Abstract
Keywords
Introduction
Dysphagia is common in idiopathic Parkinson’s disease (IPD), with a pooled prevalence of 82%. 1 Dysphagia has detrimental effects on quality of life, 2 and swallowing dysfunction contributes to the development of pneumonia—a leading cause of death in people with IPD. 3 Swallowing dysfunction in people with IPD is characterized in part by impairments in both swallowing safety (penetration and aspiration) and swallowing efficiency (pharyngeal residue).4,5
Aspiration, or the entry of swallowed material into the subglottic space and lungs, is common in people with IPD 5 and increases the risk of aspiration pneumonia.6,7 Current research demonstrates strong associations between aspiration and demographic findings 8 such as age and gender, specific patterns of swallow dysfunction such as delayed initiation of the pharyngeal swallow and reduced hyolaryngeal excursion, 9 pharyngeal residue, and patient-reported aspiration sym-ptoms. 8
Pharyngeal residue is an important clinical feature of swallowing dysfunction in IPD as it is associated with malnutrition.4,10 The causes of pharyngeal residue in IPD remain to be elucidated. Some relationships have been identified between tongue-driving force and vallecular residue, and between pharyngeal shortening and constriction and pyriform residue.11–13 While an understanding of the physiologic causes of pharyngeal residue is important for swallowing rehabilitation treatment planning, more research is needed to identify demographic factors (e.g., age, cognitive status) and clinical factors (e.g., disease duration and disease severity) associated with post-swallow residue. Identifying demographic and clinical factors associated with pharyngeal residue could help screen for those at risk of swallowing efficiency impairments in need of comprehensive instrumental swallowing assessments and swallowing rehabilitation.
Flexible endoscopic evaluation of swallowing (FEES) is a sensitive instrumental swallowing assessment tool, well-established in the identification of impairments in swallowing safety and efficiency. 14 Traditional methods for rating residue during FEES categorize residue into 3-7 severity groups, depending on the scale used.15-19 However, categorical methods for rating pharyngeal residue appear less valid, reliable, and sensitive than continuous-based numerical rating methods.20-22 To address this limitation, the Visual Analysis of Swallowing Efficiency and Safety (VASES) was recently developed and validated by Curtis et al in 2022. VASES allows us to rate the pharyngeal residue, penetration, and aspiration during FEES.23,24 VASES uses transparent and standardized anatomic and temporal boundaries, along with detailed “secondary rules” and 100-point percentage-based rating scales, to rate the Penetration-Aspiration Scale (PAS) and to rate the amount of residue filling or covering six anatomic landmarks (oropharynx, hypopharynx, epiglottis, laryngeal vestibule, vocal folds, and subglottis).
The aim of this study was to build upon recent advancements in FEES interpretation to (1) characterize residue patterns in people with IPD and (2) identify clinical and non-residue endoscopic findings that significantly relate to residue in people with IPD during FEES using VASES.
Materials and Methods
This was a prospective single-blinded cross-sectional cohort study conducted at Istanbul University-Istanbul Faculty of Medicine (IU-IFM), Department of Oto-laryngology and Head & Neck Surgery between March and May 2020. The study was approved by the IU-IFM Clinical Research Ethics Committee (Approval Number 2020/413). All methods followed the principles of the Declaration of Helsinki. Written informed consent was obtained from each patient.
Patients
In all, 60 consecutive patients with a diagnosis of IPD over 30 years of age who regularly followed up at the Parkinson’s Disease and Movement Disorders Outpatient Clinic at IU-IFM were included in the study. Exclusion criteria of the study were as follows: diagnosis of secondary or atypical Parkinsonism; history of deep brain stimulation surgery; previous surgery, trauma, or radiation to the neck; diagnosis of other diseases known to cause dysphagia and dementia and strong gag reflex that interferes with the completion of FEES. Patients with a Montreal Cognitive Assessment (MoCA) score of less than 21, which indicates severe cognitive dysfunction, were excluded from the study due to the inability to complete the tasks required for this study. All patients were evaluated during the “on” stage of their PD medication.
Clinical Variables
Clinical variables for this study included patient age, cognitive function, and measures of disease severity. They were collected by a neurologist at the time of the regular follow prior to swallowing evaluation. Patient age was collected from medical records at the time of the visit by a neurologist.
Assessment of cognitive function was completed using a validated Turkish translation of the MoCA. 25
Three measures of disease severity were included in this study: part III of the Unified Parkinson’s Disease Rating Scale (UPDRS-III), the Hoehn & Yahr Stage (HYS), and the Levodopa Equivalency Dose (LED).
