Abstract
Introduction
Hyperbaric oxygen therapy (HBOT) involves delivery of 100% oxygen to patients in pressurized chambers. 1 Indications include infection, arterial gas emboli, radiation injury, and sensorineural hearing loss. Middle ear barotrauma is the most common side effect of HBOT, occurring in 9.2% of cases. 2 Failure of the middle ear pressure regulation system formed by the Eustachian tube, tympanic cavity, and mastoid results in barotrauma. 3 Minor barotrauma will generally self-resolve. However, severe injury may cause permanent hearing and balance deficits. 4 Intubation or preexisting Eustachian tube dysfunction increases the risk of barotrauma.
Tympanostomy tubes facilitate middle ear equalization. We describe a technique of placing a modified peripheral intravenous cannula (PIVC) tympanostomy tube, which can be performed using a Voroscope® (VorOtek, Moolap, Australia), head-worn microscope, and topical anesthetic. The ability to perform this at the bedside allows for clinicians to establish timely barotrauma prevention prior to emergent HBOT. Further, it avoids theater-associated costs. The primary objective of this study is to evaluate the efficacy and safety of this technique in preventing barotrauma in patients undergoing HBOT. The secondary objective is to determine whether procedural or hyperbaric factors impact the ability of ventilation tubes to prevent barotrauma.
Patients and Methods
We recommend modifications to techniques described by 2 previous articles which utilize PIVCs for barotrauma prevention.5,6 Equipment required includes:
1. Voroscope® or conventional microscope
2. Ear speculum
3. Extended length PIVC size 18-gauge or 20-gauge. At our institution, B Braun Introcan Safety® Deep Access IV Cannulas (B Braun, Melsungen, Germany) are available
4. Scissors
5. Local anesthetic (if patient awake); EMLA® (Aspen Global Incorporated, Grand Bay, Mauritius) 5% cream (lidocaine/prilocaine) or Xylocaine® (Aspen Global Incorporated, Grand Bay, Mauritius) 10% spray (lidocaine)
6. Additional equipment which may be required includes micro-alligator forceps, Jobson-Horne wax curette, 3 ml syringe, and a ear suction cannula
We recommend the operator be seated and patient be positioned in a reclined chair or bed with the head rotated away from the operator. The external auditory canal (EAC) is toileted with curettes or suction catheters.
Apply topical anesthetic for awake patients. Apply EMLA® 5% cream topically to the tympanic membrane (TM) and EAC via a 3 ml luer lock syringe attached to a large-bore ear suction cannula. Alternatively, Xylocaine® (lidocaine) 10% spray may be used, which requires the patient to lie with the anesthetized ear upward or soaked cotton wool placed against the TM. Leave the agent for 30 minutes to ensure adequate anesthesia.
Extended length PIVCs are preferred to achieve an unobstructed view and optimize control. For PIVC preparation, with scissors, circumferentially cut around the sheath 10 mm from its end, making it independent from its proximal part. Bend the needle to 20-degrees 5 mm from the cannula hub to optimize the view of the TM (Figure 1). Consider attaching the cannula to a 3-ml syringe as a handle for extra stability.
