Abstract
Introduction
Sudden sensorineural hearing loss (SSNHL) is defined as sensorineural hearing loss of more than 30 dB over 3 consecutive wavelengths occurring within 72 hours, but no identified etiologies have been found. A diagnosis of SSNHL is excluded once the cause of hearing loss is clear. However, the pathogenesis of SSNHL remains vague. It seems that the incidence of SSNHL increased during the COVID-19 pandemic.1-3 Studies have shown that viral infection, including direct invasion, indirect reactions, stress responses, and immune-mediated hearing loss, is the main cause of SSNHL. Viral infection may play an important role in SSNHL by causing injury to the inner ear through blood or cerebrospinal fluid (mumps), to cochlear nerve fibers [herpes simplex virus (HSVs)], and to vestibular neurons through the middle ear (HSV). 4 Hand, foot, and mouth disease (HFMD) is a common, highly contagious infectious disease that usually occurs during childhood (usually under 10 years old) and is caused by infection via the fecal-oral route, ingestion of fecal material, or respiratory secretions containing the virus. 5 It is most frequently caused by coxsackievirus-A16 and enterovirus (EV)-A71, and reports of coxsackievirus B1 to B6 are increasing in adults. 6 Typical symptoms of HFMD include malaise, fever, decreased appetite, pain in the oral cavity, and lesions involving the hands, feet, oral cavity, upper extremities, lower extremities, and buttocks. 5 Nevertheless, SSNHL secondary to HFMD has rarely been reported. We describe an adult case of secondary SSNHL following HFMD.
Case Report
