from onset to treatment; SSNHL, sudden sensorineural hearing loss.
Variables Assessment
Pure-tone audiometry and tympanometry were conducted prior to admission and at the completion of treatment. Air-conduction and bone-conduction hearing thresholds were evaluated at 250 Hz, 500 Hz, 1 kHz, 2 kHz, 4 kHz, and 8 kHz.
Audiometric configuration was determined according to the Chinese SSNHL guidelines. 1 Types of hearing loss are categorized into 4 audiograms: (1) low-frequency descending type (LFDT): hearing loss below 1000 Hz with a minimum of 20 dB loss at 250 and 500 Hz; (2) high-frequency descending type (HFDT): hearing loss above 2000 Hz with a minimum of 20 dB loss at 4000 and 8000 Hz; (3) flat descending type (FDT): hearing loss at all frequencies with an average threshold of ≤80 dB from 250 to 8000 Hz; (4) total deafness type (TDT): hearing loss at all frequencies with an average threshold of ≥81 dB from 250 to 8000 Hz. The variables were audiometric configuration, age, sex, vertigo, tinnitus, duration from onset to treatment, and the laboratory variables, such as prothrombin time, activated partial thromboplastin time, platelet count (PLT), serum albumin (ALB), lymphocyte count (LYM), neutrophil count (NEU), and fibrinogen. PLT, ALB, LYM, and NEU were individual markers associated with inflammation. The neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio, platelet-to-albumin ratio, and systemic immune inflammation index (calculated as PLT multiplied by the NLR) were composite markers associated with inflammation.
Treatment and Criteria for Treatment Efficacy
All patients were hospitalized and prescribed an identical treatment regimen for average 7 days, and received intravenous administration of medications for the treatment, including glucocorticoids for anti-inflammatory effects, agents for improving microcirculation, and neurotrophic therapy. In addition, based on the type of hearing loss and treatment response, patients were also administered with batroxobin. The specific treatment protocol is as follows: methylprednisolone injection + Ginkgo biloba extract injection + alprostadil injection. Methylprednisolone was administered at a dose of 80 mg + 100 mL normal saline injection once daily for 3 consecutive days, followed by a reduced dose of 40 mg + 100 mL normal saline injection for another 3 days. Dosage reduction was considered based on individual conditions, such as age, diabetes, and hypertension. G. biloba extract injection was administered at a dose of 5 vials + 250 mL normal saline injection once daily. All patients were discharged and underwent hearing reevaluation at the end of the treatment course. After discharge, they continued oral medication and were followed up in the outpatient clinic. According to the diagnostic and treatment guidelines for SSNHL in China (2015), the efficacy of treatment is classified into 4 levels: complete recovery (hearing at the affected frequencies returns to normal or reaches the level of the unaffected ear); significant improvement (average hearing improvement of more than 30 dB at the affected frequencies); effective (average hearing improvement of 15-30 dB at the affected frequencies); ineffective (average hearing improvement of less than 15 dB at the affected frequencies). In cases of bilateral hearing loss, the improvement is calculated based on the average of both ears. In addition, based on the evaluation of treatment efficacy, patients are further divided into 4 groups: ineffective, effective, significant improvement, and complete recovery. Meanwhile, the improvement group (including complete recovery, significant improvement, and effective cases) and the ineffective group were also assessed in this study.
Statistical Analysis
Categorical data were analyzed using chi-square test or Fisher’s exact test. Continuous data did not follow a normal distribution, therefore the “median (interquartile range)” was used to represent measures of central tendency and dispersion. The Kruskal-Wallis test and Mann-Whitney U test were performed for group comparisons. Variables with a P < .05 in the univariate analysis were included in a multivariate logistic regression to examine the effects of various indicators on the prognosis of SSNHL. Based on the aforementioned analysis, 2 nomogram models for predicting the prognosis of SSNHL patients were constructed. The accuracy of the model for 2 groups was validated using receiver operating characteristic (ROC) curve analysis with the “pROC” package. The internal validation for prediction reliability was evaluated with the bootstrap approach, and the calibration curve was prepared to present the agreement result calibration curve; and decision curve analysis (DCA) measure the clinical utility of the nomogram. The C-index of the nomogram for 4 groups reflected the performance of the model. All statistics were calculated using R software (version 4.2.3) and IBM SPSS Statistics software version 19.0 (IBM, Armonk, NY, USA), and a P value < .05 indicated statistical significance. The R packages “glm,” “rms,” “haven,” “pROC,” “ggDCA,” and “ggplot2” were employed for this purpose.
