Abstract
Congenital first branchial cleft anomalies (CFBCAs) are rare cervical branchiogenic conditions, occurring at an estimated incidence of approximately 1 in 1,000,000. They account for 1%-8% of all congenital branchial cleft abnormalities. 1 Patients with CFBCAs always exhibit the following 3 syndromes: masses in Pochet’s triangle area (up to the bottom wall of the ear canal and down to the upper margin of the hyoid bone, forward to the submental region and backward to the anterior margin of the sternocleidomastoid); recurrent episodes of swelling, pain, and pus formation; and otological symptoms (ear canal overflow, hearing loss, etc). 2 Complete resection of the lesion in the quiescent stage of inflammation is the only effective means to cure this disease. According to the origin of the germ layers, CFBCAs can be divided into Work type I and Work type II. 3 Work type I lesions were cystic masses in the postauricular area, extending medially and anteriorly along the external auditory canal. Work type II lesion was located from the earlobe to the mandible angle, the lesion arised from both ectodermal and mesodermal, and might present as a fistula (Figure 1(a)) or cyst. Sinus tractography is a method of showing the alignment of the lesion (Figure 1(b)). The distinction between these subtypes is closely associated with the dissection of the facial nerve. 4 In the current study, we retrospectively analyzed the treatment outcomes of 35 cases of type II CFBCAs. Our analysis focused on evaluating the anatomical proximity between the lesions and the facial nerve, aiming to provide valuable insights for preoperative prediction and prevention of facial nerve injury.
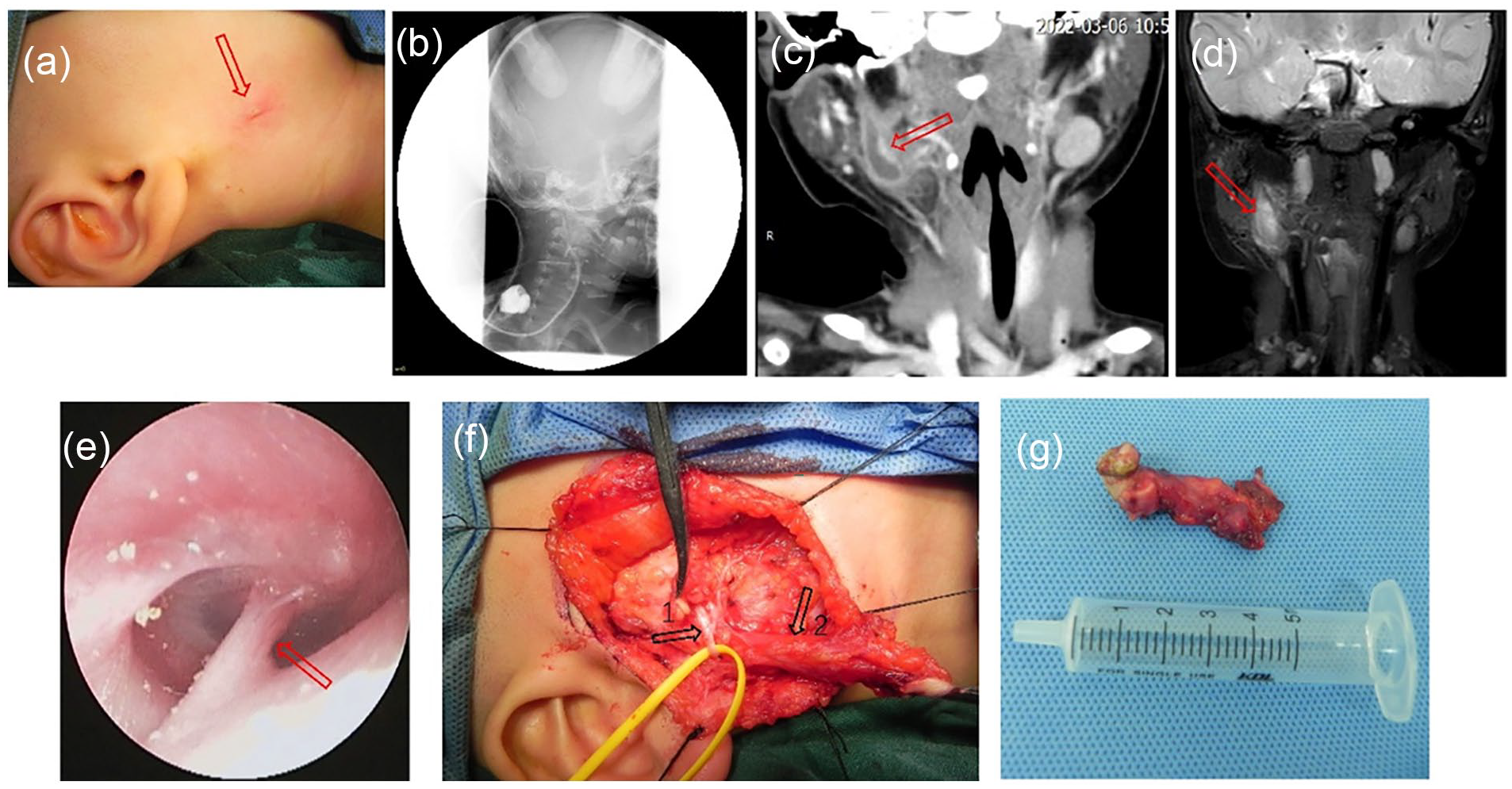
(a) Recurrent redness, swelling, and purulence in the right mandibular angle (red arrow) in a 1.5-year-old female with type II congenital first branchial cleft anomalies. (b) Positive fistula test. (c) Computed tomography (CT) scan revealed a cystic lesion lay in the right parotid gland with a tract extending to the inferior wall of external auditory canal (red arrow). (d) Magnetic resonance imaging (MRI) indicates the lesion which is characterized by high signal on T2WI (red arrow). (e) A tympanic membranous attachment (red arrow)was found by auricular endoscopy. (f) The lesion (arrow 2) ran deep to the facial nerve (arrow 1) and ended in the external auditory canal (EAC) wall. (g) The excised lesion was approximately 4.5 cm long.