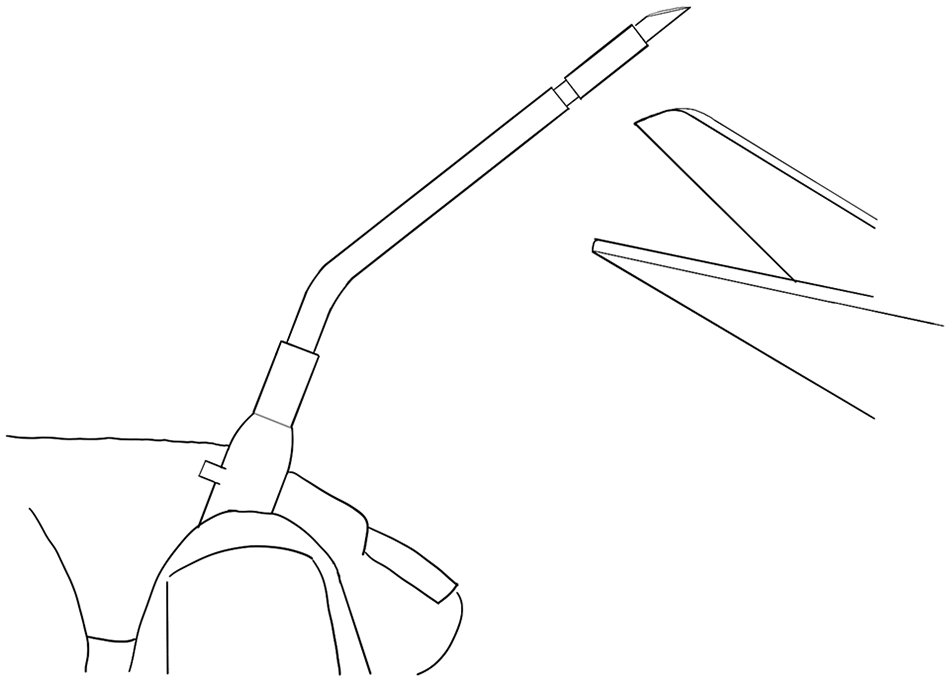
Preparation of the peripheral intravenous cannula by bending the needle and circumferentially cutting the sheath.
Perform a pinhole antero-inferior quadrant myringotomy. With the bevel of the needle through the TM, advance the cannula hub to slide the sheath into the myringotomy (Figure 2). A Jobson-Horne curette may be used to apply pressure to the hub with the opposite hand to advance the sheath. Alternatively, following myringotomy, remove the entire apparatus, and place the free sheath into the myringotomy with micro-alligator forceps.
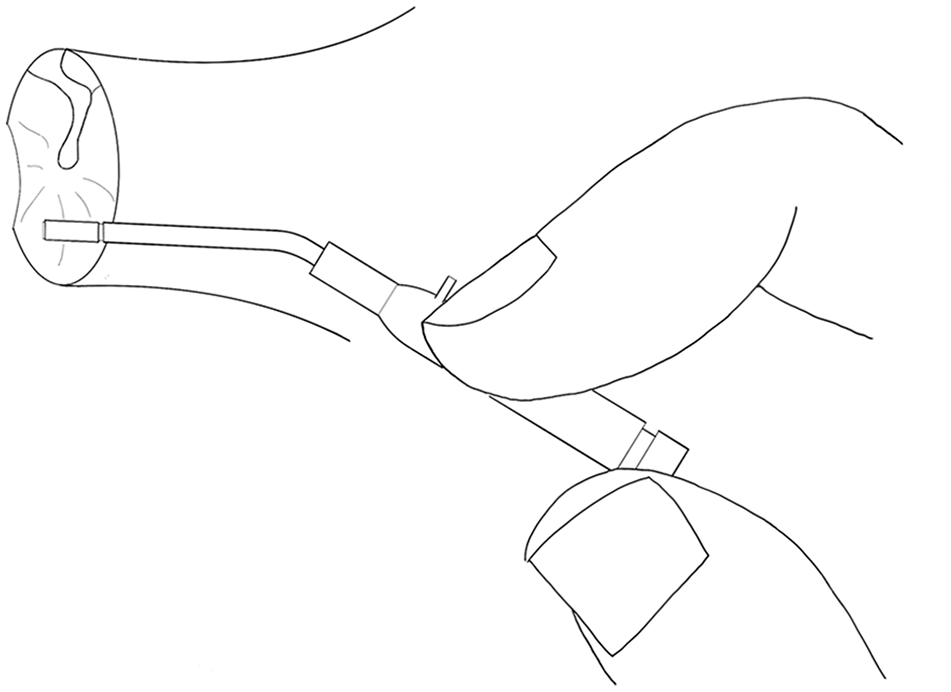
Myringotomy and insertion of the modified peripheral intravenous cannula into the tympanic membrane.
