Abstract
Introduction
Middle ear cholesteatoma secondary to chronic otitis media (COM) is commonly seen in otology clinics and can be either acquired or more rarely congenital. Primary acquired cholesteatoma arises mainly from pars flaccida in the attic and is related to invagination of the retraction pocket. Secondary acquired cholesteatoma occurs as a result of the ingrowth or migration of squamous epithelium from the edge of a tympanic membrane (TM) perforation.1,2 Chronic inflammation and the formation of intratympanic folds between the medial wall of the tympanic cavity and the TM are also important in the formation of cholesteatoma in COM.3,4
Intratympanic membrane cholesteatomas (ITMCs) are rare and occur mainly in children.5-7 ITMC was defined as the cholesteatoma confining to the TM in the specified area, but the middle ear, ossicular chain, attic, and mastoid air cells were within normal limits.5-7 Most ITMCs in children are congenital, although Jeong and Choi 8 reported an ITMC following traumatic perforation. ITMCs occur less frequently in adults. Pedruzzi et al 9 and Murphy and March 2 reported congenital ITMCs in 36- and 20-year-old adults. However, most adult ITMC occurs after otologic surgery, including the overlay, underlay, and over-underlay techniques. The mechanism of ITMC remains unclear. The best explanation of ITMC formation may be that by Ruedi, 10 who theorized that inflammatory injuries to the TM result in the proliferation of its squamous epithelial basal layer into protruding cones. Then, these cones form cholesteatomas within the TM. However, there are few reports on the management and complications of COM with ITMC in adults. Therefore, this study evaluated the surgical outcomes and complications of the endoscopic cartilage-perichondrium underlay technique for the treatment of COM with ITMC in adults.
Materials and Methods
Ethics Considerations
The study protocol was reviewed and approved by the Institutional Ethics Review Board of Yiwu Central Hospital (protocol number: K2020-IBR-027 on April 20, 2020). All participants provided informed consent.
Patient Selection
Subjects were recruited from consecutive adults diagnosed with COM and ITMC. The inclusion criteria were adult, history of COM, and ITMC with central TM perforation or atrophic TM or TM scar healing. Exclusion criteria included poor pneumatization of the middle ear and mastoid, ossicular chain abnormalities, and middle ear cholesteatoma behind the TM. Age, sex, pre- and postoperative symptoms, involved ear, cholesteatoma position, and pre- and postoperative hearing levels were recorded in all subjects. The criteria to diagnose ITMC was the cholesteatoma confining to the TM in the specified area but the middle ear, ossicular chain, attic, and mastoid air cells were within normal limits. All the patients underwent high-resolution computed tomography and magnetic resonance diffusion weighted imaging (MRDWI).
Audiometric data were assessed preoperatively and 12 months postoperatively. Pure-tone averages were calculated for both air (AC) and bone (BC) conduction by averaging the thresholds at 500, 1000, 2000, and 3000 Hz; in most cases, a threshold of 4000 Hz was used to interpolate the threshold at 3000 Hz as per the standards of the Hearing Committee of the American Academy of Otolaryngology—Head and Neck Surgery. The air-bone gap (ABG) was calculated by subtracting the AC threshold from the BC threshold. ABG closure was calculated as the preoperative ABG minus the postoperative ABG at 500, 1000, 2000, and 3000 Hz.
Surgical Approach
All surgeries were performed endoscopically under general anesthesia by the same surgeon. A single-layer perichondrium-cartilage composite graft was harvested from the ipsilateral tragus. The graft was supported medially by Biodegradable NasoPore (Stryker Canada) in all the ears.
The ITMC with the fibrous layer was removed entirely by endoscopy via a transcanal approach (Figure 1). Also, the atrophic or scar TM was removed before the myringoplasty.