Endoscopic Evaluations of Voice and Swallowing
Laryngoscopic examinations of each patient were completed by two otolaryngologists using a Xion GmbH (Berlin, Germany) flexible endoscope unit. No topical anesthetics were used during the examination. Once the scope was advanced into the oropharynx, visualization of the larynx was completed to obtain tidal breathing measurements of the vocal fold Bowing Index (BI). 26 For the FEES, the patients were seated in their usual eating position. A FEES was then completed using water (IDDSI 0), pudding (IDDSI 4), and bread (IDDSI 7). Green food coloring was used to dye all water and puree trials. The protocol included a fixed presentation of the following trials: 10 milliliters (mL) of green water via a 30 mL medicine cup (1 trial); 30 mL one-sip trial of green water via a 30 mL medicine cup (1 trial); 90 mL continuous drinking trial of green water via an 8-ounce cup (1 trial); one tablespoon of green puree via spoon (2 trials); and one bite of bread (2 trials). All foods and liquids were self-administered, non-held, and non-cued (patients were asked to take the boluses and swallow without holding them first, and all trials were performed separately). Second trials with puree and bread and higher volume trials with liquids were not conducted in the presence of major aspiration during the first trials. Laryngoscopic examinations and FEES studies were recorded separately for each patient and de-identified before being uploaded to a hard drive for future blinded evaluation. A fellowship-trained laryngologist (NE) who completed training in VASES with its developers blindly analyzed all residue (VASES 6 residue ratings) and non-residue endoscopic (PAS, premature leakage, and build-up phenomenon) outcome measures asynchronously. There was no time between the first and second analyses for the assessment of intra-rater reliability. Ten of the anonymized recordings (19%) were duplicated and included randomly among videos for rating as if they belonged to a new patient to evaluate intra-rater reliability.
Residue outcome measures
VASES residue ratings out of 100 points provided an estimation of how filled or covered a structure was with residue. Residue ratings were made for the oropharynx, hypopharynx, epiglottis, laryngeal vestibule, vocal folds, and subglottis. Higher scores represent more residue such as scores of 0% would indicate no residue filling or covering an anatomic structure and scores of 100% would indicate that residue was filling or covering an anatomic landmark in its entirety. For this study, the highest residue rating across all swallowing trials including all bolus types for each anatomic landmark was used to capture impairments in safety and efficiency.
Non-residue endoscopic outcome variables
PAS is an 8-point scale used to describe the depth of and reaction to airway invasion. 27 For this study, PAS was dichotomized into two swallowing safety categories: swallows with no airway invasion (PAS 1) and swallows with airway invasion (PAS 2-8). Due to the whiteout during swallowing, a small amount of penetration might sometimes be missed with FEES. For this reason, we distinguished between “airway invasion” to describe PAS > 2 and “no airway invasion” to describe PAS = 1.
Premature leakage of the food before the initiation of swallowing was first described by Langmore et al 28 in patients with frontotemporal lobar dementia, and later used in the IPD population by Pflug et al. 4 In the present study, premature leakage was noted as present during FEES when the rater observed material passing the base of tongue prior to the initiation of the pharyngeal phase of the swallowing and was judged. The initiation of the pharyngeal phase starts with the posterior movement of the base of the tongue and contraction of the pharynx. This was distinguished from residue from prior swallows by having patients drink undyed water to clear any residue from the previous swallow. The presence of premature leakage was noted as absent or present.
The “build-up” phenomenon is described as the accumulation of firm consistencies in the valleculae after repetitive swallowing and has been shown in patients with PD.4,8,29 This was first described by Pflug et al as a special variant of residues. 4 The presence of build-up was also judged as either absent or present.
BI values were analyzed with ImageJ visual analysis software (National Institutes of Health, Bethesda, MD, USA) using a previously described technique30,31 by two otolaryngologists.
Statistics
Statistical analyses were performed with the statistical software package SPSS, version 26 (Armonk, NY). The level of statistical significance was set to an alpha of
Intra-class correlation coefficient (ICC) was used to determine intra-rater reliability. Interpretation of ICC was judged to be “excellent” if ≥0.90, “good” if between 0.75 and 0.90, “moderate” if between 0.50 and 0.75, and “poor” if <0.50. 32
Summary statistics were used to characterize all data, with percentages/frequency distributions used to characterize all nominal and ordinal scaled data, and mean values and standard deviations were used to characterize all continuous data.
