Abstract
Primary melanoma of the parotid gland is an extremely rare and challenging tumor with a poor prognosis, and its ultrasonic characteristics have yet to be reported. This article presents a case of a 77-year-old man with a left parotid mass that was confirmed as a melanoma following surgery. The ultrasonic features of melanoma were examined in detail, with a particular focus on their diagnostic value. Furthermore, we summarized the clinical characteristics, treatment options, and outcomes associated with primary melanoma of the parotid gland based on a thorough analysis of the available literature.
Introduction
Malignant melanoma (MM), a neoplasm derived from melanocytes, represents approximately 20% to 35% of all head and neck tumors. 1 MM may occur in the skin of any part of the body and frequently metastasizes widely. The most commonly affected site in the head and neck region is the face, followed by the scalp. 2 Most cases of MM in the parotid gland are metastatic 3 ; primary MM in the parotid gland is extremely rare. Present knowledge regarding primary MM in the parotid gland is based on sporadic case reports.
We herein report a case of a 77-year-old man with primary MM in the parotid gland. Similar English-language reports published since 1990 were reviewed and summarized.3-13
Case Report
A 77-year-old man visited the oral and maxillofacial surgery department of our hospital because of a 1 month history of a painless and progressively growing mass in the left parotid gland region. He also felt numbness in the left earlobe. Physical examination revealed bilateral asymmetry in the