Abstract
Keywords
Introduction
In the last decade, the safety of magnet resonance imaging (MRI) for patients with cochlear implants (CI) has been ensured by the introduction of freely rotatable, removable diametric magnets which align with the magnetic field. These MRI-compatible devices are approved for use with MRI scanners up to 3 T field strength without a compressive dressing.1-4 The first of these devices was released by MED-EL® (MED-EL, Innsbruck, Austria), which in 2014 introduced Synchrony, a CI with a new type of internal magnet that aligns itself to the magnetic field. 5 In 2018, Advanced Bionics® (Advanced Bionics, Valencia, United States) acquired FDA approval for the HiRes Ultra 3D implant containing 4 cylindrical, separately rotatable magnets. 6 The Nucleus® Profile™ Plus Series was introduced by Cochlear® (Cochlear Limited, Sydney, Australia) with a self-aligning internal magnet in 2019. 7 All aforementioned devices have been proven to be MRI compatible in laboratory and clinical studies.8-15
Earlier generations of CI devices either contain a fixed magnet or an axial, removable magnet that does not align with the magnetic field during MRI. For some devices with fixed magnet, MRI is contraindicated. Most implants with axial, removable magnets are MRI compatible up to 1.5 T when a compressive dressing is applied to prevent dislocation of the internal magnet.1,2,4 Surgical magnet removal is necessary for MRI at 3 T or may be considered in patients requiring multiple MRI scans. However, surgical intervention carries the risk of infection or damage to the implant.16,17
Dislocations of the internal magnet, mostly due to MRI, have been described in numerous case reports and case series and can occur despite the application of a compression bandage.10,11,18-26 For the implants with axial magnets, the risk of magnet dislocation also depends on the CI device type. 9 Tilting of the magnet in relation to its silicone housing, if the lip of the housing is not traversed by the magnet, is defined as partial magnet dislocation (PMD). In contrast, in a complete dislocation, the edge of the magnet traverses the lip of the housing entirely.21,24,27 In either case, the external processor cannot be used until the dislocated magnet has been repositioned to avoid pain, skin wounds, or even extrusion of the magnet through the skin.26,28 Historically, in most cases, surgical magnet repositioning (SMR) was performed with a small incision to visualize and reposition the dislocated magnet.11,12,18-20 Endoscopic approaches to SMR have also been previously described. 29 In both cases, the need to violate the receiver stimulator pocket poses a risk of implant infection that can ultimately lead to device explantation.17,18 As an alternative to SMR, noninvasive manual magnet repositioning (MMR) can be performed by applying directed pressure on the tilted part of a partially dislocated magnet. This technique was first described by Carlson et al. in 2015. 24 We published our first experience with this method in 2021. 28 The implant can be used immediately after MMR if the skin condition over the implant is normal. 10 There has not yet been a direct comparison of SMR and MMR in the literature. The objectives of this retrospective study were therefore to compare the success rate of SMR and MMR as well as the post-interventional complication rate and CI downtime.
Materials and Methods
This retrospective study was conducted at a tertiary referral medical center (Department of Otorhinolaryngology and Head and Neck Surgery, Friedrich-Alexander-University Erlangen-Nürnberg (FAU), Erlangen, Germany). The study was approved by the institutional review board (application number: 99_21 Bc) and performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments. The requirement of informed consent for patient information to be published in this article could be waived, as the data for this study were reviewed retrospectively.
All patients with CI with dislocation of their internal CI magnet presenting at our institution between January 1, 2007 and September 30, 2022 were eligible for the study. Inclusion criteria were as follows: PMD confirmed by imaging (X-ray or ultrasound) or direct intraoperative visualization; history of MRI at 1.5 T before onset of symptoms; treatment with SMR or MMR; age ≥ 18 years; minimum follow-up of 3 months, and complete medical record. The following exclusion criteria were applied: missing confirmation of PMD; complete magnet dislocation; incomplete medical record, and spontaneous repositioning of the dislocated magnet.
