Abstract
This case report illustrates a unique presentation of Lemierre’s syndrome precipitated by Fusobacterium necrophorum. This case report describes a 20-year-old patient who developed Lemierre’s syndrome secondary to a shoulder hematoma and neck abscess with multiple systemic complications in the absence of tonsillitis or oropharyngeal infection. Two weeks prior to presentation, the patient sustained a right shoulder injury and contracted COVID-19. Due to his Lemierre’s syndrome, he developed right internal jugular vein and subclavian vein thrombosis, septic lung emboli, right sided Horner’s syndrome, disseminated intravascular coagulation, pelvic collection, septic arthritis of pubic symphysis and osteomyelitis of the right pubic bone, and proximal left femoral shaft. The patient received non-operative and operative management to manage his Lemierre’s syndrome including surgical drainage, antibiotics, and anticoagulation; he was discharged following an extended hospital stay. This case report highlights a rare presentation of Lemierre’s syndrome secondary to a shoulder hematoma in a COVID-19 positive patient, and its potential systemic and life-threatening complications. Its importance is highly relevant in the context of the COVID-19 pandemic. Further studies are warranted to explore the effect of preceding COVID-19 infections on the microbiological profile in Lemierre’s syndrome.
Introduction
Lemierre’s syndrome is a rare infection typically triggered by the gram-negative anaerobe Fusobacterium necrophorum. Within this entity, neck abscess’ commonly form after oropharyngeal infections such as tonsillitis.1,2 This syndrome can lead to various complications including septic lung emboli, pulmonary abscess, septic arthritis, osteomyelitis, and disseminated intravascular coagulation (DIC) and has a 5% risk of mortality. 2
We present the case of a 20-year-old man who was managed by the otolaryngology team for Lemierre’s syndrome secondary to Fusobacterium necrophorum with multiple complications; this developed following a shoulder hematoma without signs of tonsillitis or oropharyngeal infection. Due to his Lemierre’s syndrome, he also had associated right internal jugular vein (IJV) and subclavian vein thrombosis, septic lung emboli, right-sided Horner’s syndrome, DIC, pelvic collection, septic arthritis of the pubic symphysis, osteomyelitis of the right pubic bone body, and proximal left femoral shaft. Patient was discharged following an extended stay at the hospital.
Case Presentation
A 20-year-old man, previously fit and well, presented with a 1 week history of confusion, fever, diarrhea, drooping right eyelid, right shoulder, and neck pain. He denied any sore throat, odynophagia, dysphagia, and dysphonia at the time of presentation. The patient was diagnosed with COVID-19 15 days prior to presentation and was treated with COVID-19-related sepsis on admission. Two weeks prior to presentation, the patient sustained a right shoulder injury during rugby causing right shoulder pain and limited movement. He had no significant past medical or social history including no diabetes mellitus or intravenous drug use.
On assessment the patient was delirious but demonstrated unremarkable cardiorespiratory and abdominal examinations with soft and non-tender calves. Oropharynx revealed no signs of tonsilitis or tonsillar exudate. Neck examination unveiled a right-sided tender neck swelling with some overlying erythema. There was right eye ptosis, enophthalmos, with an unreactive right pupil 2 mm in size while the left pupil was reactive to light at 4 mm. Hence, a diagnosis of Horner’s syndrome was established.
Serology demonstrated hemoglobin of 120 g/L, C-reactive protein of 222 mg/L, white cell count of 13.4 × 109/L with neutrophilia (12 × 109/L), and lymphopenia (0.7 × 109/L). There was thrombocytopenia at 40 × 109/L which promptly decreased to 24 × 109/L necessitating 1 pool of platelet transfusion. Prothrombin–time ratio was raised at 1.33, and the prothrombin time was 18.6 s. He had normal Activated Partial Throm-boplastin Time (APTT), fibrinogen, and APTT ratios. Blood film exhibited neutrophilia with left shift, thrombocytopenia with giant platelets, no red cell fragments, and some target cells. The conglomeration of thrombocytopenia, raised D-dimer at 4536 µg/l, and prolonged prothrombin–time ratio diagnosed the patient with overt DIC in accordance with the International Society of Thrombosis and Haemostasis criteria. 3 Liver function tests showed elevated total bilirubin (30 µmol/L) and conjugated bilirubin (19 µmol/L). The patient demonstrated deranged renal function with elevated creatinine (158 µmol/L), reduced eGFR (53 ml/min/1.73 m2), and uremia (13.6 mmol/L) in addition to hyponatremia (125 mmol/L).