oral and maxillofacial region and a hard mass of approximately 4.0 cm × 3.5 cm with poor mobility in the left parotid gland region. No lesion was found in the oral cavity, and no facial paralysis or significant enlarged lymph nodes were present.
The patient agreed to undergo ultrasound (US) examination. A heterogeneous hypoechoic nodule with a lobulated and well-defined border was detected in the left parotid region. The mass was located adjacent to the skin superficially and facial nerve area deeply, and it measured approximately 3.6 cm × 3.7 cm × 3.5 cm. US also showed multiple extremely hypoechoic regions and dot-like hyperechoic foci within the tumor. In addition, color Doppler flow imaging showed punctate blood flow signals on the edge of the mass (Figure 1). A malignant tumor of the parotid gland was suspected, and surgery was advised.
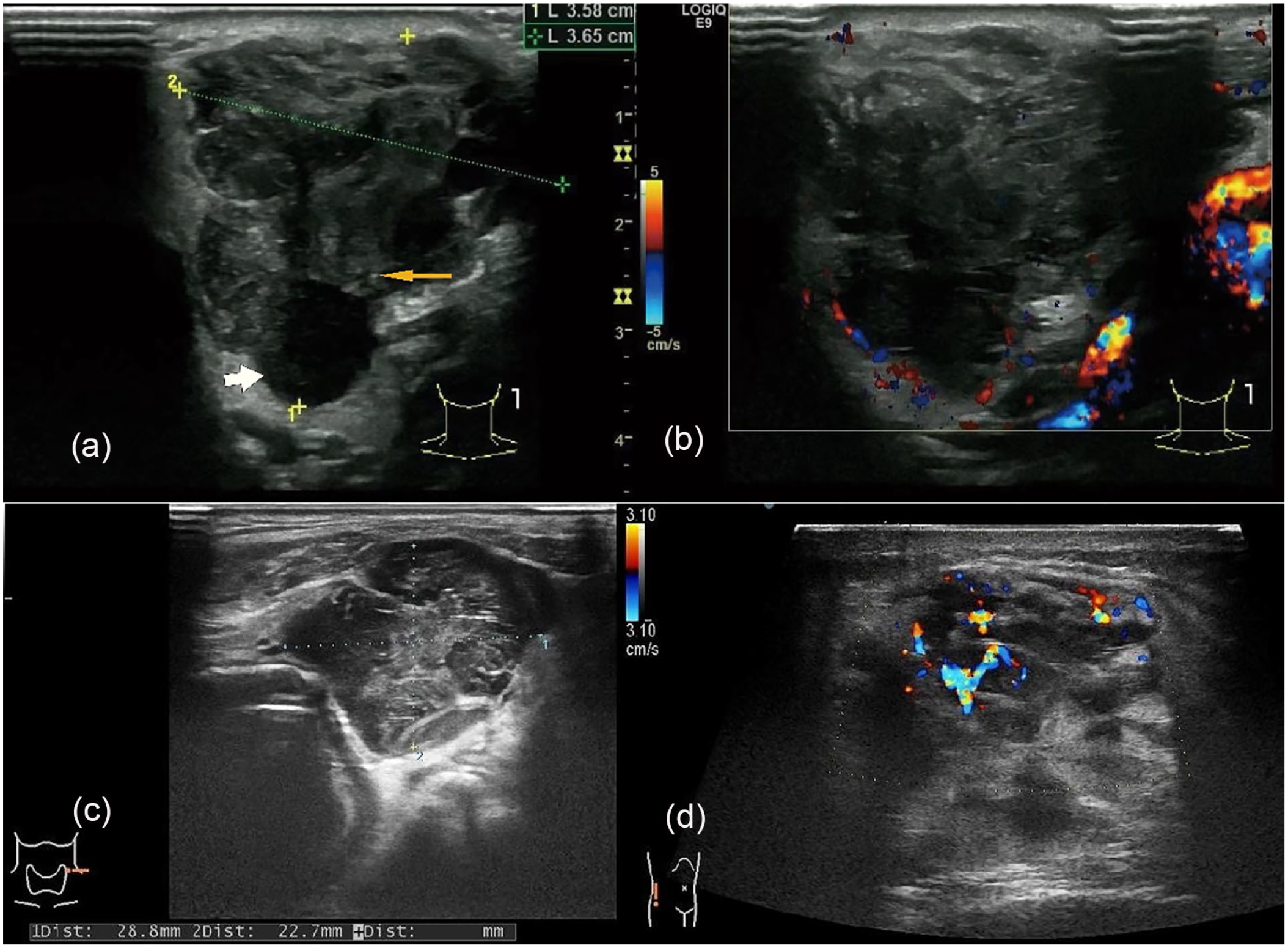
(a) Ultrasound showed a heterogeneous hypoechoic nodule with a lobulated and well-defined border. Multiple extremely hypoechoic region (white arrow) and dot-like hyperechoic foci (yellow arrow) within the parotid tumor was emphasized. (b) Color doppler flow image showed punctate blood flow signal inside the parotid tumor. (c) A heterogeneous hypoechoic nodule with a lobulated and well-defined border was detected in the left neck. (d) There was abundant blood flow signal in an ill-defined hypoechoic mass at the right lumbar [equipment: GE LOGIQ E8 with linear probe (a and b), Hitachi Aloka Arietta 70 with linear probe (c and d); deep:4.5 cm].