In general, patients who had a CI history and showed discomfort, swelling, and/or pain at the implant area were referred to an ear, nose and throat (ENT) specialist at our institution. The history was taken with special focus on recent MRI or head trauma. PMD was diagnosed by X-ray, transcutaneous ultrasound, or by clinical examination alone. SMR of the dislocated magnet was the treatment option of choice for all types of magnet dislocation until June 2019. After diagnosis, the patient was advised to abstain from wearing their processor to avoid skin breakdown. In cases of SMR, surgery was planned urgently within a few days. SMR was performed with local anesthesia as standard of care. The procedure was done under general anesthesia only for reasons of medical safety or patient preference. In all cases of SMR, a single-shot intravenous antibiotic (amoxicillin/clavulanic acid 3 g) was administered. In brief, SMR involved a skin incision of approximately 3 cm in length at the cranial edge of the palpable underlying internal magnet. The magnet was freed from the surrounding tissue to allow complete visualization. Following this, the magnet was carefully slipped back into its silicone housing using nonmagnetic instruments. Finally, the wound was rinsed with antiseptic povidone-iodine solution, closed in a multilayer fashion, and dressed with a compression bandage until the following day. A follow-up examination was scheduled after 1 week. Further follow-up examinations were scheduled if wound healing was not complete. Use of the CI processor was resumed only after the wound was fully healed.
After the observation of a spontaneous magnet repositioning in a patient with PMD at our institution in June 2019, MMR was recommended as an alternative noninvasive treatment option for PMD. It was performed without anesthesia. MMR started with palpation and ultrasound visualization of the dislocated part of the magnet. According to the direction of dislocation, that is, anterior, posterior, superior, or inferior, firm pressure was applied with the thumb at a sloped angle to the skin. The position of the repositioned magnet was subsequently controlled by ultrasound. Patients were allowed to wear their processor immediately in the absence of irritated, reddish skin, and/or swelling. Otherwise, patients were advised not to wear the processor until the first follow-up visit. In general, a maximum of 3 attempts of MMR were made with ultrasound visualization of the magnet position after every attempt, in accordance with the protocol published by Rupp et al. 28 If MMR failed or if there was a complete magnet dislocation, then SMR was performed.
Complications occurring within 4 weeks of magnet repositioning were classified as early. Otherwise, they were considered as late complications. It was expected that patients would have no pain at the implant area 1 week postoperatively and that wound healing would be completed after 2 weeks.
Statistical Analysis
Statistical calculations were performed using SPSS (IBM SPSS Statistics 28.0, IBM, New York, NY). The total CI downtime was defined as the time between the diagnosis of PMD and CI reactivation. The total follow-up time was defined as the time between treatment of PMD and the last visit of the patient at our institution. For both SMR and MMR, the time from diagnosis to treatment, treatment to CI reactivation, total CI downtime, and the total follow-up time were calculated and tested for significant differences. Additionally, the time from MRI to diagnosis was compared to evaluate the impact of a delayed diagnosis on the complication rate. Metric variables are presented as mean values ± 1 standard deviation (SD) and range [minimum (min.) and maximum (max.)] and were tested for normal distribution using the Kolmogorov–Smirnov test and by means of Q–Q plots and histograms. The independent t-test was used to compare metric variables. Non-normally distributed metric variables were analyzed using the Mann–Whitney U-test. The chi-square test was used for the comparison of nominal variables. If the reported frequency was below 5, Fisher’s exact test was conducted. A P-value ≤ .05 was considered as statistically significant. Effect sizes were reported for the Mann–Whitney U-test using r, and for the chi-square test and Fisher’s exact test using ϕ. An r or ϕ of 0.1 represents a small effect, 0.3 a moderate effect, and 0.5 a strong effect.
Results
Patients and Implants
During the study period, 68 cases of magnet dislocation were identified. Eleven cases were excluded due to complete magnet dislocation. In 4 cases, a spontaneous repositioning of the magnet was observed and no further treatment was necessary. One case was excluded because the reason for magnet dislocation remained unknown. One dislocation appeared in a 7-year-old child who did not meet the inclusion criteria.