Result
Study Participants
Of the 269 patients with SSHL enrolled in this study, 44 cases achieved complete recovery (16.4%), 28 cases showed significant improvement (10.4%), 64 cases had effective response (23.8%), and 133 cases showed no improvement (49.4%). The overall improvement rate was calculated as 50.6%. There were 265 patients (98.5%) with unilateral deafness and 4 patients (1.5%) with bilateral deafness. There were 131 male and 138 female cases. The age range was 5 to 78 years, with a mean age of 43.6 ± 16.6 years. The duration from onset to treatment ranged from 1 to 30 days. For 156 patients (58.0%), the time from the onset of illness to first treatment ranged from 0 to 7 days, for 89 patients (33.0%), from 7 to 15 days, and for 24 patients (9.0%), >15 days. About the audiometric configuration, there were 42 cases (15.6%) of LFDT, 26 (9.7%) of HFDT, 108 (40.2%) of FDT, and 93 cases (34.6%) of TDT. In terms of accompanying symptoms, 99 cases presented with vertigo and 170 cases did not have vertigo. A total of 251 cases presented with tinnitus and 18 cases did not have tinnitus. The results of the comparisons of the general data of different groups were set out in Table 1. Univariate logistic regressions showed that age, duration from onset to treatment, audiometric configuration, ALB, and NLR were potential prognostic factors of SSNHL (P < .05). The multivariate ordinal logistic regression and binary logistic regression analyses revealed that duration from onset to treatment and audiometric configuration were independent factors that significantly influenced the treatment efficacy in patients with SSNHL (Table 2 and Figure 2).
Demographic Data and Clinical Characteristics of Study Participants.
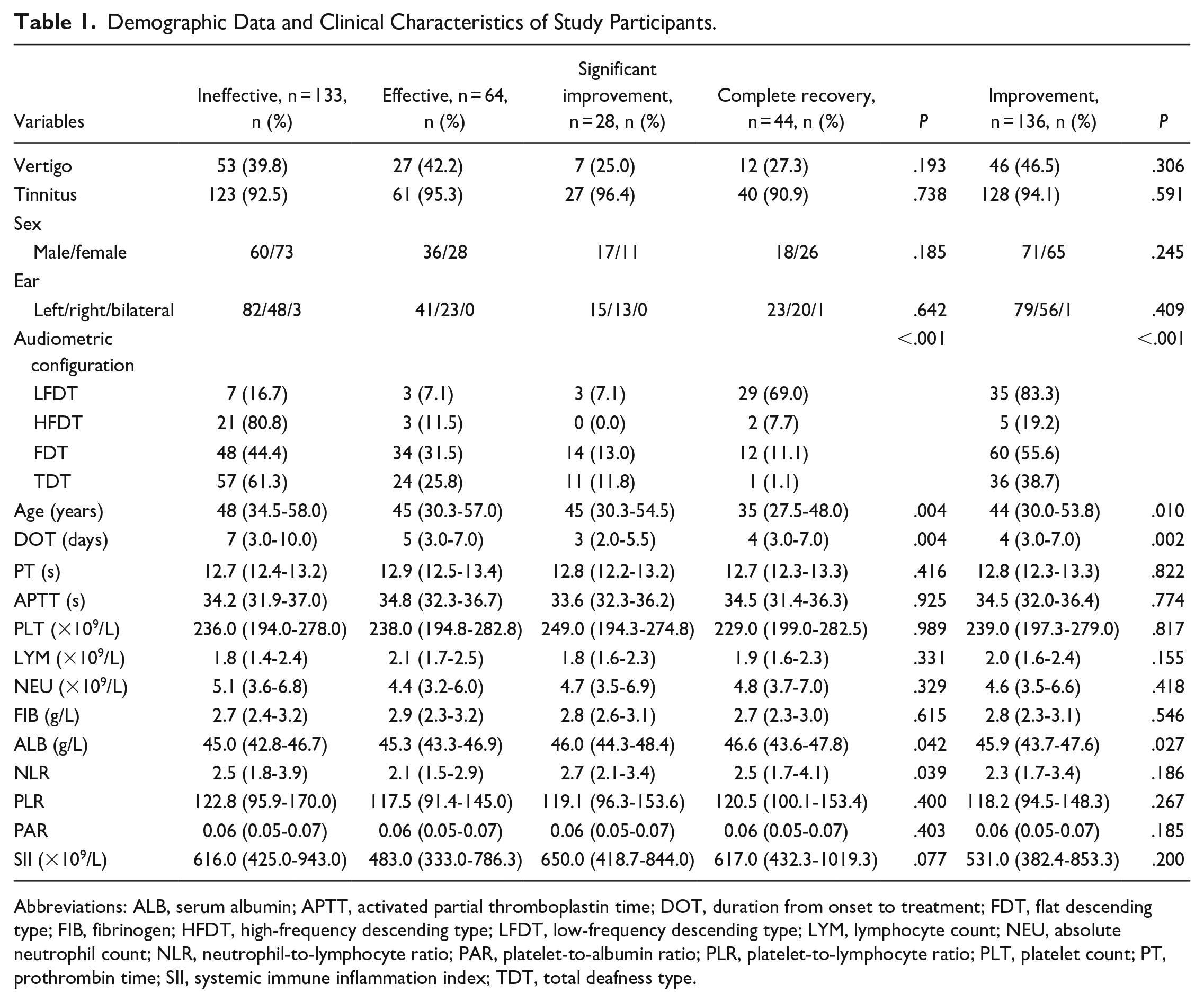