We performed an analytical observational study at a metropolitan Australian hospital with a state-wide HBOT service. Patients were identified via unit audits between October 2018 and October 2022. Data regarding patient, HBOT, procedural factors, and post-procedural outcomes were collected from electronic medical records retrospectively. Barotrauma was described in accordance with Modified Teed Grading, a 6-tiered scale based on otoscopic examination findings 7 :
- Grade 0: Normal TM
- Grade 1: Diffuse redness and TM retraction
- Grade 2: Grade 1 plus slight TM hemorrhage
- Grade 3: Grade 1 plus gross TM hemorrhage
- Grade 4: Dark and slightly bulging TM due to hemotympanum
- Grade 5: TM perforation
Modified Teed Grading was commonly documented within the medical record. In the absence of formal documentation, documented otoscopic findings were equated to the appropriate grade.
Graphpad Prism 9 was used for statistical analysis. D’Agostino-Pearson testing determined normality for all continuous variables. The cohort was divided into subgroups based on patient, hyperbaric, and procedural factors. Complication rates were compared between subgroups by using 2-sided Fisher’s exact tests with Bonferroni correction applied to accept a significance level of 5%; to mitigate type I error, only tests with a P-value of <.0026 was considered significant. Simple logistic regression was used to compare continuous variables with the presence of complications.
Results
A total of 220 among 112 patients received modified PIVC ventilation tubes over 4 years. Thirty-eight patients (33.9%) were female, 74 (66.1%) were male (Table 1). Mean age was 58.6 years (95% CI: 55.7-61.6, SD 15.9). One patient (0.9%) had preexisting Eustachian tube dysfunction and 8 (7.1%) had premorbid hearing impairment documented. Sixteen patients (31 ears) were anticoagulated with agents other than prophylactic low-molecular-weight heparin at ventilation tube insertion.
Descriptive Statistics of Patient and Hyperbaric Oxygen Therapy Factors.