In our final study cohort, 51 cases of PMD were observed in 42 patients and 43 devices (18 ♀; 24 ♂). Twelve patients had bilateral CI. The median age at implantation was 51.7 years (min. 2 years, max. 92 years.). The median age at first magnet repositioning was 53.8 years (min. 19 years, max. 93 years). The mean time from implantation to first magnet repositioning was 3.6 ± 3.1 years (min. 3 months, max. 16.7 years). The 43 different devices were all manufactured by Cochlear®. Device types were as follows: CI24R (CS) = 1 (2.3%); CI24RE(CA) = 1 (2.3%); CI532 = 9 (20.9%); and CI512 = 32 (74.4%). All devices had FDA approval for MRI at 1.5 T with the implant magnet in place. 2
MRI and Diagnosis of PMD
In 31 cases (61%), MRI had been performed at our institution with a routine head bandage. The remaining 20 cases (39%) occurred after MRI that had been conducted at external institutions. In 7 out of these 20 cases (35%), no head bandage had been applied. The mean time from MRI to diagnosis of PMD was 12.4 ± 31.7 days (min. 0 day, max. 172 days) for SMR and 14.2 ± 31.1 days (min. 0 d, max. 111 d) for MMR, with no significant difference between both groups (P = .10; r = 0.24).
PMD was diagnosed by ultrasound and X-ray examination in 38 and 4 cases, respectively. Nine cases of PMD were diagnosed by clinical examination alone. Four patients had 2 independent episodes of PMD of the same implant (CI532 in 1 patient; CI512 in 3 patients) that were caused by different MRI scans; 1 of these patients was implanted bilaterally (CI512 on both sides). One patient with bilateral CI (CI532 on both sides) showed a simultaneous bilateral PMD caused by the same MRI examination. In 1 patient with a CI512 of the left ear, 5 consecutive independent episodes of PMD occurred during 5 different MRI scans.
Treatment
The mean time from diagnosis to treatment was significantly shorter for cases treated with MMR (0.5 ± 1.5 days, min. 0 day, max. 6 days) compared to SMR (9.8 ± 7.6 days, min. 0 day, max. 36 days; P < .01; r = 0.78). After the introduction of MMR, all patients with PMD opted for that treatment. All MMR cases were treated by 1 ENT specialist. MMR was successful in 19 out of 20 cases (95%). The magnet was repositioned after the first attempt at MMR in 11 cases. A second attempt was required in 5 cases and a third attempt in 3 cases. In the 1 case in which MMR failed, SMR was performed as a secondary treatment.
Thirty-one cases of PMD were treated with SMR as primary treatment option. SMR was performed by 5 ENT specialists with several years of CI experience. In 1 case that was treated with SMR, general anesthesia was used, while in 31 cases local anesthesia was applied. In all cases of SMR, the magnets were repositioned successfully.
MMR resulted in a significantly shorter time between magnet repositioning and CI reactivation (1.4 ± 4.3 days, min. 0 day, max. 18 days) compared to SMR (13.1 ± 6.7 days, min. 7 days, max. 30 days; P < .01; r = 0.76). The mean total CI downtime was significantly shorter for MMR (1.9 ± 4.8 days, min. 0 day, max. 20 days) than for SMR (22.9 ± 11.9 days, min. 7 days, max. 66 days; P < .01; r = 0.80).
Complications
Cases of SMR had a significantly longer mean total follow-up time (48.8 ± 33.8 months, min. 2 months, max. 112 months) than those of MMR (17.7 ± 8.4 months, min. 5 months, max. 32 months; P < .01; r = 0.47). In the 19 cases of successful MMR, no complications occurred. Significantly, more complications were found in the 32 cases of SMR (8 complications; 25%; Fisher’s Z: P = .04; ϕ = 0.32). For SMR, early complications occurred in 6 out of 32 cases (19%); 5 patients complained about pain 1 week after the operation, whereas in 1 patient, wound healing was prolonged at 4 weeks. Concerning late complications, pain was reported more than 1 month postoperatively in 2 cases (6%). In no case was a polarity reversal of the internal magnet observed.
Patient characteristics and a comparison of the results of SMR and MMR are shown in Table 1.
Patient Characteristics and Comparison of the Results of Surgical and Manual Magnet Repositioning.