Materials and Methods
Clinical Data
A total of 35 children with Work type II of CFBCAs, who had complete clinical data and were treated at the Department of Otolaryngology–Head and Neck Surgery at Shenzhen Children’s Hospital from August 2017 to March 2023, were examined in this study. The inclusion criteria were as follows: a diagnosis of CFBCAs based on medical history, physical examination, auxiliary examination, and pathological confirmation of the presence of ectodermal and mesodermal tissues in the lesions (eg, the lesions were surrounded by cartilage or attached to skin appendages). The cohort included 12 boys and 23 girls with ages ranging from 1 to 12 years at the time of diagnosis and disease durations ranging from 1 to 4 years. Among the cases, 20 had left lesions, 15 had right lesions, and there were 32 primary cases and 3 recurrent cases (who had previously received excision of branchial cleft cystic fistulae). All children presented masses in the Pochet’s triangle area, recurrent swelling, pain and pus, and otological symptoms. 2 Olsen’s classification was carried out according to preoperative clinical manifestations and imaging data, 5 which revealed that all cases were of the non-cystic type (sinus type or fistula type).
Methods
Specialized examination: The lower margin of the lesion (the blind end of the sinus and external fistula) was above the mandibular angle in 27 (77.1%) children and below the mandibular angle in 8 (22.9%) children (Figure 1(a)). Otoendoscopy showed tympanic membrane attachment in 9 (24.3%) children (Figure 1(e)).
Auxiliary examination: All 35 children received otoendoscopy and imaging examinations (B-ultrasound, CT, and Magnetic resonance imaging) before surgery. They all showed abnormal cystic or tubular shadows that were closely related to the external auditory canal (Figure 1(c) and 1(d)). Enhanced scanning revealed an enhanced wall of the cystic duct and non-cystic lesions connected with the skin.
