Abstract
Introduction
Thyroid nodules (TNs) are common diseases of the endocrine system defined as scattered lesions caused by abnormal proliferation and growth of thyroid cells. TNs can be benign or malignant according to their pathological characteristics. Papillary thyroid carcinoma (PTC) is the most common type of malignant TNs, accounting for 80% to 90%. 1 Recently, the incidence of thyroid carcinoma has increased, making it the most common malignant tumor in young women (<30 years old) in China. 2 In the United States, thyroid cancer is the fifth most common cancer among females and is likely to become the most common cancer among 15- to 29-year-old females in the future. 3 China has the highest number of thyroid cancer-related deaths worldwide. The high incidence of thyroid cancer imposes a heavy burden on global healthcare systems. 4 Therefore, early diagnosis and effective treatment of TNs are crucial. High-resolution color Doppler ultrasonography has an irreplaceable role in the early diagnosis of thyroid carcinoma. 5 Thyroid Imaging, Reporting and Data System (TI-RADS) 3 TNs are mostly benign lesions but missed diagnosis of TI-RADS 3 thyroid carcinoma is always an issue in clinical practice. Therefore, some patients miss their best opportunity for surgical treatment because of cervical lymph node metastasis before diagnosis. The reason for the missed diagnosis of TI-RADS 3 thyroid carcinoma may be the overlap of ultrasound features between malignant and benign TNs. Therefore, it is important to differentiate between benign and malignant TI-RADS 3 TNs in the early stage. Currently, there are few reports on the risk factors of TI-RADS 3 thyroid carcinoma. Therefore, our goal was to detect the risk factors for malignant TI-RADS 3 TNs and to construct a predictive model.
Materials and Methods
Patients
We retrospectively collected the clinical images and pathological data of 199 patients, including 46 men and 153 women. Their age ranged from 18 to 80 years with a mean ± standard deviation (SD) of 51.79 ± 10.40 years. All patients had TI-RADS 3 TNs treated at the Department of Thyroid Cancer, Affiliated Hospital of Qinghai University, from January 2018 to September 2021. All 199 patients with TI-RADS 3 TNs underwent first-time thyroid surgery. This retrospective study was approved by the Ethics Committee of Affiliated Hospital of Qinghai University, and written informed consent was waived.
Inclusion and Exclusion Criteria
Inclusion criteria were as follows: (1) Patients having a preoperative ultrasound report of TI-RADS 3 TNs. (2) Patients with surgical indications, including confirmed/highly suspected thyroid cancer in preoperative fine needle aspiration cytology or clinical symptoms of compression. (3) Patients who underwent thyroid resection for the first time. Exclusion criteria were as follows: (1) Patients with incomplete medical records. (2) Patients with a history of other malignant tumors. (3) Patients with discordant TI-RADS grades on 2 or more preoperative thyroid ultrasonography. (4) Patients whose preoperative thyroid ultrasonography was not performed in Qinghai University Affiliated Hospital.