Computed tomography (CT) scan showed a heterogeneous solid and low-density nodule in the left parotid region with ill-defined boundary. No bone invasion was detected (Figure 2). The patient also underwent other radiological examinations, such as chest CT and echocardiography; however, no other lesion was found.
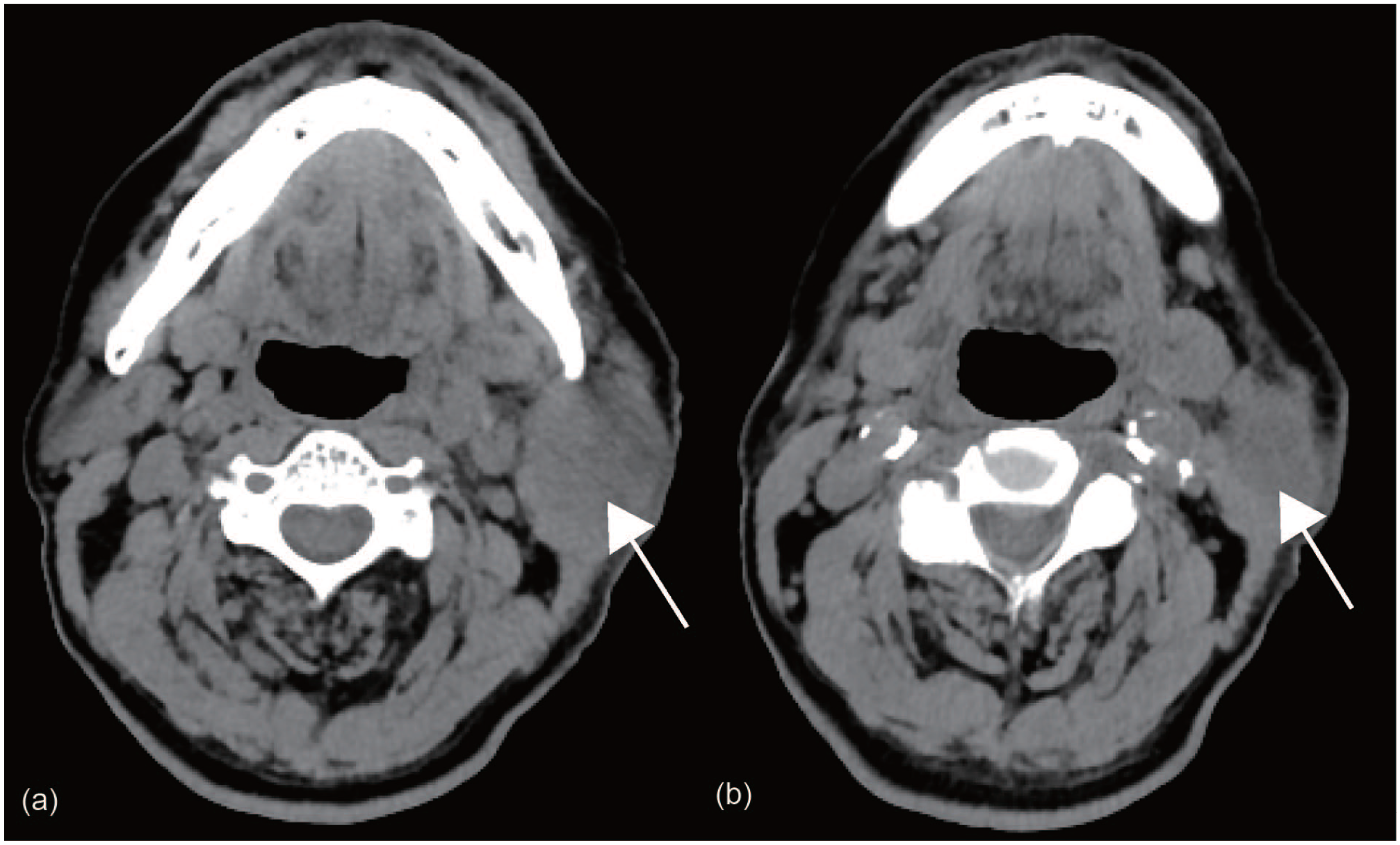
(a) Computed tomography (CT) scan displayed swollen parotid gland (arrow). (b) CT scan showed a heterogeneous solid and low-density nodule (arrow) in left parotid region with ill-defined boundary (equipment: GE Optima CT620, slice thickness: 2.5 mm, slice interval: 2.5 mm).
After thorough evaluations, the patient agreed to undergo total parotidectomy with interposition of a tissue patch and sternocleidomastoid muscle flap transfer. During the operation, the tumor was found to be invading the deep and superficial lobes of the parotid gland, and part of it was adhered to the sternocleidomastoid and skin. The total parotid gland and tumor with affected muscles and skin (4 cm × 1 cm) were completely separated and removed along the inferior surface of the external acoustic meatus and mastoid process. Although the mass was immediately adjacent to the facial nerve, we were able to achieve clear surgical margins without impinging on this nerve.
Malignant tumor was detected by intraoperative frozen pathology. Histopathological examination and immunohistochemical analysis showed positive staining for S-100, SOX10, and Ki-67 (40%), and negative staining for HMB45, CKpan, SMA, LCA, and p63. Thus MM was confirmed. The mass extensively invaded the surrounding salivary glands, striated muscles, and subcutaneous tissue (Figure 3).