Abbreviations: ALB, serum albumin; APTT, activated partial thromboplastin time; DOT, duration from onset to treatment; FDT, flat descending type; FIB, fibrinogen; HFDT, high-frequency descending type; LFDT, low-frequency descending type; LYM, lymphocyte count; NEU, absolute neutrophil count; NLR, neutrophil-to-lymphocyte ratio; PAR, platelet-to-albumin ratio; PLR, platelet-to-lymphocyte ratio; PLT, platelet count; PT, prothrombin time; SII, systemic immune inflammation index; TDT, total deafness type.
The Multivariate Ordinal Logistic Regression.
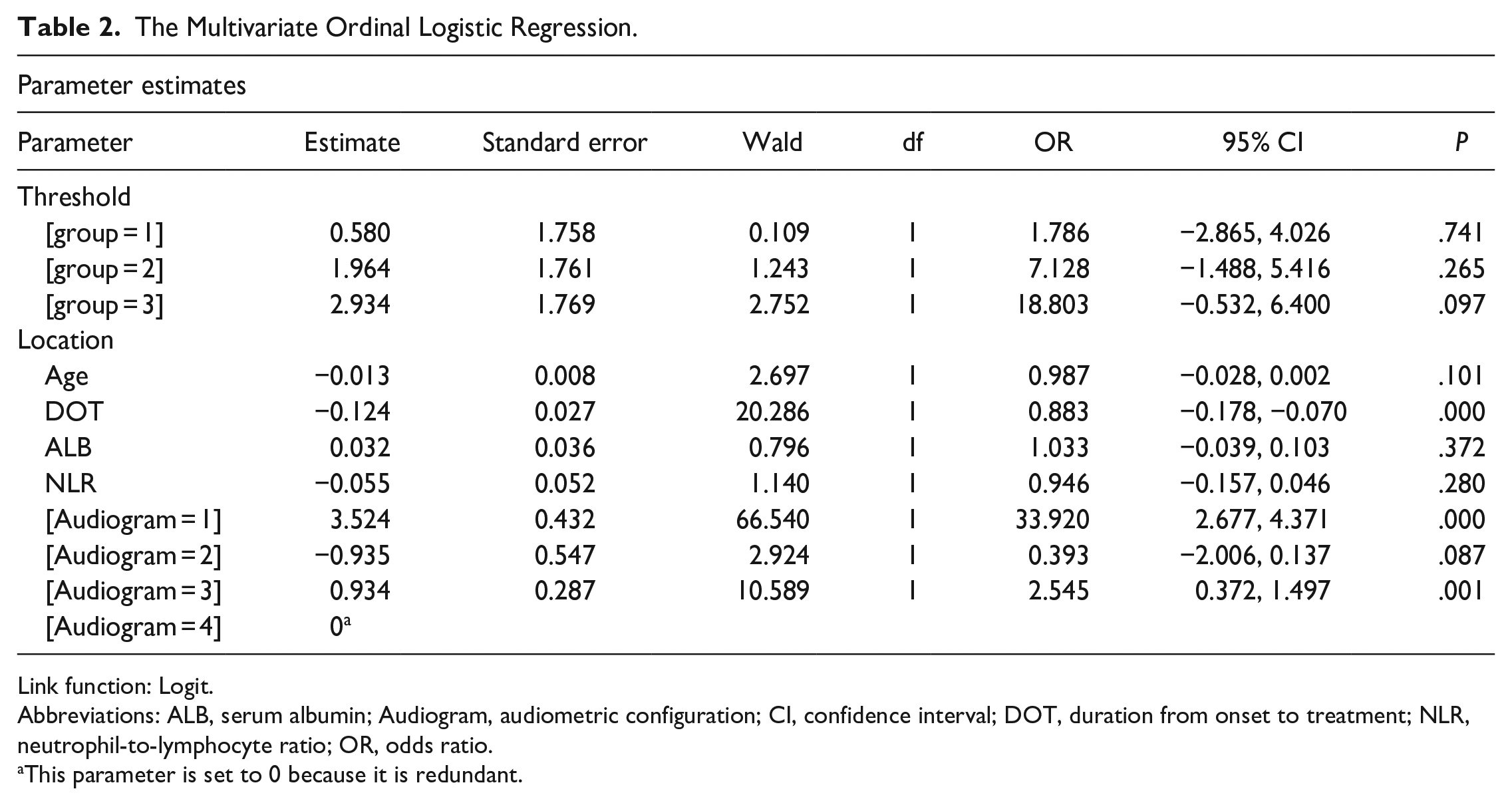
Link function: Logit.
Abbreviations: ALB, serum albumin; Audiogram, audiometric configuration; CI, confidence interval; DOT, duration from onset to treatment; NLR, neutrophil-to-lymphocyte ratio; OR, odds ratio.
This parameter is set to 0 because it is redundant.
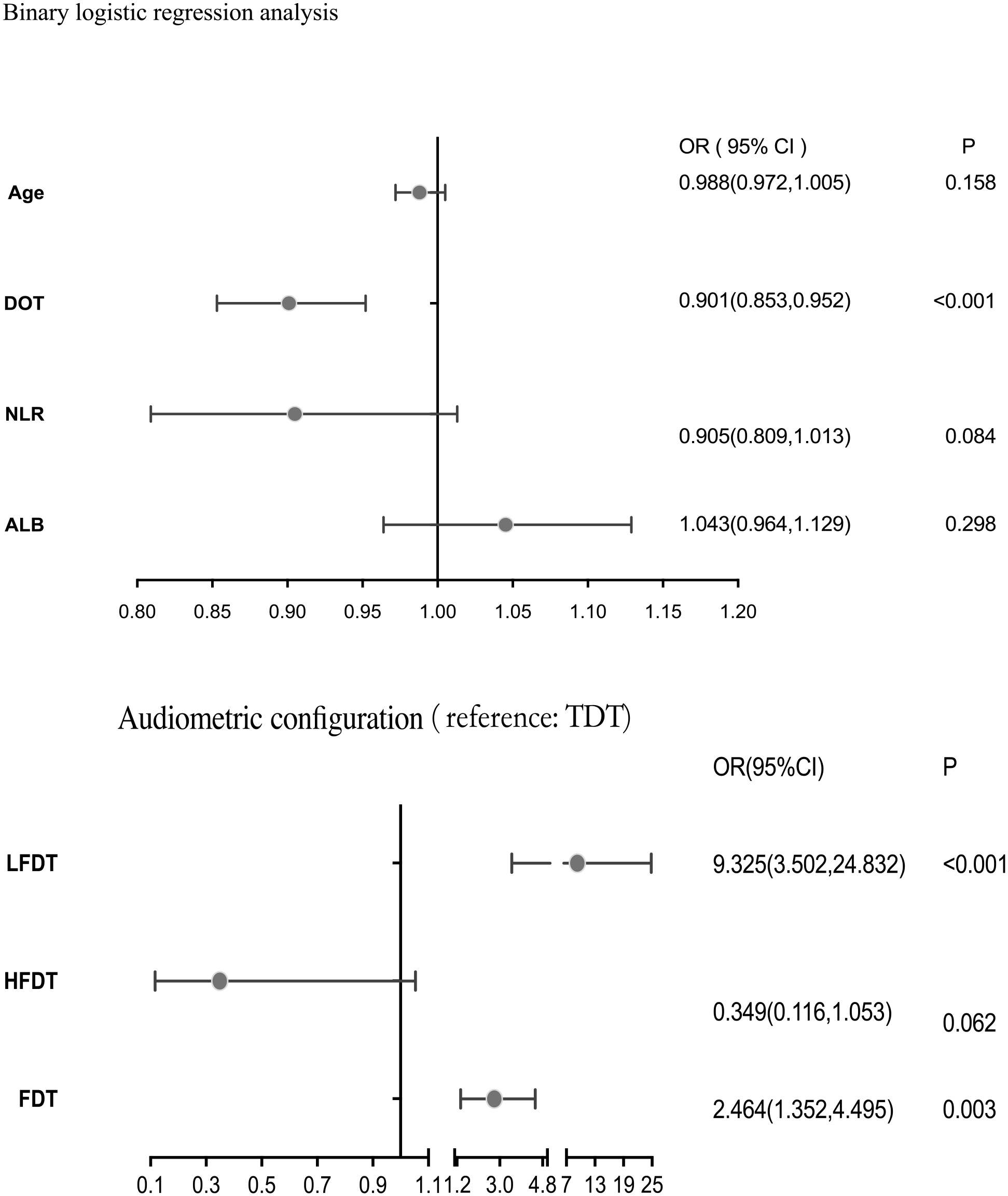
Binary logistic regression analysis.
Construction of Nomogram
Age, duration from onset to treatment, audiometric configuration, ALB, and NLR were subsequently used to construct nomograms for 2 or 4 treatment efficacy outcomes that could predict the hearing prognosis of patients (Figure 3). Although univariate and multivariate logistic regression revealed that age, ALB, and NLR were not independent risk factors but merely influencing factors in this study. They were included in the nomograms to differentiate them from the prediction ability differences of duration from onset to treatment, audiometric configuration more intuitively. To estimate the recovery rate of patients with SSNHL, the observed value of each predictor was quantified according to a predefined scoring system, wherein a vertical line was drawn to determine the corresponding points on a predefined scale. The hearing prognostic assessment for each individual patient was obtained by summing the points associated with each prognostic factor.