Covariates
Preoperative basic clinical characteristics, preoperative imaging, and lab data were collected. Gender, age, height, weight, body mass index (BMI), lesion ultrasound characteristics, and the results of fine needle aspiration cytology, thyroid function, parathyroid function, and thyroid autoantibodies were collected for each patient. In particular, total triiodothyronine (TT3), total thyroxine (TT4), free triiodothyronine (FT3), free thyroxine (FT4), thyroid-stimulating hormone (TSH), thyroglobulin (Tg), parathyroid hormone (PTH), anti-thyroglobulin antibodies (TgAb), anti-thyroid peroxidase antibodies (TPOAb), and surgical histopathology report were determined for each participant. BMI was calculated by the following formula: weight (kg)/height (m²). American College of Radiology (ACR) TI-RADS was used to evaluate the categories of TNs. The ACR TI-RADS level was determined based on the points given to all ultrasound features of a nodule, with a point of 3 being defined as TI-RADS 3. Various ultrasound features of TNs are described in ACR TI-RADS as follows: (1) Composition: (a) cystic or almost completely cystic (0 points). (b) Spongiform (0 points) composed predominantly (>50%) of small cystic spaces. Further points are not added for other categories. (c) Mixed cystic and solid (1 point): points are assigned for the predominant solid component. (d) Solid or almost completely solid (2 points): 2 points are assigned if composition cannot be determined because of calcification. (2) Echogenicity: (a) Cystic echoic (0 points) applies to cystic or almost completely cystic nodules. (b) Hyperechoic/isoechoic (1 point) compared with adjacent parenchyma. (c) Hypoechoic (2 points) compared with adjacent parenchyma. (d) Very hypoechoic (3 points): more hypoechoic than strap muscles. (e) One point is assigned if echogenicity cannot be determined. (3). Echogenic foci: (a) None or large comet-tail artifacts (0 points): V-shaped, >1 mm, in cystic components. (b) Macrocalcifications (1 point) causing acoustic shadowing. (c) Peripheral (rim) calcifications (2 points): complete or incomplete along margin. (d) Punctate echogenic foci (3 points), which may have small comet-tail artifacts.6,7 Unfortunately, there was no detailed classification report of echogenic foci in our ultrasound report. The ultrasound image features of TI-RADS 3 TNs were interpreted and reported, including the number of nodules (single/multiple), the boundary of nodules (regular/irregular), echogenic foci (no/yes), maximum tumor diameter (≥30 mm/<30 mm), the vessels of nodules (no/yes), and echogenicity. For covariate echogenicity, chi-square test was performed on isoechoic, hypoechoic, very hypoechoic, mixedechoic, hyperechoic, and cysticechoic before the experiment. The results showed that there was no significant difference in iso, hypo, and very hypo echo between the 2 groups. Similarly, the distribution of mixed, hyper, and cystic echo did not significantly differ between the 2 groups. Therefore, the echoes were divided into mixed/hyper/cystic echo group and iso/hypo/very hypo echo group. The PHILIPS EPIQ5 and PHILIPS Affiliti70 systems were used as the diagnostic apparatus, with a probe frequency of 7 to 10 MHz. The instrument settings were suitable for thyroid examination. The Roche E601 automatic chemiluminescence immunoassay analyzer was used to assess thyroid function tests and autoantibodies. All ultrasound images in this study were retained by 2 senior physicians and reviewed by the chief sonographer.
Methods Statement
All methods in this study were performed in accordance with the relevant guidelines and regulations.
Statistical methods
The data were analyzed using Statistical Package for Social Science (SPSS Inc., Chicago, IL, USA) version 25.0. The data were expressed as mean ± SD and interquartile range for continuous variables, while categorical variables were expressed as the number of cases (percentage). Differences between the 2 groups were analyzed using Student’s t-test, the Mann–Whitney U test for continuous variables, and the χ2 test or Fisher’s exact test for categorical data. A predictive model was established based on the risk factors determined by multivariate logistic regression analysis. Logistic regression was used to construct receiver operating characteristic (ROC) curves. Youden’s method was used to determine an optimal cut-off point in the ROC curve to maximize sensitivity and specificity. The tolerance (Tol) <0.1 or the variance inflation factor (VIF) >10 were considered the presence of collinearity. For all statistical tests in this study, P-values <.05 were considered statistically significant.
Results
Univariate Analysis for Postoperative Pathologically Proven Malignant TI-RADS 3 TNs
As shown in Table 1, the age of patients (P = .001), preoperative ultrasound features such as echogenicity (P = .005), echogenic foci (P = .001), tumor size (P < .001), and PTH level (P = .003) were associated with TI-RADS 3 thyroid carcinoma. However, there were no significant differences in gender, BMI, nodule number, nodule boundary, vascularization, Tg, Tg Ab, TT3, TT4, FT3, FT4, TSH, and TPOAb between benign and malignant TNs (all P ≥ .05).
Clinical Characteristics and Univariate Analysis of TI-RADS 3 Thyroid Carcinoma.