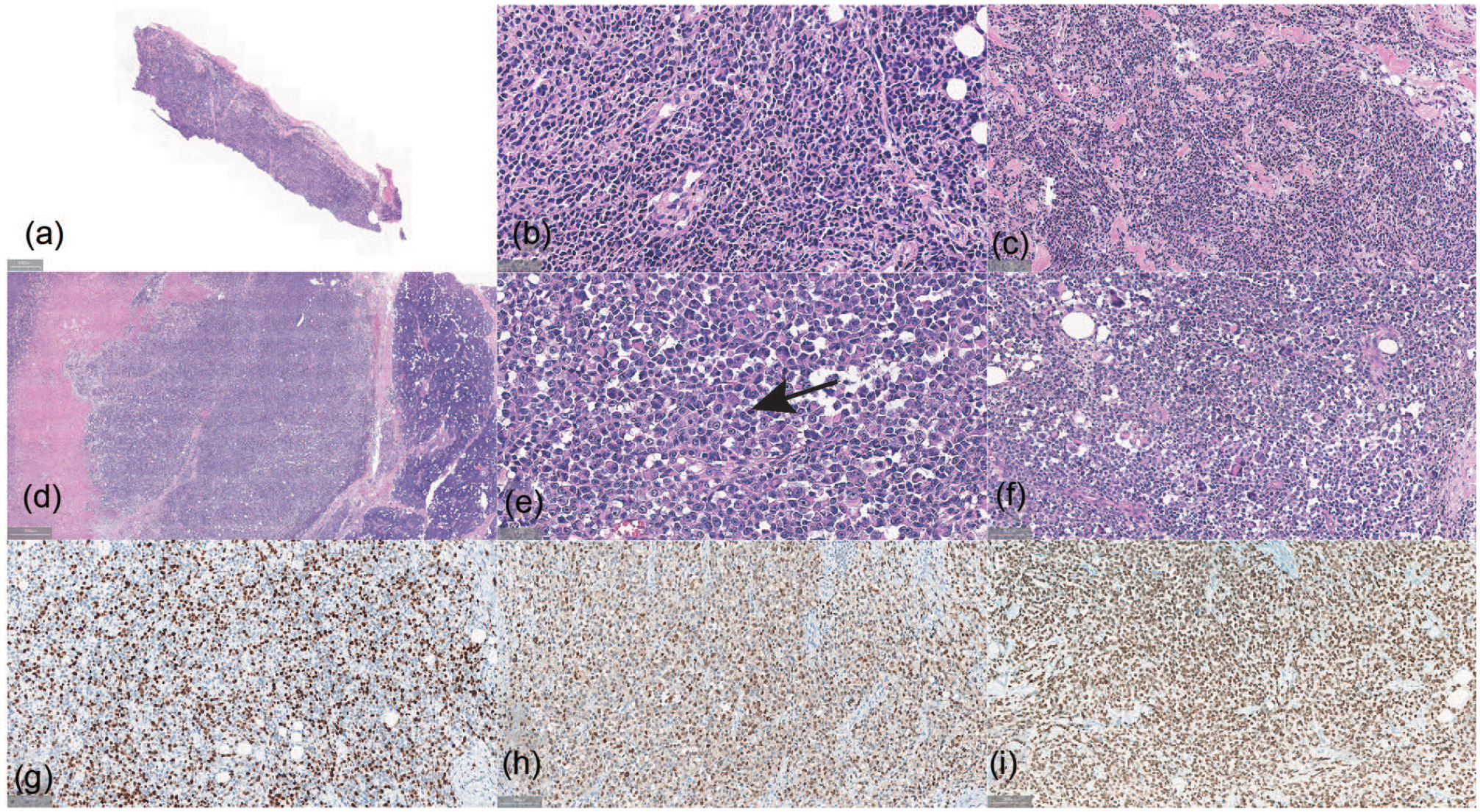
(a-c) The frozen pathological picture is examined at various magnifications (10× → 100× → 200×). Multinucleated giant cells were showed. (d-f) Photomicrographs depicting a tumor section at varying magnifications (10× → 200× → 100×). (e) Representative area of melanoma showing mitotic figure (arrow). (f) Representative area of melanoma showing multinucleated giant cell. (g-i) Histopathological image shows positive staining for (g) ki-67, (h) S-100, and (i) SOX-10.