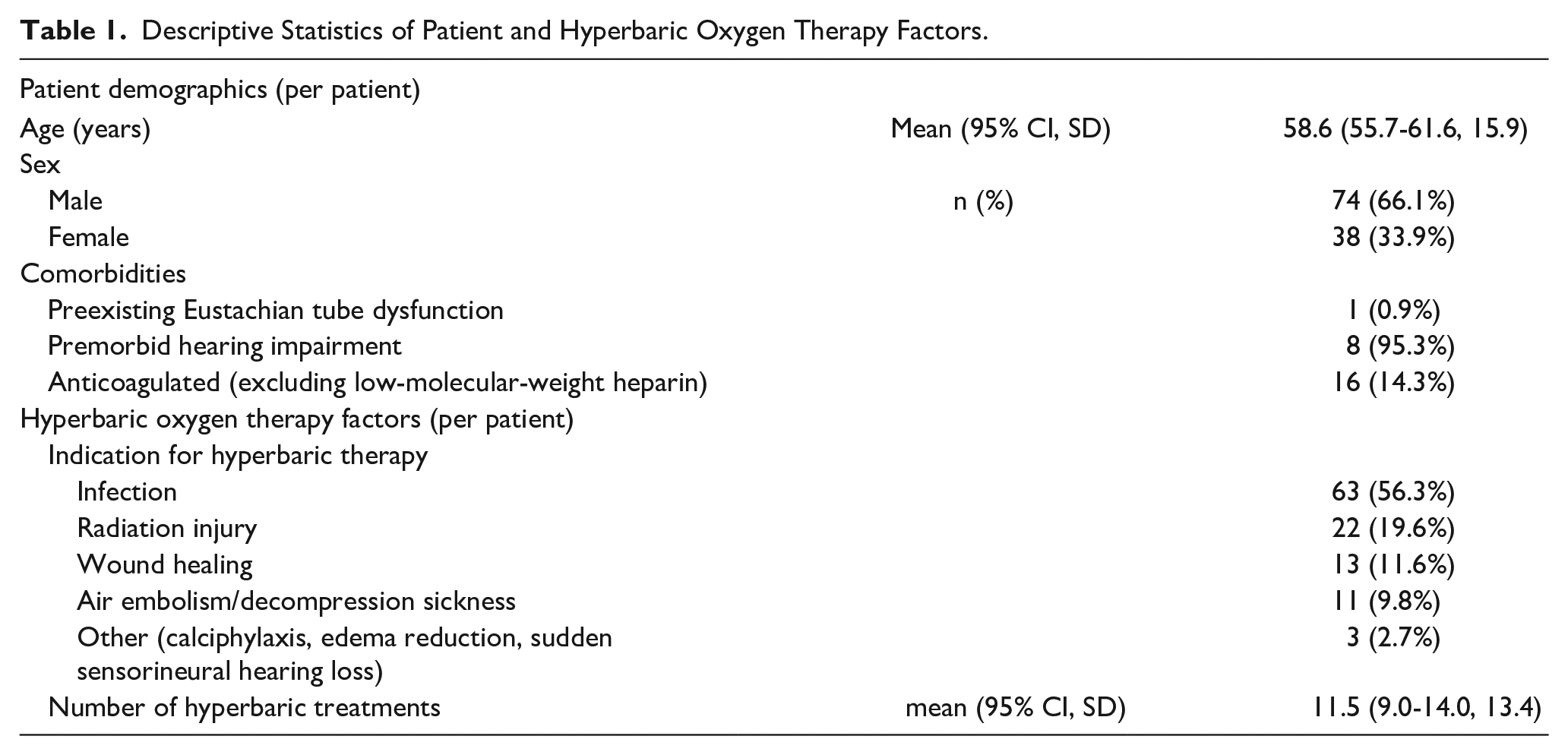
Patients underwent a mean of 11.5 (95% CI: 9.0-14.0, SD: 13.4) hyperbaric treatments. Indications included infection (56.3%), radiation injury (19.6%), wound healing (11.6%), air embolism or decompression sickness (9.8%), and other (calciphylaxis, edema reduction, sensorineural hearing loss; 2.7%).
Ventilation tubes were inserted bilaterally in 105 patients. Seven patients underwent unilateral insertion; 3 (42.9%) required contralateral insertion during their HBOT course. We defined primary insertion as the first ventilation tube insertion in an ear for a HBOT course. A total of 43 (19.5%) primary insertions were performed under microscope and 177 (80.5%) under Voroscope®. Thirty (13.6%) were performed in theater and 190 (86.4%) in other settings (intensive care, ward, emergency department, or clinic). Seventy-eight primary insertions (35.5%) were on awake patients, and 142 (64.5%) on sedated/intubated patients. Seventy-six primary insertions used topical anesthetic, with EMLA® 5% alone in 35 (46.1%), Xylocaine® 10% spray alone in 27 (35.5%), phenol in 10 (13.2%), and a combination of EMLA® and Xylocaine® in 4 (5.3%).
Thirty-four ears suffered barotrauma before primary insertion, with Modified Teed Grades of 1 in 20 (9.1%), 2 in 4 (1.8%), 3 in 3 (1.4%), and 4 in 7 ears (3.2%). Eighteen-gauge PIVCs were used for primary insertion in 131 ears (59.5%), and 20-gauge in 76 ears (34.5%). Other sizes used were 14-gauge (0.9%), 16-gauge (0.5%), and 22-gauge (4.5%). Tubes were required for a mean of 15.0 days (95% CI: 11.8-18.2, SD: 24.0) until HBOT course completion.
On primary insertion, immediate complications were described in 15 ears (6.8%; Table 2): 8 instances of canal trauma/bleeding (3.6%), 2 ventilation tubes placed into the middle ear (0.9%), 2 TM tears (0.9%), 2 patients with significant pain (0.9%), and 2 failures of insertion under Voroscope® (1 patient, 0.9%).
Complications of Modified Peripheral Intravenous Cannula Ventilation Tube Insertion (Per Ear).