A two-step process was used to determine which clinical and endoscopic variables are “significantly associated with” the amount of residue observed within the pharynx, larynx, and subglottis during FEES. First, relationships between the amount of residue across the six anatomic structures and the clinical and non-residue endoscopic variables were established using Mann-Whitney U for all dichotomous predictor variables (PAS, premature leakage, and build-up phenomenon), Kruskal-Wallis tests for all ordinal predictor variables (HYS), and Spearman’s correlations for all continuous predictor variables (age, LED, MoCA, UPDRS-III, and BI). Then, after significant clinical and endoscopic predictor variables were identified, the second step was to include all significant variables within a multiple regression model for each anatomic structure to identify continued significance and relative effective sizes.
The data that support the findings of this study are openly available.
Results
Overall 53 patients completed the study with an average age of 65 ± 12 years. Most of the patients had a Hoehn and Yahr Stage 2 disease (n: 37) with an average UPDRS-III score of 32 ± 18. Out of the 60 patients initially recruited for the study, 5 patients were excluded due to low MoCA scores and 2 due to uncontrollable gag reflexes. Clinical variable and endoscopic evaluation including the residue and non-residue outcome variable findings are summarized in Table 1. ICC ranged from 0.881 (95% CI: 0.526–0.970) to 0.997 (95% CI: 0.987–0.999), indicating “good” to “excellent” agreement across all outcomes.
Demographic Information, Stage of the Disease, and Laryngoscopic and FEES Findings of the Patient Population.
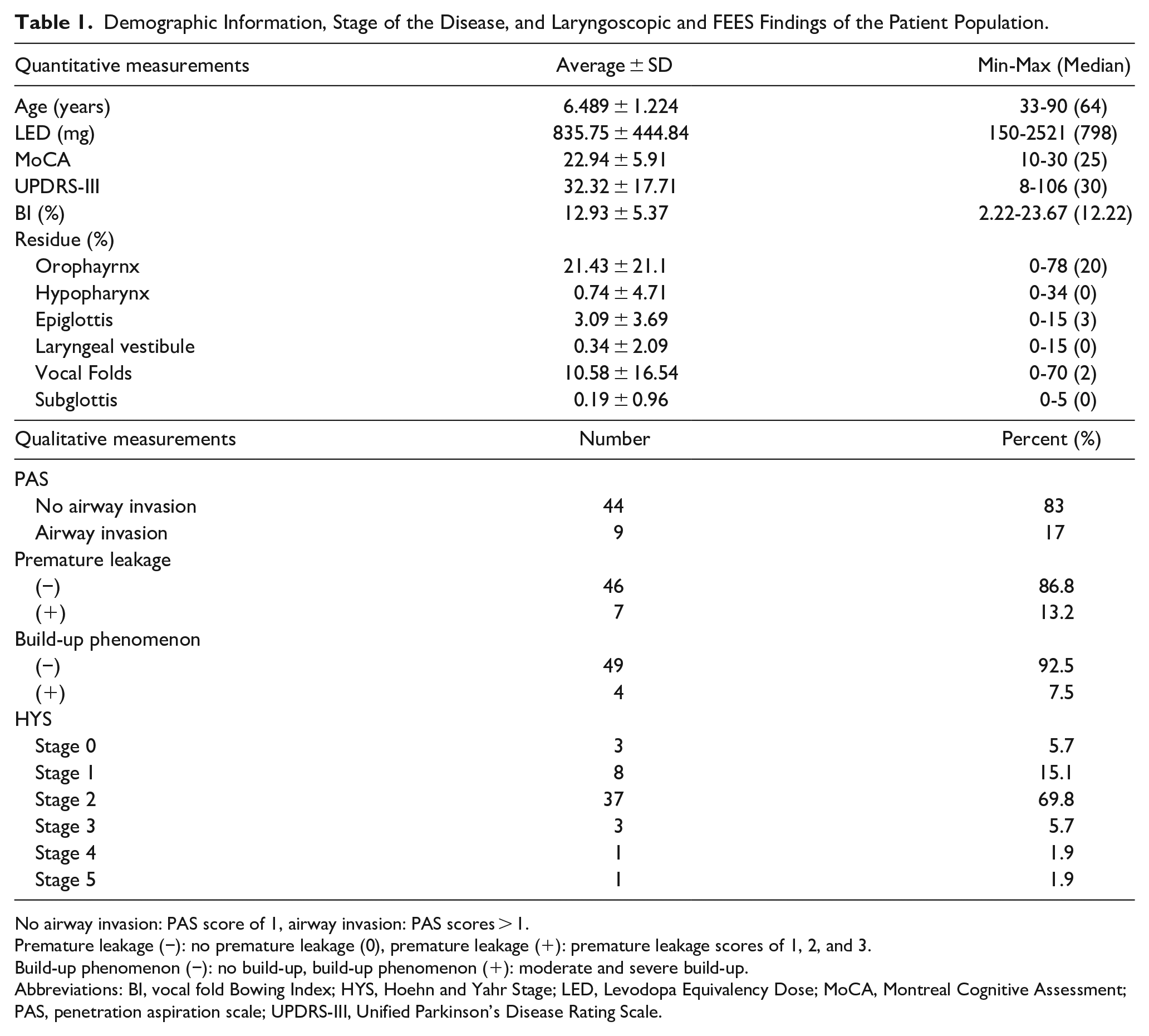
No airway invasion: PAS score of 1, airway invasion: PAS scores > 1.