After 5 months, the patient revisited the oral and maxillofacial surgery department for evaluation of 2 new masses: one in the right waist region and the other on the left neck. US examination showed that the left cervical nodule was similar to the original parotid gland mass (a heterogeneous hypoechoic nodule with a lobulated and well-defined border), while the right lumbar nodule was ill-defined, heterogeneous, and hypoechoic. Unlike the parotid gland neoplasm, both nodules showed stripy blood flow signals with high impedance (Figure 1). Core needle biopsy of the right lumbar mass was performed, and MM was again pathologically confirmed. The patient also underwent whole-body positron emission tomography-CT (PET-CT), which showed multiple active metastases throughout the body, including the bilateral neck and axilla, left pericardium, right waist, and root of mesentery (Figure 4). The patient began treatment by immunotherapy on the advice of an oncologist. No adverse or unexpected events occurred during treatment. However, the patient died of multiple metastases shortly thereafter due to the natural proceeding of the disease.
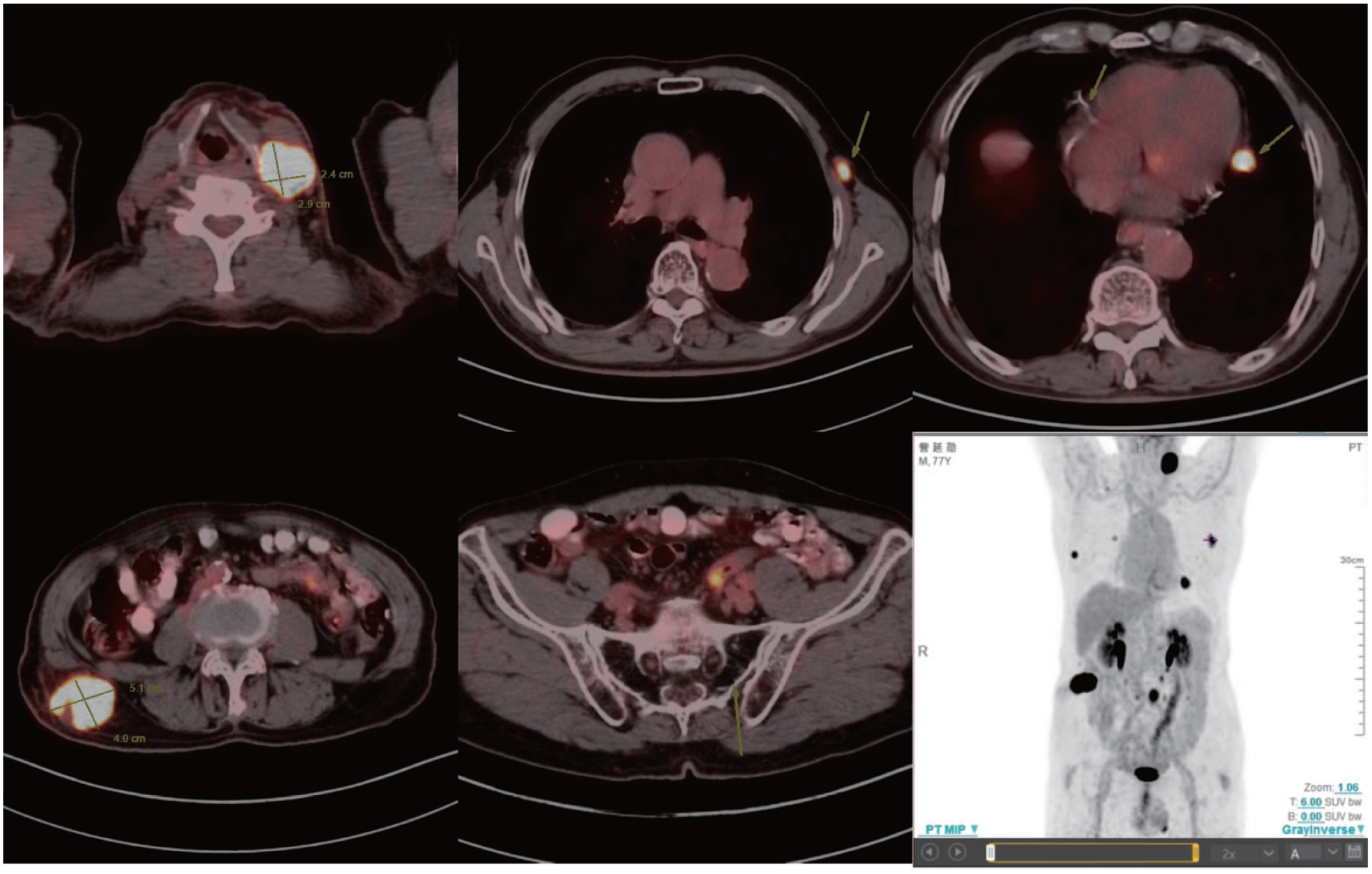
PET-CT scan showed multiple active metastases including bilateral neck and axilla, left pericardium, right waist, and the root of mesentery (radiopharmaceutical agent: fluorodeoxyglucose F^18^, dose: 0.12 mCi/kg, thickness: 3.75 mm, CT matrix: 512 × 512, PET matrix: 192 × 192, scanning speed: 29.46 mm/s, reconstructive method: VUE Point FX). PET, positron emission tomography; CT, computed tomography.