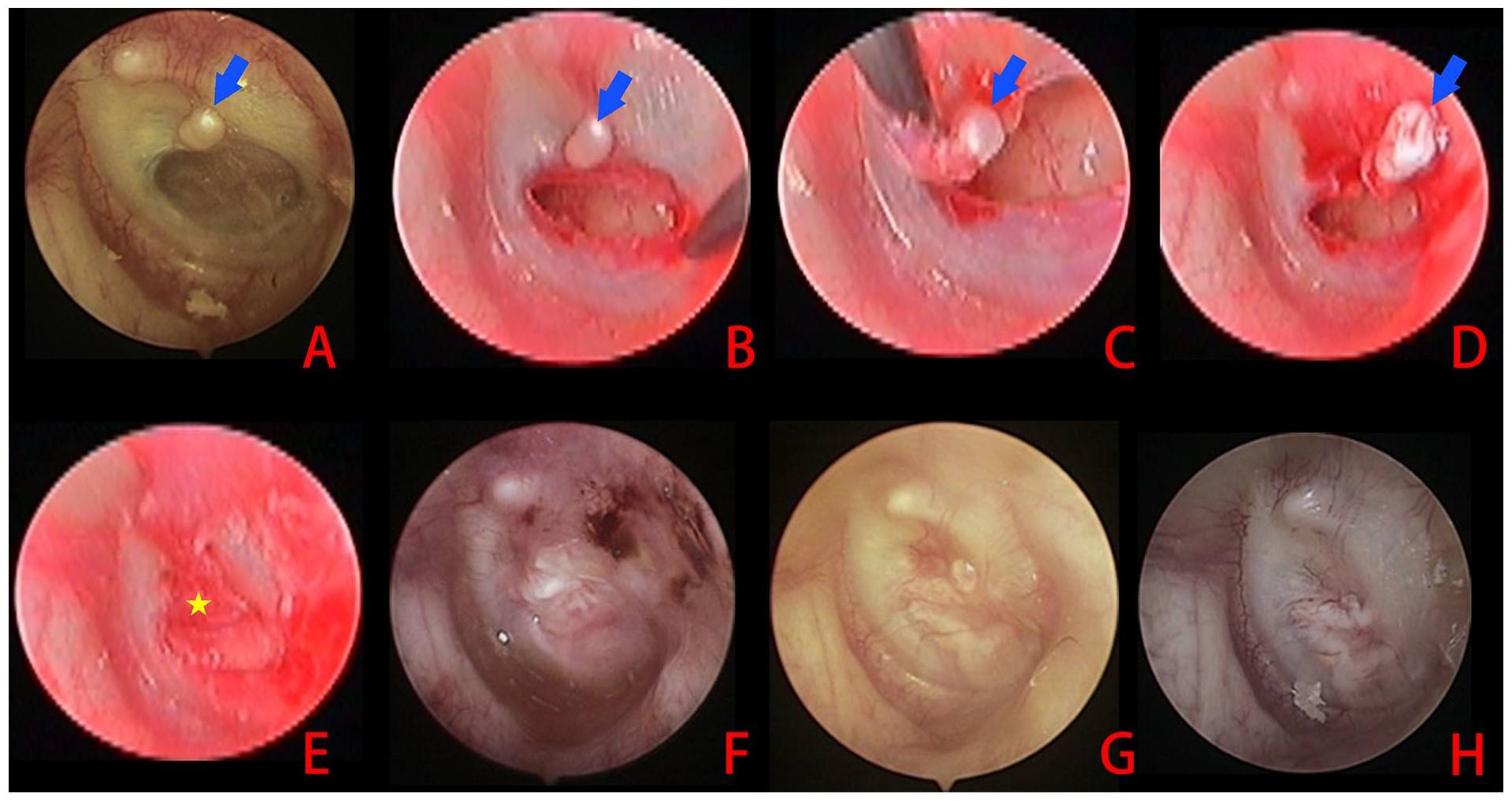
Preoperative intratympanic membrane cholesteatoma with an atrophic TM (A); removal of the atrophic TM (B); removal of the cholesteatoma (C and D); cartilage-perichondrium composite graft underlay technique (E); and 1 week (F) and 2 (G) and 3 months (H) postoperatively. Blue arrows: cholesteatoma; yellow star: graft. TM, tympanic membrane.
No raising the tympanomeatal flap was used. The lateral perichondrium was freed completely from the cartilage graft. Cartilage was removed peripherally according to the size of the perforation size, which makes it at least 1 mm larger than the perforation margins. A notch was created in the cartilage graft to accommodate the malleus handle if any. The perichondrium graft was also trimmed peripherally and was approximately 2 mm larger than the cartilage graft. The cartilage graft was placed trans-perforation medial to the remnant TM and annulus, with a notch in the cartilage to accommodate the malleus handle. Subsequently, the perichondrium graft was also placed trans-perforation medial to the remnant TM and annulus but lateral to the cartilage graft.
Postoperative Follow-Up
All patients were discharged on the day after surgery. Follow-up appointments were scheduled at 2 and 4 weeks and 3, 6, and 12 months postoperatively. The TM and graft were observed by endoscopy at each follow-up visit. The graft success, hearing improvement, postoperative symptoms, and cholesteatoma recurrence were evaluated at 12 months postoperatively. Graft success was determined by the presence of an intact graft without residual perforation or reperforation.
Results
Demographic Variables
This study included 11 patients with ITMC. Of the 11, 2 (18.2%) had TM perforation and the remaining 9 (81.8%) had an atrophic TM or TM scar healing; the left ear was affected in 8 (72.7%) and the right ear in 3 (27.3%); there were 7 (63.6%) females and 4 (36.4%) males. The average patient age was 43.6 ± 5.4 (range 21-56) years. All the patients had a history of COM. Both patients with TM perforation had a dry ear at least 6 months preoperatively.
All patients reported different degrees of hearing loss. In addition, 6 complained of tinnitus and ear fullness; 3 complained of tinnitus only; and 1 complained of ear fullness only. In endoscopy, accumulated keratin debris was noted at the superior edge of the perforation or atrophic TM in all 11 patients. CT image showed a localized mass within the TM or the thickened area of TM with no middle ear and ossicular chain extension in all the patients. MRDWI showed a localized abnormally high signal intensity within the TM. The preoperative ABG was 28.6 (range 18.3-34.7) dB. Intraoperatively, the epithelial invasion of the cholesteatoma was limited to the fibrous layer within the TM. Squamous epithelium with keratin debris in the TM was confirmed histopathologically in all cases.