Blood cultures grew Fusobacterium necrophorum 12 h after presentation sensitive to metronidazole. He was negative for HIV and hepatitis A, B, and C.
Computed tomography (CT) scan of the neck (Figure 1) on admission illustrated a 10 cm by 7 cm right-sided deep cervical abscess within the parapharyngeal space with extension into the perivertebral space. There was direct involvement of the right IJV with thrombophlebitis and an acute thrombus extending from the origin to the intracranial portion of the right IJV. Pulmonary septic emboli were also noted.
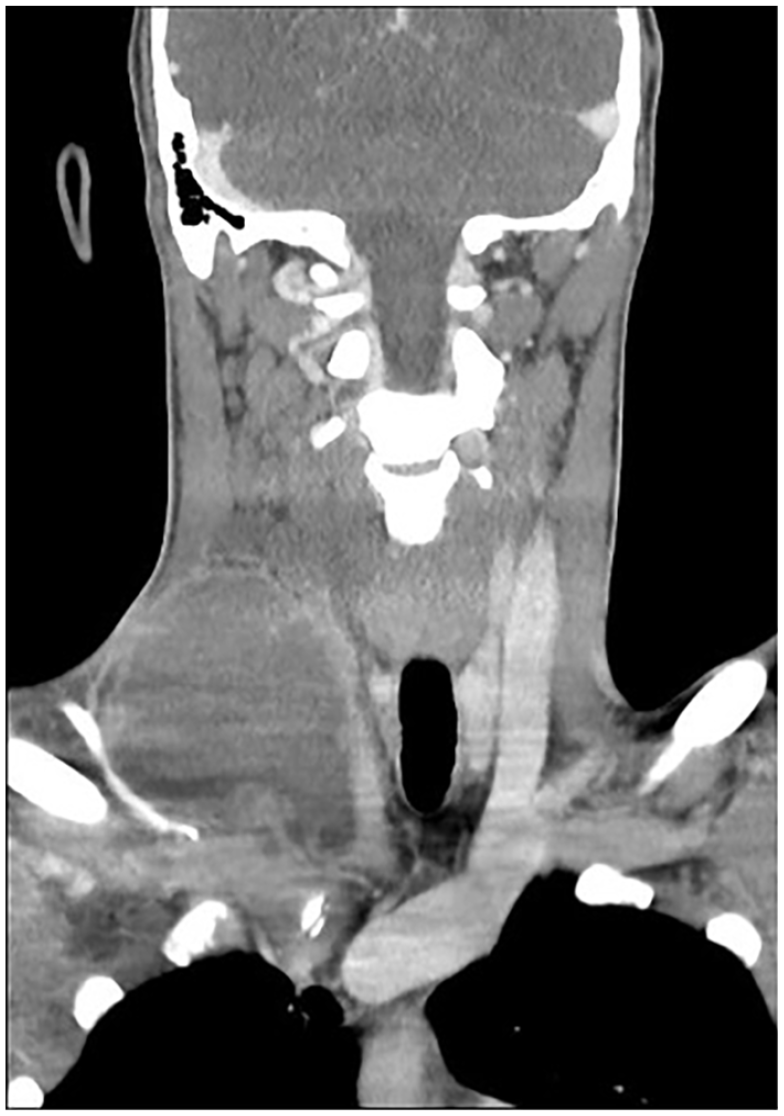
Coronal section of CT neck demonstrating right neck abscess measuring 10 × 7 cm with thrombosis of right internal jugular vein and right subclavian vein.
The patient underwent surgical drainage of the neck abscess; intra-operatively a supraclavicular shoulder hematoma was discovered, in addition to an abscess extending to the prevertebral, parapharyngeal, and carotid sheath spaces. Over 100 ml of frank pus was drained which later grew the anaerobe Fusobacterium necrophorum. Hence, the patient was started on Piperacillin-Tazobactam, Metronidazole, and treatment dose of Dalteparin. He was taken to the Intensive Care Unit for inotropic support post-operatively.
Due to the patient’s delirium and persistent right-sided Horner’s syndrome 24-48 h post-operatively, neuroimaging was performed after seeking advice from the neurology team. His CT head was unremarkable and magnetic resonance imaging (MRI) head with contrast illustrated nonspecific intracerebral white matter lesions.