Treatment principle: General anesthesia was performed in all cases during the quiescent stage of inflammation for the excision of the congenital first branchial cleft lesion, partial parotidectomy, and facial nerve dissection. A “Y”-shaped incision was designed around the earlobe for children with lesions or external fistulae in the retroauricular sulcus, and an “S”-shaped incision was used for children with lesions in the parotid region or external fistulae near the mandibular angle. Then the external fistulae, local scars, or granulation tissues were placed into the incision. 6 Subsequently, the platysma myocutaneous flap on the neck and face was overturned. In cases of primary treatment for children, dissection was performed on the superficial surface of the middle and upper segment of the sternocleidomastoid, toward the earlobe, while ensuring the identification and preservation of posterior auricular and lobular branches of the great auricular nerve. The facial nerve trunk and branches were dissected, identified, and protected using the anterograde anatomy of the facial nerve, and the lesions, scars, granulation tissues, and some involved parotid gland tissues were resected en bloc (Figure 1(f) and (g)). Three children had a recurrence and were treated with facial nerve monitoring during the whole surgical procedure. For those with lesions that were closely related to the external auditory canal, partial cartilage and skin of the external auditory canal adjacent to the lesions were resected simultaneously, and the external auditory canal was reconstructed, followed by local packing with iodoform gauze. Finally, the parotid gland was sutured intermittently to prevent saliva leakage.
Negative pressure drainage was routinely performed after surgery. When the drainage volume was <10 ml, the drainage tube was withdrawn. Subsequently, the children were asked to wear a parotid gland sheath for compression dressing for 7 days. Before surgery, children with local chronic infection were treated with sensitive antibiotics or empirical broad-spectrum antibiotics for 7 days, according to the results of drug sensitivity tests. The suture was removed 7 days after surgery, and the iodoform gauze was removed from the ear canal 14 days after surgery.
Results
Correlations Between Clinical Characteristics and Facial Nerve Location in Work Type II CFBCAs
All 35 children were operated on successfully. During the surgery, the lesions were found to be in the superficial facial nerve in 12 (34.3%) cases, between branches in 5 (14.3%) cases, and in the deep facial nerve in 18 (51.4%) cases. In female patients in whom the lower margin of the lesions was below the mandibular angle and had Work type II CFBCAs accompanied by tympanic membrane attachment, the lesions were more likely to be in the deep facial nerve or between branches compared with male patients who exhibited the lower margin of the lesions above the mandibular angle and tympanic membrane attachment. These differences were statistically significant (P < .05) (Table 1).
The Relationship Between Clinical Features of the Type II CFBCAs and the FN Location.
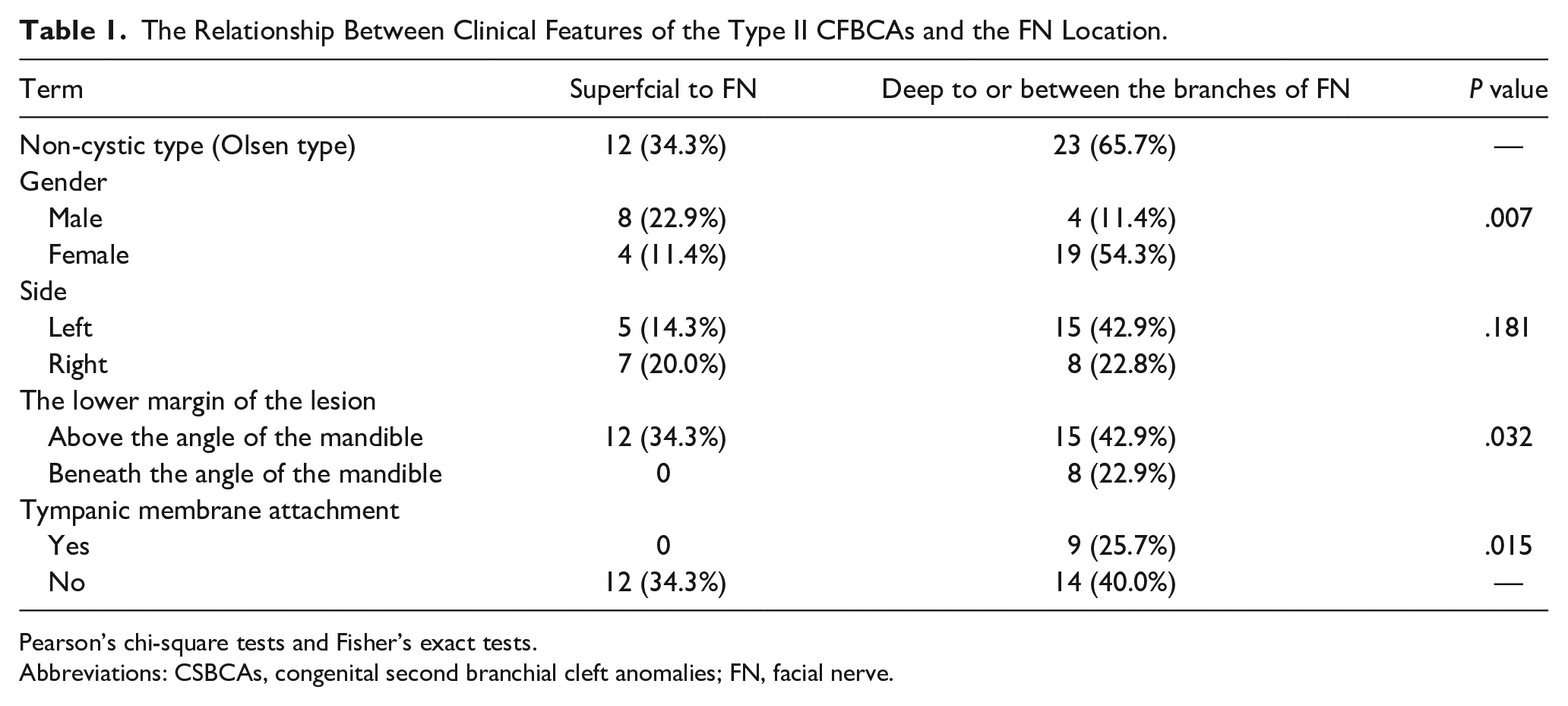
Pearson’s chi-square tests and Fisher’s exact tests.