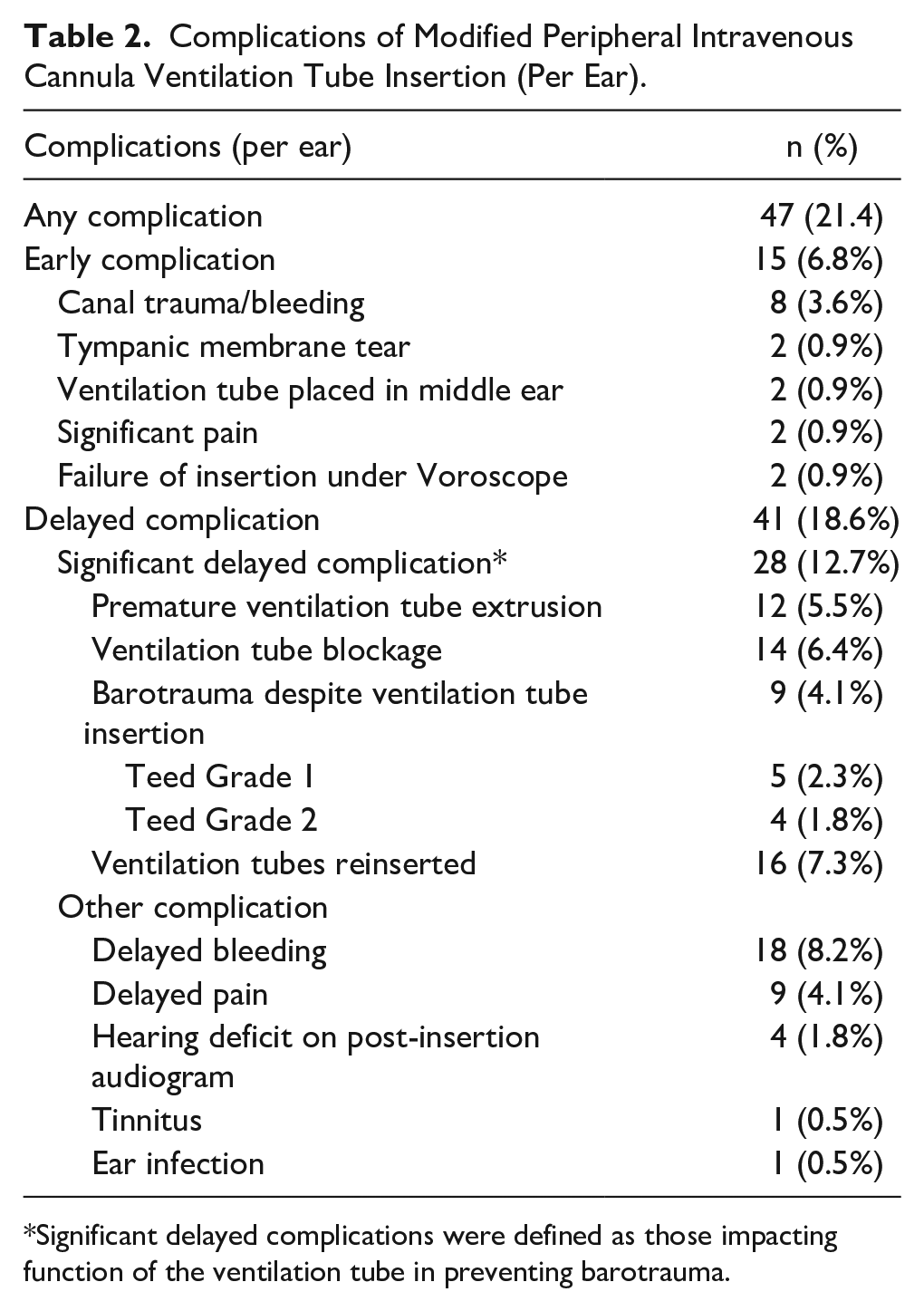
Significant delayed complications were defined as those impacting function of the ventilation tube in preventing barotrauma.
We considered significant delayed complications to include those impacting function of the ventilation tube in preventing barotrauma—premature extrusion, ventilation tube blockage, and barotrauma despite ventilation tube insertion. Twenty-eight ears (12.7%) were affected by significant delayed complications: ventilation tube blockage in 14 (6.4%), extrusion prior to completion of HBOT in 12 (5.5%), barotrauma despite ventilation tube insertion in 9 (4.1%) with Modified Teed Grade 1 barotrauma in 5 ears and Grade 2 in 4. Sixteen ears (7.3%) required reinsertion of modified PIVC ventilation tubes, with 6 ears requiring 2 reinsertions (2.7%). Delayed complications of any sort were present in 41 ears (18.6%): delayed-onset bleeding in 18 (8.2%), delayed pain in 9 (4.1%), hearing deficits on audiogram following ventilation tube insertion in 4 (1.8%), tinnitus in 1 (0.5%), and ear infection in 1 (0.5%).