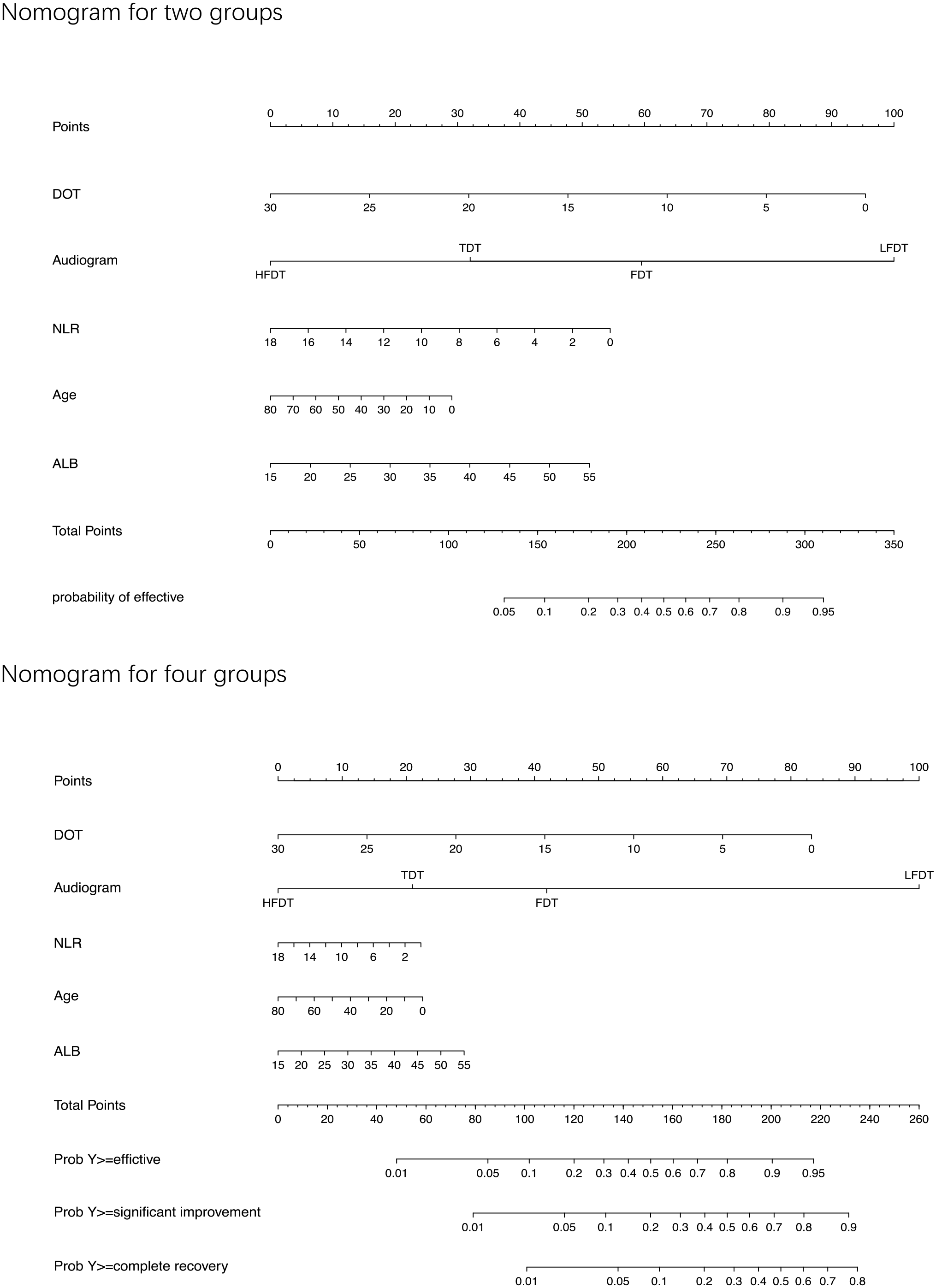
Prognostic nomogram of patients with SSNHL. DOT, duration from onset to treatment; Audiogram, audiometric configuration; SSNHL, sudden sensorineural hearing loss.
Evaluation and Verification of Nomogram
Various methods were used to validate the predictive ability of the model for ineffective and improvement groups through internal validation, as shown in Figure 4. The discriminatory power of the model, as indicated by the area under the ROC curve, was 0.752 (95% CI: 0.695-0.808). The calibration curve demonstrated consistency in a practical occurrence probability. Furthermore, DCA revealed favorable clinical effectiveness of the predictive model. The C-index was 0.756 (95% CI: 0.710-0.802) when the treatment outcome was divided into 4 groups showing the moderate discriminatory power.