Premature leakage (−): no premature leakage (0), premature leakage (+): premature leakage scores of 1, 2, and 3.
Build-up phenomenon (−): no build-up, build-up phenomenon (+): moderate and severe build-up.
Abbreviations: BI, vocal fold Bowing Index; HYS, Hoehn and Yahr Stage; LED, Levodopa Equivalency Dose; MoCA, Montreal Cognitive Assessment; PAS, penetration aspiration scale; UPDRS-III, Unified Parkinson’s Disease Rating Scale.
Post hoc power analysis yielded power >0.95 using G*Power. The effect size of the study is 0.15-0.35.
First Step Analyses
Comparison analyses (Table 2)
Residue ratings were significantly higher at all anatomic locations in patients with PAS scores > 1 (
Comparison of Post-Swallowing Residues by PAS Scores, Premature Leakage, and Build-Up Phenomenon.
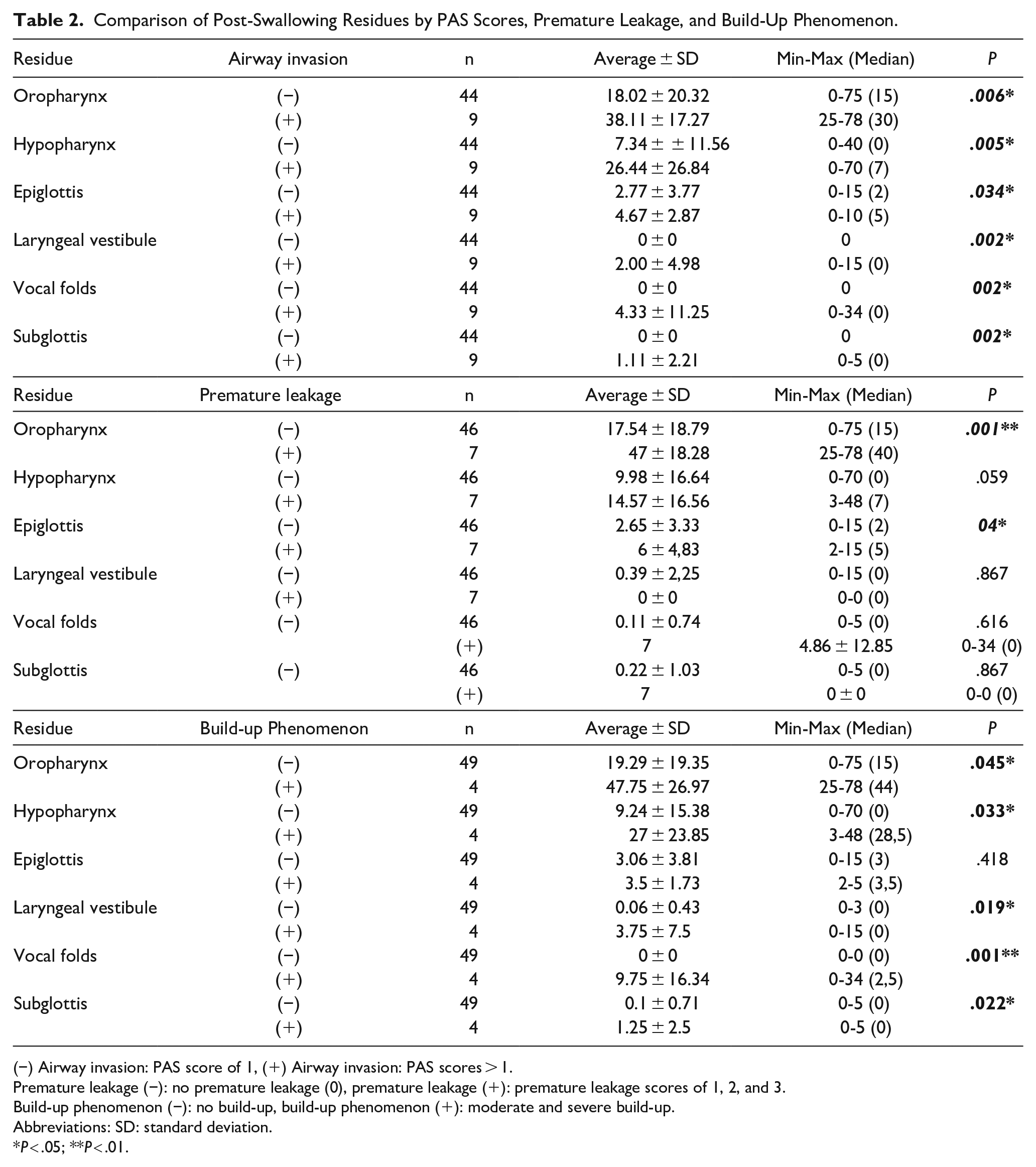
(−) Airway invasion: PAS score of 1, (+) Airway invasion: PAS scores > 1.