Abbreviations: CSBCAs, congenital second branchial cleft anomalies; FN, facial nerve.
Postoperative Pathology
All lesions exhibited cystic or luminal changes. The cyst wall was lined with stratified squamous epithelium, lymphoid tissue, and lymphoid follicles. Hair follicles, sebaceous glands, sweat glands, or cartilages (surrounded by the duct wall) were observed under the epithelium.
Postoperative Complications and Follow-Up
In total, 33 (94.3%) children presented with stage I wound healing, and 2 (5.7%) children experienced a postoperative salivary fistula, which resolved after 2 weeks of active dressing changes and pressure bandaging. Three children had an asymmetric corner of the mouth, which was recovered within 6 months, and 1 child suffered from ear canal stenosis. No postoperative bleeding or permanent facial paralysis occurred. The children were followed up for 0.5-5 years, and no recurrences were observed.
Discussion
The histogenesis, classification, and nomenclature of Branchial Cleft Anomalies (BCAs) remain a topic of great controversy. Many theories have been proposed to explain the formation of these anomalies. Based on the proposed origin, they can be divided into 2 main categories—congenital and lymph node theories. The former includes the Branchial Apparatus Theory, Precervical Sinus Theory, and Thymopharyngeal Theory. While the latter comprises Inclusion Theory. In recent years, Pupic-Bakrac proposed the Hybrid Branchial Inclusion Theory in their study, which posits that the fusion of branchial and lymph node tissues serves as a mechanism for developing BCAs. 7 They showed that the classification system based on the Hybrid Branchial Inclusion Theory is more effective than previous systems in determining the pathoanatomical extent of BCA. 8 Among the congenital theories, the Branchial Apparatus Theory is the most widely accepted. According to this theory, CFBCAs result from the incomplete closure of the ectoderm of the first branchial groove during the 6th to 8th week of gestation. The degree of incomplete closure results in different types of lesions. 6 The muscles involved in facial expression and the parotid glands that are innervated by the facial nerve are derived from the second branchial arch. So the developmental stage of the first branchial cleft aligns with that of the parotid gland and facial nerve during embryogenesis. The interlaced development of different branchial apparatuses leads to the close and variable anatomical relationship between CFBCAs and parotid glands and the extracranial segment of the facial nerve from adjacent branchial arches. 9 Embryological patterns suggest that the lesions could potentially be located superficially, deeply, or internally within the branches of the facial nerve.10,11 This poses an extremely high risk of facial nerve injury during surgery to treat this condition. Reports indicate that the incidence of permanent peripheral facial paralysis after BCA surgery ranges from 5% to 12%, while the incidence of temporary facial paralysis can be as high as 21% to 66.7%.12,13 These findings underscore the importance of understanding the relationship between BCAs and the facial nerve before surgery.