A 34-year-old man presented with sudden hearing loss in the right ear for 4 days after HFMD onset. Tinnitus, ear fullness, and a slightly heavy head appeared simultaneously as accompanying symptoms. The patient did not experience dizziness, vertigo, fever, chills, physical activity disorders, or consciousness disturbance during hearing loss. A pure tone test and acoustic immittance were performed, and the results (Figure 1) met the criteria for SSNHL in accordance with the “Guideline for the sudden sensorineural hearing loss diagnosis and treatment (2015)” in China. 7 All the frequencies were affected in the right ear, and the hearing thresholds were as follows: 75, 85, 85, 75, 70, 45 dB HL of 0.25k, 0.5k, 1k, 2k, 4k, 8k Hz, respectively, with a Pure tone average (PTA) of 79 dB Hearing Level (HL). The PTA in the unaffected ear (the left ear) was 9 dB HL (tested on August 11, 2023; Figure 1A). The patient was admitted to the inpatient department 4 days after he began to suffer from hearing loss.
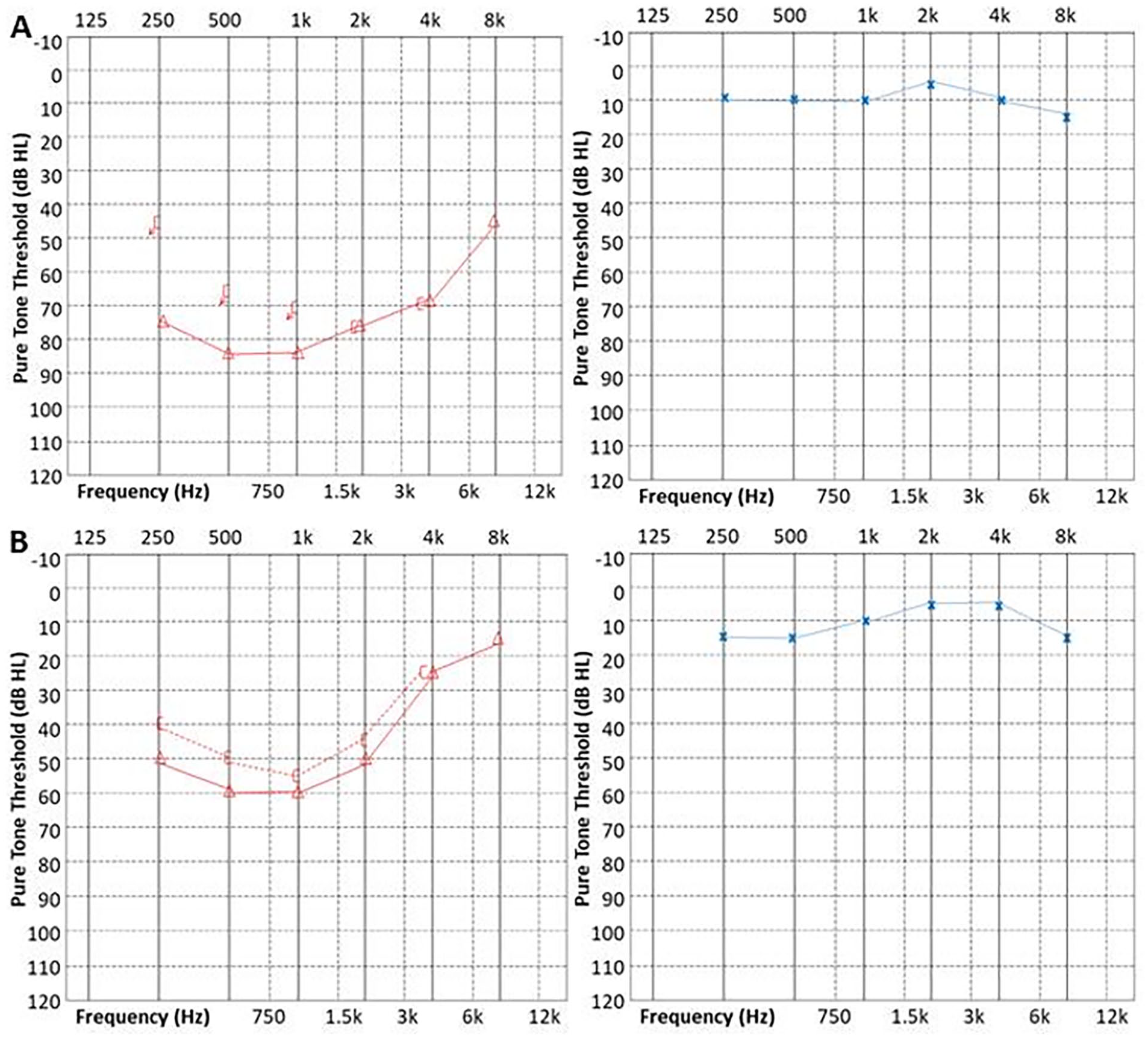
Pure tone test. (A) The first pure tone test; PTA is 79 dB HL on the left and 9 dB HL on the right. (B) The last pure tone test; PTA is 49 dB HL on the left and 9 dB HL on the right.
Before 6 days, he had a fever for 2 days (the highest temperature was 39.4°C), followed by vesicles in the oral mucosa (mainly on the soft palate) and papules on the hands and feet (Figure 2) after his child was diagnosed with HFMD. He was clinically diagnosed with HFMD after oral vesicles and hand-foot papules according to the “Guideline for the diagnosis and treatment of hand foot and mouth disease (2018)” in China. 8
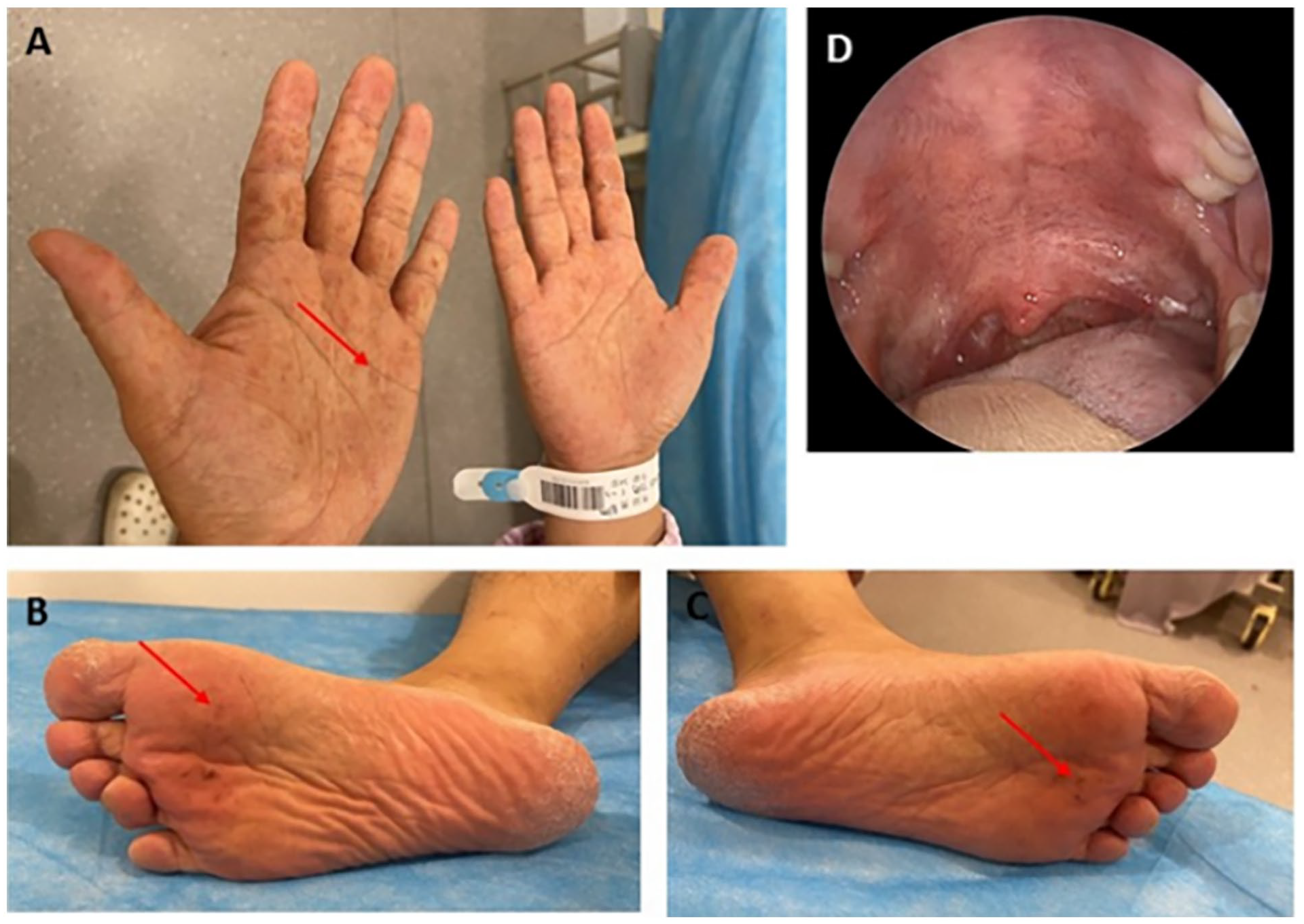
Lesions on the hand, feet, and oral mucosa. (A) The papules on hands; (B) the papules on the right foot; (C) the papules on the left foot; (D) the pigmentation on the soft palate.