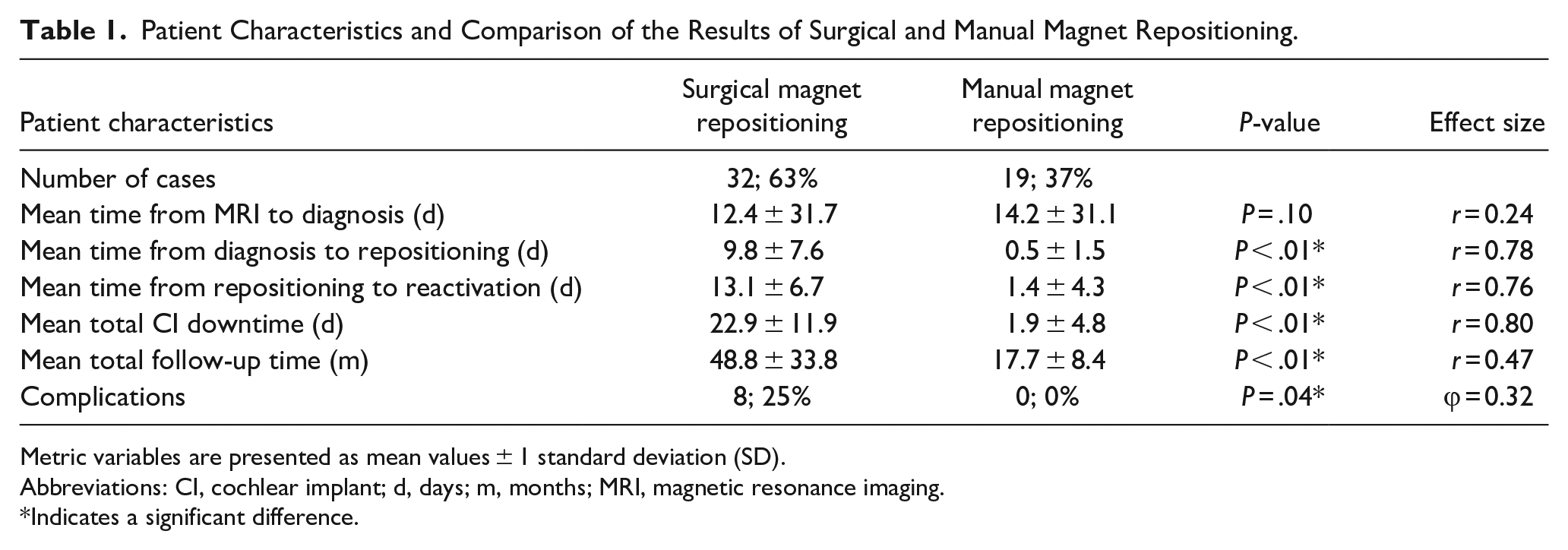
Metric variables are presented as mean values ± 1 standard deviation (SD).
Abbreviations: CI, cochlear implant; d, days; m, months; MRI, magnetic resonance imaging.
Indicates a significant difference.
Discussion
MMR was first performed successfully in 2013 by di Nardo et al., who managed a case of magnet dislocation in an Advanced Bionics HiRes 90K after MRI. 25 A series of 3 cases of PMD repositioned with MMR was published by Carlson et al. in 2015. 24 Seven cases were published with an update of this case series by Fussel et al. in 2021. 10 All cases were successfully repositioned with no complications. Our institution’s first experiences with MMR were published in 2021 and showed 9 cases of PMD that were treated successfully. 28 On the basis of these results, we aimed to compare MMR with the previous standard of care, SMR. As our study shows, the use of MMR can shorten the total CI downtime by approximately 3 weeks, allowing CI patients almost uninterrupted use of their devices. Even when SMR was performed on the day of diagnosis, there was still a mean time of almost 2 weeks until reactivation of the CI. These findings are in line with Leinung et al., who described a median time of 20 days between SMR and CI reactivation and a median CI downtime of 29 days. 18 Furthermore, Holtmann et al. recommended a period of 3 weeks from SMR to CI reactivation. 20 In our experience, wound healing is completed at least 2 weeks postoperatively in the majority of patients and the external processor can be used again. In addition to the disadvantage of not being able to use the CI for a certain time with the use of SMR, there is a risk of implant infection. In the series of 9 patients treated with SMR presented by Leinung et al., 1 experienced a postoperative implant infection requiring device explantation and consecutive reimplantation, resulting in a CI downtime of 1.2 years. 18 Device loss after SMR due to postoperative infection was also reported by Tam et al. 17 Against this background, it is important to emphasize that MMR showed significantly less complications due to the noninvasive manner with no need to expose the CI. Thus, a risk of infection and wound healing disorders as is reported for SMR can be minimized.