Work categorized CFBCAs into 2 distinct types, namely type I and type II, based on the origin of the lesion during embryonic development. 3 Type I anomalies, originating from ectodermal tissue and containing only squamous epithelium, result from the duplication of the membranous part of the external auditory canal. Work type I lesions mainly occur in adults and predominantly manifest as cystic formations, characterized by their extension from the inner side reaching the middle ear, while the outer side reaches the periauricular region or cavum conchae.14,15 Type II anomalies arise from ectoderm and mesoderm structures, characterized histologically by the presence of squamous epithelium, skin appendages, and cartilage. Type II lesions mainly occur in childhood and primarily present as sinus or fistula formations. 14 These lesions typically originate from the bottom wall of the external auditory canal and follow a course that extends forward and downward through the parotid parenchyma or the superficial surface of the masseter fascia. Ultimately, they terminate in the Pochet’s triangle area of the hyoid bone on the same side.4,11
While it has been observed that certain instances of Work type I exhibit a substantial association with the facial nerve, 16 a significant body of research indicates that facial nerve involvement is typically located deep within type II CFBCAs, suggesting no direct correlation with type I.16-19 This notion is further supported by statistical evidence. For the positional relationship between Work type II CFBCAs lesions and the facial nerve, Ertas et al. 11 have reported that only 28.6% (2/7) of Work type II lesions were on the deep surface or between the branches of the facial nerve, which is far lower than the 88.9% (8/9) reported in another review. 20 The observed disparity between the 2 groups could potentially be attributed to the limited sample size of the former group and the lack of clear classification in the latter group. In our current study, a cohort of 35 children diagnosed with Work type II CFBCAs was examined. Among these cases, 12 lesions (34.3%) were identified to be located in the superficial facial nerve, while the majority, 65.7% (23 out of 35), were found on the deep surface or between the branches of the facial nerve. Notably, the prevalence of lesions in the latter location was significantly higher than that in the former. Analyzing the positional relationship between CFBCAs lesions and the facial nerve can be intricate, particularly in the context of Work type II CFBCAs lesions. After data analysis, D’Souza et al. 13 suggested that in young females with non-cystic CFBCAs with an external opening in Pochet’s triangle area, especially those aged less than 6 months and presenting symptoms, the lesions tended to be in the deep facial nerve. The results of other studies showed that lower right non-cystic CFBCAs lesions in young patients were more likely to be in the deep facial nerve. 9 According to the analysis of Work type II CFBCAs cases performed in our study, in cases where the lower margin of the lesions was below the mandibular angle (P = .032) and in female patients (P = .007), the lesions tended to be on the deep surface or between the branches of the facial nerve, which is consistent with the reports of D’Souza et al. 13 and Trig1ia et al., 12 respectively. In addition, our study found that all cases of Work type II CFBCAs were of the non-cystic type, and lesions tended to be on the deep surface or between the branches of the facial nerve (65.7%), which are in line with the conclusion of Work. 3 Tympanic membranous attachment is a fibrous cord-like tissue that remains in the embryo and connects the bottom wall of the external auditory canal to the tympanic membrane. It may be formed in the early embryonic stage and continues into the sinus or fistula. It was deemed an essential otoscopic finding for CFBCAs, particularly among patients without symptoms. 21 Earlier studies have indicated that overlooking this finding could potentially lead to a delayed diagnosis.6,21,22 In our study, all patients with tympanic membrane retraction showed lesions located between or below the branches of the facial nerve. This observation holds statistical significance(P = .015). In conjunction with the existing body of literature, our study findings have shed light on several key factors that can serve as crucial indicators for predicting the positional relationship between Work type II CFBCAs lesions and the facial nerve before surgery. These factors encompass gender, the correlation between the lower border of the lesion and the mandibular angle, the non-cystic type as defined by Olsen, and the attachment of the lesion to the tympanic membrane. Collectively, these indices hold significant value in preoperative prognostication.