Premature leakage (−): no premature leakage (0), premature leakage (+): premature leakage scores of 1, 2, and 3.
Build-up phenomenon (−): no build-up, build-up phenomenon (+): moderate and severe build-up.
Abbreviations: SD: standard deviation.
P < .05; **P < .01.
There was no significant finding for ordinal predictor variables using Kruskal-Wallis.
Correlation analyses (Table 3)
A small positive correlation was observed between BI and residue at the level of the hypopharynx (
Correlation Analyses at the Level of the Hypopharynx.*
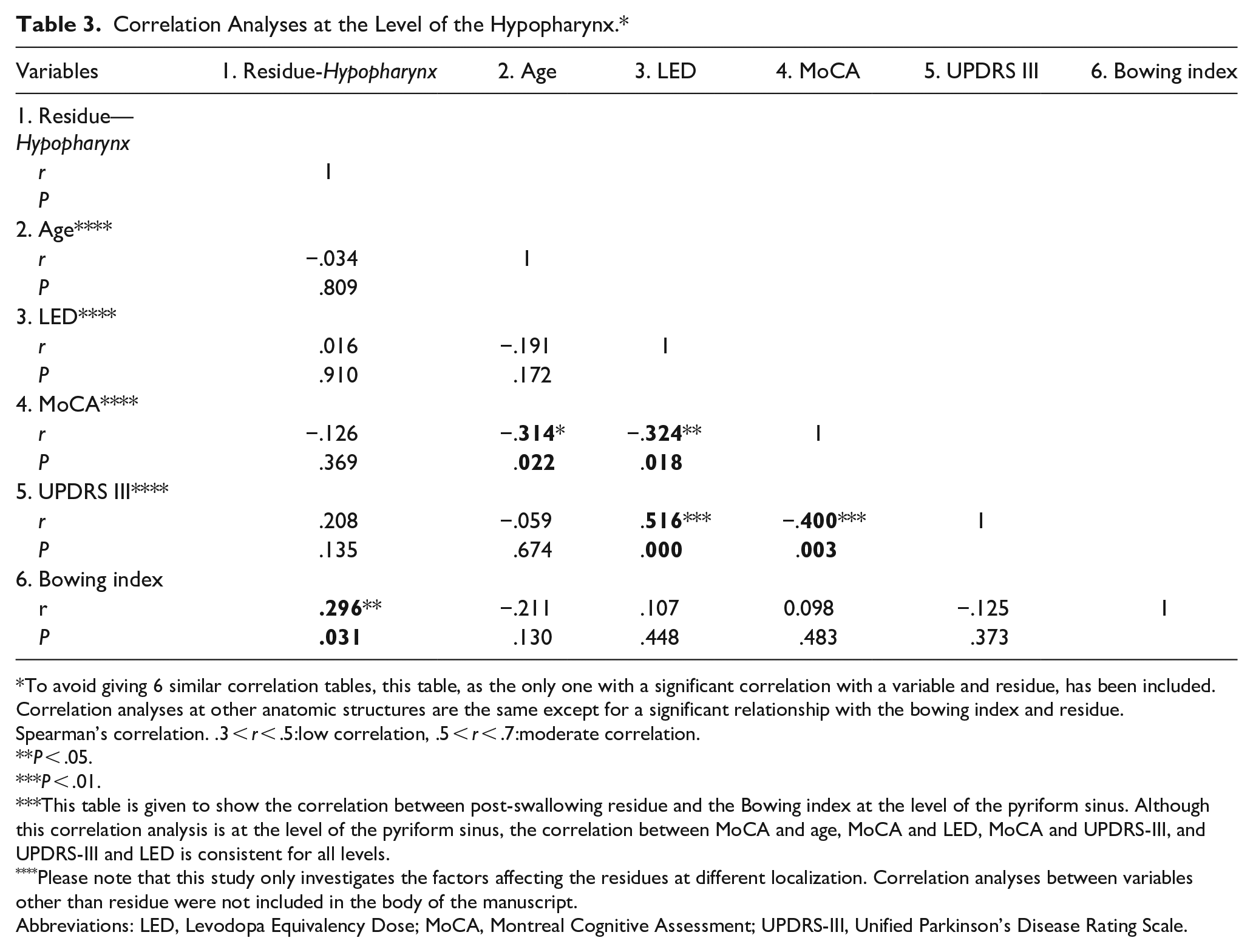
To avoid giving 6 similar correlation tables, this table, as the only one with a significant correlation with a variable and residue, has been included. Correlation analyses at other anatomic structures are the same except for a significant relationship with the bowing index and residue.
Spearman’s correlation. .3 <
This table is given to show the correlation between post-swallowing residue and the Bowing index at the level of the pyriform sinus. Although this correlation analysis is at the level of the pyriform sinus, the correlation between MoCA and age, MoCA and LED, MoCA and UPDRS-III, and UPDRS-III and LED is consistent for all levels.
Please note that this study only investigates the factors affecting the residues at different localization. Correlation analyses between variables other than residue were not included in the body of the manuscript.