On admission, physical examination revealed an intact tympanic membrane with no red or blue color or cerumen impaction. No rash or ulceration on the auricle or external ear was found. There was no facial palsy or feeling disorders. At day 6 after HFMD onset, many dark red, disseminated papules of different shapes and sizes were still observed (Figure 2A-C), while the skin remained intact. The number of vesicles on the soft palate had already partially decreased, but scattered pigmentation remained (Figure 2D). Respiratory virus antibody detection revealed no signs of Mycoplasma pneumoniae-Immunoglobulin M (IgM), Chlamydia pneumoniae-IgM, coxsackie virus B (CV-B)-IgM, adenovirus-IgM, or respiratory syncytial virus-IgM.
The patient was given intravenous batroxobin for defibrillation with indication for monitoring fibrinogen for 1 course (10 BU first time, 5 BU for the remaining 4 times) and intravenous alprostadil 10 µg daily for improving microcirculation for 1 week. After systemic administration of 10 mg of glucocorticoids daily for 5 days, 40 mg of glucocorticoids and dexamethasone were injected locally in the retroauricular position 5 times Quaque Altere Die (q.o.d). within 10 days. As the patient’s fever diminished and no serious situation or complication of HFMD occurred, no additional management was added to the HFMD treatment, though surveys were performed. After total treatment, the patient felt much better at hearing, tinnitus, and ear fullness. The papules on the palms and soles also vanished. The reexamined hearing thresholds were as follows: 50, 60, 60, 50, 25, 15 dB HL of 0.25k, 0.5k, 1k, 2k, 4k, 8k Hz, respectively, with PTA of 49 dB HL. The PTA in the left ear was 9 dB HL as before (tested on September 5, 2023; Figure 1B). As a result of the significant effectiveness of the treatment for SSNHL, the patient was released from the hospital.