Based on the prediction of the adjacent relationship between CFBCAs lesions and the facial nerve, the control of operation timing is deemed crucial. First, the facial nerve undergoes full development during the embryonic stage and reaches maturity at 1 year of age. Once mature, it becomes resilient to intraoperative stimulation and minor stretching. 23 Guo et al. proposed that conducting surgery on CFBCAs beyond 4 years of age reduces the risk of damaging the facial nerve. 20 Second, inflammatory granulation tissue and interstitial edema during infection can both hinder intraoperative dissection and neuroprotection, as well as increase surgical difficulty and the recurrence rate. A pathological examination has substantiated that a minimum duration of 4 weeks is required for the complete transition of inflammatory tissue into discernible scar tissue, facilitating surgical intervention. 24 Third, the likelihood of facial paralysis during the surgical removal of CFBCAs intensifies if a patient has a history of recurrent infections and insufficient treatment (such as incision, drainage, or incomplete excision), as these factors may lead to adhesions between the tract and the facial nerve.18,25 Consequently, considering these factors and our experience, it is crucial to conduct surgery during the noninfected phase, preferably after the age of 1 year or following 4 weeks of effectively managing acute inflammation.
The complex anatomical relationship between Work type II CFBCAs lesions and the facial nerve determines that the approach of facial nerve dissection should be different from conventional parotid gland surgery. In our present study, all 35 children diagnosed with Work type II CFBCAs underwent anterograde dissection of the facial nerve. Surgeons must acquire proficient skills and employ a flexible approach to facial nerve dissection. In cases of recurrent CFBCAs, particularly those classified as Work type II, the utilization of facial nerve monitoring can prove invaluable. 17 This monitoring technique aids surgeons in swiftly identifying and dissecting the facial nerve, which can be challenging to differentiate from inflammatory granulation and scar tissue under complex conditions using visual inspection alone. By employing these strategies, the risk of iatrogenic injury can be reduced.
Conclusion
In our study, the lesion of Work type II CFBCAs in females, with a lower edge of the lesion located below the angle of the mandible, a non-cystic type of Olsen, and the presence of tympanic membranous attachment, tends to be located deep to the facial nerve or between its branches. By taking these factors into account before surgery, it becomes possible to predict the positional relationship between the lesions and the facial nerve, determine the optimal timing for surgical intervention, employ delicate anatomical skills specific to both ear surgery and parotid surgery, and utilize facial nerve monitoring when necessary. By incorporating these measures, the lesions can be effectively treated while simultaneously preserving the function of the facial nerve.
Footnotes
Acknowledgements
Not applicable.
Authors’ Contributions
H-C.W, H-W.Y, C-Q.Z, and Y-S.T prepared the data and wrote the main text. All the authors contributed the idea of the research and reviewed the article.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author (Y-S.T,
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Guangdong High-level Hospital Construction Fund Clinical Research Project of Shenzhen Children’s Hospital (LCYJ2022063).
Ethics Approval and Consent to Participate
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institution. Due to all participants included in the study are under 18 years, informed consent was obtained from all individual participants’ parents and/or legal guardian. As a retrospective study, The Ethics and Safety Committee of our hospital has confirmed that no ethical approval is required.
Consent for Publication
Written Informed consent for publication was obtained from the patient’s parents for the use of their medical records and exam images in writing this study.