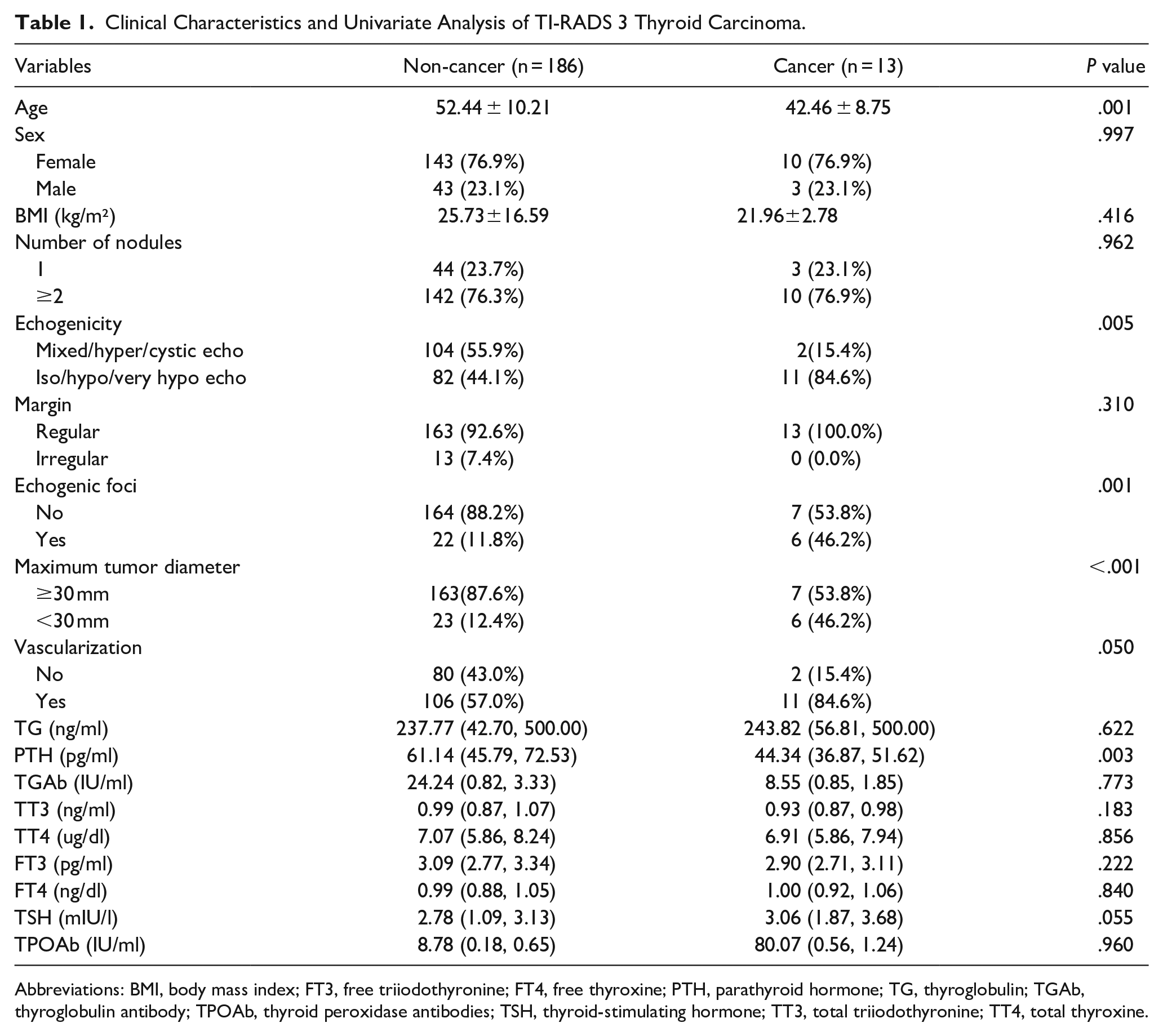
Abbreviations: BMI, body mass index; FT3, free triiodothyronine; FT4, free thyroxine; PTH, parathyroid hormone; TG, thyroglobulin; TGAb, thyroglobulin antibody; TPOAb, thyroid peroxidase antibodies; TSH, thyroid-stimulating hormone; TT3, total triiodothyronine; TT4, total thyroxine.