Abbreviations: LED, Levodopa Equivalency Dose; MoCA, Montreal Cognitive Assessment; UPDRS-III, Unified Parkinson’s Disease Rating Scale.
Second-Step Analyses—Regression Models (Table 4)
Model 1: Oropharynx
Multiple linear regression revealed a positive, significant, and moderate relationship between premature leakage and
Regression Analyses*; Simple Linear Regression Analysis Between Independent Variables and Post-Swallowing Residue at the Level of Epiglottis (Model 3). Multiple Linear Regression Analysis Between Independent Variables and Post-Swallowing Residue at the Level of Oropharynx (Model 1), Hypopharynx (Model 2), Laryngeal Vestibule (Model 4), Vocal Folds (Model 5), and Subglottis (Model 6).
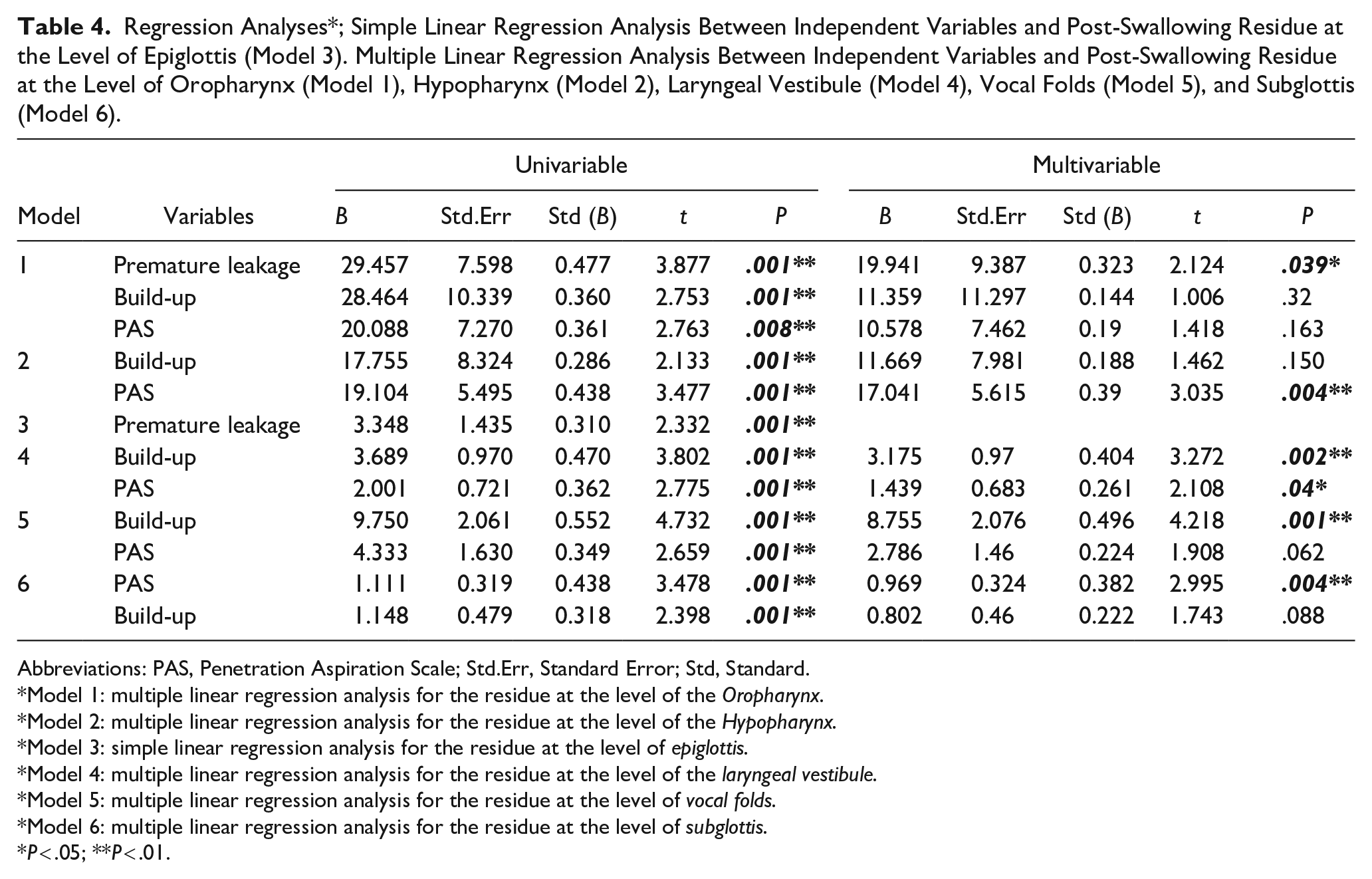
Abbreviations: PAS, Penetration Aspiration Scale; Std.Err, Standard Error; Std, Standard.
*Model 1: multiple linear regression analysis for the residue at the level of the
*Model 2: multiple linear regression analysis for the residue at the level of the
*Model 3: simple linear regression analysis for the residue at the level of
*Model 4: multiple linear regression analysis for the residue at the level of the
*Model 5: multiple linear regression analysis for the residue at the level of
*Model 6: multiple linear regression analysis for the residue at the level of
P < .05; **P < .01.
Model 2: Hypopharynx
Multiple linear regression revealed a positive, significant, and moderate relationship between airway invasion and residue at the level of the hypopharynx (
Model 3: Epiglottis
Simple linear regression revealed a positive, significant, and moderate relationship between premature leakage and epiglottic residue (
Model 4: Laryngeal vestibule
Multiple linear regression revealed a significant and moderate relationship between build-up phenomenon, airway invasion, and residue at the level of the laryngeal vestibule (
Model 5: Vocal folds
Multiple linear regression revealed a positive, significant, and moderate relationship between the build-up phenomenon and residue at the vocal folds (
Model 6: Subglottis
Multiple linear regression revealed a positive, significant, and moderate relationship between airway invasion and residue at the level of subglottis (
Discussion
This study focused on FEES findings in patients with IPD using the newly validated VASES scale, and focusing on residue patterns and their association with specific aspects of disordered swallowing. Our study had several significant findings. First, oropharyngeal residue was significantly associated with premature leakage. Second, the build-up of residue was significantly associated with residue at the level of the vocal folds and laryngeal vestibule. Third, demographic characteristics, disease stage, cognitive status, and BI were not associated significantly with residue scores at any level.