One week after drainage of the neck abscess, the patient clinically deteriorated after switching Piperacillin-Tazobactam to Co-amoxiclav, while continuing metronidazole. To investigate further, a CT neck, thorax, abdomen, and pelvis was performed which showed an abscess within the right hemipelvis, closely associated with the right pubic bone and pubic symphysis. Further imaging with an MRI pelvis demonstrated septic arthritis of the pubic symphysis with adjacent deep soft tissue abscess and surrounding cellulitis, associated with reactive marrow edema (Figure 2a). MRI femur (Figure 2b) and X-ray of femur confirmed osteomyelitis of the posterior left femoral cortex measuring 5 cm by 1 cm. The morphological appearance of this was suggestive of a septic bone infarct. The pelvic collection was drained by interventional radiology who inserted a pelvic drain. Culture from this pelvic collection showed an elevated white cell count but no bacterial growth. To exclude possible sources of infection, a transthoracic echocardiogram was conducted demonstrating no obvious mass or vegetations on the heart valves. He was treated with intravenous Benzylpenicillin and Metronidazole for his osteomyelitis, and this was later changed to oral Clindamycin and Metronidazole on discharge.
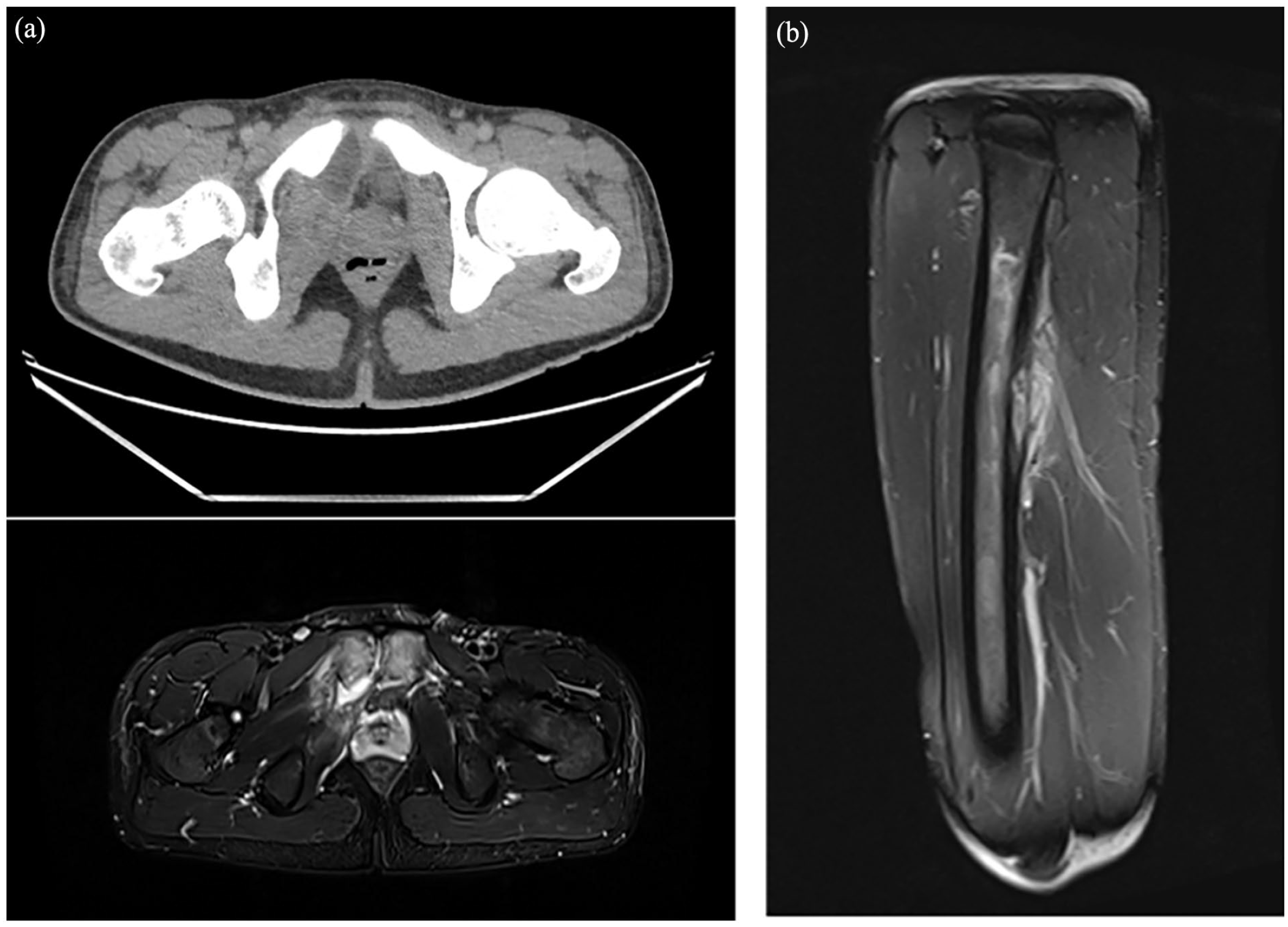
Figure 2(a) (left): Axial view of left pubic symphysis osteomyelitis and right pelvic abscess in CT (top) and MRI T2 sequence (bottom). (b) (right): Sagittal view of left femur osteomyelitis in MRI T2 sequence.