Discussion
The cause and pathophysiology of SSNHL have not been clearly elucidated; however, the predisposing factors include mental tension, stress, mood fluctuations, irregular lifestyle, and sleep disorders. The etiology of SSNHL may include vascular diseases, viral infection, autoimmune diseases, infectious diseases, and tumors. Vasospasm in the inner ear, stria vascularis malfunction, vascular embolism, thrombosis, endolymphatic hydrops, and hair cell damage account for the widely accepted pathogenesis. Previous studies have shown a possible link between viral infection and SSNHL; the virus species include HSV (HSV1, HSV2), HIV, hepatitis virus, measles virus, rubella virus, mumps virus, Epstein-Barr virus (EBV), Lassa virus (LASV), human cytomegalovirus, varicella, and EV. 9 In this case, the patient presented with SSNHL just 2 days after the HFMD. It should be deduced that SSNHL is secondary to HFMD virus infection but is rare in adults.
As mentioned above, viral infection plays an important role in sudden hearing loss. A hypothesis has been proposed that suggests that the mechanism of viral infection in SSNHL has 3 aspects. The first is direct viral invasion into the inner ear, which can cause neuritis or cochleitis (HSV, LASV). The second is damage to the inner ear tissue caused by latent reactivation of the virus (HSV, EBV). The third is indirect triggering of SSNHL, either through immune mediation triggering of antibody-antigen reactions in the inner ear or through stress responses, triggering the activation of cellular pathologic pathways within the cochlea (HSV, LASV, or EBV). 4 However, no case of EV infection as a causative agent has been found in SSNHL patients, according to a literature review. Moreover, the pathogenesis of EV infection and the role it plays in SSNHL also remain unclear. Infection caused by EV may result in a wide range of acute symptoms involving the skin, mucous membranes, cardiac and skeletal muscle, the pancreas, and the central nervous system. Coxsackievirus, a member of the EV family, is the main serotype of HFMD (especially EVA16 and EVA71) in China. HFMD is most prevalent in children. As a result of antibodies generated by inapparent EV infection, adults are not easily infected with HFMD via EV. However, outbreaks of atypical presentation in adulthood associated with the coxsackie-A6 virus have been reported.10,11 The diagnosis of HFMD in adults is imminent clinically since virus culture and laboratory identification require specialized laboratories. 11 In this case, the adult patient was diagnosed with HFMD clinically on the basis of fever, lesions on the hands, feet, or oral mucosa, and cohabitation with the diagnosed patient. PCR could not be performed because the patient was admitted 6 days after HFMD. On admission, the patient’s virus antibody detection was negative. This could be explained by the fact that CV-IgM appears from 1 to 2 weeks after infection but the patient was tested within 1 week after infection. The second reason is that the patient was infected by CV-A, but the test was for the CV-B antibody. The third reason is that the virus antibody enzyme linked immunosorbent assay (ELISA) has lower specificity than Polymerase Chain Reaction (PCR).
Reports have shown nervous system damage due to HFMD. André et al 12 reported a clinical case of typical Guillain-Barré Syndrome (GBS) after HFMD. Falk and Malik 13 reported a case of unilateral acute idiopathic maculopathy in a 32-year-old woman with a history of HFMD caused by coxsackievirus. This case may be the first report on SSNHL following HFMD infection in adults. As coxsackievirus is a common kind of EV, it may offer new insight into the pathogenesis of SSNHL, but additional studies are needed.
Footnotes
Data Availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Chinese Medicine Science and Technology Innovation Special Project of Dongzhimen Hospital Affiliated to Beijing University (Grant Number: DZMKJCX-2023-031) and Natural Science Foundation of Beijing Municipality (Grant Number: 7214290).
Ethical Approval
Ethical approval to report this case was obtained from the Ethics Committee of Dongzhimen Hospital Affiliated to Beijing University of Chinese Medicine (Approval Number: 2023DZMEC-577-01). The patient provided written informed consent prior to enrollment in the study.