Discussion
Parotid gland tumors are commonly seen in clinical practice, and 80% are benign. 14 Primary MM in the parotid gland is an exceptional rarity, constituting only 0.68% of malignant neoplasms of the parotid gland.4,15 This may be because melanocytes are derived from neural crest cells rather than being normal structural part of the salivary gland. 13 Nevertheless, Takeda 16 reported the presence of melanocytes in the interlobular duct of the parotid gland during an autopsy of male cadavers. Moreover, Greene and Bernier 17 demonstrated that these melanocytes may be related to melanoblasts that become incorporated in the parotid gland during embryonic development. They supported this claim by showing that melanin is also present in some cells of healthy parotid glands.
Diagnosis of primary MM of the parotid gland is usually exclusionary and controversial. Woodwards et al 5 suggested the following 4 criteria. First, most of the tumor is contained in the parotid gland. Second, the tumor contains no identifiable lymph node tissue. Third, there is no evidence of other MM lesions in the body. Fourth, there are no suspicious pigmented lesions or a history of excision of an MM. In our case, other than the mass in the parotid gland, no suspicious skin lesions were found during physical examination, and repeat imaging studies showed no corresponding lesions in the metastatic sites. The diagnosis of primary melanoma of the parotid gland was confirmed. However, it is regrettable that PET-CT was not performed before the surgery.
To understand the clinical characteristics of primary MM of the parotid gland, we searched the PubMed, EMBASE, Web of Science, and Cochrane Library databases. The search terms used were as follows: [(malignant melanoma OR melanoma) AND (parotid OR parotid region OR parotid gland) AND (primary)]. Case reports describing patients with a definite diagnosis of primary parotid melanoma were enrolled. Articles for which the full text was not available and articles not written in English were excluded. The references of the selected articles were also reviewed to identify additional relevant cases.
In total, 11 cases have been reported since 1990. The clinical features of the patients are summarized in Table 1. The patients’ age at presentation was variable, ranging from 27 to 80 years. However, 73% of the patients were older than 50 years. The prevalence of men and women was nearly equal (5:6). All patients reported unilateral cheek swelling. Eight patients reported gradual growth of the mass. Only 2 patients felt pain. In 2 cases, the pathology examination revealed unique amelanotic MM, which has a wide-ranging clinical appearance with no pigmentation.
Cases in the Literature of Primary Malignant Melanoma of the Parotid Gland.