Multivariate Logistic Regression Analysis for Malignant TI-RADS 3 TNs
Five variables with statistical significance in the univariate analysis were included in the multivariate logistic regression analysis as follows: echogenicity (0 for Iso/hypo/very hypo echo and 1 for mixed/hyper/cystic echo), echogenic foci (0 for no and 1 for yes), and maximum tumor diameter (0 for <30 mm and 1 for ≥30 mm). Age and PTH level were considered as continuous variables. As shown in Table 2, a collinearity diagnosis was performed on the independent variables with statistical significance. It can be concluded that there is no collinearity among the above independent variables (Tol > 0.1 and VIF < 3). Taking TI-RADS 3 thyroid carcinoma as the dependent variable, the 5 preoperative factors with statistical significance were used in the regression model as independent variables. The results showed that age [odds ratio (OR): 0.926, 95% CI: 0.865-0.992; P = .029], PTH level (OR: 0.940, 95% CI: 0.890-0.993; P = .027), and preoperative ultrasound features of TI-RADS 3 TNs, such as echogenicity (OR: 8.496, 95% CI: 1.377-52.406; P = .021), echogenic foci (OR: 8.611, 95% CI: 1.484-49.957; P = .016), and maximum tumor diameter (OR: 0.188, 95% CI: 0.040-0.888; P = .035) were independent risk factors for malignant TI-RADS 3 TNs (see Table 3 for details).
Collinearity Diagnosis.
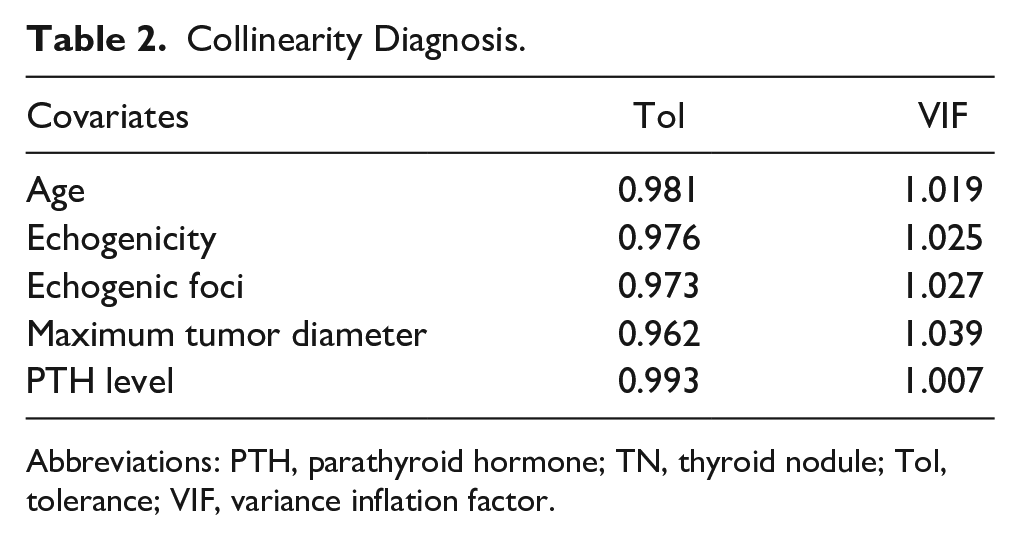
Abbreviations: PTH, parathyroid hormone; TN, thyroid nodule; Tol, tolerance; VIF, variance inflation factor.
Multivariate Logistic Regression Analysis for Malignant TI-RADS 3 TNs.