One advantage of SMR is the direct view of the magnet, allowing visual confirmation of a safe magnet relocation. In addition, the silicone implant housing can be checked for damage caused by PMD, as was reported earlier by Holtmann et al. 20 Damage to the silicone implant housing was not observed in any of our SMR cases. In the case in which MMR was not successful no abnormal findings were seen intraoperatively.
Despite the lack of anesthesia, MMR was well tolerated by patients. Nevertheless, it could be difficult to perform MMR in children, as the procedure itself may be more painful and uncomfortable than SMR.
Interestingly, 4 patients who were excluded from the study experienced spontaneous repositioning of the partially dislocated magnet. In these cases, repositioning happened unintentionally during the ultrasound examination. Spontaneous repositioning has been previously described,16,30 and could be triggered by the pressure applied by the external CI processor or by an ultrasound probe. This observation shows that some dislocated magnets can be repositioned easily, underlining the importance of MMR.
Currently, all major CI manufacturers offer CI devices containing rotatable, diametrical magnets that do not require a compression bandage up to 3 T. However, magnet dislocation will still persist in future years, as the service life of CI devices is several decades. Therefore, when performing MRI in CI patients, specific safety measures have to be applied, depending on the field strength and the implant type.1-4 In this regard, it is worth mentioning that a pressure bandage was not applied in 35% of the MRI examinations (7 out of 20) performed at external institutions. Therefore, staff at institutions dealing with CI patients have to be well trained to guarantee the best possible safety measures before and during MRI and provide immediate care in case of any complications.
In the clinical workup of patients with suspected PMD, we recommend a thorough clinical and ultrasound examination, especially in those with a history of recently performed MRI. If the ultrasound examination is inconclusive, further imaging, for example, X-ray examination or CT scan, can be considered. In cases of suspected PMD, MMR is recommended as the results of the present study show a high success rate and a low risk of complications. In the absence of any skin irritation, the CI sound processor can be