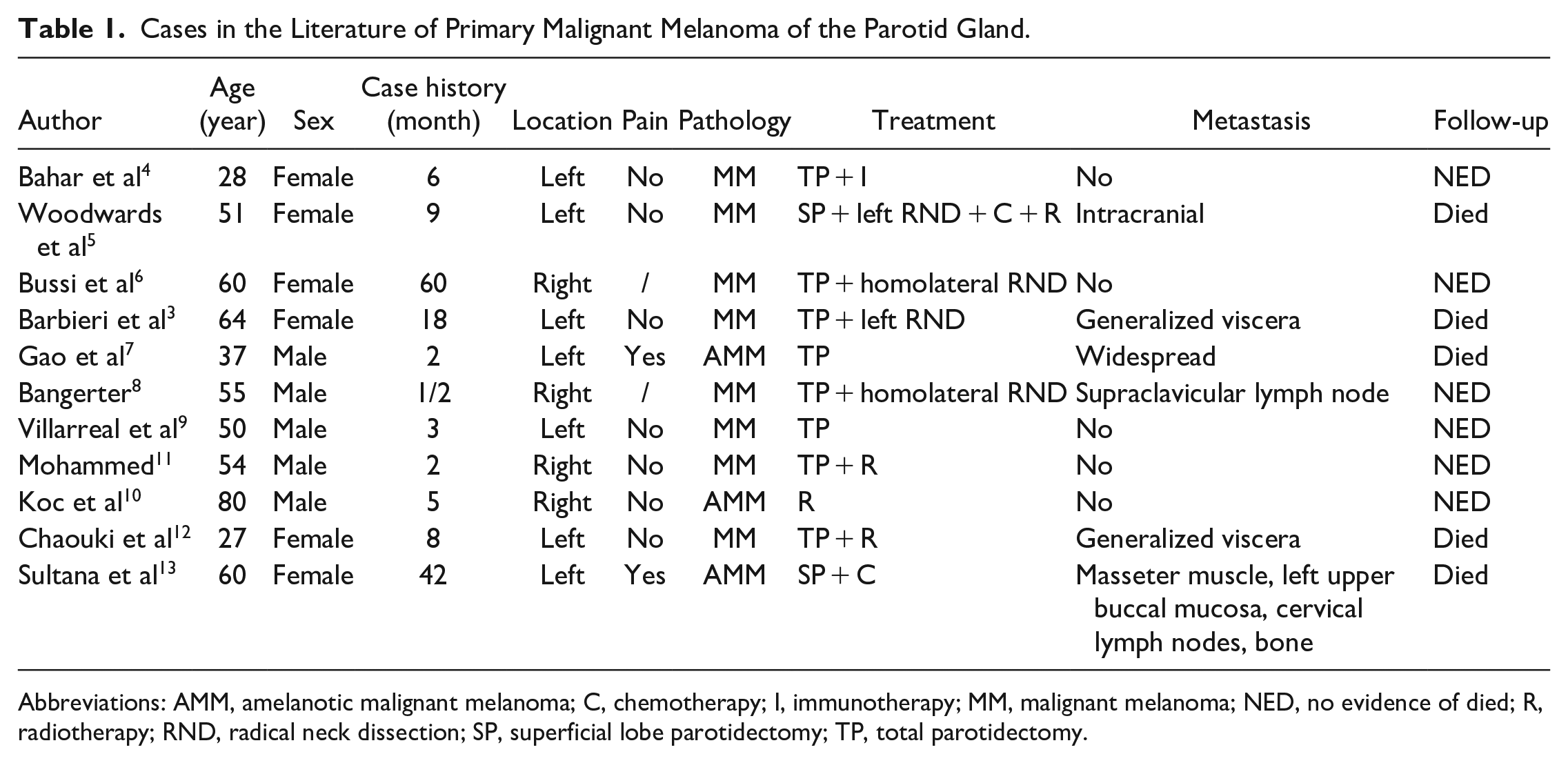
Abbreviations: AMM, amelanotic malignant melanoma; C, chemotherapy; I, immunotherapy; MM, malignant melanoma; NED, no evidence of died; R, radiotherapy; RND, radical neck dissection; SP, superficial lobe parotidectomy; TP, total parotidectomy.
US is widely accepted as an ideal tool to evaluate soft tissue masses. To our knowledge, however, no reports to date has described the ultrasonic features of primary MM in the parotid gland. In our case, the mass manifested as a hypoechoic and heterogeneous nodule with a lobulated and well-defined border. These characteristics are consistent with MM in the skin or subcutaneous soft tissue. 18 Although multiple extremely scattered hypoechoic regions and dot-like hyperechoic foci were observed within the mass, no evidence of calcification or cystic change was detected in the pathological examination. By contrast, calcification and cystic changes are commonly seen in pleomorphic adenoma19,20 and Warthin’s tumor. 20 Unlike Warthin’s tumor, which typically exhibits hyperechoic internal septation and hypervascularization, only punctate blood flow signals were seen on the edge of this tumor. Although it was nearly impossible to guess the pathologic tumor type prior to surgery, we readily suspected that it was malignant. Ultrasonic examination can clearly display morphological features, activity, blood flow, and especially the relationship between the mass with nerves and bone. Therefore, we believe that ultrasonic examination was necessary before the surgery. We look forward to similar reports to confirm this notion.