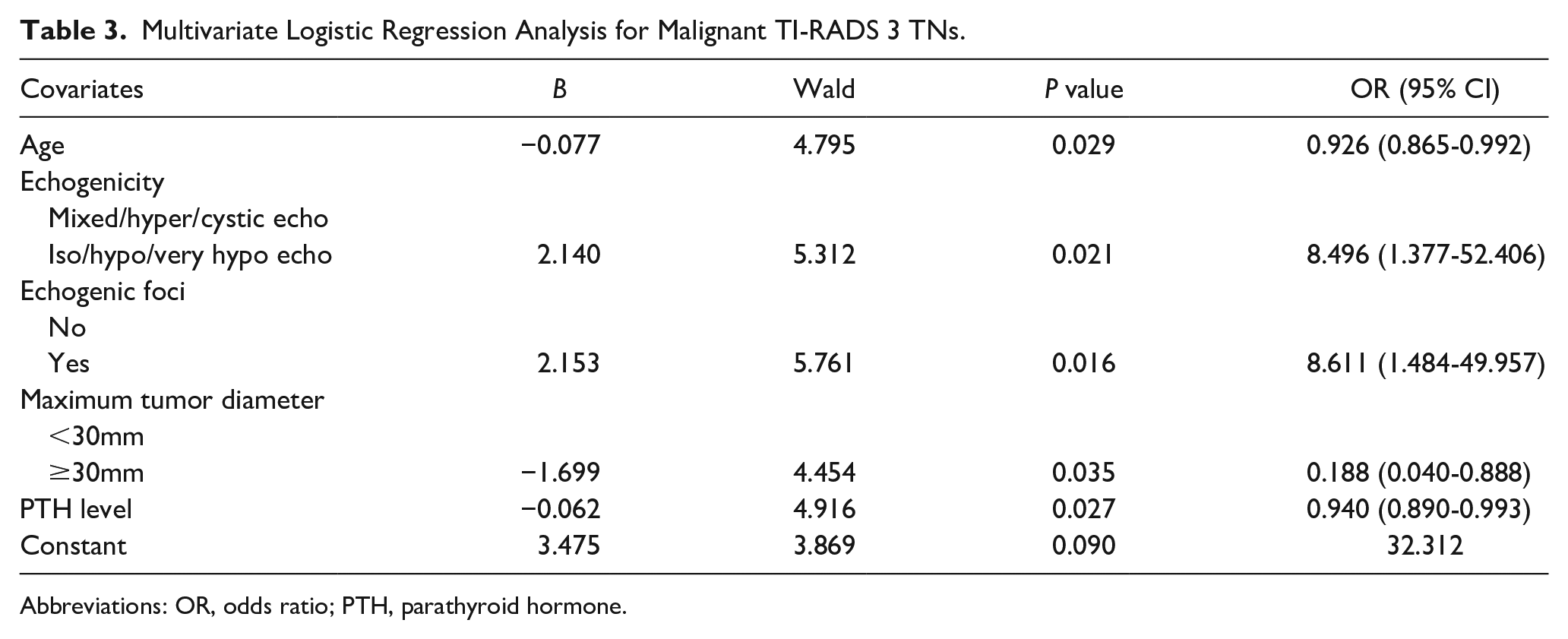
Abbreviations: OR, odds ratio; PTH, parathyroid hormone.
Clinical Value of Logistic Regression Model in Predicting TI-RADS 3 Thyroid Carcinoma
Based on the results of multivariate analysis, a logistic regression model was established using the following formula: Logistic (P) = 3.475 − 0.077 × age + 2.140 × echogenicity + 2.153 × echogenic foci − 1.699 × nodule diameter − 0.062 × PTH. The values of each factor were substituted into the equation to obtain the probability of multi-factor joint prediction, and the ROC curve was drawn (Figure 1). The area under the curve (AUC) of the prediction model for TI-RADS 3 thyroid cancer was 0.921 (95% CI: 0.856-0.986, P < .001). The maximum Youden index was 0.698. The cut-off value, sensitivity, and specificity were 0.074, 84.6%, and 85.2%, respectively. Hosmer-Lemeshow goodness-of-fit test showed that the model had a good fit (P = .946). The prediction model comprehensively reflected the predictive value of the 5 risk factors, and the diagnostic accuracy of the model was 94.0%. The AUC of age was 0.791 (95% CI: 0.680-0.902, P < 0.001), the maximum Youden index was 0.570, and the cut-off value of diagnosis was 46.5 years. The AUC of the PTH level was 0.745 (95% CI: 0.642-0.847, P = .003), the maximum value of the Youden index was 0.461, and the cut-off value of diagnosis was 53.71 pg/ml.