In previous studies, premature leakage had been associated with deficient oral motor control, although no comprehensive study investigated its pathophysiology.5,33 This is in contrast with many studies examining the pathophysiology of vallecular residue: tongue driving forces, tongue retraction 34 to contact with the posterior pharyngeal wall, pharyngeal wall movement, 35 and excursion of the hyoid for epiglottic inversion 36 have all been found to be important factors in vallecular clearance. Our study shows that premature leakage occurred in 13.2% of patients, and the most common localization of residue with the highest median scores was the oropharynx, at the level of the vallecula. Furthermore, premature leakage was found to be the only factor associated with residue at the levels of the oropharynx and neighboring epiglottis. These findings are consistent with prior studies and indicate that instead of muscle weakness, a loss of coordination between the oral and pharyngeal phases might be the main mechanism of oropharyngeal residue and premature leakage in patients with IPD.5,33
The introduction of VASES to standardize FEES interpretation has facilitated the analysis of residues at several levels. Our regression models reveal a significant relationship between residues at the laryngeal vestibule and vocal folds, and the presence of build-up phenomenon. Atrophy of the pharyngeal musculature has been associated with increased pharyngeal area, which is strongly associated with detrimental changes to swallowing kinematics, safety, and efficiency in PD. 37 Interestingly, there are not many studies available on the pathophysiology of the build-up phenomenon in the literature. Pflug et al observed that half of their study group of patients with PD had build-up, which they related to pharyngeal weakness and sensory deficit. 4 We did not measure pharyngeal muscle strength. However, we did find a significant correlation between markers of pharyngeal weakness, namely (1) build-up phenomenon as a surrogate of swallowing efficacy and (2) residue at the levels of vocal folds and laryngeal vestibule as a maker of swallowing safety compromise. These results suggest that build-up may be a risk for penetration.
We also found a significant relationship between residue at the level of subglottis and laryngeal vestibule and airway invasion. Although penetration and aspiration are common in patients with IPD, residue at these levels might also indicate a decrease in cough reflex and/or cough strength. Troche et al 38 investigated the relationship between cough and dysphagia in PD, and their analysis revealed two main findings: (1) patients with dysphagia were less likely to cough during reflex cough testing and (2) a negative correlation was present between dysphagia severity and cough sensitivity (urge to cough). Furthermore, Pitts et al 39 showed a moderate relationship between voluntary cough airflow speed and penetration/aspiration scores. Together, these two studies suggest that patients with PD and dysphagia are less likely to cough in response to aspiration, and when they do cough, they are less likely to have coughs that are effective at clearing the penetrant and aspirate residue out of the airway. Future studies on cough measurements and residue in IPD might shed light on the significance of the residue at the level of the subglottis and laryngeal vestibule as an indicator of blunted cough reflex and/or deteriorated voluntary cough strength or efficiency.
BI has been used as an indicator of vocal fold atrophy. Yiu et al 30 showed that vocal fold atrophy is predictive of swallowing safety and that the severity of atrophy is correlated with higher PAS scores in patients with PD. Although a significant correlation between hypopharyngeal residue and BI was found in the correlational analysis in our study, this significance was not present in the univariate analysis.
However, the presence of airway invasion was the only significant variable related to hypopharyngeal residue. Previous studies showed findings either consistent or disagreeing with these results. Nordio et al 40 found a significant correlation between pharyngeal residue scores and PAS scores for semisolid boluses and liquids. Controversially, Molfenter et al 41 published a paper suggesting that only the presence of vallecular residue is significantly associated with penetration-aspiration on subsequent clearing swallows, not the residue at the pyriform sinus. Further studies investigating the effects of hypopharyngeal residues on airway safety are needed.
There are several limitations to our study. No relationship was found between age, MoCA scores, HYS, LED and UPDRS-III, and residue at any level. The study group mostly included patients who were in the HYS 2 of the disease. Furthermore, selected patients were also in a good state cognitively and physically. Thus, the distribution of MoCA, LED, and UPDRS-III in the study group was limited, reducing the ability to detect an effect if present. Other limitations of the study are the low number of patients, which was due to restrictions related to the COVID-19 pandemic. Due to the low numbers, we were unable to analyze the residue scores for all consistencies. Instead, the worst residue scores were used. Furthermore, due to the scarcity of fellowship-trained physicians at the time of data analysis, only one reviewer analyzed the videos. To decrease the bias, recordings were presented to the reviewer in a blinded fashion and 19% of the recordings were also re-included blindly to evaluate intra-rater reliability, which showed high correlation at all levels. Lastly, our regression models only found a moderate association between residues and factors that were associated with residues. To get a stronger association, studies with larger populations are needed.
Conclusion
The present study shows that residues at different anatomic levels might indicate distinct pathophysiologic aspects of swallowing dysfunction in IPD. Residue at the base of the tongue might be due to loss of coordination between the oral and pharyngeal phases of swallow, and pharyngeal weakness may be associated with residue at the subglottis and laryngeal vestibule. Further studies that combine pathophysiologic evaluation, localization, and quantification of residue are needed to prove these hypotheses. Using residue localization and quantification may be a helpful tool in assessing the impact of targeted swallowing interventions in patients with IPD and dysphagia.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