The preoperative differential diagnosis of parotid tumors poses a challenge for clinicians because of the extensive histological diversity of these tumors and their overlapping histological and imaging features. Multimodal imaging, including US and magnetic resonance imaging (MRI), etc, improves the accuracy of differential diagnosis. US elastography enables quantitative or qualitative assessment of soft tissue elasticity. Conventional US examination allows for quantitative analysis of tumoral tissue microvascular perfusion and evaluation of the morphologic and distribution features of microvascularity. 21 Functional MRI methods (eg, diffusion-weighted imaging, dynamic contrast-enhanced MRI, and perfusion-weighted MRI) provide information on tissue cellularity, microstructure, vascularity, metabolite concentrations, and the behavioral patterns of tumors.22,23 PET-CT serves as a primary tool to confirm or exclude recurrences and assess the prognosis in patients with melanoma. 24 Undoubtedly, multimodal imaging provides superior visualization and management capabilities for parotid tumors.
Admittedly, the prognosis for parotid melanoma is poor. However, there is no clear consensus regarding therapy. In our review of the literature, 10 patients were treated surgically with procedures varying from superficial to total parotidectomy, and 1 patient only accepted radiotherapy. The analysis of postoperative recovery revealed that 54% of patients had metastasis and 45% died. The survival curve drawn according to the patients’ follow-up results also showed that the prognosis of this disease is poor and that the long-term survival rate is low (Figure 5). No reliable conclusions regarding the effectiveness of various treatment modalities can be determined because the follow-up date indicated no consistent relationship between the type of therapy and the survival rate. However, surgery remains the primary treatment; the benefits of radiotherapy, chemotherapy, and immunotherapy are limited and unclear.2,25-27
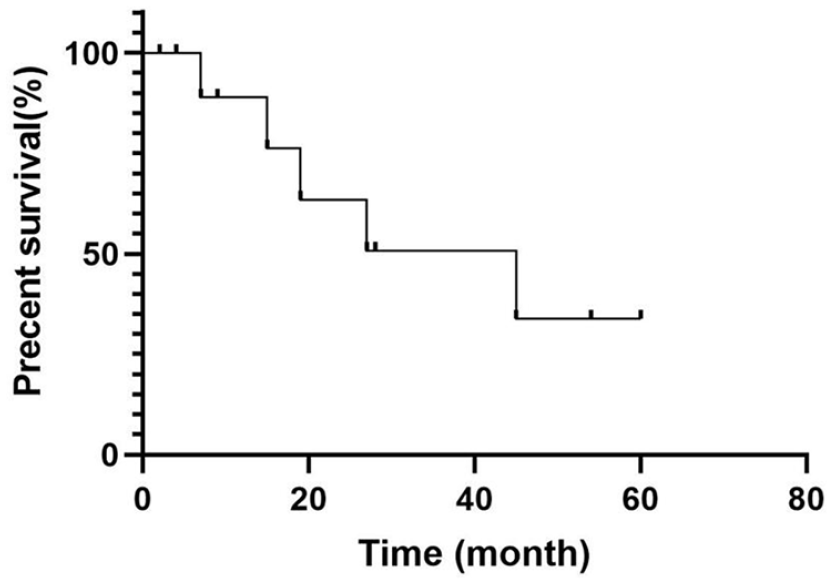
Survival curve with follow-up. Some of the patients develop/die from metastatic disease.
Conclusion
Primary MM in the parotid gland often presents as progressive enlargement of a painless mass in the unilateral parotid region, and it is characterized by metastasis and adverse outcomes. Pathological evaluation is the gold standard diagnostic technique for MM. US plays a role in determining the nature and location of parotid gland tumors. In this case report, we aim to emphasize the need for sonographers to consider melanoma as a differential diagnosis when encountering rapidly growing parotid gland tumors adjacent to or invading the skin layer, and to recommend clinicians to complete the comprehensive preoperative evaluation and appropriately extend the surgical scope if necessary.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shandong Provincial Natural Science Foundation, China [No. ZR2020MH290].
Ethical Approval
Ethical approval to report this case was obtained from the Affiliated Hospital of Qingdao University (QYFY WZLL 28035).
Informed Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Data Availability
All data generated or analyzed during this study are included in this published article.