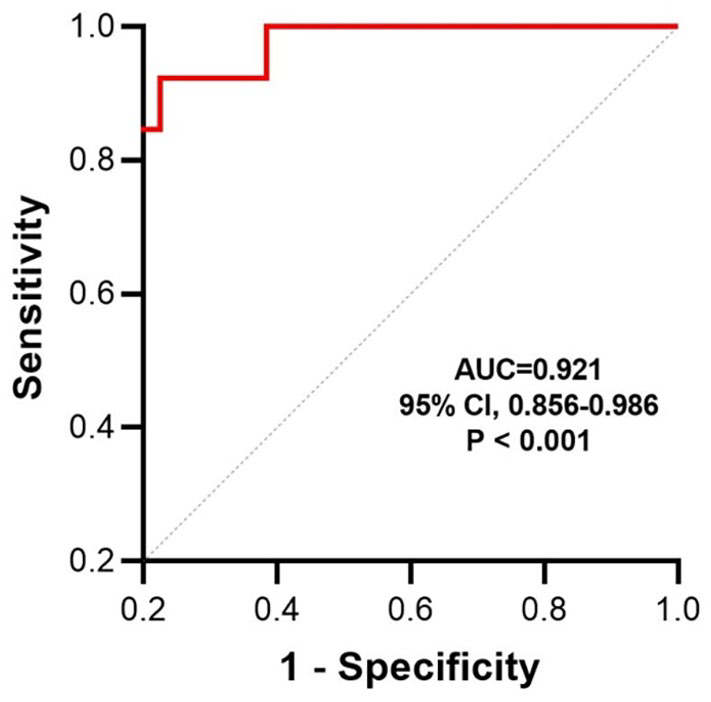
ROC curve of the logistic regression model for predicting TI-RADS 3 thyroid carcinoma.
Discussion
Thyroid carcinoma is a common malignancy of the endocrine system. At present, there are many studies on the risk factors of thyroid carcinoma, but few studies were conducted on malignant TI-RADS3 TNs. TI-RADS3 TNs are mostly benign, but postoperative pathologically-proven malignant TI-RADS3 TNs are common in some medically underdeveloped areas of China. This study showed that age, echogenicity, echogenic foci, nodule diameter, and PTH level were independent risk factors for TI-RADS 3 thyroid carcinoma.
Multivariate logistic regression analysis showed that echogenicity was significantly associated with TI-RADS 3 thyroid carcinoma. According to K-TIRADS, iso-echoic/hyper-echoic nodules and some cystic nodules can be classified into TI-RADS 3 (low-grade suspicious malignant) in the absence of suspicious features, 8 and hypo-echoic TNs are commonly considered malignant.9,10 Therefore, decreased echogenicity within TNs may imply that the follicular cells are undergoing rapid and disordered growth with the loss of normal thyroid parenchyma. 11 Our results suggest that in addition to hypoechoic and very hypoechoic, isoechoic is also a risk factor for malignant TI-RADS 3 TNs. Cancer cells were more than mesenchyme in malignant nodules thus few US reflection interfaces were created, which may be the underlying mechanism for hypoechogenicity. Fibrosis in malignant nodules may be the reason for isoechogenicity. 12 Therefore, clinicians should pay more attention to iso-echo Ti-RADS 3 TNs.
Echogenic foci refer to the highly reflective spot or light strip in ultrasound images. In vivo, cancer tissue polarization, necrosis, and calcium deposition can manifest as echogenic foci. In different versions of TI-RADS guidelines, calcifications of TNs are listed as malignant features.6,13-15 Deng et al 12 suggested that the echogenic foci inside the nodule are a hybrid ingredient in iso-echoic or slight hypo-echoic TNs, which may be caused by the reflection of fibrosis or mesenchyme inside the cancerous tissue. Echogenic foci can better represent tissue type than calcification. Our results show that echogenic foci are an independent risk factor for TI-RADS 3 thyroid carcinoma. Therefore, using the echogenic foci as a risk factor for TI-RADS 3 thyroid carcinoma may improve the sensitivity of diagnosis.
Nodule size also had predictive value for malignancy in several studies.16,17 Al-Hakami et al 18 suggested that nodule size <2 cm is associated with an increased risk of well-differentiated thyroid cancer. Our results suggest that nodule diameter <3 cm is an independent risk factor for TI-RADS 3 thyroid cancer, which is consistent with previous findings 12 . However, a meta-analysis reported an increased risk of thyroid cancer in nodules measuring 3 to 5.9 cm compared with those measuring less than 3 cm and a decreased risk of thyroid cancer in nodules measuring more than 6 cm compared with those smaller than 3 cm. 19 A systematic review has also reported that nodule diameter >3 cm is a risk factor for thyroid cancer, and surgery is a reasonable option for nodules >3 cm. 20 Other studies have shown that there is no correlation between the prevalence of malignancy and nodule size when nodule size is ≥3 cm. 20 Therefore, nodule size cannot simply differentiate between benign and malignant TNs.