Twelve patients (5.5%) died, precluding them from follow-up. Only 36 ears (16.4%) were followed-up in outpatient clinic. Eight ventilation tubes (3.6%) were manually removed following HBOT completion. Only 14 ears were followed-up with audiology.
On subgroup analysis, anticoagulation excluding prophylactic low-molecular-weight heparin was found to significantly increase the odds of delayed bleeding (OR 8.9, 95% CI: 2.9-30.1, P = .0005; Table 3). However, it did not significantly impact the rates of intra-procedural canal trauma/bleeding, significant delayed complications, or complications overall. Ears with preexisting barotrauma were significantly correlated with increased complications overall (OR 4.4, 95% CI: 2.0-9.8, P = .0004). As well, patients with preexisting barotrauma trended toward having more significant delayed complications (OR 3.9, 95% CI: 1.6-9.4, P = .0038); however, this result was not statistically significant.
Subgroup Analyses of Procedural/Patient Factors and Complications (Per Ear).
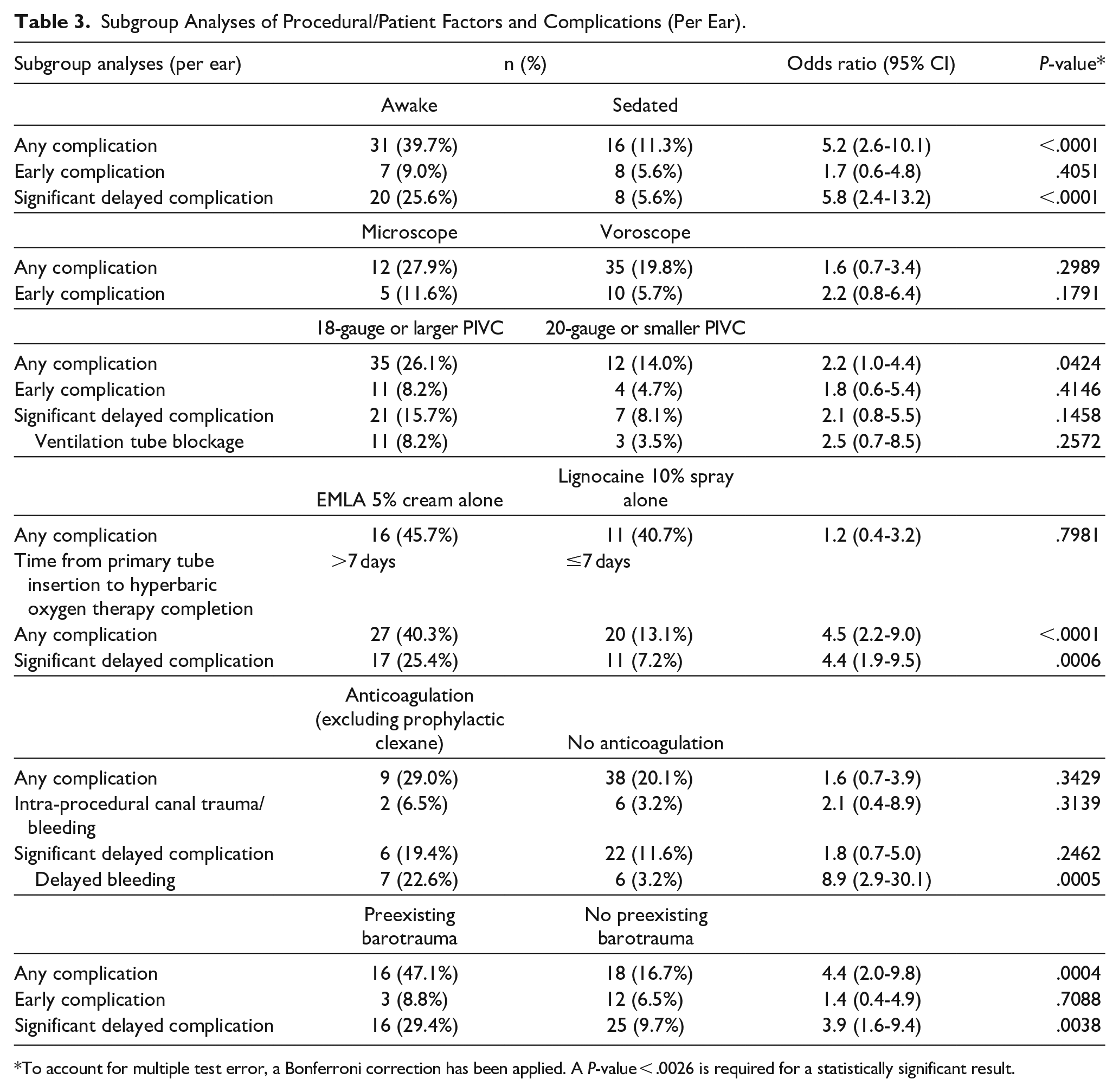
To account for multiple test error, a Bonferroni correction has been applied. A P-value < .0026 is required for a statistically significant result.