Follow-up was arranged post-discharge with otolaryngology, hematology, and vascular surgery teams; repeat ultrasound neck confirmed resolution of right IJV thrombosis after 6 months of anticoagulation.
In summary, this patient had multiple complications secondary to Lemierre’s syndrome.
Discussion
Our case of Lemierre’s syndrome involved a young healthy male with a preceding traumatic sports injury leading to a prolonged hospital admission. Complicated by COVID-19, the patient’s unusual presentation of delirium and diarrhea in the absence of sore throat made the diagnosis of Lemierre’s more clinically challenging. Traditionally, the basic elements forming the diagnosis of Lemierre’s syndrome include pharyngeal infection, septic emboli, and positive growth on blood cultures with or without IJV thrombosis. 4 Over the past few decades, variations in the presentation of Lemierre’s syndrome have surfaced since Courmont’s first description in 1900. 5 Lemierre’s syndrome has been described to arise from various foci including odontogenic, mastoid, nasopharyngeal and sinus infections, tonsillar and peritonsillar infections, gastrointestinal tract pathology, otological infections, and those with endometrial pathology.1,6 There is a spectrum of ages that Lemierre’s syndrome has been reported in, from infants below 1 year of age to older patients in their sixth decade.7,8
Our case report is the first to demonstrate a suprascapular shoulder hematoma instigating Lemierre’s syndrome in the presence of COVID-19. Our case highlights the need for clinicians to associate preceding orthopedic related injuries to the other diagnostic criteria of Lemierre’s syndrome and the need for a multidisciplinary approach inviting appropriate specialties to offer opinions with the specific clinical issues at hand. Other unusual causes have been documented including a sternocleidomastoid hematoma leading to Lemierre’s syndrome with Staphylococcus aureus as the causative organism 9 ; viruses including influenza A and Epstein–Barr virus have also been associated with Lemierre’s syndrome. 10
Our case implicated Fusobacterium necrophorum, an anaerobe which initially responded to metronidazole alone. However, as the patient’s clinical status deteriorated, the need for additional antibiotic coverage and escalation became apparent. The resistance of fusobacterium to anti-anaerobic cover and its possession of beta-lactamases often necessitates change in the choice of antibiotic.11-13 It is this recognition and understanding of antibiotic resistance that may explain the improved survival outcomes in cases of Lemierre’s syndrome since it was first described in the early 20th century. 1
The decision to utilize anticoagulation in the management of thromboembolic events in Lemierre’s syndrome remains a subject of debate. In our case, the patient received treatment dose low molecular weight heparin on the basis that dissolving infected thrombi would permit enhanced bacterial exposure to incoming antibiotics; other published cases have taken similar approaches in their management.14,15 However, one may question whether dissolving septic thromboemboli may lead to a septic shower thus temporarily resulting in acute clinical deterioration 16 ; hence, benefit and risks of anticoagulation must be evaluated in different patients groups.
Further studies are warranted to explore the influence of preceding COVID-19 infections on the microbiological profile in Lemierre’s syndrome, and whether COVID-19 is an independent prognostic factor in those diagnosed with Lemierre’s syndrome.
Conclusion
This rare case report features a COVID-19 positive 20-year-old male with Lemierre’s syndrome secondary to a shoulder hematoma. The patient was confronted with multiple systemic and life-threatening complications secondary to Lemierre’s syndrome. The patient’s treatment comprised of surgical drainage of shoulder hematoma and neck abscess, therapeutic anticoagulation, and systemic antibiotic therapy.
Footnotes
Acknowledgements
Authors give credit to all those involved in the care of the patient at Royal Surrey County Hospital, UK.
Contributorship Statement
Shivanchan Rajmohan and Chuanyu Gao contributed equally to the concept, data acquisition, interpretation, and writing the manuscript and are joint first authors. MG, IM, and LP involved in data acquisition and editing of manuscript. All authors agreed for the article’s submission.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
Patient was treated in accordance with ethical standards in the Helsinki Declaration (2013).
Statement of Informed Consent
Written informed consent was obtained from the patient for their anonymized information and images to be published in this article.