In this study, age was inversely associated with the risk of TI-RADS 3 thyroid carcinoma, and age less than 46.5 years increased the risk of malignancy in patients with TI-RADS 3 TNs, which was consistent with the results of previous studies.21-23 In Bessey’s study, the frequency of malignant nodules in patients with ≤45 years of age was 2 times more than that in those with >45 years of age. 24 Meanwhile, young age is also an independent risk factor for central lymph node metastasis in patients with PTC.1,25,26
A study reported that most non-medullary thyroid carcinomas were detected synchronously with primary hyperparathyroidism. 27 Many theories, including genetic mutations and goitrogenic effects of calcium, have been proposed to explain this coexistence. 28 However, others still believe that coexistence is casual.28,29 Interestingly, our findings revealed that patients with Ti-RADS-3 TNs with lower levels of PTH within the normal range have a higher risk of thyroid cancer than those with higher levels of TSH. This correlation has been previously described by Lehwald et al. 27 Therefore, the author suggests that PTH can be used as a preoperative indicator for malignant TI-RADS 3 TNs. Whether the normal level of PTH in Ti-RADS-3 thyroid carcinoma is redefined remains to be discovered. Its mechanism also needs to be further investigated.
TPOAb is a serological marker for predicting thyroid autoimmune diseases, such as Hashimoto’s thyroiditis (HT). Clinical studies have shown that HT and PTC are similar regarding their genetic background and biological characteristics. HT can increase the risk of PTC. 30 In 2005, Boi et al 31 retrospectively found a higher prevalence (18.8%) of suspicious/malignant cytology in patients with positive serum TPOAb than in patients with negative anti-thyroid autoantibodies (ATA) (9.28%), and univariate analysis showed that ATA positivity indicates a significant risk (OR: 2.29) of suspicious/malignant cytology independent of age. This correlation has been approved by other researchers.32-34 Other studies have shown that high concentrations of TPOAb are associated with occult PTC, 20 and TPOAb > 1300 IU/ml is an indicator of multifocal PTC in HT patients. 35 In this study, the results showed that there was a non-significant difference in TPOAb between TI-RADS 3 benign and malignant TNs. Therefore, the value of TPOAb in predicting TI-RADS 3 thyroid carcinoma needs further studies.
After determining the risk factors of malignant TI-RADS 3 TNs, we established a prediction model. The goodness-of-fit test showed that the model had a good fit, with a prediction accuracy of 94.0%, a sensitivity of 84.6%, and a specificity of 85.2%. Therefore, clinicians can apply this model to evaluate the risk of malignant Ti-RADS 3 TNs before surgery and recommend reasonable treatment for patients with Ti-RADS 3 TNs.
This study had some limitations. First, it was a retrospective study with inherent selection bias. Second, our study merely reflected the experience of a single center. As a result, a multicenter study from different institutions and regions is expected in the future. Third, there were no external validation data, which reduces statistical power.
In conclusion, young age, iso/hypo/very hypo echo, echogenic foci, nodule diameter <30 mm, and low level of PTH are independent risk factors for TI-RADS 3 thyroid carcinoma. This prediction model has a high sensitivity and specificity. A prediction model value of more than 0.074 implies that the Ti-RADS 3 TN has undergone a malignant transformation, and fine needle aspiration is recommended in these cases.
Footnotes
Acknowledgements
Author Contributions
BP and SZ: conceptualization. BP: data curation. BP and FD: methodology. BP: writing—original draft. SZ and BP: writing—review and editing.
Availability of Data and Materials
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This retrospective study was approved by the Ethics Committee of Affiliated Hospital of Qinghai University in accordance with the Declaration of Helsinki, but without an ethics number. Written informed consent was waived by the Ethics Committee of Affiliated Hospital of Qinghai University.