Patients awake during the procedure were found to suffer more overall (OR 5.2, 95% CI: 2.6-10.1, P < .0001) and significantly delayed (OR 5.8, 95% CI: 2.4-13.2, P < .0001) complications compared to sedated patients. PIVCs 18-gauge or larger trended toward having more complications (OR 2.2, 95% CI: 1.0-4.4, P = .0424); however, this result was not statistically significant. Notably, there was no significant difference in ventilation tube blockage rates between PIVC sizes. Further, minimal difference in complication rates was found between topical anesthetic agents.
The time to completion of HBOT profoundly affected complication rates. On simple logistic regression, each added day of HBOT increased the odds of significant delayed complications by 1.034 (95% CI: 1.02-1.05, P < .0001) and complications overall by 1.036 (95% CI: 1.02-1.05, P < .0001).
Discussion
Placing ventilation tubes for patients undergoing HBOT provides a unique scenario; the tubes facilitate the passage of air to equalize pressures across the TM as opposed to draining fluid and must only remain in situ for a limited time period. Indications for insertion in the context of HBOT include obtunded patients, Eustachian tube dysfunction, or barotrauma sustained during HBOT. Middle ear aeration should be achieved via a technique that is easily and readily performed, has few complications, and is simply reversible; 8 a flangeless ventilation tube that can be placed at the bedside appears ideal in this circumstance, allowing for HBOT to be expedited. In addition to time efficiency, this method foregoes the costs associated with theater.
We propose alterations to techniques only twice described in previous literature. When first described by Mooney et al., 6 an 18-gauge PIVC was recommended with anesthetic via a regional EAC ring blockade with 1% lignocaine and 1:100,000 adrenaline, or 10% lignocaine solution topically. Contrastingly, Zhang et al recommended a 24-gauge PIVC and anesthetic with 80% phenol. 5
Like Mooney et al., 6 we recommend bending the needle to 20° to optimize visualization during the procedure. As the cannula hub cannot be pushed past the bend of the needle, bending the PIVC 5 mm from the hub reduces the risk of over-advancement of the cannula and loss of the tube within the middle ear. To further optimize handling, we recommend a syringe be attached to the PIVC for added stability.
The PIVC sizes used in our service were mostly 18 and 20-gauge. Counterintuitively, trends favored larger PIVCs to have increased rates of blockage and complications overall. However, these results were not statistically significant. This may be partially accounted for by lower rates of canal trauma with smaller cannulas, an outcome perhaps underreported considering the retrospective nature of the analysis.
We found no significant differences in complication rates between EMLA® and Xylocaine® 10% spray. Phenol is a suitable alternative which works via rapid tissue chemical cauterization with immediate analgesic effect. 9 However, its limitations include inadequate EAC analgesia and the requirement for a precise myringotomy to be made. 10 Further, phenol is not readily available in most institutions and poses challenges regarding appropriate storage and potential toxicity if spilt. In comparison, EMLA® and Xylocaine® 10% are readily available compounds that do not require specialized handling or storage. They can broadly anesthetize the EAC and TM, the downside being their slower onset.11,12
Barotrauma prevention is required expeditiously in multimorbid patients to facilitate immediate HBOT. Consequently, our results reflect real-world practice where on-call otolaryngology trainees perform this procedure, often in after-hours settings, instead of experienced clinicians routinely inserting these ventilation tubes. While the effect of clinical experience is difficult to gauge due to yearly changeover of trainees, education may prove valuable in decreasing the heterogeneity of procedural factors, including the local anesthetic used, ventilation tube size, and cannula preparation, which may further reduce complication rates.