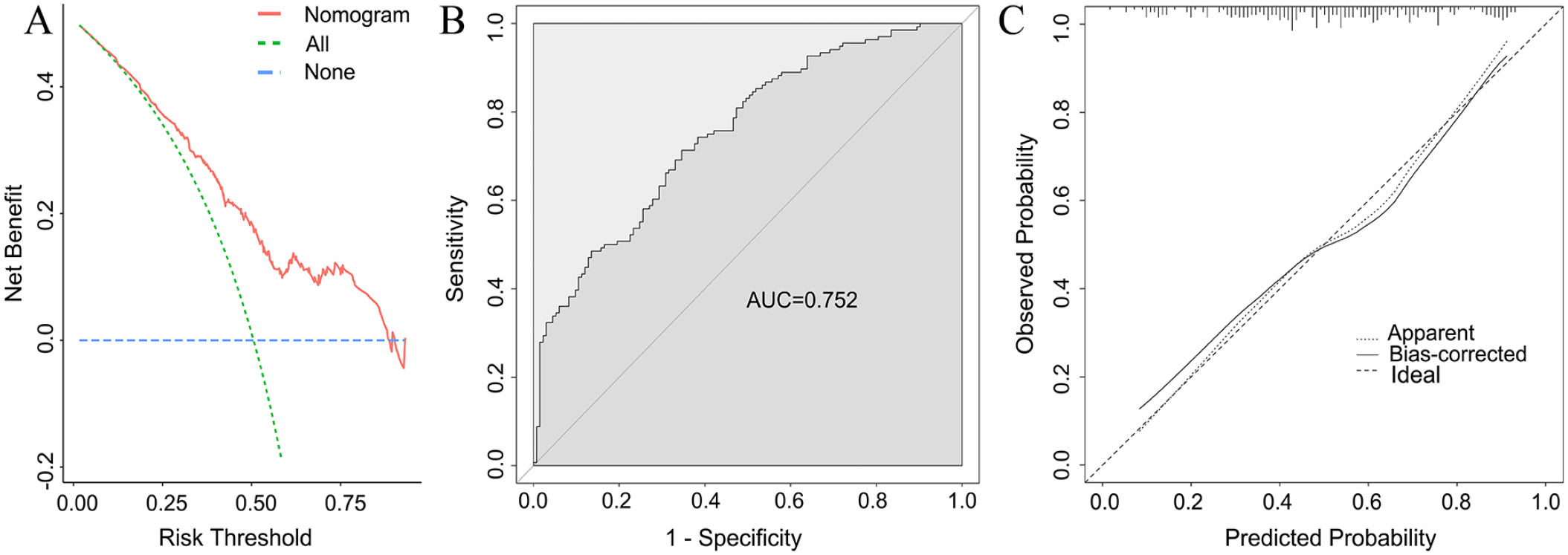
Evaluation of the nomogram model for 2 groups. (A) Decision curve analysis for the prediction model. Red line: prediction model. Green line: assume all patients have hearing recovery. Blue line: assume no patients have hearing recovery. (B) Receiver operating characteristic curve. AUC = 0.752 (95% CI: 0.695-0.808). (C) Nomogram calibration plot using bootstrap resampling (1000 times), the solid line represents the performance of nomogram. AUC, area under the receiver operating characteristic curve; CI, confidence interval.
Discussion
This study is based on the results of multivariate regression analysis of different treatment efficacy groups. Age, duration from onset to treatment, audiometric configuration, NLR, and ALB were included to construct nomograms for predicting SSNHL. In recent years, nomogram was used in different medical fields for predicting prognostic, overall survival rate, recurrence rate, and so on. Wu et al 6 established the nomogram to assist clinicians in evaluating the overall survival in low-grade endometrial stromal sarcoma (LG-ESS) patients. Eisner and Goldfarb 7 constructed a nomogram for the prediction of kidney stone recurrence. The nomogram for predicting SSNHL prognostic was few. Zhang et al 8 constructed a predictive model using a nomogram and found that patients younger than 20 years, those with a low-frequency descending hearing loss curve, mild hearing loss at admission, and duration from onset to treatment within 3 days, had the highest total score on the nomogram and the highest probability of improvement. The audiometric configuration and duration from onset to treatment are recognized as independent factors affecting the improvement of SSNHL and are well-documented prognostic indicators.1,3 Psillas et al 9 and Choo et al 10 demonstrated that patients who had SSNHL that exclusively affected low frequencies had a significantly better prognosis than patients who had losses at high frequencies.Our study revealed that the worst treatment outcomes were observed in cases of TDT and HFDT of SSNHL, followed by FDT, while the best outcomes were seen in cases of LFDT. The possible rationale for this observation could be that the basal turn of the cochlea predominantly affects high frequency, while the apical turn primarily influences low frequency. Prior studies have indicated that the base of the cochlea may possess a higher susceptibility to injury compared to the apical region, potentially attributed to differences in vascular perfusion and metabolic requirements. 11
Numerous studies have shown that the longer the time between the onset of SSNHL and treatment, the worse the prognosis.12-14 Patients who started treatment within the first 7 days gained the best prognosis, and a therapeutic effect between 8 and 14 days, and a little better than those who sought treatment after 14 days. The possible reason behind these observations is that once there is microcirculation blockade in the inner ear, ischemia and hypoxia occur in the inner ear tissues, leading to damage to hair cells.11,15 If left untreated for an extended period, the recovery of hair cell function becomes difficult, and hearing improvement becomes challenging.