Postoperative Outcomes
The cartilage-perichondrium graft underlay technique or overlay-underlay technique with perichondrium placed lateral to the malleus was performed after removing the atrophic TM and cholesteatoma in the 9 patients with an atrophic TM, while the cartilage-perichondrium graft underlay technique was applied after removing the cholesteatoma in both patients with TM perforations. All grafts were successful, and the perforations had closed completely by the final 12 month follow-up in all 11 patients. Endoscopy revealed no recurrence of cholesteatoma (Figure 1).
Of the 9 patients with preoperative tinnitus, the tinnitus disappeared in 3 (33.3%), was relieved in 4 (44.4%), and was unchanged in 2 (22.2%). Of the 7 patients with preoperative ear fullness, the ear fullness disappeared in 6 (85.7%) and was relieved in 1 (14.3%). The postoperative ABG was 16.2 (range 11.6-22.4) dB. None of the patients reported surgery or graft-related complications during follow-up.
Discussion
Congenital ITMC occurs mainly in children, and less frequently in adults5-7; only 4 authors have reported congenital ITMC.2,9,11,12 Characteristically, keratin debris accumulates between the outer epidermal layer and middle fibrous layer of the TM without extending into the middle ear cavity. 9 Most ITMC in adults is acquired from previous otologic surgery or COM.5,13 Whether the ITMC is secondary to chronic suppurative otitis media (CSOM) or a type of CSOM is unclear. All the patients had early history of CSOM in this study; we speculated that these ITMCs were secondary to CSOM and acquired. COM causes recurrent inflammatory injury to the TM, which leads to the proliferation of the basal layer of the squamous epithelium, which forms protruding cones. Then, the cones form cholesteatomas within the layers of the TM. 10 Myringoplasty is another important pathogenic factor in acquired ITMC. 14 The ITMC usually occurs after tympanoplasty with the overlay technique or combined overlay-underlay technique.14-16 Adult ITMC is also seen with the underlay technique, and Nejadkazem et al 14 reported an ITMC occurrence rate of 0.8% after this technique.
Most ITMC in adults is asymptomatic and is only discovered during a physical examination. Our findings are similar to studies that report that the ITMC is confined to the fibrous layer of the TM and does not involve the ossicular chain or middle ear invasion.2,6,9,11,12 A few authors have reported mild conductive hearing loss.2,6,9,11,12 In the current study, most patients complained of tinnitus and ear stuffiness and all the patients reported different degrees of conductive hearing loss. We believe that these symptoms are not related to the ITMC but rather to the TM perforation, scar healing, or Eustachian tube dysfunction.
Some scholars have recommended that ITMC be simply observed at first in asymptomatic patients; they believe that the ITMC may not induce symptoms or invade the middle ear because it can grow outward into the external auditory canal. 8 Other scholars suggest early removal; they believe that it can grow and compress the TM and advance into the middle ear and ossicular chain, which can cause symptoms such as hearing loss.5,6 Some authors suggest removing the cholesteatoma and preserving the intact fibrous layer; however, graft myringoplasty should be performed if the fibrous layer is injured.
In this study, all the patients had TM perforation or an atrophic TM accompanied by symptoms at the initial visit; the symptoms were confounded because of simultaneous atrophic membrane and ITMC. Consequently, atrophic TM was removed following complete excision of the ITMC and fibrous layer; then the perforations were repaired by cartilage-perichondrium graft technique without raising a tympanomeatal flap. During the 12 month follow-up, all 11 patients obtained well graft success and no cholesteatoma recurred. These results agreed with those of a previous study. 5 Several previous studies have suggested that endoscopic cartilage-perichondrium graft technique without raising a tympanomeatal flap that had a high graft success rate.15,16 []Although revision cases were not encountered in this study, we believed that this technique is also applicable to revision cases.
In this study, 77.8% of the patients with tinnitus and all the patients with ear stuffiness had symptom improvement. The mean ABG improved from 28.6 dB preoperatively to 16.2 dB postoperatively. In addition, none of the patients reported surgery or graft-related complications during the follow-up period. Limitations of this study were the small sample size and short follow-up. In addition, all results are confounded by membrane perforation so would be for all symptom improvement.
Conclusions
Endoscopic complete excision of an ITMC and cartilage-perichondrium graft without raising a tympanomeatal flap can lead to successful graft intake and improve the symptoms for the patients with ITMC combined with perforation or atrophic TM.
Footnotes
Author Contributions
Data Availability Statements
All data generated or analyzed during this study are included in the published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Health Commission of Zhejiang Province, China (Grant #2021KY1186), and Technology Agency of Jinhua, China (Grant# 2022–3–042).