Of complications described, many did not confer lasting functional consequence, including intra-procedural canal trauma/bleeding (3.6%), delayed bleeding (8.2%) and delayed pain (4.1%). While the rate of significant delayed complications in an ear was 12.7%, it is unlikely that ventilation tube blockage (6.4%) or extrusion prior to HBOT completion (5.5%) would confer long-term sequelae in the absence of barotrauma. These complications were troubleshooted by reinserting (7.3%) or using ototopical drops to unblock the ventilation tube. The rate of barotrauma despite ventilation tube insertion was ultimately low (4.1%) and limited to minor injury (Modified Teed Grade 2) only. We believe that this finding confers the procedure to be safe.
Few studies describe complication rates of conventional grommets for barotrauma prevention. A retrospective analysis of 31 patients, who received either Shah flanged or Shepard models for HBOT, reported blockage in 2 cases—comparable to our rate (6.4%). 13 A further study of 16 patients undergoing T-tube insertion for HBOT described no cases of barotrauma, but higher rates of late complications such as otorrhoea. 8
The greatest influence on complication rates was the duration of HBOT. Using logistic regression, for each added day of HBOT, the odds of a significant delayed complication occurring increased by 1.034 (95% CI: 1.02-1.05, P < .0001). Patients awake during insertion were found to be at higher risk of both overall and significant delayed complications; this is likely confounded by sedated patients having fulminant illnesses, such as necrotizing fasciitis, which required far shorter HBOT courses. The finding that preexisting barotrauma correlated with increased complication rates shares a similar risk of confounding; most patients with preexisting barotrauma were awake patients undergoing extended HBOT courses, as obtunded patients were routinely referred for ventilation tubes prior to HBOT commencement.
A limitation of our study was the poor long-term follow-up of patients. This was in part due to patients dying (10.9%), the geographical barrier following repatriation from our state-wide hyperbaric service, and the ventilation tube being overlooked as a minor component of the patient’s overarching presentation. Routine audiology was not performed but ordered to investigate aural symptoms. The etiology of hearing loss in 4 of 14 ears (28.6%) on follow-up audiogram is difficult to determine. However, a mix of sensorineural and conductive losses suggests that deficits found may be multifactorial—perhaps from a combination of barotrauma, premorbid hearing loss, or ventilation tube insertion. Prospective study with routine audiology may enhance our understanding of hearing loss post HBOT and PIVC ventilation tube insertion. Time to tube extrusion is difficult to determine based on our data and would be best investigated via prospective study. Notably, Zhang et al.’s 5 study found 88% to be self-extruded at a median follow-up time of 6 weeks. Further, that study found only a small incidence of persistent perforation post-extrusion, in a single TM out of 257 ears.
Conclusion
The modified PIVC technique can be performed at the bedside with safe and reproducible results. In patients planned for prolonged HBOT courses, we advocate counseling on increased risks, including blockage, extrusion, and the potential need for tube replacement. Prospective investigation would assist determining the effect of factors such as PIVC size on complications, and to establish an expectation of tube extrusion times.
Footnotes
Presentation Declaration
This article has been presented as an oral presentation at the International Federation of ORL Societies Dubai ENT World Congress, in Dubai, United Arab Emirates, January 17 to 23, 2023.
Data Availability Statement
The datasets generated during and/or analyzed during the current study will not be made publicly available due to the risk of patient identification in a cohort having undergone a rare procedure. However, data may be made available on reasonable request made directly to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval to report this case series was obtained from the Alfred Health Office of Ethics & Research Governance (Project Number: 587/22).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Alfred Health Office of Ethics & Research Governance approved protocols.
Statement of Informed Consent
Informed consent for patient information to be published in this article was not obtained because all information collected was completely de-identified, previously available to treating clinicians via our Electronic Medical Record system, and analyzed as a cohort.