used immediately after magnet repositioning. Follow-up after 1 week is recommended to repeat the clinical examination and to confirm the correct position of the magnet by ultrasound visualization. If MMR fails or if complete magnet dislocation is suspected initially, SMR should be planned as soon as possible. A flowchart of a suggested treatment algorithm is given in Figure 1.
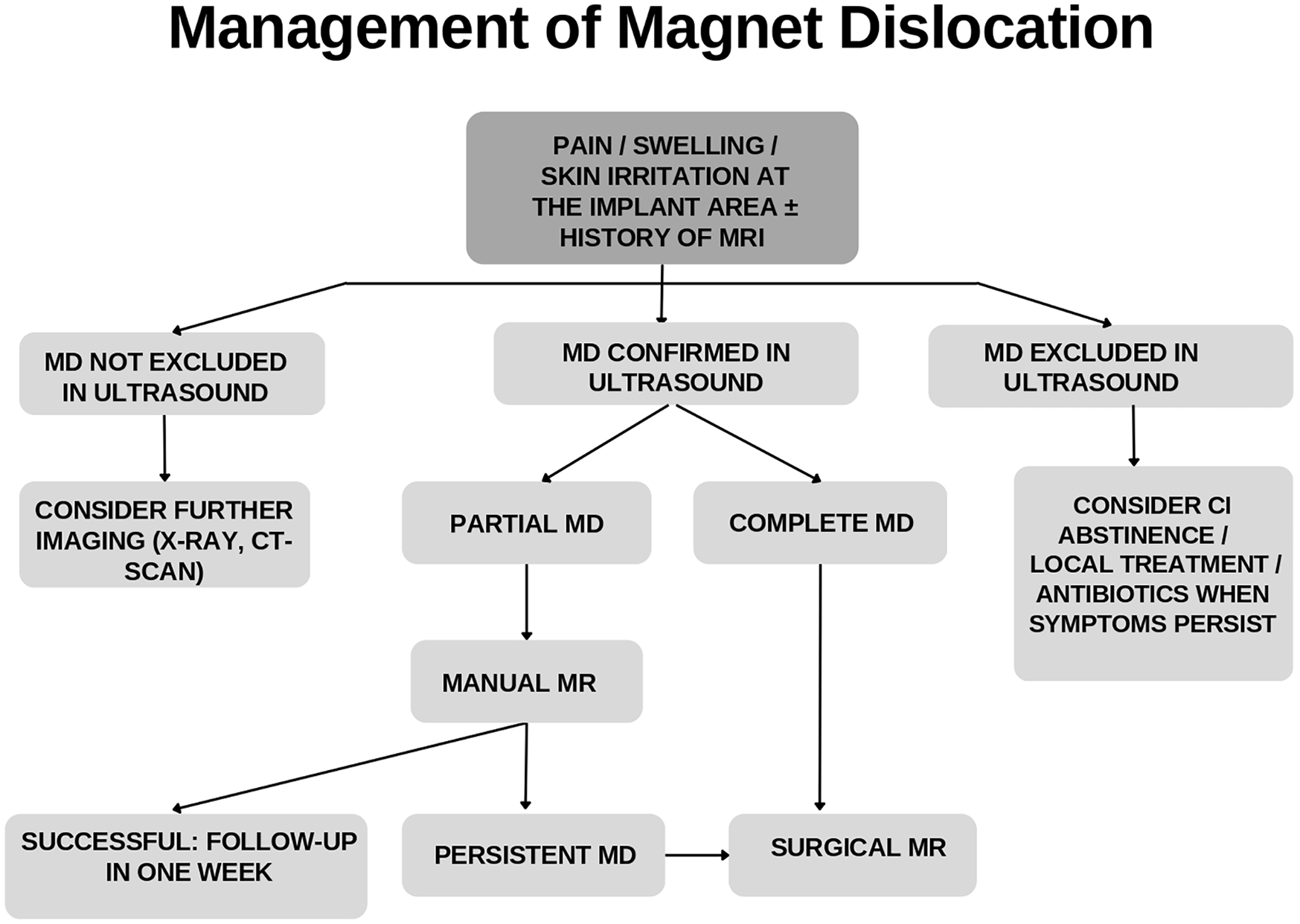
Flowchart for managing suspected magnet dislocation in cochlear implant patients. CI, cochlear implant; CT, computed tomography; MD, magnet dislocation; MR, magnet repositioning; MRI, magnetic resonance imaging.
Limitations
There are several limitations of the study that should be addressed. First of all, the presented results are limited by the retrospective character of the study. Furthermore, only CI devices from 1 manufacturer were included. Also, it is worth mentioning that SMR was performed by 5 different ENT specialists whereas MMR was performed by 1 specialist only. Moreover, the reason why MMR was not successful in 1 case could not be elucidated in this study. In addition, total follow-up time was much longer for SMR than for MMR, as the noninvasive method was first implemented in 2019. Future studies should aim to include devices from different manufacturers.
Conclusion
MRI is the most common cause of PMD in patients with a CI. Noninvasive MMR shows a high success rate with a shorter total CI downtime as well as a lower complication rate compared to the previous standard of care, SMR. MMR should be attempted as first line treatment for PMD, with SMR reserved only for refractory cases. Therefore, practitioners dealing with CI patients should be educated about MMR techniques.
Footnotes
Availability of Data and Materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval to report this study was obtained from the institutional review board (Ethik-Komission der FAU; application number: 99_21 Bc). The study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the institutional review board’s (Ethik-Komission der FAU; application number: 99_21 Bc) approved protocols.
Statement of Informed Consent
Informed consent for patient information to be published in this article was waived, as the data for this study were reviewed retrospectively.
Trial Registration
Registration in a public trials registry was waived due to the purely retrospective character of the study.