Inflammation is one of the many causative factors of SSNHL, which increases the risk of the cochlear vasculature damage and ischemia. Many authors have studied the role of laboratory parameters especially inflammation parameters as risk factors and potential prognostic biomarkers for SSNHL, often with contradictory conclusions. NLR was the most researched factor. A meta-analysis concluded that NLR was a novel potential marker for SSNHL 16 associated with a chronic subclinical inflammatory status and they were considered as relatively stable composite markers of inflammation 17 compared to the other single markers associated with inflammation. 18 However, Lee et al 19 utilized artificial intelligence to develop prognosis models for predicting the prognosis of SSNHL, and revealed that the correlation between NLR and the prognosis of SSNHL was not substantial. This study also found that NLR was only an influencing factor in the 4 groups for SSNHL prognosis but not an independent risk factor for SSNHL prognosis.
The relationship between age and prognosis in SSNHL has been reported differently in various studies. Although the metabolic and regenerative capacity of the human body declines with age, research has shown that age is an unstable prognostic factor for SSNHL. Zadeh et al 20 demonstrated that patients younger than 40 years have a statistically significant increased rate of recovery. 20 While Cinamon et al 21 argued that age was not a significant factor, which was consistent with this study. In univariate analysis of this study, age showed differences among different treatment efficacy groups but not a significant indicator for SSNHL prognostic.
In addition, this study demonstrates that ALB is an influencing factor but not an independent predictor for SSNHL. The involvement of ALB in inflammatory responses may influence the development of the disease by affecting hemodynamic changes. Both acute and chronic inflammation, as well as malnutrition, can lead to decreased albumin levels.22,23 Zheng et al 24 suggested that low ALB concentration may be an independent risk factor for the prognosis of SSNHL, and the ALB level is positively associated with hearing outcomes according to the pure tone average (PTA) of impaired frequencies. Currently, there is limited information available about ALB in the prognostic of SSNHL. The value requires further confirmation through large-scale studies and basic experimental research.
In this study, there was no statistically significant difference among patients with vertigo and tinnitus in different treatment efficacy groups. This finding differs from previous studies25,26 but aligns with the findings of Neto et al 13 and Zadeh et al. 20 Further evidence is needed to confirm these findings by increasing the sample size and extending the follow-up period. In addition, Wu et al 27 found that blood lipids were a high-risk factor for SSNHL, with low-density lipoprotein (LDL) being an independent risk factor. Zhou et al 28 found that abnormal sensation around the ear and ear fullness sensation were protective factors for SSNHL. Although our study did not specifically investigate these factors, their findings may have some guiding implications for clinical practice. The clinical data in this study were obtained from a single-center medical institution, lacking external validation data. The nomogram for 4 groups is just evaluated by C-index. Therefore, increasing the sample size, extending the follow-up period, and conducting a multicenter study on SSNHL, as well as strengthening the external validation of the data, would be beneficial.
Conclusion
Our study demonstrated that duration from onset to treatment and audiometric configuration are authentically independent risk factors for SSNHL and suggested a lack of association between hematologic parameters and SSNHL prognosis, which warrants further investigation. The prognosis of SSNHL was divided into 4 outcomes: ineffective, effective, significant improvement, and complete recovery as well as a comparison between ineffective and improvement for both univariate and multivariate analysis. This approach provided a more comprehensive understanding of the prognostic factors in different outcome groups, which differs from previous studies.
Footnotes
Data Availability Statement
All data supporting the findings of this study are available within the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was performed according to the ethical standards of the human subjects and was approved by the Ethics Committee of Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University.
